Abstract
Objective
Pleurospermum candollei is an edible plant, used traditionally to treat several ailments. This study aimed to explore P. candollei's potential as a phytotherapeutic and functional food with increased yield of bioactive contents.
Methodology
Ultrasound-assisted extraction (70% methanol) to increase the yield of phenolic-rich extract. Spectroscopic and HPLC-PDA analysis for the quantitation of polyphenols. The GC-MS identified phytochemicals selected for molecular docking and ADMET studies. DPPH, ABTS, H2O2, and nitric oxide free radical (antioxidant), Protein denaturation and 5-lipoxygenase (anti-inflammatory), and monoamine oxidase-A and cholinesterase inhibition assays (cerebral disorders) were used to assess the therapeutic properties of the extract.
Results
The extract yield: 23.33%. The total phenolic content: 204.92 ± 2.52 mg GaE/g DE. HPLC-PDA revealed significant quantities of benzoic acid, caffeic acid, p-coumaric acid, coumarin, kaempferol, and chlorogenic acid. The molecular docking scores (binding energies in kcal/mol) for 5-lipoxygenase (−6.3 to −9.8), myeloperoxidase (−5.7 to −9.1 versus baicalein −7.7), monoamine oxidase-A (−6.1 to −9.2 versus clorgyline −5.5), acetylcholinesterase (−6.6 to −10.0) and butyrylcholinesterase (−6.6 to −12.5). In silico ADMET studies predicted the pharmacokinetics and safety of docked compounds. The IC50 values for DPPH, ABTS, H2O2, and NO free radicals were 168.06 ± 1.72, 123.24 ± 0.84, 158.54 ± 1.18, and 149.64 ± 1.02 µg/mL, respectively and hemolysis was 5.18 ± 0.52%. Protein denaturation inhibition: 45.86 ± 1.15% versus diclofenac sodium: 56.79 ± 1.45%. Enzyme inhibition assays showed percent inhibition of 5-lipoxygenase: 59.82 ± 1.89% versus baicalein; 96.56 ± 1.05%, acetylcholinesterase: 58.12 ± 1.81%, butyrylcholinesterase: 37.15 ± 0.98%, and MAO-A: 53.45 ± 0.18%.
Conclusion
Based on current findings, P. candollei's phenolic-rich extract, obtained with significant yield and showing health benefits in age-related and chronic ailments, encourages its industrial exploitation, especially in the food and nutraceutical sectors.
Introduction
Ageing-related illnesses that lower the quality of life in the elderly population are reported to be linked to oxidative stress, inflammation, and neurological abnormalities.1,2 The financial burden of geriatric healthcare and hospice care may increase to $1.1 trillion in 2050. It is substantial to promote functional capabilities among older people through a healthy lifestyle, functional foods, and bioactive natural products to prevent chronic diseases arising in old age. 3 Inflammation defends from local injury; however, it may lead to physiological damage (due to the alteration of inflammatory balance), which ultimately develops chronic disease,4,5 including neurodegenerative diseases, which poses a significant threat to public health as evidenced by the WHO. 6 Alzheimer's disease is prominent among neurodegenerative diseases related to ageing, inflammation, and oxidative stress. Neuroinflammation is often mediated by the LOX enzyme and other pro-inflammatory markers. 1 The 5-LOX enzyme has a crucial role in neurodegeneration because of the expression of LOX protein in the CNS, which is also related to ageing. 7 In addition to the decreased acetylcholine levels in the elderly, reactive oxygen species (ROS) are the main participating agents of inflammation along with other mediators, which may cause damage or death of neurons in the brain and lead to the development of AD and other cerebral disorders (depression). Hence, retarding inflammation and oxidative stress may be crucial in elderly people to prevent the chronic disease, and the concomitant inhibition of cholinesterases becomes pivotal in managing Alzheimer's disease.4,8
Medicinal plants and edible medicinal herbs are utilised to prevent and treat chronic illnesses due to their bioactive secondary metabolites, and many pharmaceutical drugs are derived from these biodiverse sources of nature.9,10 Research on the ethnobotany of traditional medicinal plants offered us a basic understanding of how local communities generally use them and how they treat associated diseases. 11 The researchers investigated various medicinal plants and their bioactive secondary metabolites for age-related chronic diseases, including antioxidant, anti-inflammatory, and anti-Alzheimer's disease activities, which may be attributed to the presence of Polyphenols and other bioactive phytochemicals.12,13 According to evidence, the Apiaceae family has important economic and medicinal value. Pentacyclic terpenoids and other bioactive chemicals from this family have been reported to have anti-inflammatory qualities as well as to improve memory and cognition in relation to Alzheimer's disease.14,15 The Pleurospermum benthamii has been reported to have good inhibition against the cholinesterase enzymes. 16 Pleurospermum candollei is an edible plant used for taste, nutrition, and therapeutic purposes; moreover, it was reported to possess constituents with anti-inflammatory activities and is employed in traditional medicine for the prevention/management of cerebral disorders. 17 The hydroalcoholic extracts of P. candollei were previously reported for antidiabetic and hepatoprotective activities.17,18 Previous literature also reported the identification of phytochemicals from this plant.19,20 However, additional research was required to determine the health effects of phytochemicals obtained from P. candollei and their relationship to different disorders. The ultrasound-assisted extraction (UAE) also reported to provide better yields of bioactive extracts from medicinal plants and gaining importance to overcome the limitations of conventional extraction. 21 UAE generates acoustic cavitation due to the propagation of ultrasound waves through a liquid medium. This mechanism of alternating high- and low-pressure cycles leads to the formation of microscopic cavitation bubbles. These bubbles on collapse produce intense localised shear forces, high temperatures, and pressures, which disrupt the cell walls and facilitate the release of intracellular bioactive compounds (flavonoids, polyphenols, carotenoids, etc) from the plant matrix into the solvent. 22 So, the ultrasound-assisted extraction is advantageous over the conventional extraction techniques in providing better yields and minimising the consumption of time and solvent, which makes UAE an economical technique. The current study was designed to analyze the anti-inflammatory, antioxidant, anti-monoamine oxidase-A, and anticholinesterase properties (in vitro and in silico) as well as their relationship to polyphenols and non-polar phytochemicals. The findings could potentially support the commercial production of P. candollei for exploitation in nutraceuticals and dietary interventions for age-related chronic illnesses.
Materials and Methods
Chemicals and Reagents
Methanol, gallic acid, Folin-Ciocalteu's phenol and other standard polyphenols obtained from Sigma-Aldrich (Steinheim, Germany), acetonitrile, and acetic acid of HPLC grade purchased from Merck (Darmstadt, Germany) and ultrapure water for HPLC was prepared in the laboratory from Milli-Q water purification system of Merck (Darmstadt, Germany). DPPH, ABTS, and Griess reagent were obtained from Sigma-Aldrich, USA. The enzymes were obtained from Merck (Merck KGaA, Darmstadt, Germany). All other chemicals and solvents were of the analytical/ highest commercial grade and obtained from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). The specifications and details of each instrument/equipment are also provided in the relevant sections of the methodology.
Plant Material and Ultrasound Assisted Extraction
Aerial parts of P. candollei with the submitted specimen in the herbarium of Hazara University Mansehra, Pakistan (voucher no. 516/ 1885) were taken. Shade-dried plant material (10 g) was then coarsely powdered. 23 The extracts were prepared by weighing 1.5 g of dry grounded plant, added to 30 mL of aqueous methanol (70% methanol) and placed in a 250 mL conical flask. A rectangular ultrasonic bath (Model 2510, Branson) was used for extraction. It contained a 40 kHz transducer fitted under the bottom and operated at an input power of 220 W for 30 mins at 50 °C. The reported procedure of de Sousa et al was used for ultrasound-assisted extraction. 24 The obtained extract was filtered, concentrated, dried, and stored in air-tight containers without adding preservatives and used for further analysis.
Quantification of Phenolics by Spectrophotometric and HPLC Methods
Total phenolic contents determined using a method of Folin-Ciocalteu reagent and the results presented as mg equivalents of gallic acid per g of dried extract. 25 Sample for HPLC–PDA analysis was prepared as follows: plant extracts were weighed on analytical balance and solubilized in mobile phase A (Milli-Q water + 3% acetic acid): B (acetonitrile +3% acetic acid) (93:7, v: v). Samples were prepared at concentration of 1 mg/500 µL. The sample was vortexed (Scilogex vortex mixture MX-S) for 30 s, sonicated for 15 min, centrifuged (Scilogex D2012) for 10 min at 10,000 g. After filtration through membrane filter, the supernatant was injected (20 µL) in HPLC system for the analysis.
HPLC analysis were performed on a Waters liquid chromatograph equipped with a model 600 solvent pump and a 2996 photodiode array detector (PDA), and Empower v.2 software (Waters Spa, Milford, MA, USA) was used for acquisition of data. C18 reversed–phase packing column (Prodigy ODS (3), 4.6 × 150 mm, 5 μm; Phenomenex, Torrance, CA, USA) was used for the separation and the column was thermostated at 30 ± 1 °C using a Jetstream 2 Plus column oven. The UV/Vis acquisition wavelength was set in the range of 200–500 nm with quantification at each compound's peak wavelength. 26
Tentatively Identified Secondary Metabolites by GC-MS
The secondary metabolites were previously identified from methanolic extract by Ahmad et al using GCMS analysis. 20 GC-MS equipment GC-MS (Agilent, 6890 series and Hewlett Packard, 5973 mass selective detector) was used. Separations were achieved quipped with an HP-5MS column (30 m in length × 250 μm in diameter × 0.25 μm in thickness of film). Spectroscopic detection by GC–MS involved an electron ionization system that utilized high-energy electrons (70 eV). Injector temperature-220 °C; transfer line-240 °C; oven temperature programmed 60 °C to 246 °C @ 3 °C/min. 1.0 µL prepared extracts diluted with respective solvents were injected in a splitless mode at 250 °C. Pure helium gas (99.995%) was used as the carrier gas with a flow rate of 1.02 mL/min. The initial temperature was set at 50 −150 °C with an increasing rate of 3 °C/min and a holding time of about 10 min. Finally, the temperature was increased to 300 °C at 10 °C/min. Detection was done using full scan mode between 35 to 600 m/z, with a gain factor of 5, and the identification of the Bioactive compound was performed using NIST 2011 MS Library, and retention indices were searched from the literature.
In Silico Studies
Molecular Docking Study with 5-LOX, Myeloperoxidase, Monoamine Oxidase-A, and Cholinesterase Enzymes
The interaction between the bioactive compounds identified in P. candollei methanolic extract and enzymes was computed by PyRx software by following the previously reported protocol. 27 The 5-lipoxygenase (6ncf), myeloperoxidase (6wy7), butyrylcholinesterase (6sam), acetylcholinesterase (4m0e), and monoamine oxidase-A (2bxr) were obtained from the protein data bank. Autodock vina embedded in PyRx was used due to its fast, easy, and comprehensive processing. 28 The 3D structure of enzymes was obtained (downloaded file) from the Protein Data Bank.
Discovery Studio 2021 Client (BIOVIA) was used to prepare the proteins for docking. All the H2O molecules and attached ligands were detached from enzyme proteins and saved in PDB formats. Structures of ligand were downloaded in SDF format from PubChem. Ligands were loaded using the Open Babel software and converted to PDB files. The prepared and optimized ligands were docked blindly in the protein's grid box to allow them to find any suitable binding location. 29
ADMET Prediction Study
Absorption, distribution, metabolism, excretion, and toxicity (ADMET) are the essential parameters to be investigated for any compound before being selected as a drug candidate. The online tools are available for quick in silico screening of compounds for ADMET parameters. The online web tool swiss ADME (http://www.swissadme.ch) was used to obtain pharmacokinetic (ADME) properties of the docked compounds, which showed greater efficacy than standard drugs, and the pharmacokinetic scores were predicted using the online web tool pkCSM (http://biosig.unimelb.edu.au/pkcsm/prediction).30,31 Protox-II (https://tox-new.charite.de/protox_II/) was employed to check the toxicity. This program forecasts compounds’ toxicology due to the similarity in chemical structures and compares them to other chemicals with known toxicities. 30
In Vitro Biological Activities
In Vitro Assessment of Antioxidant Potential
Antioxidant activities of plant extract were assessed by the potential to capture the reactive oxygen species produced in biological system. For the determination of this potential the already reported procedures by Sahoo et al were utilized with minor modification. 31 The ascorbic acid was used as positive control in the investigation of antioxidant activities. The sample solution for analysis was prepared at a concentration of 1 mg/mL, using methanol as a solvent and the same solution was used for making dilutions from dried extract (100, 200, 400, 800, and 1000 μg/mL) for all antioxidant assays.
For DPPH free radical scavenging assay, the extract solution of 10 µL volume in methanol solvent with 40 µL of DPPH was mixed in 96 well microtiter plate. This mixture was placed in an incubator at an appropriate temperature for 30 min in dark. The absorbance of the test solution was measured by setting the wavelength of 517 nm with the help of the BioTek Synergy HT microplate reader. Results of the experiment were calculated by following formula (equation I) and repeated in triplicate. The mean value was expressed as IC50 values.
Where the control represents negative control (contain solvent and all reagents except test sample) and the test represents extract/ positive control.
For the ABTS assay, cation radicals in the form of ABTS+ were obtained when a blend of 7 mM ABTS with the 2.45 mM K2S2O8 (potassium persulfate) was kept at ambient temperature in darkness. The prepared stock solution of ABTS+ was then diluted until its absorbance reached 0.700 ± 0.02 at 734 nm. Then 10 µL of extract solution (1 mg/ mL) and 20 µL of ABTS+ solution were taken in a 96 microtiter plate. The mixed solution was stored in an incubator for 30 min at room temperature. Afterward, the absorbance was recorded at 734 nm with the help of the BioTek Synergy HT microplate reader and radical scavenging activity was calculated by above stated formula (equation I). The results finally expressed as IC50 values.
Sodium nitroprusside used to produce nitric oxide and Griess reagent for measuring its concentration. Nitric oxide reacts with oxygen to produce nitric ions that can be assessed by using Griess reagent. 10 mmol/L of sodium nitroprusside in phosphate buffer saline (PBS) was mixed with different concentrations of the extract and incubated (25 °C for 150 min). The samples were added to Griess reagent (1% sulphanilamide, 2% H3PO4 and 0.1% napthylethylenediamine dihydrochloride). The absorbance of the chromophore formed, was read at 546 nm and referred to the absorbance of standard solutions of ascorbic acid treated in the same way with Griess reagent as a positive control. All the experiments were executed in triplicate and the graph was plotted (mean value). The percentage of inhibition was calculated by the above mentioned formula (equation I) and results finally expressed as IC50 values.
The H2O2 (4 mmol/L) solution was prepared in phosphate buffer saline (pH 7.4). 0.3 mL of a 4 mmol/L H2O2 solution made in PBS (after incubation for 10 min) was mixed with 2.0 mL of plant extract that had been prepared in distilled water at various concentrations. At 230 nm, the solution's absorbance was observed in comparison to a blank solution (plant extract + PBS without H2O2) and ascorbic acid (positive control). Using the previously stated formula (equation I), the amount of H2O2 radicals inhibited by the extract was taken into account and expressed as IC50 values.
Toxicity Assessment by Hemolytic Assay
The hemolytic activity of the extract was investigated by using the documented method with minor modifications.
32
A volume of 10.0 mL of fresh blood from human volunteers was collected and dropped into a top-screwed EDTA tube and did centrifugation for 5.0 min. The supernatant was separated by aspiration with a syringe. Then RBCs were washed many times with a 10.0 mL cool, sterilized isotonic solution of PBS having a pH of 7.4. Washed erythrocytes were suspended in a 20.0 mL volume of PBS, and the sample extract was mixed with this buffer solution at a concentration of 1 mg/mL. Each sample extract was added to prepare the separate analyte solution and incubated at 37 °C for sixty minutes. The hemolysis rate was determined by recording the absorbance of hemoglobin present in the supernatant layer at the wavelength of 540 nm. The 0.10% triton X-100 was used as a + ve control and PBS as a -ve control. The percentage of hemolysis was calculated by using the given formula.
Where Abs. control is absorbance of blank sample, Abs. sample include the absorbance of plant extract, and Abs. positive control is the absorbance of triton X-100.
Anti-Inflammatory Activity
The anti-inflammatory activity was checked through the inhibition of the albumin denaturation assay. Previously reported protocols were employed to investigate in vitro anti-inflammatory activity.
33
A one percent solution (aqueous) of bovine albumin serum was prepared. The pH of the solution was then accommodated to 6 with 1.0 M hydrochloric acid. The stock solution of the test sample was made at a concentration of 5.0 mg/mL (in DMSO). The standard drug (diclofenac sodium), in a concentration of 1.0 mg/mL, was employed to make the stock solution of standard. The stock solution of samples and the standard drug was diluted to achieve the final concentrations of μgmL−1. The solution of standard and extracts were kept in an incubator for 20 min at a temperature of 37 °C and then heated at seventy degrees centigrades by using a water bath for five minutes. The turbidity was measured by taking the absorbance at 660 nm with a spectrophotometer after the reaction mixtures were cooled to room temperature. The %age inhibition of albumin denaturation was computed using the equation given below, and the experiment was repeated to get values in triplicate.
Where, Abs. control is absorbance of solution containing solvent and all reagents except sample. Abs. test is absorbance of sample/ standard.
Lipoxygenase Inhibitory Activity
Lipoxygenase inhibition of ultrasound-assisted methanolic extract (UPCM) was measured by the spectrophotometric technique of the 5-LOX assay. 3 It measures the amount of hydroperoxide ((13S)-13-hydroperoxyoctadeca-9, 11-dienoate) released by the 5-LOX enzyme after reacting with its substrate. The UPCM extract solution was prepared (1 mg/mL). Then prepared, tribasic sodium phosphate buffer solution (50 mM) of pH 8.0 and 5-LOX enzyme (10,000 U/mL). The extracts (1 mg/mL) solutions were mixed with 250.0 μL of prepared buffer and 250.0 μL 5-LOX enzyme solutions. The mixture was put in an incubator at 25 °C (5 min). Afterward, a 1000 μL substrate solution (linoleic acid 0.6 mM) was to the previously made enzyme solution and incubated for 5 min at 25 °C. Absorbance was measured at 234 nm. The percent inhibition values were determined by established protocols. Baicalein was used as the standard, and phosphate buffer solution as a negative control. The percentage of enzyme inhibition by the UPCM was calculated by the formula given above as equation II.
Cholinesterases Inhibitory Activities
For the acetylcholinesterase inhibitory activity, a reaction mixture was prepared, which comprised a volume of 50.0 µL of the extract solution (Conc. 1 mg/mL), 125.0 µL of DTNB (3 mM), and 25.0 µL of acetylcholinesterase solution (0.265 U/mL AChE) in a buffer of Tris-HCl with pH 8.0, and this mixture was kept in the incubator at 35 °C temperature for fifteen minutes. Then, the substrate (25.0 µL of 15.0 mM acetylthiocholine iodide) was added to this mixture. The absorbance of the prepared solutions was measured at 405 nm after 15 min of the incubation period. Galantamine was the standard drug used in acetylcholinesterase inhibition activity. Similarly, a blank solution was prepared from the buffer, which did not contain a sample extract or standard. In case of butyrylcholinesterase the butyrylthiocholine chloride was used as substrate and analyzed according to the procedure. 34 The readings were taken in triplicate, and percentage inhibition was calculated by the formula given in equation II.
Monoamine Oxidase (MAO-A) Inhibition Assays
The inhibitory effects of the UPCM extract (0.05-1.0 mg/mL) against MAO-A was determined using previously described protocols. 35 Each well of a 96-well plate was used for the assays. The reaction mixture contained 60 µL of buffer (NaHPO4, pH 7.4), 10.0 µL of test substance (1 mg/mL, in DMSO), and 10 µL of enzyme (26.0 g protein for MAO-A), and it was incubated for 20 (MAO-A). A fluorescence plate reader (BMG Labtech GmbH, Ortenberg, Germany) was used to assess the variation in fluorescence (320 nm) after adding 10 μL of substrate (p-tyramine) and 10 μL of freshly prepared Amplex Red after the incubation period. Clorgyline (10 µL of 1 mM solution) was used as positive control for MAO-A. All tests were performed in triplicate, and the percentage of MAO-A inhibition was calculated by the formula given in equation II.
Statistical Analysis
The mean ± SD (after 3 repetitions of procedure) was used to express the results obtained in all the experiments. MS excel and GraphPad Prism software was used to analyse the data.
Results and Discussion
Extraction Yield and Quantification of Phenolic Compounds by HPLC-PDA
The UPCM extract showed a 23.33% yield (0.35 g of extract obtained from 1.5 g dried plant powder). This was a greater yield than the previous study, in which the methanolic extract showed a 16.40% yield. 17 So, the ultrasound-assisted extraction help to improve the yield of extract by releasing more intracellular contents compared to simple extraction techniques. The quantity of phenolic compounds was investigated by spectroscopic method, which showed 204.92 ± 2.52 mg GaE/g dried extract. HPLC-PDA analysis of the studied extract of P. candollei indicated a number of important bioactive components belonging to the general class of polyphenols. Among the identified compounds, chlorogenic acid was eluted at 2.76 min at a concentration of 9.35 µg mg−1 dried fraction, whereas p-coumaric acid, caffeic acid, kaempferol, coumarin, and benzoic acid were detected at 3.13, 8.30, 14.98, 16.00, and 18.31 min at concentrations of 12.32, 32.40, 21.26, 33.02, and 25.26 µg mg−1, respectively. Although six compounds were detected (Table 1), many extra peaks in the chromatogram indicated the presence of other compounds (Figure 1). These compounds were recorded in comparison to the peaks of standard phenolic acids. A previous study also showed the significant but lesser quantities of similar phenolic acids (gallic acid; 0.94, coumaric acid; 0.4, caffeic acid; 1.22, and kaempferol; 2.82 µg mg−1) in the methanolic extract of P. candollei. 17 A previous study from our project also reported a significant quantity (240.69 ± 2.94 mg GaE/g) of phenolic compounds using spectroscopic method (TPC), which is also substantiated by the current work with little difference. The significant values of TPC are also in agreement with the HPLC results that indicate the presence of phenolic compounds. In 2022, Ahmed et al also identified the phytochemicals belonging to different groups of chemicals including terpenoids, phenolics, flavonoids, steroids and alcohols. 42 The results of polyphenolics analysis by HPLC are provided in Table 1, while the chromatogram is presented in Figure 1. The aqueous methanol (solvent) may be contributing to the extraction of polyphenols in higher quantities. The demonstrated phenolic acids are well known for their health benefits, antioxidant, and other biological activities. So, their significant quantities in the UPCM forecast the bioactive nature of the extract that may be used in various ailments.

Chromatogram Obtained from HPLC-PDA Analysis of UPCM Extract.
Polyphenols Quantified by HPLC-PDA from UPCM Extract of P. candollei.

Identification of Plant Secondary Metabolites by GC-MS
GC-MS (gas chromatography-mass spectrometry) analysis is preferable for non-polar and volatile compounds. 43 In our previous project, the methanol extract of P. candollei was subjected to gas chromatography-mass spectrometry (GC-MS) to search for non-polar and volatile bioactive phytochemicals. 20 There were 22 natural compounds, which were tentatively identified using the NIST library (Table 2). We also searched the literature for identified compounds’ anti-inflammatory and antioxidant properties (Table 2). The methanol extract contains nearly 99% (by peak area) of the previously identified phytochemicals’ combined anti-inflammatory and antioxidant properties. Various studies are available supporting phytochemicals with anti-inflammatory properties. 63 Terpenoids were found to be the major portion of methanolic extract (82.61%) of this plant in our previous study, and terpenoids are also bioactive compounds that show anti-inflammatory effects. 64 The parameters of retention time (minutes), compound name, molecular formula, molecular weight, chemical class, retention indices, and the % of peak area. Major constituents of methanol extract were the terpenoids which constitute 82.61% of the non-polar components of extract, and hopanoids were the main subclass of the terpenoids, while the other compounds were steroids, alcohols, and flavonoids. Pleurospermum amabile, in which, Wangchuk et al revealed many phenolics, terpenoids, and other bioactive compounds of pharmacological interest. 65 The UPCM extract, being the ultrasound-assisted methanol extract (the solvent of extraction was the same as that used in the previous project), was considered to contain similar non-polar phytochemicals. So, they are considered for molecular docking against the target proteins of the current study. It further encouraged the evaluation of the UPCM extract for in vitro studies.
Secondary Metabolites Identified from UPCM Extract of P. candollei Through GC-MS Analysis Along with Their Reported Antioxidant and Anti-Inflammatory Properties.

In Silico Studies
Although In vitro pharmacological screenings (bioassays) are extensively employed for new drug development, experimental techniques are insufficient to explore drug-target interactions. 66 It is evident from past research on drug design that the conventional methods of drug discovery may help in successfully designing drug molecules. However, a prolonged period is required from the identification of lead molecules to the conduction of clinical trials (typically twelve years of study), and it became an economic burden with the huge expenditure on the resources employed for these studies. So, the in silico studies attract researchers by accelerating drug discovery in terms of economy, labor, and time. This ultimately results in the development of new drug molecules with the help of computational methodologies. 67 These computational strategies can help to limit the use of animal models for in vivo biological studies and rational designing of novel and safer lead molecules, which result in the discovery of novel drugs. Further, computer-aided drug designing also helps in the repositioning of marketed drugs to support medicinal chemists and pharmacologists during the discovery processes of novel drugs. 68 From this evidence, we were inspired to conduct the in silico prediction studies of the identified compounds from the methanolic extract of P. candollei, so we included molecular docking studies for the 5-LOX enzyme to predict the anti-inflammatory as well as acetylcholinesterase inhibitory effects of those molecules. We also included in silico ADMET studies to predict the pharmacokinetics and toxicity of docked ligands.
Molecular Docking Study with 5-LOX Enzyme
Recently, molecular docking has become the most important and common method among the theoretical methods to evaluate the experimental work for the comparison of the biological activities of bioactive natural compounds with the enzymes, which is found by numerical values of docking parameter, and these findings of molecular docking ultimately help to develop the virtual libraries of drug-like phytochemicals. 69 Major compounds identified from the methanolic extract of P. candollei (Table 2) were docked against the 5-LOX enzyme to predict their anti-inflammatory effect, and the results are presented in Table 3. The interactions of the standard drug and a compound are presented in Figure 2. Eight docked ligands showed higher binding affinity than the standard drug as represented by the high negative value of binding energies (or lower binding energies), and values of binding energies range from −8.2 to −9.8 kcal/mol as compared to the standard drug (diclofenac sodium; −6.4 kcal/mol). However, cedran-diol, 8S,14- showed binding energy (−6.4 kcal/mol) equal to that of diclofenac sodium. So, the anti-inflammatory activity of the UPCM extract may be due to the presence of these compounds, but a stronger activity must be observed based on docking results because none of the compounds showed a lesser binding affinity with the 5-lipoxygenase enzyme. The authors observed moderate in vitro and in vivo anti-inflammatory activities. Hence, the docking study further strengthened our prediction about the possible antagonism among the identified compounds from the methanolic extract of this plant. So the isolation of the compounds may lead to the discovery of novel anti-inflammatory agents that help prevent and cure chronic inflammation and consequent chronic diseases.

Binding Energies After Molecular Docking of Previously Identified Compounds from Methanolic Extract of P. candollei.

Standards a; diclofenac sodium, b; baicalein, c; galantamine, and d; clorgyline.
Molecular Docking Study with Myeloperoxidase Enzyme
Docking of selected ligands showed lower binding energies (range; −7.8 to 9.1 kcal/mol) compared to the standard baicalein (−7.7 kcal/mol), except Cedran-diol, 8S,14- which showed higher binding energy than standard (−5.7 kcal/mol). It is attributed to the good binding affinity of the ligands with the enzyme active site except for one ligand. These findings substantiated the significant antioxidant activity of the UPCM extract as investigated by different assays. The docking score in terms of binding energy is provided in Table 3 and major interaction in Figure 3.
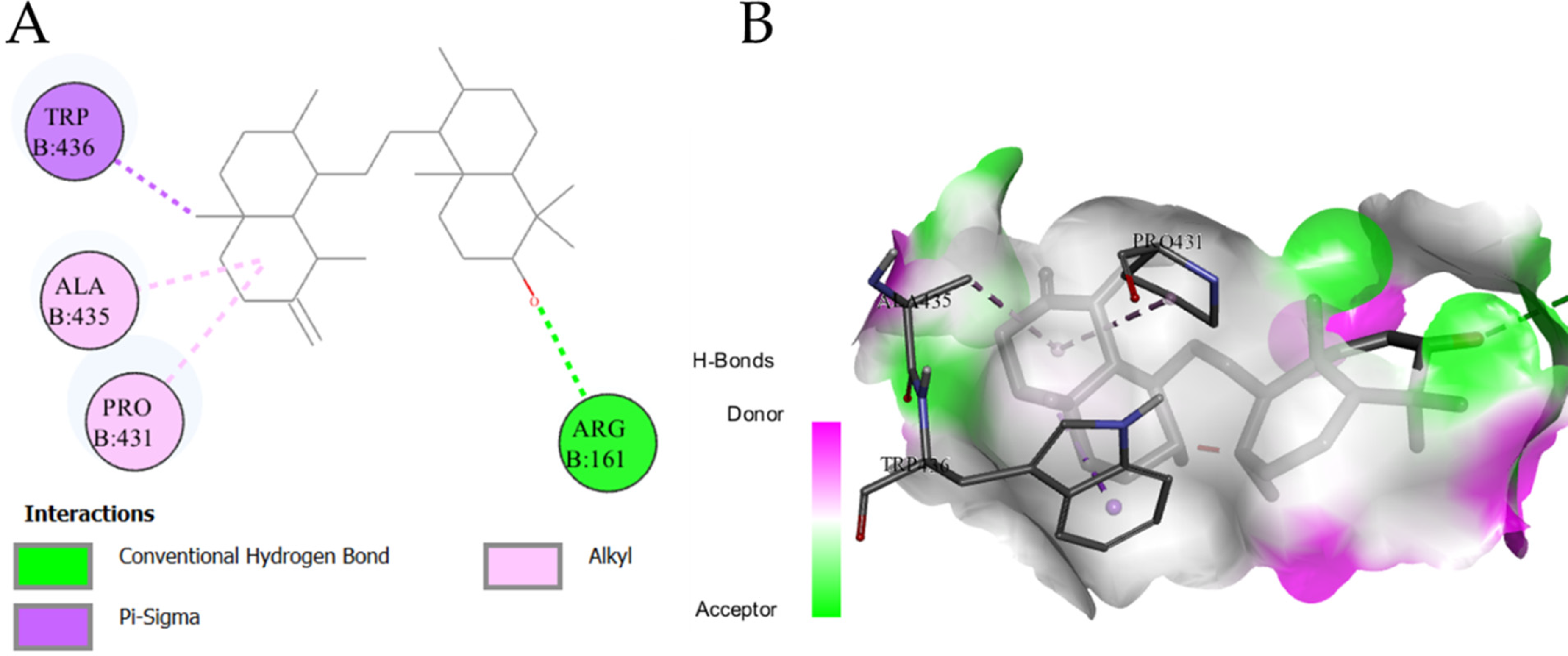
Myeloperoxidase (MPO) is considered a marker that is released by stimulated polymorphonuclear leukocyte (PMN) and contributes to oxidative stress by generating oxidant species, especially HOCl (hypochlorous acid) and H2O2. These are imperative microbial killers through dual action (oxidation and chlorination reactions) and oxidants. MPO is released by highly stimulated and dying neutrophils in response to acute and chronic inflammation. Under these pathological conditions, MPO is able to exert oxidant activity on adjacent cells and tissues. 70 Polyphenols, including phenolic acids, are known to possess antioxidant activity that inhibits oxidative damage and may consequently prevent inflammatory conditions, which accelerate ageing and neurodegenerative diseases. 71
Molecular Docking Study with Cholinesterase Enzymes
A molecular docking study analyzed acetylcholinesterase inhibitory properties of the selected (nine) ligands. The results are shown in Table 3, and some interactions are presented in Figure 4. Eight of the nine docked ligands showed better binding energies (−8.5 to −10.0 kcal/mol) as compared to the binding energy of a standard drug (galantamine; −7.4 kcal/mol). So, the ligands showed higher binding affinities than galantamine and predicted their anticholinesterase activity, which also supports the traditional use of this plant for anti-AD effects. However, cedran-diol, 8S,14- showed higher binding energy (−6.7 kcal/mol) as compared to the standard drug (so the lower binding affinity), which suggests that this compound may have no role in acetylcholinesterase inhibition. The binding energies of btuyrylcholinesterase with the 13 ligands were in a range of −6.1 to −12.5 kcal/mol. In this case the lowest affinity for the enzyme was expressed by coumaric acid acid; −6.1 kcal/mol. While all the ligands showed higher binding affinities with the btuyrylcholinesterase except for the four ligands Table 3, while, the Figure 5 showed the highest interacting ligand with enzyme active site residues. This docking study predicted the vital relation of the terpenoids to their anticholinesterase effects. So, a method for the targeted extraction of terpenoids may be beneficial for the development of phytotherapeuticals for AD. Moreover, the isolation of pure compounds may lead to the discovery of novel drugs for AD and other cerebral disorders.
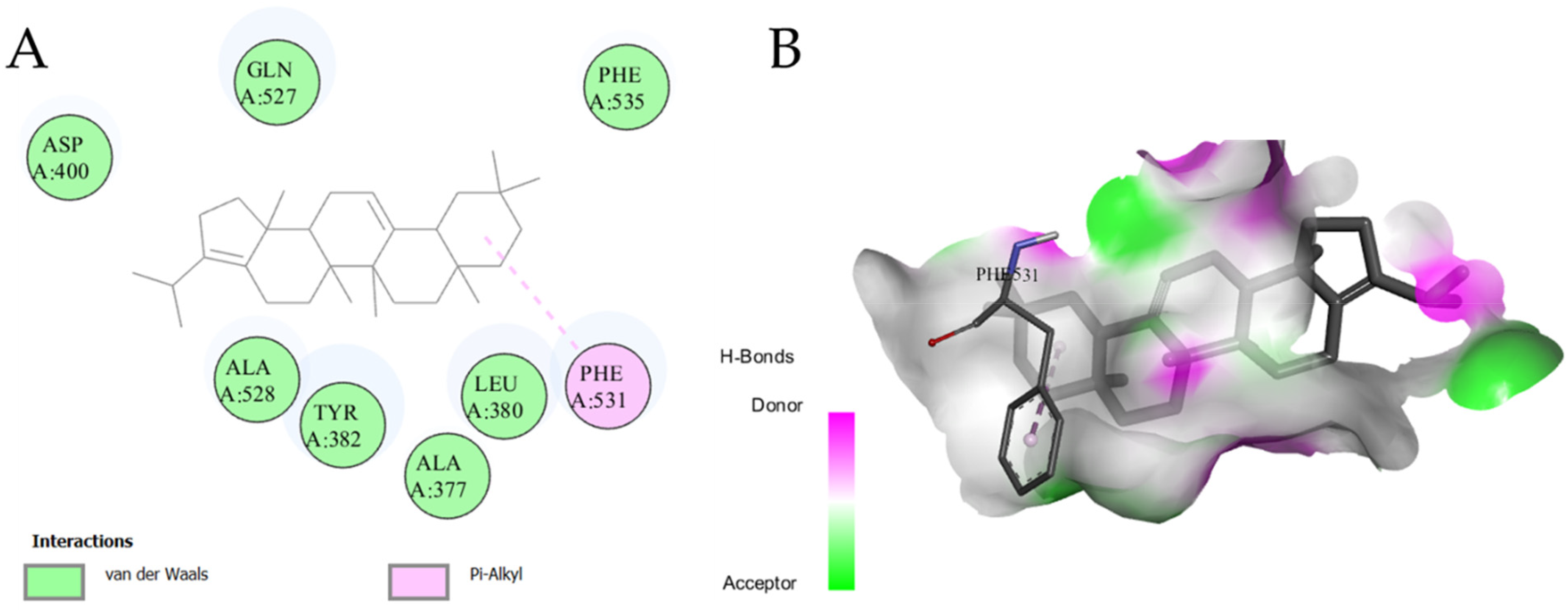

Molecular Docking Study with MAO-A
The results showed that all the docked compounds demonstrated higher binding affinities (−6.1 to −9.2 kcal/mol) with MAO-A enzyme compared to the clorgyline (−5.5 kcal/mol). So, the identified non-polar compounds predicted to have MAO-A inhibitory property. Their presence in UPCM may confer to the antidepressant effect to the extract. Moreover, the phenolic compounds demonstrated in significant quantities from UPCM extract. These compounds have already been reported for various biological activities including neurological disorders (Table 1). Therefore, the UPCM extract may also be expected to have antidepressant effects due to the presence of phenolic compounds. The result of this molecular docking study are presented in Table 3 and the interactions of ligand (with highest affinity) with MAO-A active site residues is shown in Figure 6.

ADMET Prediction Study
ADMET prediction study plays an essential role for every type of bioactive molecule before its transformation into a drug. 72 Results of absorption, distribution, metabolism, and excretion are given in Table 4. All the compounds showed high gastrointestinal absorption (GI absorption of more than 90%), and none of the compounds could cross the blood-brain barrier (BBB). Among the ADME parameters, absorption (intestinal or CNS) is crucial to be checked before a compound gets the status of “drug” and enters into pharmaceutical formulation or clinical trial, and the compounds which cross the BBB may exert adverse effects so it is desirable for a compound that it should not cross BBB. 73 All the compounds show high GI solubility and absorption and no penetration from BBB, which means the compounds have good potential for GI absorption in humans and minimal possibility of CNS side effects. Compounds show skin permeation in the range of log kp −2.58 to −2.82. The higher the negative score of the Kp, the lesser the skin permeation of the biomolecule will be. 74 So, the compound Urs-12-en-3-ol, acetate, (3beta) displayed the highest skin permeability with log kp −2.82, and all other compounds were less permeable to the skin.
ADME Parameters Predicted from major Identified Phytochemicals of P. candollei.
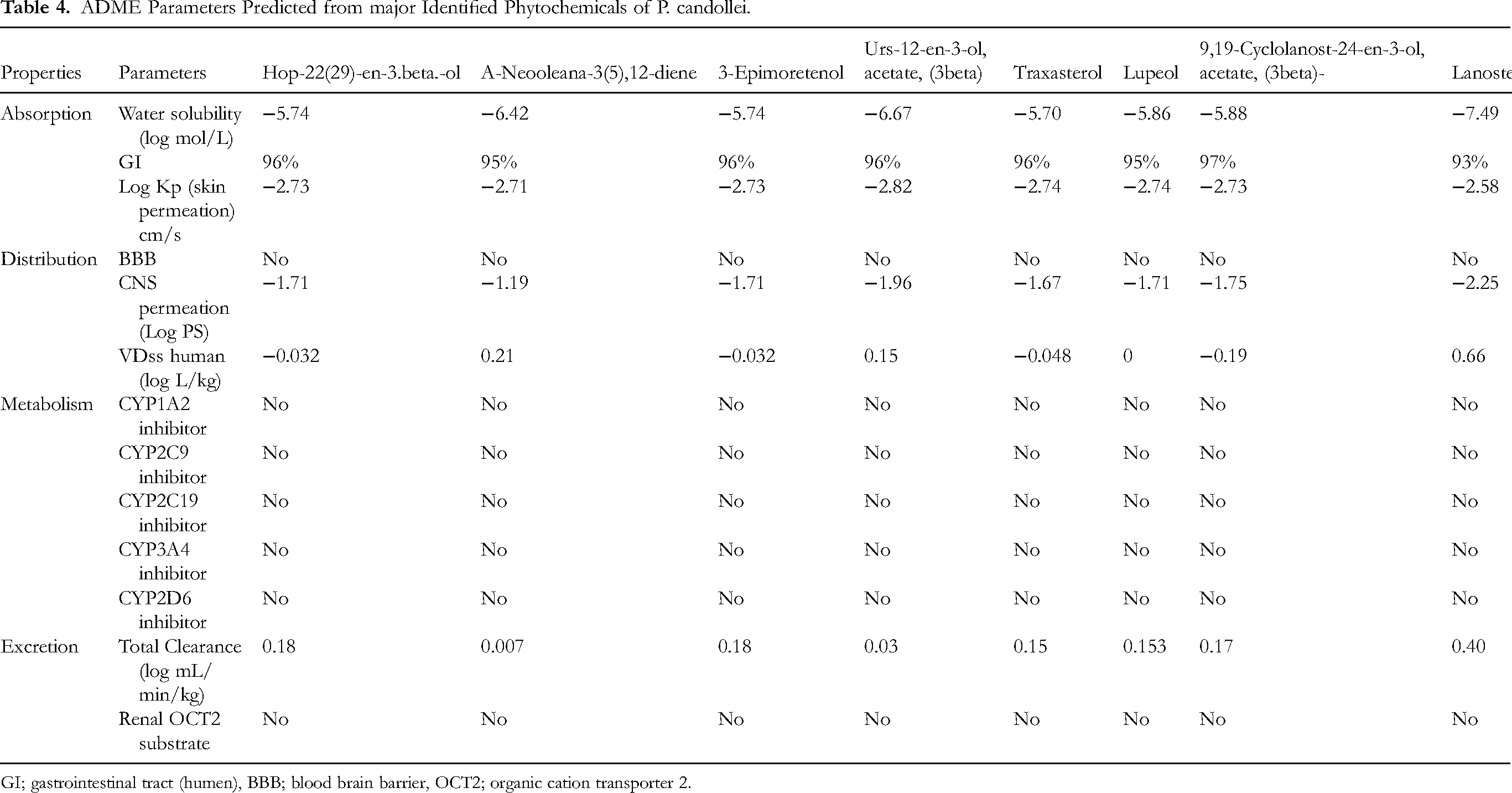
GI; gastrointestinal tract (humen), BBB; blood brain barrier, OCT2; organic cation transporter 2.
There are five main cytochrome (CYP) isoforms, which show an auxiliary benefit to forecasting the metabolism of compounds. This enzymatic system plays a pivotal role in the excretion of drugs/compounds from the body. Moreover, they control the metabolism of nearly 75% of the medicines existing in the market. Inhibition of any of these enzyme isoforms may lead to drug interactions. 75 As shown in Table 5, compounds did not inhibit any of the CYP enzyme isoforms and were metabolized rapidly. Rates of administration of the dose of a drug depend on its clearance for the achievement of steady-state concentrations, which can be investigated by determining the excretion rates from the liver as well as kidney, and additional values will represent toat clearance, and clearance values of the studied compounds were insufficient. 76 Undesired drug interactions and adverse effects may occur due to the impact of organic cation transporter 2 (OCT2) and its intermediates with the concomitant use of OCT2 inhibitors and their substrates. It has been predicted that the compounds of study would not perform as a substrate for OCT2, which hypothesize the safe excretion with minimal or no drug interactions. 75
Lipinski's Rule of Five and Solubility of Best-Docked Compounds.

HBA; hydrogen bond acceptors, M.R; molar refractivity, HBD; hydrogen bond donors, MWT; molecular weight.
The bioavailability radar plots (Figures 7,8,9, and supplementary figures A, B, C, D, E, F, G, H, I) were also drawn for the docked compounds and the results are shown in Table 5. The results showed that eight compounds (Hop-22(29)-en-3.beta.-ol, A-Neooleana-3(5),12-diene, 3-Epimoretenol, Urs-12-en-3-ol, acetate, (3beta), Taraxasterol, Lupeol, 9,19-Cyclolanost-24-en-3-ol, acetate, (3beta)-, Lanosterol) showed a maximum of 2 violations of Lipinski's Rule, while all the other compounds showed a maximum of one or no violations of the mentioned rule. A chemical is a underprivileged candidate for oral administration if it disrupts two or more of Lipinski's Rule of Five, which consist of molar refractivity, hydrogen bond donors, molecular weight, lipophilicity (LogP), and hydrogen bond acceptors. 77 The results predicted that the bioavailability of seven (Cedran-diol, 8S,14-, Chlorogenic acid, Coumarin, p-Coumaric acid, Kaempferol, Benzoic acid, and Caffeic acid) out of fifteen compounds is good. However, the bioavailability of other compounds may be poor and can be improved by using expertise in biopharmaceutics.

Bioavailability Radar of Chlorogenic Acid (Left) and Coumarin (Right).

Bioavailability radar of p-Coumaric acid (left) and Kaempferol (right).
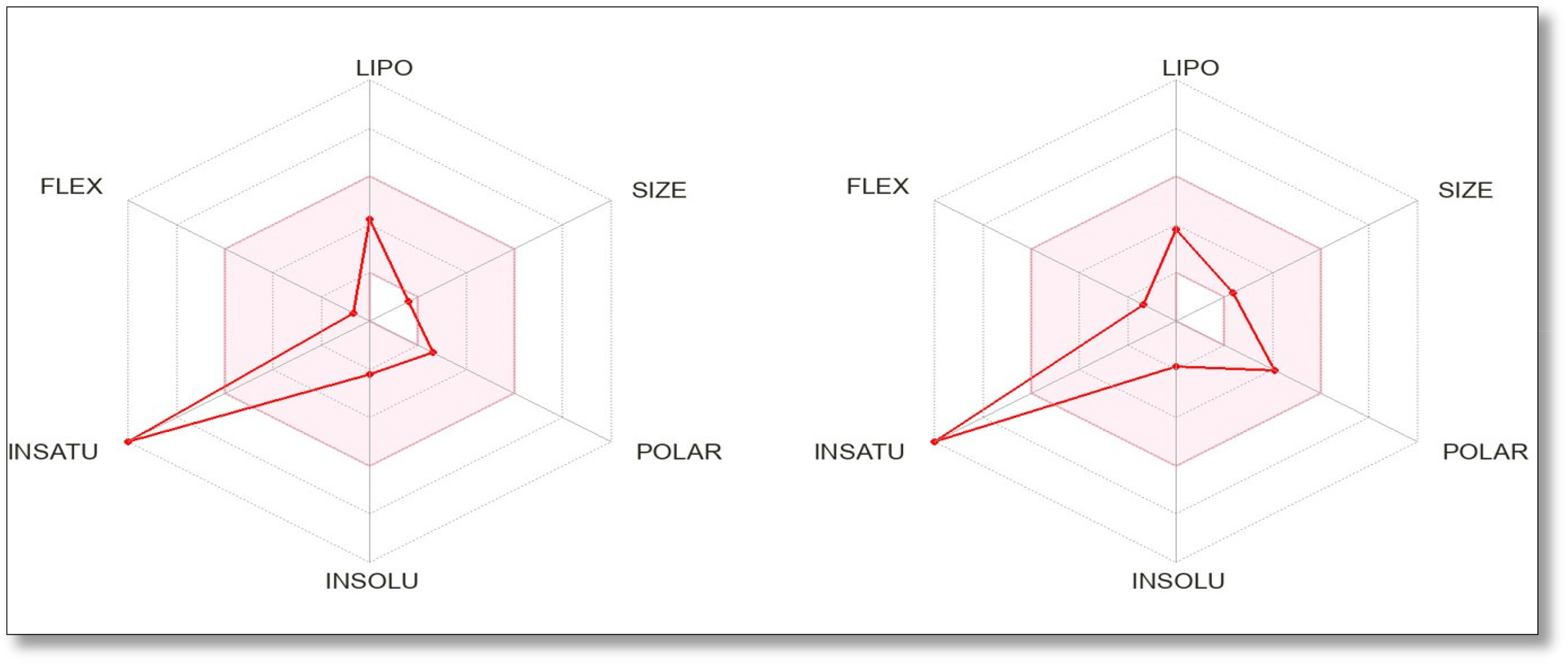
Bioavailability Radar of Benzoic Acid (Left) and Caffeic Acid (Right).
The toxicological profile of the compound becomes critical when it seeks approval for a clinical trial or is ready to be used in a pharmaceutical formulation. 78 The toxicity of the compounds in the study was predicted by using ProTox-II online tool, which classified the compound by predicting their oral, hepatic toxicities and carcinogenicity. Moreover, various Tox21-Nuclear receptor signaling pathways are included in this toxicity prediction approach. 79 The observed toxicity parameters are given in Table 6, which shows LD50, toxicity class, hepatotoxicity, and carcinogenicity of the major docked compounds. Compounds a-neooleana-3(5),12-diene, urs-12-en-3-ol, acetate, (3beta), traxasterol, 9,19-cyclolanost-24-en-3-ol, acetate, (3beta)-, (LD50; 5000,3460, 5000, and 4300 mg/kg respectively) are predicted as “may be harmful” with the toxicity class V, while other compounds belong to the toxic class IV (harmful). However, their values on the higher end of this class (300-2000 mg/kg) predict their lesser toxicity, as mentioned for this class. 80 None of the compounds were found to be hepatotoxic (predicted inactive for hepatotoxicity), and the compound Urs-12-en-3-ol, acetate, (3beta) was supposed to be “active” for carcinogenic toxicity. However, the probability of the predicted carcinogenicity was moderate (51%), and it may not be a definitive finding without further investigations. 81 All the other compounds were observed to be inactive for carcinogenicity.
in Silico Prediction of Toxicity.

IV and V; toxicity class four and five, * actively toxic with probability score 0.51 (51%).
In Vitro Biological Activities
In vitro biological activities were performed after observing a significant potential of phytochemicals from literature-based evidences and in silico docking studies. Antioxidant assays were performed to find a relation of radical scavenging potential predicted by docking study, anti-inflammatory effects were determined by protein denaturation as well as lipoxygenase inhibitions. Inhibition of acetyl and butyryl cholinesterases showed the anti-Alzheimer's disease effects predicted from molecular docking studies. In silico safety of the extract containing docked phytochemicals was also evaluated by in vitro hemolytic assay.
In Vitro Assessment of Antioxidant Potential
The free radical scavenging assays were performed to assess the antioxidant potential of the UPCM extract. The results are shown in Figure 10 as the percentage inhibition of free radicals. The IC50 values were also calculated and presented in Table 7. The antioxidant properties of the extract were investigated by four different radical scavenging assays, which showed 68.74 ± 1.25%, 83.15 ± 1.42%, 58.83 ± 1.09%, and 78.45 ± 1.52% percent inhibitions for DPPH, ABTS, H2O2 scavenging assay, and NO scavenging assays, respectively. The radical scavenging activities of UPCM were comparable to the standard antioxidant ascorbic acid (DPPH; 84.56 ± 2.06%, ABTS; 95.75 ± 2.15%, H2O2 scavenging; 85.45 ± 1.81%, and NO scavenging; 91.54 ± 1.58%) compared to the UPCM extract. The higher antioxidant activities may be due to a more significant amount of polyphenolic compounds in the methanol extract. Moreover, It is a proven fact that methanol extract has more substantial antioxidant activity and phenolic compounds than water extract. 82 We analyzed the antioxidant activities to assess this plant's general health-promoting effects and the link between anti-inflammatory properties and antioxidant effects. In 2022 Ahmed et al, reported that P. candollei possessed good antioxidant capacity expressed by reducing power (FRAP and CUPRAC). 42 Reducing antioxidants also show anti-inflammatory properties, which can be explained based on the 5-LOX enzyme, which contains an iron (non-heme iron) in the active site. So, redox inhibitors inhibit this enzyme by reducing ferric (Fe3+) from the active state to the inactive form of ferrous ion (Fe2+). Moreover, the phenolics have radical scavenging properties and may disrupt the ferric ion redox cycle of the enzyme, resulting in 5-LOX enzyme inhibition. 83 A description of the experimental results given in Table 1, which shows a significantly higher quantity of polyphenols.

Free radical Scavenging Antioxidant Assays of UPCM Extract Presented as Inhibition (%). DPPH (2,2-Diphenyl-1-picrylhydrazyl assay), ABTS (2,2'-Azino-bis(3-ethylbenzothiazoline-6-sulfonic Acid) Assay), HRSA (Hydroxyl Radical Scavenging Assay), NOSA (Nitric Oxide Scavenging Assay).
Total Phenolic Contents and Radical Scavenging Antioxidant Activities of UPCM Extract.
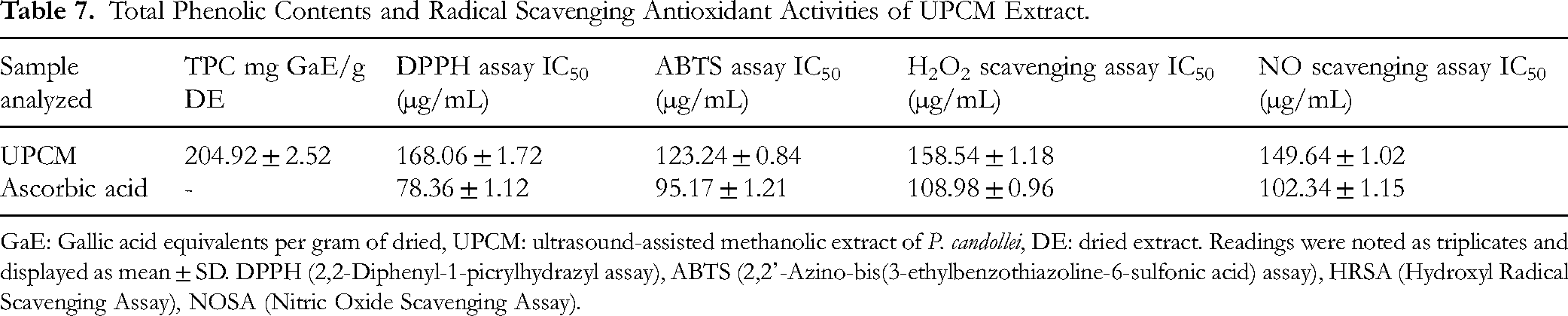
GaE: Gallic acid equivalents per gram of dried, UPCM: ultrasound-assisted methanolic extract of P. candollei, DE: dried extract. Readings were noted as triplicates and displayed as mean ± SD. DPPH (2,2-Diphenyl-1-picrylhydrazyl assay), ABTS (2,2'-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) assay), HRSA (Hydroxyl Radical Scavenging Assay), NOSA (Nitric Oxide Scavenging Assay).
Toxicity Assessment by Hemolytic Assay
In the current study, the haematological model of RBCs was used to screen the safety of the UPCM extract, using triton X-100 as a reference hemolytic agent. It was observed that the extract showed less significant hemolysis (5.18 ± 0.52%) compared to the standard chemical, which caused 98.66 ± 1.14% lysis of RBCs. The results of this experiment are given in Table 8.
In Vitro Toxicological, Anti-Inflammatory, and Enzyme Inhibitory Activities of P. candollei (% of Inhibition).
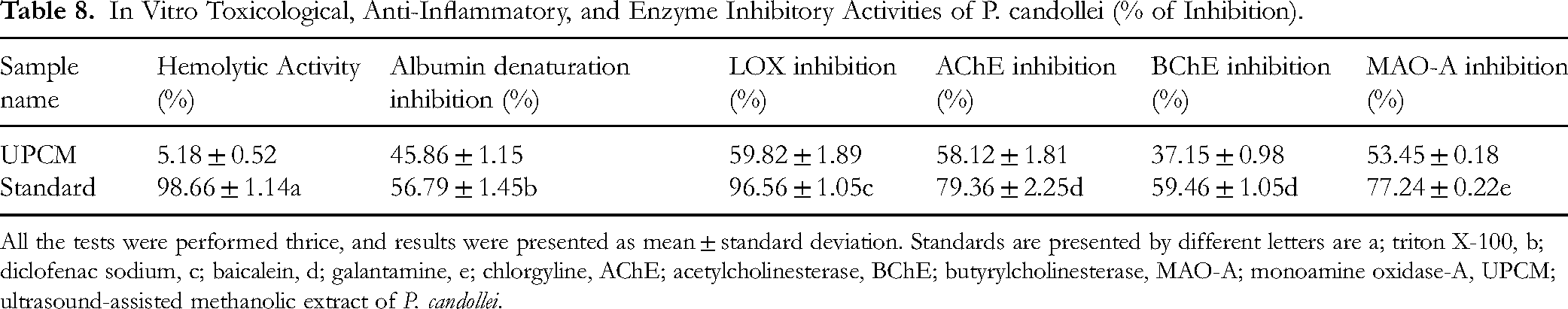
All the tests were performed thrice, and results were presented as mean ± standard deviation. Standards are presented by different letters are a; triton X-100, b; diclofenac sodium, c; baicalein, d; galantamine, e; chlorgyline, AChE; acetylcholinesterase, BChE; butyrylcholinesterase, MAO-A; monoamine oxidase-A, UPCM; ultrasound-assisted methanolic extract of P. candollei.
Toxicity assessment is used to establish the safety of medicinal plants, herbs, extracts, and phytochemicals, so people may use them as a drug without suffering from their side effects. 84 Moreover, safety studies are also necessary before the in vivo activities on animal models, and the RBCs (erythrocytes) induced hemolysis is a reliable and simple screening technique to assess the safety of natural products in terms of compatibility with blood. 85 The extracts or the compounds causing hemolysis 30% or above are generally considered toxic. 86 Hence, the current study predicted the safety of P. candollei, which may be investigated further through in vivo safety studies. After the final establishment of safety, the extract of this plant may be used in therapeutic applications.
Anti-Inflammatory Activity
The anti-inflammation effect was determined by inhibition of the bovine albumin from denaturation. We found that the UPCM again exhibited significant inhibition values (45.86 ± 1.15% inhibition) compared to the diclofenac sodium (standard drug; 56.79 ± 1.45% inhibition), as shown in Table 8.
Our study aimed to get insights into the anti-inflammatory properties also of P. candollei, so we started with the in vitro screening by using this simple, reliable, and well-documented method, and the mechanism is that the denaturation of tissue proteins causes the production of auto-antigens in the inflammatory diseases. 87 Although we found a significant anti-inflammatory activity of P. candollei, it substantiates the presence of anti-inflammatory compounds in this plant. Ali et al also separated two phytochemicals with anti-inflammatory properties, and the reported compounds were trans-asarone and α-linolenic acid. 88 Compound asarone was also identified in our previous study through the GC-MS technique from the n-hexane fraction of methanolic extract of this plant, and this compound was present in a significantly high concentration (6.30% peak area). 42 Although the second compound (α-linolenic acid) was not identified in our studied fraction, the ester of similar fatty acid was found (2-Chloroethyl linoleate). These compounds have already been reported for antioxidant effects, which might be contributed to their anti-inflammatory properties due to their highest peak area (39.69%). In the same study, some other compounds were identified with anti-inflammatory effects were; methyl iso-eugenol 2; bicyclo[6.3.0]undec-1(8)- en-3-ol, 2,2,5,5-tetramethyl; 3-Hydroxy-4-methoxycinnamic; 2H-1-benzopyran-2-one; 7- methoxy, 7,10,13-hexadecatrienoic acid, methyl ester; 11,14,17-eicosatrienoic acid, methyl ester; n-hexadecanoic acid; 9,12-octadecadienoic acid, methyl ester; 9,12,15-octadecatrienoic acid, methyl ester; 9,12-octadecadienoic acid (Z,Z); flavone; N-hydroxy-N'-[2-(trifluoromethyl)phenyl]pyridine3-carboximidamide; 3β-acetoxy-pregna-5,16- dien-20-one; vitamin E. We also observed the significantly high antioxidant activity of methanolic extract compared to fractions, which may contribute to anti-inflammatory effects due to antioxidant based anti-inflammation, which can also be correlated with the high quantities of total phenolics and flavonoids. 89 Furthermore, the GC-MS analysis of methanolic extract also identified various compounds with anti-inflammatory effects, strengthening the results of the studied extract. So, the plant showed anti-inflammatory potential and may be considered in dietary intervention to prevent aging-related chronic ailments.
Lipoxygenase Inhibitory Activity
The lipoxygenase activity was demonstrated by 5-LOX assay, and results were given in Table 8. The UPCM extract showed significant inhibition of the enzyme (59.82 ± 1.89%) compared to standard natural compound baicalein (96.56 ± 1.05%). Various plants from the family Apiaceae was reported for lipoxygenase inhibitory activities. 90 This evidence substantiated the current findings from P. candollei, that plant has the potential to inhibit lipoxygenase enzyme. Moreover, the association of antioxidants with inflammation comes from the mechanism by which bioactive antioxidant compounds scavenge free radicals which are produced during the inflammatory process by macrophages. Reactive Oxygen Species (ROS) produced during an inflammatory response can also provoke the arachidonic acid-mediated lipoxygenase pathway. 91
Lipoxygenase can generate inflammatory mediators such as leukotrienes and prostaglandins by interaction with lipids, which can lead to several chronic diseases such as bronchial asthma, allergic rhinitis, cardiovascular diseases, rheumatoid arthritis, neurodegenerative diseases and certain types of cancers. 92 The possible mechanism of anti-inflammatory effects related to the 5-LOX enzyme was explained by Doiron et al; they reported that radical scavenging (DPPH and ABTS) and reducing (CUPRAC and FRAP) antioxidant properties of phytochemicals inhibit this enzyme by disrupting its ferric ion redox cycle, which also substantiates the higher anti-inflammatory activity of UPCM. 83 A previous study on the anti-inflammatory and antioxidant activities of methanolic and aqueous extracts also concluded that methanolic extract showed more potent antioxidant and anti-inflammatory activities than the aqueous extract due to the presence of higher bioactive contents. 93 It supports our findings of anti-inflammatory activity of UPCM. Based on previous phytochemical investigations reported to date, the anti-inflammatory activities are supposed to be stronger; however, moderate results for this activity predict a possible antagonism among the various phytochemicals. It encourages the isolation of these compounds and to study their anti-inflammatory activities individually. So, we suggest that the isolation of compounds may lead to the finding of potent anti-inflammatory natural products for use in alternative medicine, and pharmaceuticals.
Cholinesterases Inhibitory Activity
Management of Alzheimer's disease (AD) focuses on inhibiting the acetylcholinesterase and butyrylcholinesterase enzyme, and they are documented as good candidates for the development of drugs against AD. 94 So, we conducted the cholinesterase inhibition assays to assess the anti-Alzheimer's disease effects of P. candollei. It found that UPCM showed significant inhibition of acetylcholinesterase which was 48.12 ± 1.81% in comparison with the galantamine (79.36 ± 2.25%), as shown in Table 8. While the butyrylcholinesterase inhibition was 37.15 ± 0.98% compared to 59.46 ± 1.05% inhibition by galantamine.
Many plant extracts have been investigated for their potential to prevent or manage cognitive disorders and other neurological diseases. 95 The cholinesterase inhibitory effect of similar specie Pleurospermum benthamii has been investigated previously, and good results showed the potential of this genus for AD. 16
Ahmed et al also reported some compounds with antioxidant, anti-inflammatory, sedative, and neuroprotective properties from the methanolic extract of P. candollei. They also identified a coumarin (7H-Furo(3,2-g)(1)benzopyran-7-one, 4,9-dihydroxy) with anti-acetylcholinesterase as well as anti-butyrylcholinesterase activity, which was 1.47% in terms of peaks area. 42 Based on all this discussion, it can be concluded that the significant inhibition of cholinesterases by UPCM is due to the described compounds and other compounds with antioxidant and anti-inflammatory activities. In the same study, the antioxidant effects of the methanolic extract were also reported by various mechanisms of reduction properties, which may contribute to the reduction in memory problems by protecting the brain cells from oxidative damage. 96 Based on current findings of anticholinesterase activity, the plant of study may be utilized in the formulations or dietary approaches to prevent or manage ageing related illness including AD and depression, which may rationalize its traditional use in cerebral disorders.
Monoamine Oxidase-A Inhibitory Activity
The findings of MAO-A inhibitory activity demonstrated significant inhibition (53.45 ± 0.18%) compared to the standard inhibitor of MAO-A (clorgyline; 77.24 ± 0.22). The results are presented in Table 8. Various compounds from UPCM detected in previous studies and the significant affinities of non-polar compounds with MAO-A enzyme may explain the in vitro enzyme inhibition by the UPCM extract. Moreover, the significantly higher quantity of phenolic compounds in UPCM compared to previously analysed methanol extract also showed the link of these bioactive compounds with antidepressant effect. Another study also reported the neuroprotective effect of a similar species (Pleurospermum kamtschaticum) from essential oil (that may contain non-polar bioactive compounds). 97 This finding may also support the ethanobotanical value of P. candollei in cerebral disorders, as it is used by native populations of the Himalayan region.
Conclusions
The objective of the current study was to reveal the health merits of P. candollei as a functional food/dietary adjuvant for the ageing population. Ultrasound-assisted extraction facilitated the yield of phenolic-rich extract. The molecular docking of previously identified non-polar compounds and quantified polyphenols predicted antioxidant, anti-inflammatory, anticholinesterase, and MAO-A inhibition. So, the bioactive contents detected by HPLC may be contributing to its biological activities along with non-polar compounds. In silico ADMET studies predicted the pharmacokinetic parameters and toxicological profile of major compounds, followed by insignificant results of the in vitro haemolytic assay. In vitro screening of the UPCM extract showed antioxidant, anti-inflammatory, antidepressant and anti-Alzheimer's (AD) effects. Overall, the results of this study provide knowledge of the plant's therapeutic potential, especially for ageing-related illnesses. It also encourages the traditional use in cerebral disorders and commercial cultivation of P. candollei to exploit it in the functional food and nutraceutical industry.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251378072 - Supplemental material for Phenolic Quantification, In Vitro Biological Activities and In Silico Studies of Ultrasound Assisted Extract of Pleurospermum candollei to Evaluate its Functional Food Potential
Supplemental material, sj-docx-1-npx-10.1177_1934578X251378072 for Phenolic Quantification, In Vitro Biological Activities and In Silico Studies of Ultrasound Assisted Extract of Pleurospermum candollei to Evaluate its Functional Food Potential by Jinghong Wei, Kashif-ur-Rehman Khan, Maqsood Ahmad, Sobia Tabassum, Rizwana Dilshad and Abdul Basit in Natural Product Communications
Footnotes
Acknowledgments
Acknowledged the resources and technical support from: Department of Pharmacy, 923 Hospital of the Joint Service Support Force of the Chinese People's Liberation Army China and Department of Pharmaceutical Chemistry, The Islamia University of Bahawalpur, Pakistan.
Ethical Statement
The animal/human models of study are not included in this article, so the statement is not applicable. However, publication ethics were followed according to journal's instructions.
Informed Consent/ Patient Consent
Not applicable.
Author Contributions
Conceptualization, M.A. and K.U.R.; methodology, J.W. and K.U.R.; software, M.A. and S.T.; validation, M.A.; formal analysis, A.B.; investigation, K.UR.; resources, J.W; data curation, A.B.; writing—original draft preparation, M.A. and R.D.; writing—review and editing, M.A. and S.T.; visualization, M.A.; supervision, K.U.R.; project administration, K.U.R.; funding acquisition, J.W. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Not applicable.
Trial Registration Number
Not applicable.
Supplemental Material
The bioavailability radars of nine compounds were provided as Supplemental material (supplementary figures A, B, C, D, E, F, G, H, I).
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



