Abstract
Objectives
Flemingia prostrata and Scutellaria baicalensis are vital components of Chinese herbal medicine, renowned for their diverse therapeutic applications attributed to rich flavonoid content. Accurate quantification of these flavonoids is crucial for ingredient quality control and pharmacological studies.
Methods
This present study reports the development and validation of a rapid and sensitive high-performance liquid chromatography coupled to mass spectrometry (HPLC-MS) method for the simultaneous quantification of eight key flavonoid compounds in traditional Chinese medicine, including genistein, prunetin, flemichin D, kushenol E and so on. The assay method utilized a Phenyl column based liquid chromatography coupled to triple-quadrupole mass spectrometer as the instrument for quantification.
Results
The assay demonstrated excellent linearity, precision, accuracy and stability. The validated method was successfully applied to quantify the eight flavonoids in multiple batches of Flemingia prostrata, Scutellaria baicalensis and Lycium barbarum, revealing variations in flavonoid profiles.
Conclusion
This reliable and efficient assay provides a valuable tool for the comprehensive evaluation and quality assessment of ingredients in Chinese herbal medicine, contributing to the standardization of their medicinal use.
Introduction
Flemingia prostrata and Scutellaria baicalensis are traditional Chinese medicinal plants extensively used for their therapeutic properties. F. prostrata is a perennial herb that has been employed in traditional medicine for its anti-inflammatory, analgesic, and wound-healing properties.1,2 Its extracts contain various bioactive flavonoids and phenolic compounds responsible for these pharmacological effects. S. baicalensis, commonly called “Huang Qin”, is a perennial herb renowned for its broad pharmacological activities, including antimicrobial, anti-inflammatory, and anticancer effects.3,4 The roots of S. baicalensis are rich in flavonoids such as baicalin, baicalein, and wogonin, which have been extensively studied for their antioxidant and immunomodulatory actions. 5 Both herbs have long-standing applications in traditional Chinese medicine formulations aimed at clearing heat, detoxification, and enhancing immune function. 6 Modern research has begun elucidating their bioactive constituents and mechanisms of action, supporting their traditional uses and suggesting potential for developing novel therapeutic applications. Given their extensive historical use and emerging scientific evidence, F. prostrata and S. baicalensis remain significant in the ongoing search for plant-based medicinal compounds.
Flavonoids are a diverse group of polyphenolic compounds widely distributed in the plant kingdom and known for their broad spectrum of bioactivities. These natural compounds exhibit significant pharmacological effects, including antioxidant, anti-inflammatory, antiviral, anticancer, cardioprotective, and neuroprotective activities.7,8 Their antioxidant properties are primarily due to their ability to scavenge free radicals and modulate oxidative stress-related signaling pathways, which contributes to the prevention of chronic diseases such as cardiovascular disorders and cancer.9,10 Given these multifaceted bioactivities, flavonoids are gaining increasing attention in drug discovery and the development of novel therapeutic agents derived from natural products.
The quality and efficacy of many Chinese herbal medicine ingredients, such as F. prostrata and S. baicalensis, are directly related to the concentration and composition of its flavonoid components. Therefore, accurate and reliable methods for quantifying these compounds are essential for quality control, standardization, and pharmacological research. High-performance liquid chromatography-mass spectrometry (HPLC-MS) has emerged as a powerful technique for the separation and quantification of complex mixtures.11,12 While several HPLC or LC-MS methods have been reported for the analysis of individual or a limited number of flavonoids in Chinese herbal medicines,13,14 there is a need for a comprehensive assay that can simultaneously quantify a wider range of key flavonoid compounds. This is particularly important for accurately reflecting the overall chemical profile and potential synergistic effects of these compounds.
This study aims to develop and validate a rapid, sensitive, and reliable HPLC-MS method for the simultaneous quantification of eight major flavonoid compounds in samples. The developed method will be applied to analyze multiple batches of F. prostrata, S. baicalensis and Lycium barbarum samples from different places of origin, providing valuable insights into the variations in their flavonoid profiles. This comprehensive approach will contribute to the establishment of robust quality control standards and facilitate further research into the pharmacological mechanisms of flavonoids.
Materials and Methods
Chemicals and Reagents
Reference standards of eight flavonoid compounds, as listed in Table 1, were purchased from MUST (Cheng Du) Biotechnology Co., Ltd with purity ≥ 98%. Acetonitrile (LC-MS grade), methanol (LC-MS grade), and formic acid (LC-MS grade) were obtained from Fisher Scientific (Waltham, MA, USA). Ultrapure water used for mobile phase was prepared using a Milli-Q water purification system (Millipore, Billerica, MA, USA). All other chemical reagents were of analytical grade.
Information and MS Parameters of the Eight Flavonoids.

Abbreviations: EP, Entrance Potential; CE, Collision Energy; CXP, Collision Cell Exit Potential.
Preparation of Standard Solutions
Stock solutions of each reference standard were prepared by dissolving 2 mg of each compound in methanol to a final concentration of 2 mg/mL. Working standard solutions were prepared by serial dilution of the stock solutions with methanol to obtain a series of concentrations within the linear range of the method.
Preparation of Chinese Herbal Medicine Samples
F. prostrata were collected from 6 places of origin (Province of Yunnan, Sichuan, Guangdong, Hainan, Hunan and Guizhou). For compound extraction, dried roots (0.5 g) were homogenized in 10 mL of methanol and subjected to ultrasonic extraction for 60 min at room temperature. Then mixture was centrifuged at 12,000 rpm for 15 min, and the supernatant was diluted 10 times with methanol before LC-MS analysis. S. baicalensis were collected from 4 places of origin (Province of Sichuan, Shanxi, Shandong and Neimenggu). For compound extraction, dried roots (0.1 g) were homogenized in 2 mL of methanol and subjected to ultrasonic extraction for 60 min at room temperature. Then mixture was centrifuged at 12,000 rpm for 15 min, and supernatant was submitted to LC-MS analysis. L. barbarum (Wolfberry) were collected from 4 places of origin (Province of Ningxia, Xinjiang, Gansu and Qinghai). For compound extraction, dried fruits (0.1 g) were homogenized in 2 mL of methanol and subjected to ultrasonic extraction for 60 min at room temperature. Then mixture was centrifuged at 12,000 rpm for 15 min, and supernatant was submitted to LC-MS analysis. Three biological replicates were prepared for each sample type.
LC-MS Analysis
The LC-MS analysis was performed on an SCIEX 7500 QTRAP system (SCIEX, Framingham, MA, USA) equipped with an ExionLC UHPLC (Shimadzu Corporation, Kyoto, Japan). The LC separation was achieved on an XBridge Phenyl 2.1 mm×10cm,3.5 um (Waters Corporation, Milford, MA, USA) column at a temperature of 40 °C. The mobile phase A consisted of 0.1% formic acid in water and mobile phase B was 0.1% formic acid in acetonitrile. A gradient elution was used as follows: 0–0.5 min, 5% B; 0.5–10 min, 5–95% B; 10–12 min, 95% B; 12–12.5 min, 95–5% B; 12.5–15 min, 5% B. The flow rate was 0.5 mL/min, and the injection volume was 10 μL. Mass spectrometer was operated in MRM mode targeting 16 transitions representing the eight compounds (Table 1). The MRM assay was performed in a positive ion mode without scheduling. Ion source was operated in ESI mode. Curtain gas, 40 psi; ion source gas 1, 70 psi; ion source gas 2, 35 psi; CAD gas, 9 psi; source temperature, 350 °C. The ion spray voltage was 1.5 kV. Q0 Dissociation (Q0D) was set to simple mode at −10 V for all compounds. One technological replicate was performed for each sample.
Peak Extraction
Raw MS data files were imported to the OS software for peak extraction (SCIEX). One transition for each compound was selected for quantification, and the peak area corresponding to each transition was calculated by the software.
Method Validation
The method was validated for linearity (dynamic range), low limit of quantification (LLOQ), precision, accuracy (recovery), and stability. The LLOQ can be defined as the lowest concentration with a coefficient of variation (CV%) lower than 20% obtained from three replicates. Linearity was assessed by constructing calibration curves using standard solutions at different concentrations. Precision was evaluated by intra-day and inter-day repeatability, expressed as relative standard deviation (RSD, CV%). Intra-day precision was performed and accessed using six replicates of one medium concentration analyzed in one day. Inter-day precision was performed and accessed using three replicates on three consecutive days. Accuracy (recovery) was determined by a spike recovery experiment. Known amounts of standard solutions of three concentrations (low, medium and high) were spiked into a sample with known analyte concentration. Stability was evaluated by preparing a sample and store it in room temperature for 1, 2, 4, 6, 8 and 12 h before analyzed, and a coefficient of variation (CV%) lower than 20% was considered acceptable.
Statistical Analysis
Coefficient of variation (CV%) and RSD were Calculated by dividing the SD (Standard Deviation) by the average. The calibration curves (y = ax + b) were constructed using the weighted regression method (1/x2) of the peak area of the analyte (y) versus actual concentrations (x). An acceptance criterion of the coefficient of determination (R2) value being greater than 0.99 and the accuracy at each concentration (accuracy is presented as a percentage of the calculated concentration over theoretical concentration) being between 85%-115% was employed to assess the linearity of the calibration curves. Recovery was calculated by comparing the actual concentration to theoretical concentration after spiking, and a recovery between 80% and 120% was considered acceptable.
Results
Method Development
Firstly, mass spectrometer parameters were optimized for each compound by infusing standard solutions into MS via syringe. Key parameters were optimized including transitions, collision energies, exit lens voltage and Q0D. At least two transitions were selected for each compound, using one as quantitative transition and the other as qualitative transition (Table 1). Chromatographic conditions were optimized in terms of column type, mobile phase composition and gradient. Finally, the assay method achieved the simultaneous quantification of the eight flavonoid compounds in a 15 min LC-MS run as shown in the extracted ion chromatography (Figure 1).
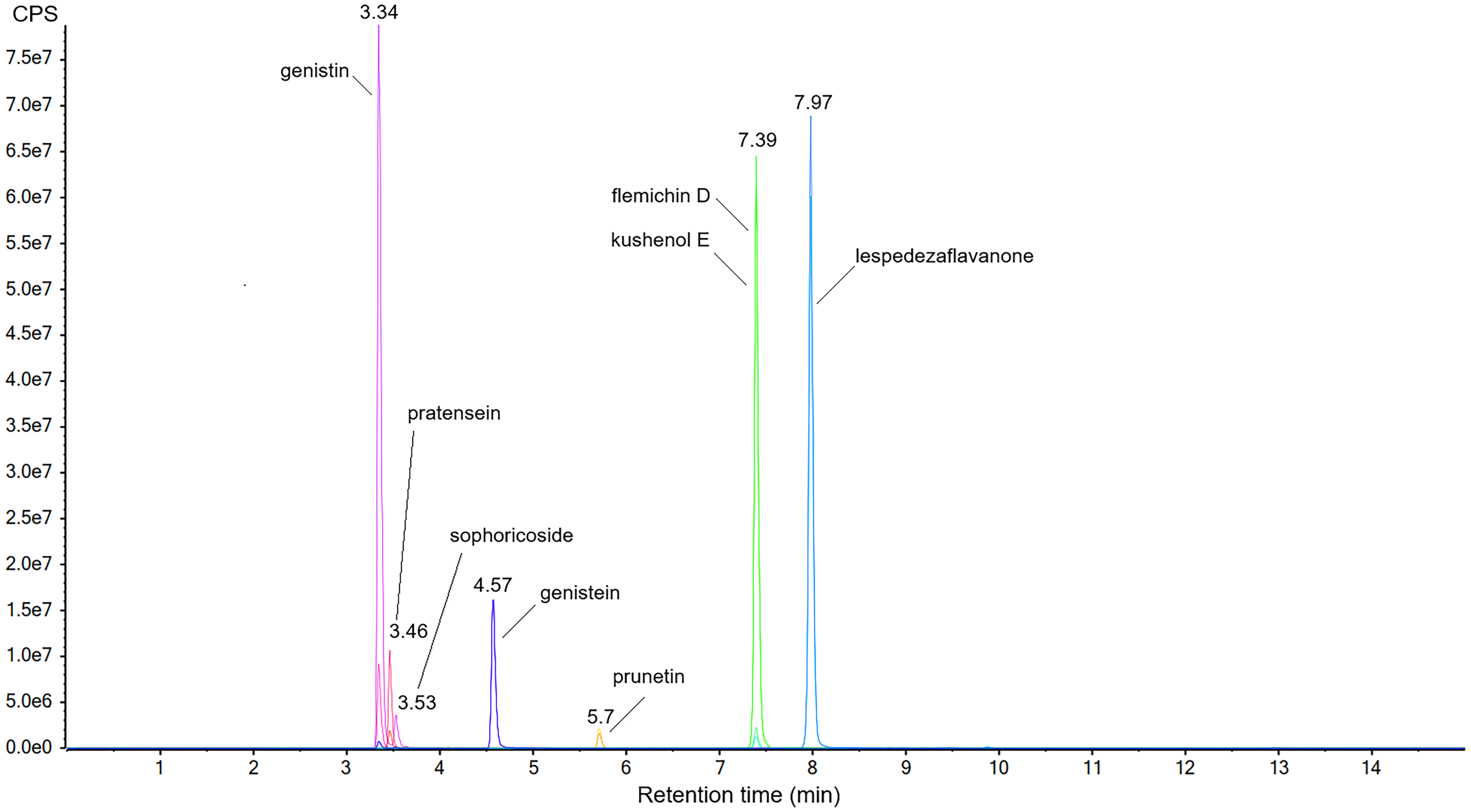
Extract Ion Chromatography of the Eight Flavonoids.
Method Evaluation
The developed assay method demonstrated excellent linearity for all eight flavonoid compounds within the tested concentration ranges from 1 ng/mL to 1 ug/mL, with correlation coefficients (R²) exceeding 0.99 (Table 2). The lower limits of quantification (LLOQs) were determined to be ranging from 0.2 ng/mL to 3.9 ng/mL depending on certain compound, indicating the high dynamic range and sensitivity of the method. The intra-day and inter-day precision, expressed as RSD, were found to be less than 6%, demonstrating good repeatability (Table 2). The accuracy of the method, assessed by recovery studies, ranged from 80% to 120%, indicating satisfactory accuracy (Table 2). The stability of the eight flavonoids in the sample solution was confirmed for at least 12 h at room temperature (Table 2).
Method Evaluation Result of the Eight Flavonoids.
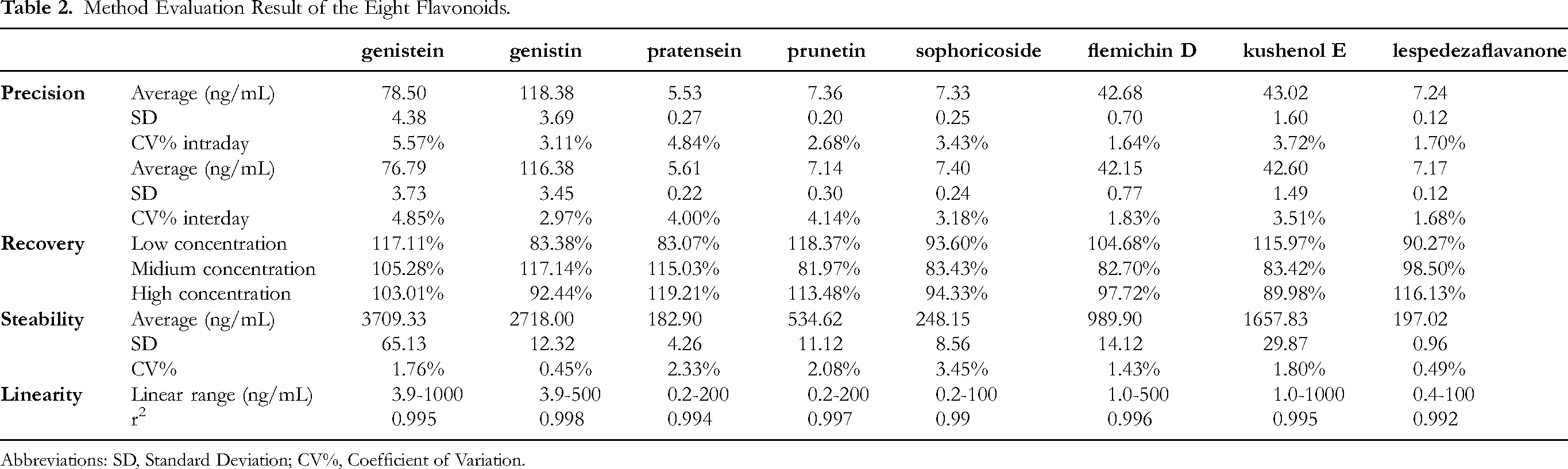
Abbreviations: SD, Standard Deviation; CV%, Coefficient of Variation.
Application to Chinese Herbal Samples
The developed assay method was successfully applied to quantify the eight flavonoids in F. prostrata collected from six places of origin, as well as S. baicalensis and L. barbarum collected from four places of origin. These three categories of plants showed different expression profiles in terms of the eight flavonoids (Table 3). For instance, all the eight kinds of flavonoids were detected in the F. prostrata, but S. baicalensis mainly contained five of the eight flavonoids (genistein, genistin, pratensein, prunetin and sophoricoside). As for L. barbarum, flavonoids were basically not detectible except genistin and pratensein being detected at extremely low level from two geographical origins (Ningxia and Xinjiang). For most of the detected flavonoids, F. prostrata showed a higher concentration (ug/g) than S. baicalensis. Principal component analysis (PCA) and hierarchical clustering analysis were used to illustrate the relations among the samples. As shown in Figure 2A, PCA score plot showed distinct separation among these different medicines as well as among F. prostrata samples collected from different places of origin. The heatmap in Figure 2B showed that F. prostrata showed an overall higher concentration (ug/g) than S. baicalensis but presented distinct differences among samples collected from different places of origin.
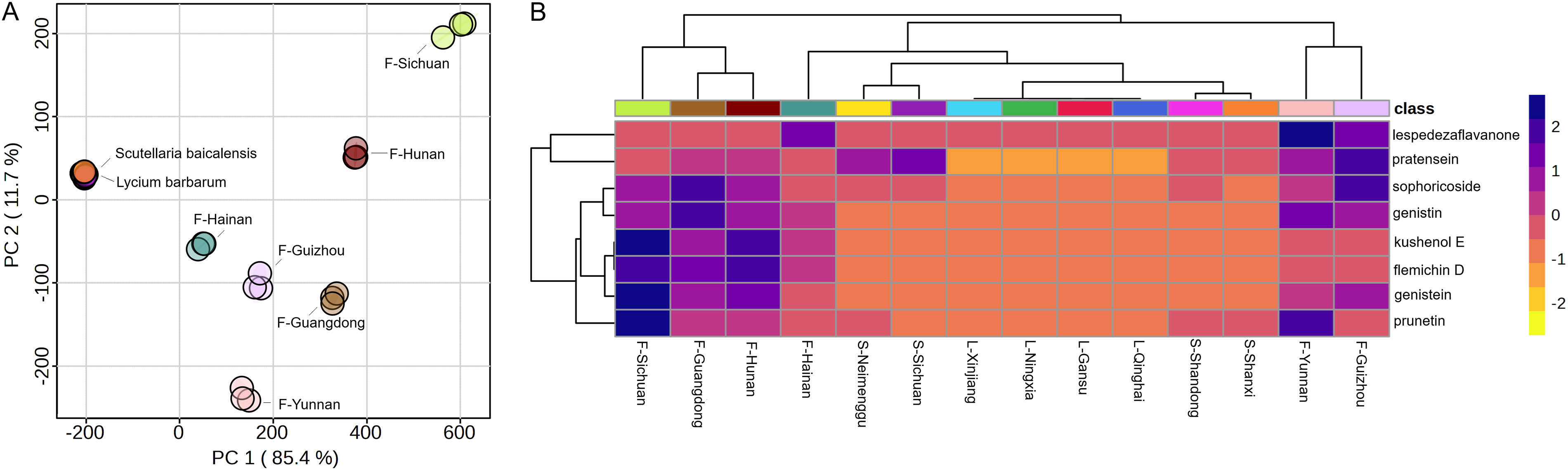
Flavonoid Profiles of the Samples. Abbreviations: F-, Flemingia prostrata; S-, Scutellaria baicalensis; L-, Lycium barbarum.
Concentrations of 8 Flavonoids in Ingredients of Tradition Chinese Medicine.

Discussion
Flavonoid is a group of natural products having biomedical promise and valuable bioactivity, thus making the accurate analyses of flavonoid in biological samples meaningful. The developed LC-MS method in this study provides a rapid, sensitive, and accurate tool for the simultaneous quantification of eight key flavonoid compounds in traditional Chinese herbal medicine F. prostrata and S. baicalensis. This assay method showed great potential in applications for quality control and research purposes.
After parameter and condition optimizations for the liquid chromatographer and mass spectrometer, the finalized assay method achieved a high analytical performance. The employment of high-performance liquid chromatographical separation of targeted compounds using phenyl column achieved excellent separation, thus enabled better sensitivity, and less matrix effect caused by co-elution compounds. Compared to previous reported assay methods using UV absorption in liquid chromatographical analysis as detector, the assay method we developed here used triple quadrupole mass spectrometer as the detector. Mass spectrometer measures molecular weight of targeted compound as well as its fragments, thus can discriminate targeted compounds from impurities which have similar liquid chromatographical retention times or chemical structures. 15 Thus, the mass spectrometry-based assay method enabled better analytical specificity and reliability. To summarize, the excellent linearity, precision, accuracy, and stability of this developed assay method demonstrate its performance and reliability for analyzing biological samples in a high throughput setting. The LLOQs indicate the high sensitivity of the method, enabling the detection of even trace amounts of these compounds.
The successful application of the method to analyze multiple batches of F. prostrata, S. baicalensis and L. barbarum samples revealed significant variations in the concentrations of the eight flavonoids. These variations may be attributed to several factors, including plant species and geographical origin. For instance, F. prostrata has higher flavonoid concentrations for all the eight kinds of flavonoids, while S. baicalensis mainly contained five kinds of flavonoids (genistein, genistin, pratensein, prunetin and sophoricoside), containing lower concentrations than F. prostrata except for pratensein. As for L. barbarum, flavonoids were basically not detectible except genistin and pratensein being detected at extremely low level from a few geographical origins. These results indicate that F. prostrata serves as a better source of certain flavonoids than S. baicalensis, while L. barbarum is not a suitable source of flavonoids. What's more, for a given kind of plant species, geographical origins also play an important role in terms of flavonoids concentration, as can be seen in the result of F. prostrata (Figure 2). This finding further shows the importance and necessity of quality test of raw ingredients of Chinese herbal medicine collected from different geographical origins or batches to ensure the content of bioactive compounds.
Flavonoid, a diverse group of polyphenolic compounds widely distributed in the plant kingdom, have attracted significant scientific interest due to their broad spectrum of pharmacological activities. These compounds are well-recognized for their antioxidant properties,16,17 which stem from their ability to scavenge free radicals and chelate metal ions, thereby reducing oxidative stress-a common pathway involved in numerous chronic diseases such as cancer, 18 cardiovascular disorders, 19 and neurodegenerative conditions. 20 Moreover, flavonoids exhibit anti-inflammatory effects by modulating key signaling pathways, including inhibition of cyclooxygenase (COX) enzymes and downregulation of pro-inflammatory cytokines such as TNF-α and IL-6, contributing to their potential in managing inflammatory diseases. 21 Their antimicrobial activity has also been documented, with flavonoids demonstrating inhibitory effects against various bacterial and viral strains, possibly through disruption of microbial enzyme systems and interference with nucleic acid synthesis.22,23 Overall, the diverse pharmacological activities of flavonoids, underpinned by their molecular interactions with various biological pathways, position them as promising agents for the development of new therapeutic strategies across multiple health conditions. Further research is warranted to elucidate their precise mechanisms of action, bioavailability, and the potential for clinical application, paving the way for optimized use of flavonoid-rich extracts or derivatives in medicine.
The observed variations in flavonoid profiles highlight the importance of establishing robust quality control standards for Chinese herbal medicine ingredients. The developed method can be used to assess the quality of different plant species to identify samples with high concentrations of the target flavonoids, or to assess the flavonoid concentrations in different batches of samples to ensure the high quality of ingredients. This will contribute to the standardization of raw ingredients and ensure the consistency of their medicinal efficacy. The method could also further facilitate scientific researches such as analyzing samples from different sources to study the effects of various factors on the flavonoid composition, or investigate the synergistic effects of the eight flavonoids and their interactions with other bioactive compounds in plants.
This assay method has great potential for further optimization and extension. First, sample preparation procedure could be further optimized to increase the extraction efficiency for target compounds depending on sample types. For example, homogenization procedure is different between fresh herbal samples and dried ingredients such as the roots we used in this study. Additional soaking or ultrasonic extraction in solvents would be necessary for samples that are dry and tough. Our present study is a proof-of-concept study which only included three kinds of herbs for flavonoid extraction, so if this assay method would be used for other types of samples, the sample preparation procedure should be optimized accordingly. Second, considering the high-throughput feature of LC-MS analysis which could quantify hundreds of targets in one single run, more targets of interest could be included in this assay method depending on need, such as other compounds in flavonoid category or other bioactive compounds coexisting in the raw ingredients.
There are several limitations in the present study. First, internal standards were not included due to the inaccessibility of isotope-labeled chemical standards. As a suboptimal alternative, analogues (structurally similar compounds to the analytes) could be used as internal standards. However, their performance must be thoroughly evaluated in terms of retention time during liquid chromatographic separation and ionization efficiency in mass spectrometry. Therefore, the selection and evaluation of suitable internal standards were not addressed in this study but will be considered in future work. The second limitation is that sample preparation recovery and matrix effects during LC-MS analysis were not assessed, primarily due to the lack of an ideal blank matrix—ie, a sample free of all target analytes. A suboptimal approach to evaluate these parameters would involve using structural analogues for which blank matrices are available, and this will be explored in future studies. Lastly, due to the limited sample size, our findings and interpretations regarding differences among plant species or their geographic origins are preliminary. Additional experiments with larger sample sets are necessary before drawing definitive conclusions.
Conclusion
In conclusion, the developed HPLC-MS method provides a valuable tool for the comprehensive evaluation of flavonoid profiles in common ingredients of Chinese herbal medicine. The method can be used for quality control, standardization, and further scientific research on the pharmacological mechanisms of bioactive components in plants.
Footnotes
Acknowledgements
The Authors would like to thank the staffs at the mass spectrometry facility in the Center of Medical and Health Analysis in Peking University Health Science Center for maintenance of LC-MS instruments and technical support.
Authorship Contributions
BZ, JL and JL conceived and designed the experiments. BZ, ZZ YL and LM conducted the sample collection and preparation. DW and JZ developed the assay method. JL, JZ and JL wrote the manuscripts. JL and JL oversaw the completion of this study and edited the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Natural Science Foundation of Hebei Province, Major Science and Technology Support Programme Projects in Hebei Province, Hebei Provincial Administration of Traditional Chinese Medicine Research Plan Project, Scientific Research Project of Colleges and Universities of Hebei Province, (grant number 31700898, H2023209038, 24297701Z, 2020222, QN2025501).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
