Abstract
Standardizing traditional herbal formulations poses significant challenges due to inherent variability in plant sources, environmental conditions, and processing methods. This narrative review explores how metabolomics a powerful high-throughput analytical approach for comprehensive chemical profiling addresses these issues by enabling the identification and quantification of bioactive compounds in complex herbal mixtures. Advanced analytical tools such as nuclear magnetic resonance (NMR), liquid chromatography–mass spectrometry (LC-MS), and gas chromatography–mass spectrometry (GC-MS) provide detailed metabolic fingerprints that underpin quality control and therapeutic consistency. Emerging techniques including desorption electrospray ionization–mass spectrometry (DESI-MS), direct analysis in real-time mass spectrometry (DART-MS), single-cell metabolomics, and stable isotope ratio analysis (SIRA) enhance spatial and geographical specificity, offering rapid and minimally invasive profiling capabilities. Integration of metabolomics with chemometric tools such as principal component analysis (PCA) and partial least squares–discriminant analysis (PLS-DA), facilitates authentication and batch-to-batch consistency. Furthermore, artificial intelligence (AI) and machine learning (ML) algorithms are increasingly employed to automate metabolite identification, predict pharmacological properties, and accelerate biomarker discovery. These advances collectively support evidence-based standardization and sustainability of herbal medicines, contributing to global healthcare integration. Key outcomes of this review include: identification of metabolomics as a cornerstone for quality assurance and authenticity of herbal products, evaluation of emerging analytical platforms for improving formulation reproducibility, and recommendations for interdisciplinary collaboration and regulatory alignment. Literature included in this study was sourced from peer-reviewed articles published over the past decade in databases such as PubMed, Scopus, and Web of Science. Overall, the review underscores the pivotal role of metabolomics in transforming traditional herbal practices into standardized, scalable, and personalized therapeutic interventions.
Keywords
Introduction
Herbal medicine refers to the use of plant-derived substances for therapeutic purposes. It encompasses a wide range of plant-based products, including extracts, powders, tinctures, and decoctions, used to prevent, manage, or treat diseases. 1 Herbal medicine is a key component of traditional medicine systems such as Traditional Chinese Medicine (TCM), Ayurveda, and African Traditional Medicine. A herbal formulation is a combination of different medicinal plant materials or their extracts, designed to enhance therapeutic efficacy. 2 These formulations may contain multiple herbs in specific proportions to achieve synergistic effects, reduce toxicity, and improve bioavailability. Examples include polyherbal formulations (multiple herbs combined), standardized extracts, and herbal capsules or tablets used in modern phytotherapy.3,4 Medicinal plants are plants that contain bioactive compounds with therapeutic properties. These plants are the primary sources of herbal medicine and may be used in their crude form (leaves, roots, stems, flowers, or seeds) or processed into extracts. Examples include Curcuma longa (turmeric), Zingiber officinale (ginger), and Withania somnifera (ashwagandha). 3
Traditional herbal formulations have been integral to healthcare systems worldwide, particularly in regions where natural products serve as primary therapeutic agents. These formulations, often derived from complex plant mixtures, exhibit diverse pharmacological properties and have been used for centuries in traditional medicine. 1 Herbal medicine has been used for thousands of years, dating back to the Palaeolithic era. 5 In 2021, the global herbal formulation market attained a value of $83 billion, underscoring its substantial expansion. 6 Although herbal medications are frequently favored for their presumed safety, a major challenge in their widespread adoption and integration into modern healthcare lies in the lack of standardization, which leads to inconsistencies in efficacy, safety, and quality control. Variability in plant sources, processing methods, and environmental factors further complicates the reproducibility of herbal medicines. Recent initiatives have aimed at integrating traditional knowledge with contemporary scientific methodologies to assess the safety, effectiveness, and bioactivity of herbal treatments. 7 This amalgamation of traditional wisdom and modern research seeks to improve world health by utilizing herbal medicine as a significant adjunct to contemporary healthcare methods. 8
Sustainability and ethical sourcing of medicinal plants are critical considerations in the standardization of herbal formulations, particularly as global demand rises. Unsustainable harvesting practices, habitat degradation, and loss of biodiversity threaten the long-term availability of many high-value medicinal species. 9 Additionally, ethical issues such as biopiracy and inadequate benefit-sharing with indigenous communities have raised concerns over the exploitation of traditional knowledge. 10 Addressing these challenges requires integrative strategies that prioritize conservation and fair-trade practices. Metabolomics plays a pivotal role in promoting sustainability by enabling the identification of metabolite-rich cultivars, supporting selective harvesting, and reducing overdependence on wild plant populations. 11 It also allows for the authentication of plant species and detection of adulterants, discouraging illegal trade and substitution. 11 By facilitating the development of standardized, reproducible, and traceable herbal products, metabolomics supports ethical supply chains and aligns with global biodiversity and access-benefit sharing (ABS) frameworks such as the Nagoya Protocol. 12 Efforts should encourage the integration of metabolomics data with geographic and ecological metadata to inform sustainable sourcing policies and protect medicinal plant ecosystems
Metabolomic profiling has emerged as a powerful analytical tool for comprehensively characterizing the chemical composition of herbal formulations. This high-throughput method facilitates the systematic examination of intricate phytochemical combinations, correlating them with biological activities without the need to isolate individual molecules. 13 By leveraging advanced techniques such as nuclear magnetic resonance (NMR), liquid chromatography-mass spectrometry (LC-MS), and gas chromatography-mass spectrometry (GC-MS), metabolomics enables the identification and quantification of bioactive compounds with high precision. 14 This approach provides a deeper understanding of the molecular fingerprint of herbal medicines, facilitating the development of standardized formulations with reproducible therapeutic effects.
In this review article, the role of metabolomics in addressing the standardization challenges associated with traditional herbal formulations is highlighted. By integrating metabolomic data with chemometric tools, researchers can enhance quality control measures, ensure batch-to-batch consistency, and authenticate herbal products. The adoption of metabolomic strategies represents a significant step toward bridging the gap between traditional and modern medicine, ultimately contributing to the global acceptance and regulatory recognition of herbal therapeutics.
Methodology
In this narrative review, a comprehensive literature search was conducted across major scientific databases, including PubMed, Scopus, and Web of Science. Keywords used included: “metabolomics”, “herbal medicine”, “LC-MS”, “NMR”, “GC-MS”, “standardization”, “traditional formulations”, and “quality control”. Articles published in English within the last 10 years (2015-2025) were included. Priority was given to peer-reviewed journal articles, reviews, and book chapters that directly addressed metabolomic technologies applied to herbal medicine. The findings were presented and discussed into the following categorized thematic areas: analytical tools in herbal standardization, spectroscopic techniques in standardization, applications of metabolomics in standardization, standardization challenges and solutions, future perspective, and recommendations. This segmented result and discussion approach was adopted so as to ensure a thorough, evidence-based foundation for discussing how metabolomics can support standardization, authentication, and quality assurance of traditional herbal formulations
Metabolomics in Natural Product Research: Analytical Tools in Herbal Standardization
Metabolomics has revolutionized natural product research by enabling the comprehensive analysis of small-molecule metabolites in complex biological systems. Metabolomics facilitates the thorough identification and analysis of both targeted and untargeted metabolites in medicinal plants, supporting herbal medicine discovery and quality control. 15 Unlike conventional analytical methods that focus on a limited set of compounds, metabolomics captures the entire metabolic fingerprint of herbal preparations, offering deeper insights into their pharmacological potential. 16 Several advanced techniques are employed in metabolomic studies of herbal medicines. Nuclear Magnetic Resonance (NMR) spectroscopy provides highly reproducible spectral fingerprints, allowing for the identification of key metabolites and their structural elucidation. 17 Liquid Chromatography-Mass Spectrometry (LC-MS) and Gas Chromatography-Mass Spectrometry (GC-MS) are widely used for their high sensitivity and resolution in detecting a broad range of phytochemicals. 15 LC-MS is considered superior to NMR and GC-MS in understanding the metabolomics analysis of herbal medicine. 18 LC-MS detects metabolites at lower concentrations, making it ideal for analyzing complex and low-abundance bioactive compounds in herbal extracts. 19 It can analyze both volatile and non-volatile metabolites, including large polar compounds like flavonoids, alkaloids, and glycosides. 19 LC-MS requires minimal sample preparation and can analyze crude herbal extracts directly, unlike NMR which requires a large amount of purified sample and a deuterated solvent. LC-MS also better characterizes polar and thermally labile compounds, as many herbal metabolites are highly polar and degrade at high temperatures. 19 LC-MS, especially high-resolution mass spectrometry (HRMS) with tandem MS, enables detailed fragmentation analysis to elucidate molecular structures with high accuracy. LC-MS is compatible with databases like METLIN, HMDB, and GNPS, aiding in the rapid identification of known and novel herbal metabolites. 20 It also supports large-scale metabolomics studies and can be integrated with chemometric tools to analyze herbal metabolite variations. LC-MS is generally preferred for the metabolomics analysis of herbal medicine due to its high sensitivity, broad metabolite coverage, minimal sample preparation, and ability to analyze both polar and non-polar compounds without derivatization. 21
While LC-MS is widely regarded for its high sensitivity, broad metabolite coverage, and minimal sample preparation, it is not without limitations. 22 One of the primary drawbacks of LC-MS is its high operational and maintenance cost, which includes expenses related to solvents, columns, and specialized technical expertise. 23 Additionally, data interpretation from LC-MS can be complex due to matrix effects, ion suppression, and the need for extensive spectral libraries for accurate metabolite identification. 24 These challenges can limit its accessibility and scalability, especially in low-resource settings. In contrast, NMR spectroscopy, though less sensitive, offers unmatched reproducibility, quantitative capabilities, and minimal sample preparation without the need for derivatization or separation. 25 It is highly robust for fingerprinting and structure elucidation, particularly useful when comparing metabolomic profiles across laboratories. GC-MS, on the other hand, remains a cost-effective alternative for analyzing volatile and thermally stable compounds, although it requires derivatization for polar metabolites. 26 Therefore, the choice of technique should be guided by the specific analytical goals, compound classes of interest, and available infrastructure. A balanced approach integrating LC-MS with complementary techniques such as NMR and GC-MS provides a more comprehensive and robust framework for herbal metabolomic profiling. These techniques, when coupled with computational and chemometric tools, enable the classification and authentication of herbal formulations based on their unique metabolite profiles. 16 Figure 1 below is a flowchart illustration for metabolomics pipeline for herbal formulation standardization.

A Flowchart Illustration for Metabolomics Pipeline for Herbal Formulation Standardization. (Created in https://BioRender.com).
Additional techniques such as High-Performance Thin-Layer Chromatography-Mass Spectrometry (HPTLC-MS) and High-Performance Liquid Chromatography (HPLC) offer complementary advantages, particularly in qualitative and quantitative profiling. HPTLC-MS combines thin-layer chromatography (TLC) with mass spectrometric detection, allowing for direct identification of separated compounds without extensive sample preparation.27,28 This technique provides advantages such as speed, cost-effectiveness, and minimal solvent usage. Unlike conventional TLC, HPTLC involves automated sample application, controlled development conditions, and densitometric analysis, making it more reproducible and quantitative. 29 Advantages of HPTLC-MS in Herbal Metabolomics include rapid separation and detection, high sensitivity and selectivity, minimal solvent usage and eco-friendliness, fingerprinting and quality control, and the ability to differentiate between formulations based on bioactive content. 30 Applications of HPTLC-MS include analyzing alkaloids and flavonoids, terpenoid and phenolic compounds, and traditional herbal formulation analysis. HPLC is widely employed in the metabolomic study of multi-herb formulations, enabling differentiation between formulations based on bioactive content. 31
HPLC is one of the most widely used chromatographic techniques in the metabolomic analysis of herbal formulations, offering high resolution, reproducibility, and versatility in detecting a wide range of compounds. 13 It is particularly suitable for polar and thermolabile compounds that are not amenable to GC-MS analysis. HPLC allows for both fingerprinting (qualitative) and quantitative assessment of herbal constituents, and precise concentration measurements of bioactive compounds can be achievable with external or internal standards.32,33 Versatile detection methods, such as HPLC-UV, UV, PDA (Photodiode Array), Fluorescence, or MS detection, enhance specificity. Reproducibility and robustness are enhanced by automated sample injection and controlled mobile phase flow. HPLC plays a critical role in the batch-to-batch consistency and authentication of herbal formulations. 34
Integration of HPTLC-MS and HPLC with other techniques enhances the robustness of herbal medicine analysis. HPTLC-MS provides rapid screening, eco-friendliness, and fingerprinting capabilities, while HPLC ensures high-resolution, quantitative analysis. 35 The integration of these techniques with advanced mass spectrometric and spectroscopic methods further strengthens the analytical framework for herbal metabolomics, ensuring authenticity, efficacy, and quality control of traditional medicines. In sum, HPTLC-MS and HPLC serve as essential tools in metabolomic profiling of traditional herbal formulations. HPTLC-MS provides rapid screening, eco-friendliness, and fingerprinting capabilities, while HPLC ensures high-resolution, quantitative analysis. 35 The integration of these techniques with advanced mass spectrometric and spectroscopic methods further strengthens the analytical framework for herbal metabolomics, ensuring authenticity, efficacy, and quality control of traditional medicines.
Spectroscopic Techniques in Standardization
The standardization of natural drugs is crucial for ensuring their quality, safety, and efficacy. Ultraviolet-Visible (UV-Vis) Spectroscopy, Infrared (IR) Spectroscopy, and Raman Spectroscopy are essential tools for quality control in the pharmaceutical and nutraceutical industries including herbal development. 36 These spectroscopic methods provide non-destructive, rapid, and accurate means for detecting, identifying, and quantifying bioactive compounds in natural drugs. UV-Vis Spectroscopy is based on the absorption of ultraviolet (200-400 nm) and visible (400-800 nm) light by molecules. 37 When light passes through a sample, specific wavelengths are absorbed due to electronic transitions in the molecules. The amount of absorbed light follows Beer-Lambert's law, which states that absorbance is directly proportional to the concentration of the absorbing species in the sample. 38 Applications in standardization of natural drugs include identification of active compounds, quantitative analysis, purity assessment, quality control, and monitoring herbal extracts and formulations. UV-Vis spectroscopy helps in qualitative identification of compounds, while IR spectroscopy measures the absorption of infrared radiation by molecules, causing vibrational transitions in chemical bonds. 36 Different functional groups in a molecule absorb IR radiation at specific frequencies, producing a unique spectral fingerprint that can be used for identification. 36
IR spectroscopy is valuable for identifying functional groups in bioactive compounds, such as hydroxyl (-OH) groups in flavonoids, carbonyl (C = O) groups in terpenoids and alkaloids, and aromatic rings in polyphenols. 39 It also allows for authentication and fingerprinting, detection of adulterants and contaminants, and monitoring herbal extracts and formulations. However, limitations of IR spectroscopy include water interference, overlapping bands, and limited quantitative capabilities. 39 Raman spectroscopy is based on inelastic scattering of light by molecules, known as Raman scattering. When a laser beam interacts with a sample, most light is scattered elastically (Rayleigh scattering), but a small fraction undergoes energy shifts due to molecular vibrations. 40 This energy shifts provide detailed molecular information. Applications in standardization of natural drugs include identification of bioactive molecules, authentication and detection of counterfeit herbal medicines, analysis of drug-polymer interactions, and real-time monitoring of extraction and processing. 40 Raman spectroscopy allows in situ monitoring of herbal extraction processes, optimizing conditions for maximum yield and purity. However, limitations of Raman spectroscopy include fluorescence interference, expensive instrumentation, and low sensitivity for some compounds. 36 In sum, UV-Vis, IR, and Raman spectroscopy play vital roles in the standardization of natural drugs by ensuring their authenticity, purity, and consistency. Combining these techniques enhances the reliability of natural drug standardization, ultimately improving their therapeutic efficacy and safety. As advancements in spectroscopic technology continue, these methods will become even more robust in ensuring the quality control of herbal medicines and natural drug formulations.
Emerging Analytical Frontiers in Herbal Medicine Standardization
Mass Spectrometry Imaging (MSI)
Mass Spectrometry Imaging (MSI) is an innovative analytical technique that combines molecular identification with spatial visualization, enabling the direct mapping of metabolite distributions in plant tissues without requiring extraction or labeling. 41 Unlike traditional mass spectrometry, MSI allows determination specific metabolites location within a sample, offering insights into tissue-specific biosynthesis, accumulation, and storage of bioactive compounds. 42 This spatial resolution is crucial in herbal medicine standardization, particularly when targeting specific parts of plants (eg, roots, leaves, or flowers) for pharmacological use. Common MSI techniques include Matrix-Assisted Laser Desorption/Ionization (MALDI-MSI) and Desorption Electrospray Ionization (DESI-MSI), both of which are compatible with complex matrices like dried plant sections or frozen tissues.42,43 By correlating metabolite maps with plant histology, MSI can help distinguish between genuine and adulterated herbal samples and identify localized patterns of contamination or degradation. Moreover, MSI can uncover the co-localization of synergistic phytochemicals, supporting the rationale for polyherbal formulations. 44 As the demand for precision herbal products grows, MSI is emerging as a valuable tool not only for quality control and authentication but also for phytochemical discovery, functional validation, and standardization of traditional herbal formulations at a much higher resolution.
Ambient Ionization Techniques (DESI-MS and DART-MS)
Ambient ionization techniques, such as Desorption Electrospray Ionization Mass Spectrometry (DESI-MS) and Direct Analysis in Real Time Mass Spectrometry (DART-MS), are novel analytical platforms that allow rapid, direct analysis of complex herbal samples under atmospheric conditions.45,46 These methods eliminate the need for extensive sample preparation, making them ideal for time-sensitive applications like point-of-care quality control, field authentication, or on-site screening of commercial herbal products. 46 In DESI-MS, charged solvent droplets are directed onto a sample's surface, desorbing and ionizing analytes for mass spectrometric analysis. In contrast, DART-MS uses a heated gas stream to ionize compounds directly from a surface. 46 These techniques are particularly useful for detecting low-molecular-weight bioactive compounds, contaminants, or adulterants in finished herbal formulations like capsules or powders. 46 Their high sensitivity and real-time output allow rapid fingerprinting and comparison across product batches. 47 Moreover, ambient ionization can be integrated with portable MS instruments, facilitating mobile metabolomics. 47 This is particularly valuable in regions where laboratory access is limited. As regulators push for improved traceability and consumer protection in the herbal industry, DESI-MS and DART-MS provide cutting-edge, cost-effective tools for ensuring authenticity, safety, and compliance with quality standards in herbal medicine supply chains.
Stable Isotope Ratio Analysis (SIRA)
Stable Isotope Ratio Analysis (SIRA) is a powerful technique that evaluates the natural abundance of stable isotopes such as carbon-13 (¹³C), nitrogen-15 (¹5N), oxygen-18 (¹8O), and hydrogen-2 (²H) within herbal products to determine their geographical origin, cultivation conditions, and authenticity. 48 These isotopic fingerprints arise from environmental factors like soil composition, climate, water sources, and photosynthetic pathways, which vary across regions and influence the isotopic composition of plant metabolites. 48 In the context of herbal standardization, SIRA serves as a forensic tool to trace adulteration, verify sourcing claims, and ensure label integrity, especially for high-value medicinal plants like ginseng, saffron, or turmeric.30,49,50 For example, a formulation claiming Himalayan origin can be isotopically matched against known reference profiles. SIRA is non-destructive and requires only small sample volumes, and its results are highly reproducible. 51 It is increasingly being adopted in food fraud investigations and is now expanding into botanical and herbal product authentication. When combined with metabolomics, SIRA enhances quality control by linking chemical composition with origin-specific markers, thereby supporting supply chain transparency and regulatory compliance. 51 As herbal trade becomes globalized, SIRA offers a scientific basis for validating provenance, deterring fraud, and strengthening consumer confidence in natural products.
Capillary Electrophoresis–Mass Spectrometry (CE-MS)
Capillary Electrophoresis–Mass Spectrometry (CE-MS) is an advanced hyphenated technique that integrates the high-resolution separation capabilities of capillary electrophoresis with the sensitive detection power of mass spectrometry. 52 CE-MS is particularly suited for the analysis of polar, charged, and thermally labile phytochemicals often found in herbal formulations, such as organic acids, amino acids, alkaloids, and flavonoid glycosides. 52 Unlike liquid chromatography, which relies on column interactions and can sometimes overlook small ionic compounds, CE separates analytes based on their charge-to-size ratio in an electric field, making it an excellent complementary method. 52 The minimal sample volume requirement (nanoliters) and fast run times make CE-MS especially attractive for precious or limited herbal samples. 53 Furthermore, CE-MS operates with aqueous buffers, reducing the need for organic solvents and enhancing its environmental friendliness. 54 In herbal standardization, CE-MS can be employed for fingerprinting, quantitative profiling, and identification of marker compounds, particularly in formulations with a wide polarity range. 54 It has also been used to differentiate between species or chemotypes of the same herb. Although still underutilized in herbal research, CE-MS is gaining attention due to its unique selectivity and compatibility with high-throughput metabolomics workflows.
Single-Cell Metabolomics
Single-cell metabolomics is a cutting-edge technology that analyzes the metabolite content of individual plant cells, offering unprecedented resolution into the cellular heterogeneity of medicinal herbs. 55 Traditional metabolomics assesses bulk tissue extracts, potentially masking important biochemical variations between different cell types or tissues. In contrast, single-cell metabolomics uses techniques like micro-sampling, capillary electrophoresis, or droplet-based microfluidics combined with high-resolution mass spectrometry to quantify the metabolite content of a single cell. 56 This approach is especially relevant for medicinal plants with complex tissues, such as roots containing vascular bundles and resin ducts, or leaves with secretory trichomes. By identifying cell-specific biosynthetic hotspots, researchers can pinpoint where critical bioactive compounds are synthesized and accumulated. 57 For example, alkaloids or terpenoids might be enriched in specific glandular cells, knowledge of which can inform selective harvesting or genetic engineering strategies. 57 Furthermore, single-cell metabolomics can be used to study stress responses, developmental stages, or interactions between cells, offering new insights into how environmental factors impact metabolite biosynthesis. 58 Though still in its infancy, the integration of single-cell approaches into herbal research promises to enhance our understanding of phytochemical dynamics, optimize cultivation strategies, and enable more targeted standardization and extraction protocols.
Metabolomics-Guided Genome Mining
Metabolomics-guided genome mining is an integrated approach that links metabolomic data with genomic information to identify the biosynthetic pathways and gene clusters responsible for the production of bioactive metabolites in medicinal plants. 59 By aligning metabolic fingerprints with transcriptomic and genomic datasets, researchers can uncover the genetic basis of key phytochemical traits, enabling precise marker-assisted selection, metabolic engineering, and synthetic biology applications. 59 This approach is especially useful for uncovering cryptic or low-abundance compounds that may have therapeutic potential but are difficult to isolate through conventional methods. Using tools like antiSMASH, BiG-SCAPE, and GNPS, researchers can predict biosynthetic gene clusters for alkaloids, flavonoids, and terpenoids, and validate their expression patterns through metabolomics.60,61 In the context of herbal medicine standardization, metabolomics-guided genome mining offers the ability to establish genetic “barcodes” for quality control and authentication. 62 It also enables the development of engineered plant lines or microbial hosts for the sustainable production of high-value compounds. 62 This is particularly useful for endangered or slow-growing medicinal plants. As sequencing costs continue to decline, and multi-omics integration becomes more routine, this technique is poised to redefine how herbal formulations are discovered, optimized, and standardized.
Microfluidics-Based Metabolomics (Lab-on-Chip)
Microfluidics-based metabolomics, also known as lab-on-chip technology, represents a transformative advancement in the field of analytical herbal medicine. 63 This technique involves the miniaturization of analytical processes into microfluidic devices, which handle extremely small fluid volumes (in the range of nanoliters to microliters). 64 These systems integrate sample preparation, separation, reaction, and detection on a single chip, drastically reducing analysis time, reagent consumption, and cost. For herbal standardization, microfluidic platforms can be coupled with spectroscopic or mass spectrometric detectors to perform rapid screening of complex phytochemical mixtures. 63 Applications include fingerprinting of herbal extracts, monitoring batch-to-batch consistency, and detecting adulterants or degradation products. 63 Moreover, microfluidic chips can be designed to mimic physiological environments, enabling on-chip bioassays for antioxidant or cytotoxic activity screening. 65 This makes them highly adaptable for high-throughput pharmacological testing. Their portability and speed are especially advantageous in resource-constrained settings where large-scale instrumentation is impractical. Despite their early-stage adoption in herbal research, ongoing developments in chip fabrication, automation, and integration with machine learning are expected to revolutionize point-of-care diagnostics and on-site quality control in traditional medicine. 66 Microfluidics holds promise as a rapid, eco-friendly, and highly scalable tool for metabolomic analysis and herbal standardization.
Table 1 categorizes and summarizes both conventional and emerging techniques based on their analytical purpose, strengths, limitations, and recent advancements. It highlights how these techniques contribute to quality control, metabolite profiling, authentication, and regulatory validation of traditional herbal formulations. The integration of advanced methods such as mass spectrometry imaging, ambient ionization, single-cell metabolomics, and AI-enhanced data analysis represents a paradigm shift from conventional bulk profiling to more precise, scalable, and high-resolution standardization. This categorization offers a valuable reference for researchers, regulatory bodies, and manufacturers aiming to select appropriate analytical platforms for specific standardization goals in phytopharmaceutical development.
Categorized Summary of Analytical Techniques and Advances in Herbal Medicine Standardization.

Applications of Metabolomics in Standardization
Recent metabolomic investigations have shown considerable promise in enhancing the standardization and quality control of herbal medications. Studies on widely used traditional formulations, such as Ayurvedic, TCM, and African herbal remedies
Standardization Challenges and Solutions
Despite the growing recognition of traditional herbal formulations in healthcare, their widespread acceptance is hindered by significant challenges in standardization (Table 2). These challenges span across technical, regulatory, and practical domains, each presenting unique barriers to the integration of herbal medicine into mainstream healthcare.
Key Challenges in Herbal Formulation and Standardization: Implications, Solutions, and Some Affected Herbs.
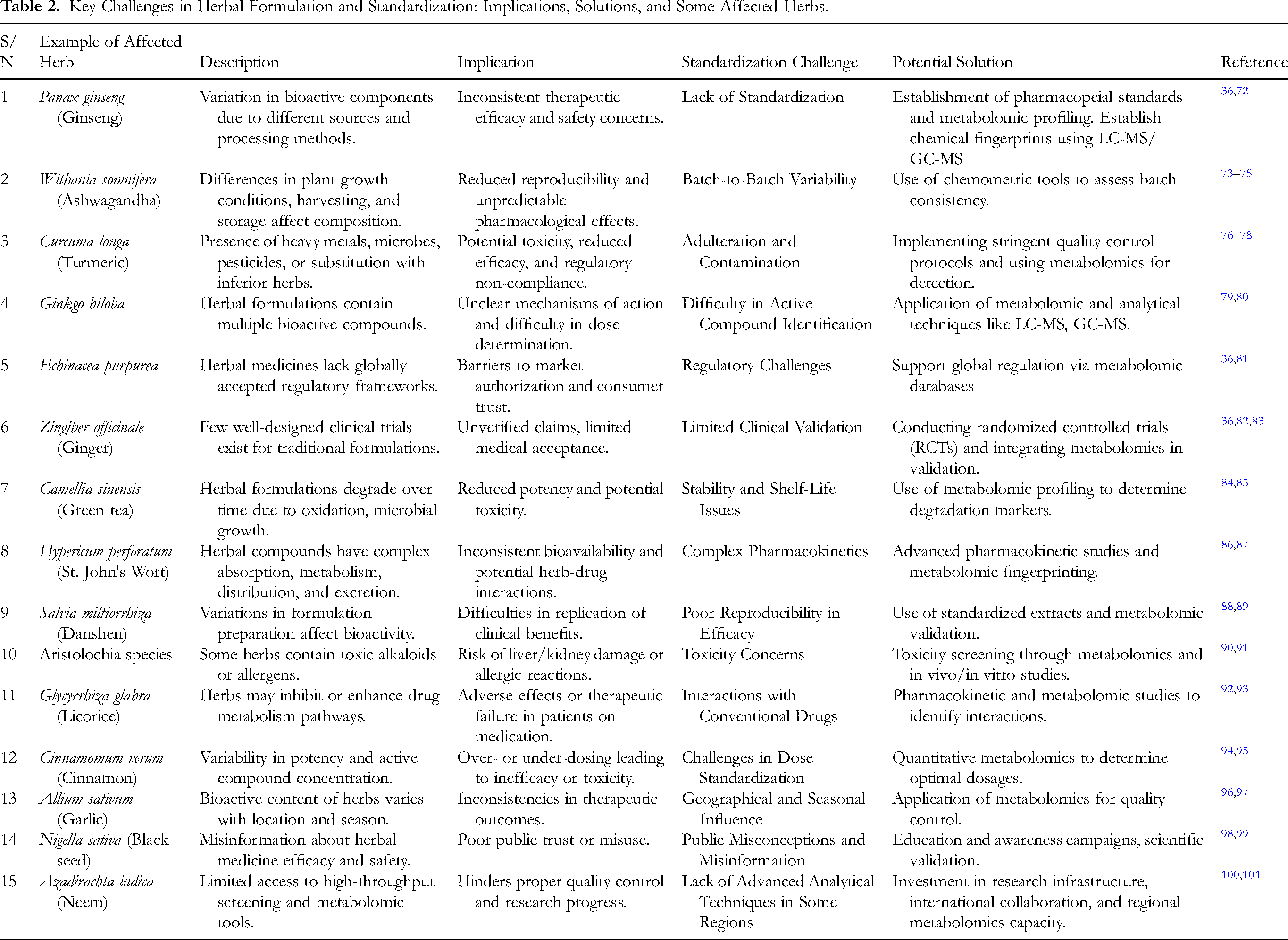
Technical Challenges and Solutions
Variability in plant sources, geographical conditions, harvesting techniques, and processing methods contributes to inconsistencies in the chemical composition of herbal medicines.102,103 Additionally, the presence of contaminants, adulterants, and variations in bioactive compound concentrations complicates quality control efforts. 103 These inconsistencies lead to fluctuating therapeutic efficacy and safety profiles. Metabolomics provides a promising technical solution to these issues by offering a comprehensive and reproducible approach to analyzing the complete metabolic profile of herbal formulations. This allows for the creation of chemical fingerprints that serve as consistent quality benchmarks.104,105 Chemometric tools, such as principal component analysis (PCA) and orthogonal partial least squares-discriminant analysis (OPLS-DA), further enhance metabolomic applications by enabling the classification and authentication of formulations based on their metabolite profiles. 106
Regulatory Challenges and Solutions
Regulatory gaps remain a major obstacle to standardization, as there is often no unified framework to assess and certify the quality and efficacy of traditional herbal formulations. 107 These gaps hinder the development of internationally recognized guidelines, delaying the integration of herbal medicines into conventional healthcare systems. Metabolomics can play a central role in regulatory advancements by contributing to the development of standardized reference databases and quality control frameworks for herbal products.108,109 Through the establishment of validated metabolic fingerprints and thresholds, regulatory agencies can more accurately assess product authenticity, safety, and therapeutic potential.
Practical Challenges and Solutions
On a practical level, limited awareness and accessibility of advanced analytical tools such as metabolomics in developing regions pose barriers to implementation. Furthermore, the traditional knowledge base often lacks documentation, complicating efforts to modernize and validate herbal practices. 110 The integration of bioactivity-guided metabolomic approaches bridges the gap between traditional medicine and modern evidence-based practices. By aligning traditional formulations with measurable biological activities and consistent metabolomic profiles, manufacturers and researchers can improve product standardization. 111 This not only ensures batch-to-batch consistency but also boosts consumer confidence and facilitates the global acceptance of herbal products as safe and effective therapeutic alternatives. 36
Future Perspectives
The integration of metabolomics into the standardization of traditional herbal formulations marks a significant step toward bridging the gap between traditional knowledge and modern scientific practices. As the field of metabolomics continues to evolve, there is growing potential to refine and optimize these methodologies for more accurate and reproducible results. The development of high-throughput platforms
Looking ahead, the standardization of herbal formulations through metabolomics can contribute to the creation of comprehensive, globally accepted databases that could serve as reference points for regulators, manufacturers, and consumers alike. Regulatory bodies may increasingly rely on metabolomic data to set quality standards and authenticate herbal medicines, fostering trust in natural products and supporting their integration into modern healthcare systems. Moreover, biotechnology innovations
Herbal medicine's future hinges on the creation of an evidence-based model combining modern technologies with ancient wisdom. In this sense, metabolomics will not only be essential for standardizing but also for releasing novel therapeutic possibilities for herbal compositions. Metabolomics could direct the creation of personalized and focused herbal treatments that fit individual health profiles by offering molecular knowledge of how bioactive components interact in complicated systems.
Application of Artificial Intelligence and Machine Learning in Plant Extract Metabolomics
The analysis of plant extracts using metabolomics has gained significant attention due to the complexity and diversity of phytochemicals present in medicinal and functional plants. Artificial intelligence (AI) and machine learning (ML) approaches are increasingly being employed to manage, interpret, and extract meaningful biological insights from the vast and multidimensional data generated during metabolomic profiling of plant materials. 113 These advanced computational techniques enable the identification of key bioactive compounds, classification of plant species or varieties, and prediction of pharmacological activities based on metabolic signatures. 113 Supervised ML algorithms such as Random Forest, Support Vector Machines (SVM), and Partial Least Squares-Discriminant Analysis (PLS-DA) have been widely used to differentiate between plant species, growth conditions, or treatment groups based on their metabolite compositions.114,115 These models also facilitate biomarker discovery by ranking metabolites according to their importance in classification, which is particularly useful in quality control and authentication of herbal products. 115 Deep learning methods, including convolutional neural networks (CNNs) and recurrent neural networks (RNNs), are now being applied to uncover complex, non-linear patterns within large-scale datasets obtained from techniques such as LC-MS, GC-MS, and NMR spectroscopy. 116
Unsupervised approaches like principal component analysis (PCA), hierarchical clustering, and t-distributed stochastic neighbor embedding (t-SNE) are commonly used to visualize metabolic variation and explore relationships between samples without prior class labels. 117 These methods help identify patterns and outliers, offering insights into the influence of environmental factors, geographic origin, or processing techniques on phytochemical profiles. Moreover, AI-driven integration of metabolomics with transcriptomic or genomic data enables researchers to link metabolic phenotypes with gene expression profiles, advancing our understanding of biosynthetic pathways and regulatory networks in medicinal plants. 118 Platforms such as MetaboAnalyst, XCMS Online, and KNIME, as well as programming frameworks like Python (scikit-learn, TensorFlow) and R, provide flexible environments for implementing ML models tailored to plant extract studies.119,120
In practical terms, AI and ML are being used to predict antioxidant, antimicrobial, or anti-inflammatory potential of plant extracts based on metabolite fingerprints, thereby accelerating the screening and validation of plant-derived compounds for therapeutic use. To ensure robustness and reproducibility, rigorous model validation using cross-validation, bootstrapping, or independent test sets is essential. By integrating AI and machine learning into plant metabolomics, researchers can enhance the accuracy, efficiency, and scalability of phytochemical analysis, ultimately contributing to the discovery and development of novel bioactive compounds from natural sources.
Recommendations
To strengthen the standardization of herbal medicine using metabolomics, the following recommendations are proposed (Table 3).
Strategic Recommendations for Metabolomics-Driven Standardization of Herbal Medicine.

Conclusion
The integration of metabolomics into the standardization of traditional herbal formulations has revolutionized how these complex mixtures are analyzed, authenticated, and validated. However, advancing this field requires not only technological adoption but also targeted research strategies. One critical research direction is the implementation of longitudinal stability studies
Additionally, future work should focus on multi-omics integration, particularly combining metabolomics with transcriptomics, proteomics, and microbiomics to decipher the underlying biosynthetic pathways and host-plant interactions that influence bioactivity. This systems-level approach can uncover novel biomarkers and synergistic compounds that conventional techniques may overlook.
Further research is also needed in metabolomics-informed clinical validation, where metabolomic profiles are linked to pharmacodynamic and pharmacokinetic outcomes through human or animal trials. These studies will help translate laboratory findings into clinically relevant standards.
Finally, the development of AI-driven predictive models, real-time portable metabolomics tools, and region-specific metabolomic databases for herbal authentication represent promising innovations. Prioritizing these directions will strengthen regulatory acceptance and global integration of herbal medicine in evidence-based healthcare.
Footnotes
Acknowledgment
The authors are grateful to Kampala International University for its support.
Ethics Approval
Not applicable
Consent to Participate
Not applicable
Consent to Publish Declaration
Not applicable
Authors’ Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Sharing Statement
All data are utilized in the manuscript.
Clinical Trial Date of Registration
Not applicable.
Clinical Trial Registration Number
Not applicable.
Clinical Trial Registry
Not applicable.
