Abstract
Objective
This study aims at identifying the chemical structure of hydromethanolic extracts from Capparis spinosa L. aerial part. In addition, it evaluates the antioxidant activity, studies the synergistic interactions of the various extracts and fractions, and evaluates the extracts’ inhibitory activity against alpha-amylase enzyme.
Methods
We used maceration and decoction to obtain hydro-methanolic extracts, and liquid chromatography LC-MS/MS to ascertain the extracts’ chemical structure. Three techniques were employed to assess antioxidant activity, amylase inhibition was assessed in vitro, and compounds that could inhibit amylase were found using in silico docking simulations.
Results
LC-MS/MS investigation revealed 17 phytochemical compounds, 03 of them, namely salicylic acid, salicin, and curcumin have not before been found in the aerial parts of the caper plant in North African countries. The butanolic fraction F3 showed the highest antioxidant activity, with IC50 values of 74.77 ± 6.08 ug/mL and 41.59 ± 5.89 ug/mL in DPPH and ABTS, respectively. Besides, A0.5 value was 28.64 ± 0.15 ug/mL in the O-Phenanthroline. In addition, most of the mixtures showed synergy, as in f2 + f4 + CSED (1:1:1) with CI effect = 0.22 and IC50 values (43.00 ± 0.76 ug/mL, 19.42 ± 2.36 ug/mL) in DPPH and ABTS, respectively. The results of alpha-amylase in vitro inhibition showed a high ability for all extracts and fractions to inhibit the enzyme compared to acarbose.
Conclusion
Findings show a significant variety of phenolic compounds in the extracts of C. spinosa aerial parts. Based on the molecular docking study, eight compounds were found to be able to occupy the enzyme's active site and form strong bonds with alpha-amylase; this is how they inhibit the enzyme.
Introduction
Due to their therapeutic potential and fewer adverse effects, than synthetic drugs, natural compounds have drawn attention of the pharmaceutical industry. 1 Remarkably, natural sources account for 45% of anti-cancer medications, including plant-based substances like galantamine (used to treat Alzheimer's disease) and artemisinin (antimalarial and anticancer). 2 Polyphenols, a broad class of secondary metabolites, are among these bioactive substances that have become well-known for their numerous pharmacological characteristics, mainly the neuroprotective, antimicrobial, anti-inflammatory, and antioxidant effects.3,4 In the last decade, more than 8000 compounds have been identified by more than 120 000 studies on polyphenols.3–5 These molecules play a crucial role in combating oxidative stress, a key contributor to chronic diseases such as diabetes, cancer, and neurodegenerative disorders.6,7
Capparis spinosa (caper plant), a Mediterranean medicinal herb, is rich in phenolic compounds and has demonstrated significant antibacterial, antioxidant, antidiabetic, and anticancer activities.8,9 While previous research has explored its individual bioactive properties, the synergistic interactions between its phenolic compounds remain underexamined. Understanding these interactions can foster enhanced therapeutic effects, offer novel approaches for drug development. This study advances the field through:
Investigating the synergistic effects of phenolic compounds in C. spinosa, a topic that has not got much attention before.
10
Combining computational (in silico docking) and experimental (in vitro) techniques to provide a thorough examination of antidiabetic activity. Focusing on a geographically rare (semi-desert) variety of C. spinosa that, as a result of environmental stressors, may show distinctive phytochemical profiles.
This study fills in these gaps by offering fresh perspectives on the combinatorial and mechanistic actions of phenolic compounds, paving the way to more potent natural remedies.
Materials and Methods
Plant Materials
C. spinosa aerial parts (leaves and twigs) were collected between May and July 2023 from Djellal region in Khenchela (34° 55′ 9″ N, 6° 53′ 34″ E). Then, they were dried at room temperature away from light and moisture and stored for further use. The laboratory study was conducted between July 2023 and January 2025 at Abbas Leghrour University in Khenchela.
The laboratory study was conducted between July 2023 and January 2025 at Abbas Leghrour University in Khenchela.
Extraction and Fractionation of Phenolic Compounds
We obtained hydromethanolic extract (CSMeoH) using the method suggested by Alsarayreh et al, 11 with slight modifications. In this regard,100 grams of C. spinisa aerial part dry powder was soaked in 400 mL of hydromethanolic (80/20 methanol-water) solution and then left at the ambient temperature away from light for 72 h, The solvent was renewed every 24 h after filtering the mixture using filter paper. The three were combined and concentrated by rotary evaporation at 45 °C to remove the solvent.
The crude hydromethanolic extract (CSMeoH) was fractionated by liquid-liquid separation based on increasing polarities of the solvents (hexane, ethyl acetate, n-butanol, and water) as reported by Rajhi et al 12 The crude extract (15 g) was dissolved in 200 mL of hot water (40 °C) and subjected to a series of liquid-liquid extractions in a separation funnel with solvents immiscible with the aqueous extract. We added 100 mL of hexane to the aqueous extract to eliminate lipids and chlorophyll and left it until two separate phases were formed, the process was repeated 3 times. The remaining aqueous phase, in turn, was subjected to three extractions with ethyl acetate using 100 mL of solvent in each process. The remaining aqueous phase was mixed with n-butanol following the same steps. The ethyl acetate (F2), n-butanol (F3), and aqueous fractions (F4) were dried at reduced pressure at 45 °C and stored at 4 °C.
The second hydromethanolic extract (CSED) was extracted according to the Kada et al 13 method with some modifications. Briefly, 10 g of dry powder of the aerial part of the caper plant was placed in 100 mL of hydromethanolic solution (50:50; methanol: water) and boiled for 20 min. After centrifugation at 3000 rpm for 15 min, the supernatants obtained were filtered and concentrated.
Estimation of the Amount of Phenolic Compounds
The Folin–Ciocalteu method is the most popular and reliable for determining the amount of phenolic compounds, In this regard color development is mostly determined by the oxidation of these chemicals. 14 In this study, the Folin-Ciocalteu approach described by Hayat et al method. 15 measured the phenol concentration in hydromethanolic extracts (CSMeoH and CSED) and other fractions (F2, F3, and F4) of C. Spinosa aerial part. Gallic acid was used as a reference compound to create a typical curve for calculating gallic acid equivalents (GAE). The results were represented in (µg GAE/mg).
The total flavonoid content (TFC) was determined using the AlCl3 colorimetric method, as previously described by Bouriche. 16 Results were presented as micrograms of quercetin equivalents per milligram of dry weight (µg QE/mg). To calculate the quercetin equivalents, a calibration curve was developed with quercetin as a reference.
LC-MS/MS Analysis
A qualitative evaluation was conducted using the LC-MS/MS technique to detect chemicals in the CSMeoH, CSED extracts, and F2, F3, and F4 fractions, as described in Zahnit et al 17 The LC-MS/MS procedure was carried out using a tandem MS system and the Shimadzu 8040 Ultra-High sensitivity with UFMS technology. We used a 3 µm x 150 × 2.1 mm reversed-phase C18 Restek Ultra column to separate chemicals. For the elution gradient, two mobile phases were employed: phase A (water + 0.1% formic acid) and phase B (methanol). The chemicals were eluted using the gradient of these two mobile phases. The injection volume was 5 µL, and the flow rate was set at 0.2 mL/min. We used the following electrospray ionization ESI conditions: 250 °C for the desolvation line (DL), 230 KPs for the CID gas, 6.00 Kv°C for the conversion dynode, 3.00 L/min for the nebulizing gas flow, 400 °C for the heat block, and 10 L/min for the drying gas flow. The gradient was 0 to 1 min A 85%, 1 to 10 min A 5%, 10 to 18 min A 5%, and 18 to 23 min A 85%.
LC-MS/MS analysis was performed using a Shimadzu 8040 tandem mass spectrometer equipped with an electrospray ionization (ESI) source. The analysis was conducted in multiple reaction monitoring (MRM) mode in both positive and negative ionization modes.
Compound identification was achieved by injecting authentic standards (eg, rutin, quercetin, and salicylic acid) and comparing their retention times and MS/MS fragmentation patterns with those of analytes detected in the samples. Additional confirmation was obtained by referencing published spectra and mass-to-charge (m/z) values from the literature.
For calibration, external standard solutions were prepared at different concentrations and injected under the same conditions to confirm system performance and to allow for semi-quantitative comparison across fractions based on peak area integration. Data acquisition and processing were conducted using LabSolutions software.
Antioxidant Activity Assessment
DPPH Scavenging Test
Some minor modifications were made to the protocol described by Ahmed et al 18 to evaluate the 1.1 - diphenyl - 2 – picryl hydrazyl (DPPH) assay. A volume of 40 µL of each extract or fraction at varying concentrations (ranging from 12.5 to 800 µg/mL) was added to 160 µL of a 0.1 mM methanolic DPPH solution. The mixture was incubated for 30 min in the dark at room temperature. Absorbance was measured by A 96-well Perkin Elmer Multimode Plate Reader En Spire at 517 nm. Besides, Butylated hydroxyanisole (BHA) and butylated hydroxytoluene (BHT) were used as positive controls at the same concentration. The results were shown as 50% inhibitory concentration (IC50, µg/mL).
ABTS Cation Radical Assay
The capacity of the methanolic extracts and fractions to scavenge free radicals in the presence of 2,2-Azinobis-(3-Ethylbenzthiazolin-6-Sulfonic Acid), (ABTS), was evaluated using the technique technique outlined by Re et al 19 In brief, a volume of 40 µL of extract or fraction was mixed with 160 µL of pre-prepared ABTS solution (7 mM ABTS radical cation pre-incubated with 2.45 mM potassium persulfate for 12-16 h). After 10 min of incubation at room temperature in the dark, absorbance was measured at 734 nm. BHA and BHT were used as positive controls. and IC50 values (µg/mL) were calculated from dose-response curves.
O-Phenanthroline Activity
The O-Phenanthroline Chelating Activity was conducted using the Szyd lowska-Czerniak et al method. 20 In this regard, we used 10 µL of extract or fraction at various concentrations, added to 80 µL of a mixture containing 30 µL of 0.5% O-phenanthroline and 50 µL of FeCl3 (0.2%). Then 110 µL of methanol was added, the mixture was incubated for 20 min at 30 °C, and absorbance was measured at 510 nm. As in previous assays, BHA and BHT were used as positive references and the solvent as a blank control. ), the results were reported as a A 0.5 (µg/mL).
Synergistic Interactions
To study the synergistic antioxidant activity between different fractions, three fractions (F3, F3, F4) and CSED extra cts were selected and tested with three methods (DPPH, ABTS, and phenanthroline assay), In this context, The fractions (F3, F3, F4) and CSED extract CSED were selected based on their high content of phenolic and/or flavonoid compounds, promising antioxidant activity observed in individual assays, chemical profiles, and solubility behavior.
As described previously by Chou. 21 CompuSyn® software was utilized to calculate the combination index (CI values).
The combination index (CI) values were interpreted according to the method of Chou, where CI < 1 indicates synergy, CI = 1 indicates additivity, and CI > 1 indicates antagonism. In this study, we used the following classification:
– CI < 0.3: strong synergy (SS) – 0.3 ≤ CI < 0.7: synergy (S) – 0.7 ≤ CI < 0.9: moderate synergy (MS) – 0.9 ≤ CI ≤ 1.1: additive effect (A) – CI > 1.1: antagonism (AN)
The software CompuSyn® software automatically calculates these values and generates isobolograms to illustrate the interactions between combinations. This approach is widely used to assess the interaction of antioxidants in plant extracts.
In Vitro Alpha-Amylase Inhibtion
Alpha-amylase inhibition assay was conducted using the DNSA method, as described by Ahmed et al 22 with slight modifications. Type VI-B alpha-amylase enzyme from porcine pancreas (Sigma-Aldrich, A3176) was used. The reaction mixture consisted of 50 µL of enzyme solution (2 units/mL), and 20 µL of plant extract or fraction, and was incubated at 37 °C for 10 min. Subsequently, 50 µL of 0.1% (w/v) soluble starch solution was added and the mixture was incubated again for 10 min. The reaction was stopped by adding 80 µL of DNSA reagent, followed by heating at 95 °C for 10 min. Absorbance was measured at 540 nm using a 96-well microplate reader.
Acarbose was used as the positive control under the same conditions, while the negative control contained the enzyme and starch without any inhibitor. The percentage inhibition was calculated using Equation 1. All experiments were conducted in triplicate.
1Absc = Absorbance control
2(Absb = Absorbance with extract.
3Abse = Absorbance blank
Molecular Docking
A molecular docking research conducted to examine the binding mode of 17 natural compounds from C. spinosa within the active site of the alpha-amylase enzyme. These 17 compounds were selected based on LC-MS/MS analysis of the plant extract, ensuring that only experimentally confirmed constituents were assessed for their potential biological activity. The crystal structure of this enzyme was received from the Protein Database (PDB code: 4GQR) and created with LeadIT version 2.1.8 (available at www.biosolveit.com). During the preparation process, all co-factors, hetero atoms, and water molecules were removed, except for those water molecules located in the active site of the enzyme. Formal charges were applied to the protein and missing atoms of hydrogen were restored. 23 The active site was defined by selecting amino acid residues within a 6.5 ˚A radius around the reference ligand in the crystal structure. 24 This selection process was further refined by including residues beyond 6.5 ˚A which are critical for the cavity's continuity. 25 Protonation states and side-chain orientations of amino acids in the active site were adjusted, followed by energy minimization. The prepared enzyme structure was then exported as a Mol2 file. 26
The SDF formatted three-dimensional structures of the substances under investigation were obtained from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/). Ligand preparation was made using the LigPrep tool in Schr¨odinger's Maestro software (version 11.3). 27 This included the generation of all possible tautomers and ionized states for each compound, along with geometry optimization and partial charge assignment. 28
Molecular docking calculations were carried out using FlexX software based on a semi-flexible docking approach. 29 In this protocol, the protein structure was treated as rigid to maintain its crystallographic integrity while flexibility was permitted for the ligands to explore their potential binding conformations.
The docking parameters were set to generate up to 30 poses per ligand using the incremental construction algorithm of FlexX, with default settings for placement and scoring. The scoring function used is based on empirical and knowledge-based terms accounting for hydrogen bonding and hydrophobic interactions. The docking process evaluated the binding energies and poses of the compounds within the defined active site of the enzyme.
To estimate the potential inhibitory strength of the compounds, docking scores and interaction patterns with key catalytic residues of α-amylase (notably Asp197, Glu233, and Asp300) were examined. Compounds displaying docking scores below −8.0 kcal/mol and establishing stable hydrogen bonds or hydrophobic interactions with these residues were considered to exhibit strong inhibitory potential.
To validate the docking results, the reliability of the docking protocol was assessed using the Root Mean Square Deviation (RMSD) metric. In this context, RMSD values of less than 2.0 ˚A between the docked poses and the reference ligand were used as a benchmark to confirm the accuracy of the docking simulations. 30
ADMET Predictions
The ADMET-AI predictions for eight molecules with strong potency inhibitory of the alpha-amylase enzyme were conducted using the methods described by Swanson et al. 31
Data Analysis
All experiments were carried out in triplicate. Data were presented as mean ± SD. Outliers were assessed using boxplots and Grubbs’ test, and values identified as significant outliers were excluded only when justified by a technical error. The data was processed using the GraphPad Prism® 8 software and compared using one-way ANOVA with Tukey's multiple comparisons post-hoc test. The statistical significance was established at p < 0.05.
Results
Extracts Yield of C. spinosa
The dry plant material and extract amount obtained after removing the solvent were used to compute the average yield of extracts according to the relationship 2, for CSMeoH and CSED extracts, as well as the other fractions (F2, F3, and F4). As shown in Table 1, Which shows the extraction yield, total phenolic content, and total flavonoid content of the hydromethanolic extracts and fractions. The extract yield of CSMeoH extract (24.81 ± 2.16%) was considered much higher than that of CSED extract (11.00 ± 0.80%). As for the fractions, F2 was the least (0.41 ± 0.03%), while F4 was the largest (5.80 ± 0.35%), followed by F3.
Total Phenolic, Total Flavonoids and Extracts Yield of Hydromethanolic Extracts and Fractions of the C. spinosa Aerial Plant.

1Extract yield.
2Extract dry weight
3Plant material dry weight
Estimation of Polyphenolic Compounds Amounts
The amounts of total phenolic compounds, flavonoids, in the C. spinosa aerial part hydromethanolic extracts and fractions were estimated with milligram equivalents of gallic acid and quercetin per gram of extract, respectively. Besides, the obtained the obtained calibration curves were: gallic acid y = 0.004x + 0.091; R² = 0.9969, quercetin y = 0.0048x; R² = 0.997. The obtained results (Table 1), showed a significant difference between the different extracts and the different fractions. The amount of phenolic compounds in CSMeoH extract was 209.19 ± 4.41 (µg GAE/mgEX) compared 138.21 ± 6.41 (µg GAE/mgEX) in CSED extract. Regarding the fractions, the largest amount was recorded in F3, estimated at 304.58 ± 2.05 (µg GAE/mgEX), followed by F2,), while the weakest was in F4, estimated at 104.88 ± 5.88 (µg GAE/mgEX). As for the flavonoid content, the estimated largest amount was in F2, estimated at 204.30 + 8.39 (µg QE/1 mg), while the weakest amount was in F4 estimated at 29.09 + 9.87 (µg QE/1 mg).
LC-MS/MS Analysis
The CSMeoH, CSED extracts, and F2, F3, and F4 fractions from C. spinosa aerial parts were subjected to LC-MS/MS chromatography to identify their chemical composition; Thus, total of 17 phytochemicals was identified, as shown in Table 2 provides detailed information on the phytochemicals identified through LC-MS/MS, including their chemical names, molecular formulas, retention times (RT), and mass-to-charge (m/z) ratios. The table also shows the relative abundance (%) of each compound across the different extracts, and Figures (1,2,3,4, and 5), Chromatograms obtained by LC-MS/MS of different extracts and fractions from the aerial parts of C. spinosa aerial parts. In this context, 14 compounds were identified in the aqueous fraction (F4) (Figure 5), 13 compounds in the CSMeoH extract (Figure 1), and 13 compounds in the ethyl acetate fraction F2 (Figure 3), and 11 compounds were identified in the n-butanol fraction F3 (Figure 4) and 09 in CSED extract (Figure 2).

LC-MS/MS Chromatogram of Crude Hydromethanolic Extract CSMeoh.

LC-MS/MS Chromatogram of Hydromethanolic Extract CSED.

LC-MS/MS Chromatogram of F2 Fraction.

LC-MS/MS Chromatogram of F3 Fraction.

LC-MS/MS Chromatogram of F4 Fraction.
Chemical Composition of Extracts and Fractions of the C. spinosa Aerial Part of, Obtained by LC-MS/MS Technique.
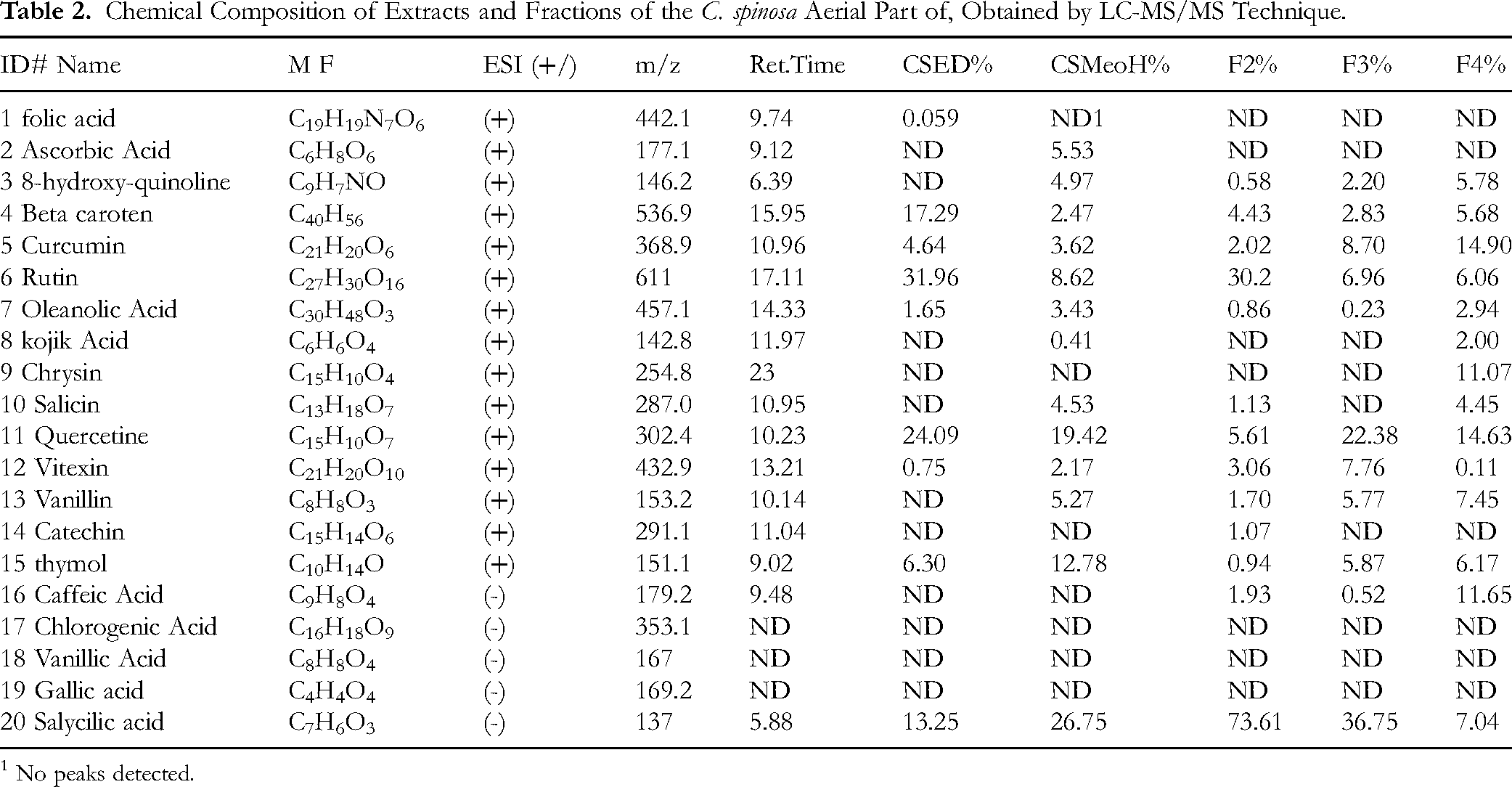
1 No peaks detected.
Antioxidant Activity
Antioxidant
The activity of the C. spinosa aerial parts extracts was assessed using DPPH, ABTS, and phenanthroline techniques, and the antioxidant values were compared to those of BHA and BHT as standards. The results showed that, regarding the hydromethanolic extracts, the CSED extract gave a higher antioxidant activity than the CSMeoH extract under DPPH method (p < 0,0001). The obtained results were as follows: IC50 values were (124.56 ± 3.85 µg/mL for CSED VS (207.89 ±8.93 µg/m) for CSMeoH, and BHT 13.43 ± 0.65 µg/mL, BHA 6.24 ± 0.2 µg/mL for the reference compounds.. With significant differences (p < 0,0001) as shown in the Table 3 summarizes the antioxidant potential of extracts and fractions, as evaluated using DPPH, ABTS, and Phenanthroline assays. Furthermore, synergistic interactions between selected fractions were reported, and Figure 6 The histogram illustrates the mean IC50 values (µg/mL) representing the antioxidant activity of extracts, fractions, and reference compounds, as determined by the DPPH free radical scavenging assay. Each bar corresponds to the average IC50 of a sample, with error bars indicating the standard deviation (± SD) of three independent experiments..

DPPH Radical Scavenging Activity of Different Extracts and Fractions of C. spinosa Aerial Parts, Obtained by GraphPad Prism® 8 Software.
Antioxidant Activity of Extracts and Fractions of the C.spinosa Plant, Synergistic Interactions Between Different Extracts and Fractions.

1 a,b,c,d,e,f Express significant differences between different groups. Similar letters: there are no significant differences. Different letthers there are significant differences.
2 SS Strong Synerdy, S Synergy, MS Moderate synergy, NA Additive non interactions, SLA Slight Antagonism, A Antagonism.
In ABTS, IC50 values of CSED and CSMeoH were 37.60 ± 1.52 µg/mL and 84.00 ± 11.67 µg/mL, respectively, while those of the reference compounds BHT and BHA were (1.50 ± 0.30 µg/mL) and (1.90 ± 0.20 µg/mL), respectively, as shown in Table 3 and Figure 7 histogram presents the mean IC50 values (µg/mL) of antioxidant activity for different plant extracts and reference compounds, as assessed by the ABTS radical cation decolorization assay. Each bar represents the average IC50 value for a given sample, with error bars showing the standard deviation (± SD) from three independent measurements. Besides, under phenanthroline method, CSMeoH extract showed a value of A0.5 (41.43 ± 1.04 µg/mL), while CSED extract showed a value of 94.17 ± 0.41 µg/mL; the two values are greater than those of BHA and BHT, which are 0.96 ± 0.06 µg/mL, 2.43 ± 0.22 µg/mL; respectively (p < 0,0001); see Table 3 and Figure 8 The histogram displays the mean IC50 values (µg/mL) of antioxidant activity for various plant extracts and reference compounds, evaluated using the phenanthroline assay. Each bar corresponds to the average IC50 value for a specific sample, with error bars indicating the standard deviation (± SD) of three independent replicates.

ABTS Antioxidant Activity of Different Extracts and Fractions of C. spinosa Aerial Parts, Obtained by GraphPadPrism® 8 Software.
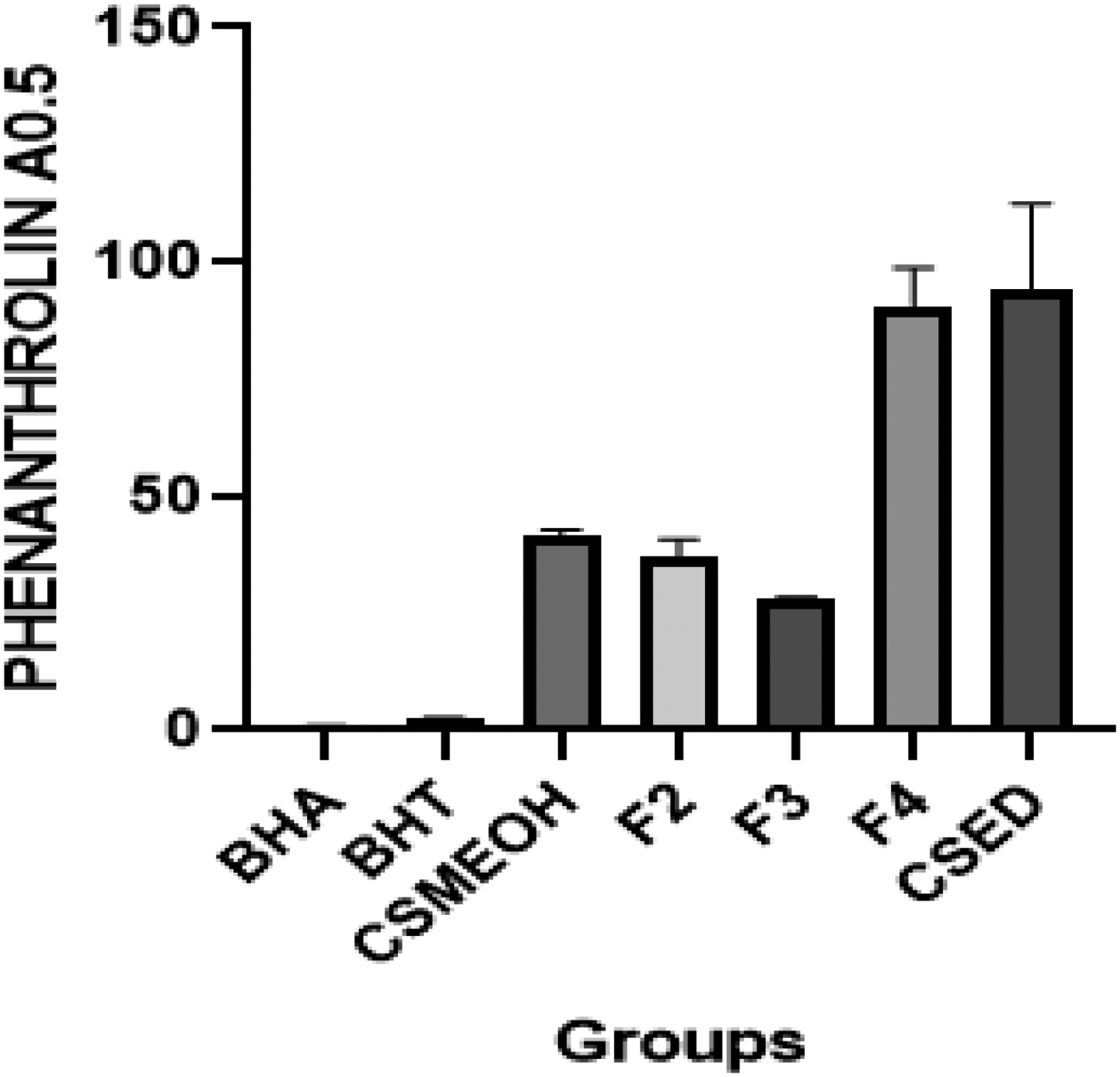
Phenanthroline Assay of Different Extracts of C. spinosa Aerial Parts, Obtained by GraphPad Prism® 8 Software.
As for the fractions, the results show a high antioxidant activity. In this regard, F3 showed the highest activity, followed by F2 and F4; the latter recorded the weakest antioxidant activity. The highest activity was registered in the phenanthroline method, as showns in the Table 3 and Figure 8 where the A0.5 value approval for the F3 was 28.34 ± 0.15 µg/mL, and the highest value was 90.75 ± 7.96 µg/mL for F4. Under ABTS (Figure 7), the value of IC50 is 41.59 ± 5.89 for F3 and 78.80 ±10.83 µg/mL for F4. Regarding DPPH (Figure 6), the value is 74.77 ± 6.08 µg/mL for F3 and 496.90 ± 23.39 µg/mL for F4 (See Table 3). There were also significant differences between BH versus F3 (p = 0,0089), BHT versus F3 (p = 0,0136) in the phenanthroline method, and (p = 0,0001) in DPPH and ABTS.
Synergistic Interactions
We chose F2, F3, F4, and CSED to study the synergistic interactions between different fractions and hydromethanolic extract and, then, processed the results using CompuSyn software to calculate the combination index CI.
Findings showed a very strong synergy in the mixtures F2 + F4 + CSED (1:1:1) and F2 + F3 + F4 + CSED (1:1:1:1) in ABTS scavenger assay. In this context, CI effect was 0.22 and IC50 was 19.42 ± 2.36 µg/mL for mixture 01, and 0.28 and 22.73 ± 2.8 µg/mL for mixture 02. Besides, the first mixture showed antagonism (CI = 1.47) while the second showed a strong synergy (CI = 0.66) in phenanthroline method. As for the rest of mixtures, they showed diverse interactions, including synergy, antagonism, and additivity. In this regard, strong synergy was found in many mixtures (See Table 3 and Figure 9 Isobolygram of synergetic interactions in antioxidant activity (DPPH and ABTS assay) at 50% free radical scavenging. Generated with CompuSyn software.), such as in F3 + CSED (1:1) (0.49 = CI) and F3 + F4 + CSED(1:1:1) (0.42 = CI) under ABTS method, and in F2 + F3 (1:1), (0.68 = CI) and F2 + F3 +CSED (1:1:1)(CI = 0.59) under phenanthroline method

Isobolograms Generated by CompuSyn Software for Synergetic Interaction in DPPH and ABTS Assay. Fa = 0 .5: Fraction Affected by the Dose of the Combinations at 50% Radical Scavenger. Dose A = Fraction 1or Extract 1 ; Dose B = Fraction 2 or Extract 2.
Antagonism interactions were reported in some mixtures under the three methods, such as in F3 + F4(1:1) in the ABTS, showing a strong antagonism, as the CI was equal to 1.41. As for the phenanthroline method, the highest CI (1.87) was reported in F2 + F4(1:2), showing a strong antagonism as well.
Alpha Amylase Inhibition in Vitro
The extracts and fractions showed high inhibitory activity for the alpha-amylase enzyme, more than acarbose (Table 4 Which displays alpha-amylase inhibitory activity by extracts, fractions and acarbose, which is expressed as IC50, and Figure 10, the figure displays the mean IC50 values (µg/mL) of Alpha-amylase inhibitory activity for various plant extracts and reference compounds). The activity in the fractions was higher than the activity in the hydromethanolic extracts, In this context, the highest activity IC50 was 5.63 ± 0.99 µg/mL, and it is specific to F3. Besides, CSED extract showed a strong activity (IC50= 9.2 ± 0.42 µg/mL) compared to CSMeoH extract (IC50= 34.44 ± 8.44 µg/mL).

Alpha-Amylase Inhibitory Activity of Different Extracts and Fractions of C. spinosa Aerial Parts, Obtained by GraphPadPrism® 8 Software.
Alpha-Amylase Inhibitory Activity of Hydromethanolic Extracts and Fractions of C.spinosa Aerial Parts.

1 a and b Express significant differences between different groups. Similar letters: there are no significant differences. Different letters: there are significant differences.
Molecular Docking
Molecular docking analysis of 17 natural compounds from C. spinosa aerial part, within the active site of alpha-amylase, revealed that eight of them showed stronger interaction energies compared to acarbose, which is the reference inhibitor (−20.46 kJ/mol). This finding highlights their significant inhibitory potential (Table 5), which shows the binging energy values of different compounds as well as acarbose.
Binging Energies Values of 13 Natural Compounds Derived from C. spinosa Aerial Part. Within -Amylase, Compared to Acarbose as Standard Inhibitors.

The results show that these compounds can occupy the active site of the enzyme, as shown in Figure 11, It shows the position of acarbose and chrysin in the active site of alpha amylase. And create multiple reactions within the active site of the enzyme, as shown in Figure 12, which shows the binding mode interactions between acarbose and chrysin at the active site of amylase.

Positioning of Acarbose (A) and Chrysin (B) in the -Amylase Active Site. the Active Pocket is Represented in Cyan “Surface” Whereas the Ligand Atoms are Color-Coded as Follows: Carbon in Green, Oxygen in red and Nitrogen in Blue.

Binding Mode Interaction of Acarbose (A) and Chrysin (B) into the -Amylase Active Site. Purple Broken Lines Show the Hydrogen Bonds and the Green Area Shows the Hdrophobic Interactions.
ADMET-AI Analysis
As shown in the Tables 6 and 7, they present ADMET predictions for eight compounds from caper plant extracts, as well as the reference compound. ADME predictions revealed that the majority of compounds showed moderate levels of absorption in the human intestine compound chrysin, salicylic acid, and quercetin (1.00), to weak rutin (0.09), and relatively high oral bioavailability salicylic acid (0.81) to weak rutin (0.18), making them well above average in terms of absorption capacity. Water solubility was relatively high to moderate for salicylic acid (0.49) and ascorbic acid (−1.64 log(mol/L)), indicating no major difficulties in dissolution for most compounds. In addition, most compounds showed poor lipid solubility (salicylic acid −0.68 log-ratio, salicin −0.94). negative hydration free energy, and low cell permeability for most compounds, such as for rutin (−6.82 log(10-6 cm/s)) to moderate in some compounds, such as for chrysin (−4.79log(10-6 cm/s)). Moreover, it showed poor permeability to PAMPA’ (0.07, 0.09, 0.11) in rutin, salicylic acid, and vitexin, respectively, and moderate rutin (0.14) to relatively high chrysin (0.57) in P-glycoprotein interaction, indicating moderate efflux activity. Penetration across the blood-brain barrier is generally low, rutin and vitexin (0.06, 0.07), respectively.
ADMET Physico-Chemical Properties of Eight Strong Compounds That Inhibit Alpha Amylase in Silico.

1 chr:Chrysin,Rut:Rutin,Sal:SalicylicAcid,Vit:Vitexin,qur:qurcitin,Sln:Salicin,Acs:AscorbicAcid,Cur:Curcumin,Aca:acarbose.
2 Prd:predictionvalue,DBP:DrugBankPercentile.
Follow Table No. 6, ADMET Physico-Chemical Properties of Eight Strong Compounds That Inhibit Alpha Amylase in Silico.

1 chr:Chrysin, Rut: Rutin, Sal: Salicylic Acid,Vit:Vitexin, qur:qurcitin,Sln:Salicin,Acs:AscorbicAcid,Cur:Curcumin,Aca:acarbose.
2 Prd:predictionvalue,DBP:DrugBankPercentile.
In addition, we found out moderate plasma protein binding of, salicylic acid ((52.73%), to high chrysin(89.84%), indicating significant binding to circulating blood. The constant volume of distribution of chrysin and vitexin (1.24-1.28 L/kg) results in moderate extravascular spread.
Besides, except for the compound chrysin, most of the compounds showed slight to moderate inhibition of cytochrome P450 enzymes, including CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4. In addition, the prediction results showed weak to moderate potentials for the compounds to be substrates for cytochrome P450 enzymes. The effect on metabolic processes involving these enzymes will not be significant.
The prediction process showed that most of the compounds have very short half-lives; ascorbic acid and curcumin (00 h), and salicin, and salicylic acid (1.43 . 4.84 h). This indicates that their residence time in the human body is very short. Moreover, half-lives of rutin and vitexin are 49.51 and 49.81 h, respectively.
Besides, findings showed moderate to high clearance rates in both hepatocytes and microsomes However, the acute toxicity of most of the compounds is rather low, as LD50 values of curcumin, ascorbic acid, salicin, and salicylic acid are 1.44, 11.55, 11.86, and 22.04, respectively, unlike rutin whose value is slightly high. In addition, the mutagenic risk was moderate to high, while the cancer risk was negligible in chrysin, rutin, quercetin, and salicin (0.03, 0.02, 0.06, and 0.03, respectively).
Discussion
The extraction process was made in two different ways, namely (maceration and decoction), and we selected the 80:20 and 50:50 methanol–water ratios to maximize the extraction of phenolic compounds with varying polarity from the study plant material. Literature has consistently shown that aqueous methanol mixtures outperform pure solvents in extracting polyphenols. For instance, Inhwan & Jihyun. 32 found out that 80% methanol achieves significantly higher total phenolic yields and antioxidant activity compared to lower methanol ratios in mulberry fruit extracts. Additionally, extensive reviews indicate that solvent polarity adjustments directly impact phenolic recovery, making ratios of 50:50 and 80:20 common choices for optimizing extraction efficiency. We included both ratios to compare their efficiency and ensure optimal recovery of a broad profile of polyphenolic compounds.
Polyphenolic compounds are characterized with a very important biological activity, and flavonoids are one of the most important classes of polyphenols. Caper is a plant rich in secondary metabolites, including phenolic compounds and flavonoids. According to Hosseini Ravesh et al, 8 liquid-liquid extraction is the process of diluting or concentrating the components of the crude extract. In this study, 304.58 ± 2.05 µg GAE/mg and 204.30 ± 8.39 µg QE/mg are the largest obtained quantities of phenolic compounds and flavonoids; they were found in the F3 and F2 respectively. They are larger than the quantities obtained in a study conducted in Tunisia, which found out values of 259.31 ± 6.177 µg GAE/mg and 107.291 ± 8.65 µg QE/mg for phenolic compounds and flavonoids, on caper leaves hydroalcoholic extract obtained by maceration, respectively. 12 Moreover, F3 showed a higher amount of phenolic compounds compared to other fractions and to CSMeoH and CSED extracts, while F4 showed the least amount of phenolic compounds and flavonoids.
Besides, F3 showed the highest number of phenolic compounds while F2 the highest number of flavenoids. On the other hand, the same study in Tunisia showed that the n-butanol fraction had the highest content of flavonoids, and that the crude hydromethanolic extract CSMeoH obtained by maceration revealed a greater concentration of flavonoids and phenolic substances compared to CSED extract. Our results are consistent with those of Tagnaou et al 33 for CSMeoH extract. The physicochemical nature of the used solvents and the chemical properties of the plant compounds, mainly the solubility, could explain the different distribution of polyphenolic compounds in the different fractions. It is also assumed that the phenolic compounds of the caper plant are highly polarized due to the hydroxide groups. Another study in Tunisia, by Rajhi et al, 34 showed great agreement with our results, indicating that ethyl acetate fraction is rich in phenolic compounds and flavonoids. This means that the selectivity of the solvents ethyl acetate and n-butanol is generally consistent with the polyphenolic compounds present in the aerial part of caper plant. The slight difference from one study to another is due to geographical differences, growth stage, and genetic factors of the plant. 35
The components of the aerial part of C. spinosa were qualitatively analyzed using UPLC-ESI-MS-MS, allowing for the identification of 17 compounds. The results of our study are consistent with those of Yahia et al, in Tunisia, where 18 compounds were identified using LC/MS technique; eight compounds were phenolic acids and ten were flavonoids.
Table 2 shows that most of the compounds in CSMeoH extract are salicylic acid, thymol, quercetin, and rutin, while in CSED extract, they are salicylic acid, beta carotene, quercetin, and rutin. Some of caper biomolecules have been detected by other researchers. Yahia et al, 36 reported that the catechins, rutin, and quercetin were identified in extracts of caper leaves obtained by three different extraction methods: reflex, maceration, and ultrasonic. Catechin, caffeic acid and rutin were identified in the caper's hydromethanolic extract using HPLC, 37 and vanillin. 38 The most abundant flavonoid, both in the buds and fruits, appears to be rutin, 39 and beta-carotene. 40
To the best of our knowledge, other compounds in caper plants have not yet been identified. According to the study's chemical analysis, the aerial portion of caper plant has a lot of phenolic chemicals, such as flavonoids, which are crucial for the pharmaceutical industry. The presence of phenolic chemicals in therapeutic plants is crucial because of their antioxidant effect on inflammation, cancer, heart problems, and stroke. 41 Rutin is a flavonoid that shows remarkable biological and pharmacological activities. 40
Moreover, based on our in-depth investigation, our study identified three compounds in C. spinosa aerial part, namely salicylic acid, chrysin, and curcumin, unlike previous studies in the North African region of Algeria, Tunisia, Libya, and Morocco.
We evaluated the antioxidant activity of hydromethanolic extracts of caper plant aerial parts, and other fractions, in three methods and found out a high antioxidant capacity, as shown in Table 3. To assess the reliability of the methodology, IC50 and A0.5 of the obtained reference compounds were compared to those of the reference compounds of other studies, showing no significant differences. For example, IC50 or A0.5 for BHT obtained in DPPH, ABTS, and Penanthroline assay was (IC50: 13.43 μg/mL, IC50: 1.50 μg/mL C, and A0.5: 2.43) respectively compared to (IC50: 15.74 μg/mL, IC50: 7.54 μg/mL, and A0.5: 2.42 μg/mL) obtained by Elkolli e al. 42
Besides, There was also a link between polyphenol concentration in extracts and the antioxidant activity; many previous studies have confirmed this. 43 Our results agree with Elshibani et al 44 The strong antioxidant activity of caper extracts is explained by the high content of phenolic compounds; they are effectively considered antioxidants for their composition. Many studies have shown a strong relation between free radical scavenging activity and total phenols.36,44,45 Moreover, the antioxidant activity of polyphenols is mainly related to their composition. Many studies have also confirmed that; the number of functional groups and their status: hydroxyl OH, carboxyl COOH, and methoxy OCH3, 46 indicate a relation between the number of OH groups and the antioxidant activity. Skroza et al 47 also mentioned that inserting OH in the para or ortho position enhances the activity of the compounds.
Catechins derive their ability to be an antioxidant from the hydroxyl group of ring B, as the hydroxyl groups on ring B and ring C of quercetin are more effective in scavenging free radicals than those on ring A. Gallic, syringic, and caffeic are phenolic acids that were reported to be effective in scavenging free radicals, due to the phenolic group. 48
Catechins derive their ability to be an antioxidant from the hydroxyl group of ring B, as the hydroxyl groups on ring B and ring C of quercetin are more effective in scavenging free radicals than those on ring A. Gallic, syringic, and caffeic are phenolic acids that were reported to be effective in scavenging free radicals, due to the phenolic group. 48
It is easy to know the biological activity of the extract, but it is difficult to determine the origin of this activity and decide whether the source of this activity is all the compounds of the complex, many of them, or a single compound because the plant extract consists of a variety of compounds in different concentrations. 49
To deepen knowledge about the properties of antioxidants and understand the interaction between the biologically active compounds present in the extracts of C. spinosa plant aerial parts and their interaction with free radicals present in the reactive medium such as DPPH and ABTS, we studied the synergistic interactions between F2, F3, and F4 fractions, and CSED extract using DPPH, ABTS, and phenanthroline assay. The results are presented in Table 3 above, showing various reactions. Some mixtures showed a strong synergy, as is the case in F2 + F4: CSED (1:1:1) CI = 0.22 in ABTS, some were additive, while some, as F3 + F4 (1:1) CI = 2.42, showed antagonism in DPPH.
The synergistic interactions can be explained by the cooperation occurring between two or more compounds. Many previous studies examined the interactions between polyphenols in the antioxidant activity. Skroza et al, 47 studied the interactions between ten phenolic compounds, tested many of their mixtures, and showed a synergistic interaction in most of the mixtures. For example, the mixture of p-coumaric and ferulic acid showed the highest difference (synergy), at 311%. In addition, caffeic acid and sinapic acid showed a high difference, estimated at 218%. Some mixtures also resulted in antagonistic reactions, as is the case with gallic + vanillic acid and gentisic + syringic acid. 47 Palafox-Carlos et al, 50 reported the synergistic interactions between gallic acid and protocatechuic acid, gallic acid and chlorogenic acid, and protocatechuic acid and chlorogenic acid. 46 In another study, Hu et al 51 tested the synergistic interactions between polyphenols and carotenoids in mango plants using DPPH and ABTS. The study showed high synergy, especially in lutein and gallic acid.
The synergistic interactions in most cases occur through several mechanisms: enzyme activity changes, as do repair and regeneration; coupling oxidation; oxygen absorption; and metal ion complexation.
On the other hand, the rivalry between antioxidant regeneration, the substitution of the microenvironment of one antioxidant by another antioxidant, and the creation of antioxidant free-radical adducts factors can influence the antagonism between antioxidants. 10 Previous studies have also confirmed the strong synergy between phenolic compounds in the antioxidant activity, although sometimes there is an antagonistic interaction, for example, as previously mentioned (gallic + vanillic acid and gentisic + syringic acid). In another study, the results showed Rutin and hydroquinone with strong antagonism. 52 In addition, the interpretation of synergistic and antagonistic interactions is not limited to qualitative criteria only; even the concentration of antioxidant compounds in the mixture has a major role in these interactions, and the dominant compound is not always responsible for the antioxidant activity. Sometimes the activity is due to synergy between two or more compounds. 47
The results of alpha-amylase inhibition in vitro of the statistical test showed significant differences between the extracts and fractions compared to acarbose (P < 0.0001). and no significant differences between fractions and extracts.. These results confirm the inhibitory activity of all C. spinosa plant extracts against alpha-amylase. Acarbose was an effective reference compound in our assay, allowing for comparison and validation of the inhibitory effects observed in the plant extracts. In our assay IC50 of acarbose was 528 µg/mL, unlike 130 µg/mL in the work of Zheng et al 53 These differences in results may be due to the source of alpha-amylase, the composition of the test buffer, the incubation time, and the substrate concentration.
Previous studies have also confirmed the inhibitory activity of C. spinosa against the alpha-amylase enzyme, 54 due to the high effectiveness of phenolic compounds found in extracts of the aerial parts of the caper plant, 50 In a study on blueberry leaf polyphenols, the compounds could competitively engage to the enzyme and compete with the substrate to create complexes that would inactivate the enzyme. 53 In addition, phenolic compounds have different mechanisms to prevent diabetes, such as inhibiting the digestion of carbohydrates and the absorption of glucose by inhibiting the enzymes amylase and glycosidase, leading to a decrease of blood glucose. 41
The results of alpha-amylase inhibition in silico (molecular docking) align with the experimental evaluation of biological activity, which demonstrated that the IC50 values of the hydromethanolic extracts and fractions were notably better than those of acarbose, further confirming their superior inhibitory efficacy.
Chrysin demonstrated a remarkable binding affinity thanks to its ability to establish multiple interactions within the enzyme's active site. Specifically, it formed six hydrogen bonds: two with Glu233, two with Asp197, one with Arg195, and one with a water molecule located in the binding site (Figure 12). In comparison, acarbose formed seven hydrogen bonds, three of which involved Asp197, while the remaining bonds included Thr163, Asn298, Arg195, and Asp300. Although acarbose formed one more hydrogen bond than chrysin, the latter showed superior binding energy thanks to additional key factors beyond the number of hydrogen bonds. Notably, chrysin formed two strong hydrogen bonds with Glu233, critical residue for the catalytic activity of alpha-amylase., 55 which substantially enhanced its binding strength. Furthermore, chrysin showed a robust hydrophobic interaction profile, engaging residues such as Tyr62, Asp197, Asp300, His305, Trp59, and Trp58 within the enzyme's hydrophobic pocket. While acarbose also formed hydrophobic interactions with residues like His299, His305, Trp58, Trp59, and Tyr62, the distribution and effectiveness of these interactions were less optimal compared to those of chrysin (Figure 11). Collectively, these findings underscore the enhanced interaction profile of chrysin, making it as a promising candidate for alpha-amylase inhibition.
The results of the in silico study show that eight compounds showed stronger interaction energies compared to acarbose, as found by the in vitro study, which showed strong inhibition of different fractions and extracts against alpha-amylase. These results confirm the great effectiveness of phenolic compounds in inhibiting alpha-amylase in particular, and diabetes in general.
The absorption capacity of the biologically active elements in biological tissues is important and can enhance the absorption of oral compounds. 56 Lipinski's rule introduces 5 criteria to evaluate the absorbability of a compound. 57 According to this rule, our study found out that seven out of eight compounds showed greater absorbability than acarbose, namely chrysin, salicylic Acid, salicin, ascorbic acid, and curcumin (4.00) with a percentage of (63.80%), vitexin (3.00) with a percentage of 20.92%, and quercetin (2.00) with a percentage of 9.85%, while rutin and acarbose (1.00) showed a percentage of 2.91%, as shown in Tables 6 and 7.
Moreover, all compounds have a higher oral availability than acarbose, and salicylic acid, salicin, and ascorbic acid have a lower potential for P-glycoprotein inhibition than acarbose.
Cytochrome P450 is vital in metabolic processes, mainly in body detoxification, and has a major influence on the drugs’ effect on the body, as it may activate or inhibit many of them. 58 The inhibition of cytochrome P450 by the eight compounds is not strong, as the results showed that the inhibition probability is generally weak to moderate; the same applies to their being a substrate. In contrast, acarbose showed lower probabilities in both cases. In addition, the risk of mutagenicity is relatively high in most compounds, while the risk of carcinogenicity is low, and most compounds have a short half-life compared to acarbose.
Notwithstanding the encouraging outcomes, this study has a number of limitations. First, polarity gradient-based fractionation offers an initial chemical separation but does not ensure the isolation of pure active compounds. Second, chemical characterization using LC-MS has enabled the initial identification of phytochemicals, but many compounds remain unidentified or have not been confirmed using reference standards, what may affect the accuracy of the phytochemical profile. Third, the extraction was carried out using straightforward and commonly used methods, which may not be as effective as other sophisticated extraction techniques in isolating refractory or low-concentration compounds.
Although molecular docking provided insights into potential interactions between selected compounds and target enzymes, the results are predictive and need experimental validation. Therefore, further studies involving in vivo models, compound isolation, toxicity assays, and mechanistic analyses are needed to confirm and extend the current findings.
Conclusions
This study investigated the chemical structure of C. spinosa aerial part, the antioxidant synergistic interactions between different fractions and extracts, and the extracts’ capacity to inhibit alpha-amylase. Findings show that C. spinosa aerial part extracts are rich in phenolic compounds, extracts show antioxidant activity potency, and butanol fraction has the strongest antioxidant activity. Moreover, synergism was found in the antioxidant activity in most of the mixtures, and antagonism was reported in other mixtures. Regarding the in vitro inhibition of amylase, the study showed a stronger inhibitory activity for the different extracts and fractions than acarbose. Eight compounds were found to be able to occupy the active site of alpha-amylase, form strong bonds with it, and, thus, inhibit the enzyme. This study enriches the literature on local caper and can be used in the pharmaceutical field for the development of natural products. Its results highlight the possibility of using caper plant's aerial parts as a natural alternative to diabetes medications, and as a natural antioxidant in nutritional supplements thanks to its richness in effective compounds. Further studies are necessary to better understand the synergistic mechanisms that are complicated due to the chemical composition of the extracts.
Footnotes
Acknowledgments
We would like to thank the Algerian Ministry of Higher Education and Scientific Research and the General Directorate for Scientific Research and Technology Development of Algiers for their financial support.
Author Contributions Statement
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All relevant data are included in the manuscript. Additional details or raw data can be provided by the corresponding author upon reasonable request.


