Abstract
Background
Central precocious puberty (CPP) results from the early activation of the hypothalamus-pituitary-gonadal axis, and its prevalence is increasing. Several studies have indicated that traditional Chinese medicine (TCM) and Chinese herbal medicine (CHM) may benefit CPP. However, the efficacy of CHM in treating CPP patients is unclear. Recently several investigations have assessed the efficacy of TCM and CHM along with Western Medicine (WM). Nevertheless, the efficacy of these products in reducing CPP symptoms is unclear.
Methods
We performed a systematic review and meta-analysis to evaluate the effectiveness of CHM as an adjuvant therapy in alleviating the symptoms of CPP patients. We searched Six main databases including Web of Science, PubMed, and Scopus, CNKI, Wan-fang Database, and CBM using the related search keywords.
Results
Our data showed that the CHM significantly decreased the uterine volume (SMD = −0.97, 95% CI [−1.34, −0.61), ovarian volume (SMD = −0.74, 95% CI [−1.09, −0.39],), and bone age index (SMD = −1.69, 95% CI [−2.58, −0.80],). There was also a significant decrease in FSH (SMD = −1.84, 95% CI [−2.33, −1.36],), LH (SMD = −2.07, 95% CI [−2.78, −1.37],), and E2 (SMD = −1.49, 95% CI [−1.89, −0.94], I2 = 92) in CHM receiving group. The subgroup meta-analysis demonstrated that the Zhibai dihuang formula was more efficient in reducing primary sexual traits and serum levels of sex hormones.
Conclusion
This study shows that Chinese herbal medicine is an effective adjuvant therapy along with Western medicine in treating patients with CPP. CHM benefits patients with CPP as an adjuvant therapy in combination with Western medicine.
Keywords
Introduction
Precocious puberty, a frequently encountered pediatric endocrine condition, is characterized by the premature occurrence of primary sexual characteristics before the ages of 8 and 9, in girls and boys respectively. 1 CPP affects approximately one in every 5000 children, with every ten female to one male ratio. 2 The pathophysiology of CPP is attributed to the premature stimulation of the axis of hypothalamus-pituitary-gonadal, the elevation in pulsatile gonadotropin-releasing hormone (GnRH) secretion, and the premature release of sex hormones, resulting in premature epiphyseal closure.3,4 Children with CPP face long-term health risks, including short stature, diabetes, cancer, and cardiovascular diseases in adulthood, significantly affecting their physical, social, and psychological development.5,6 The first line treatment for children with CPP is gonadotropin-releasing hormone analogs (GnRHa) which inhibit gonadotropin secretion and negatively regulate GnRH receptors, resulting in a decrease of gonadal steroids to prepubertal levels. 7 While GnRHa treatment slows bone maturation and helps improve adult height,8,9 it has limitations, especially in children with advanced bone age, and its use is associated with several adverse effects, such as central obesity, erythema, vaginal bleeding, hyperlipidemia, and decreased bone density.10–12 Moreover, the long-term effects of GnRHa on children's weight gain, ovarian function, and reproductive health still require further investigation.4,13 Therefore, novel therapeutic agents seem to be necessary to optimize the efficacy of current therapeutic strategies.
Traditional Chinese medicine (TCM) practitioners have relied on herbs (Chinese herbal medicine) to manage a wide range of pediatric illnesses for thousands of years. 14 Despite this, historical literature contains relatively little information on the treatment of precocious puberty, possibly due to its lower incidence compared to infectious, respiratory, and gastrointestinal disorders. Traditional Chinese medicine has been commonly employed as a therapeutic approach for the treatment of precocious puberty in recent years. According to TCM principles, the underlying cause of this condition is attributed to a deficiency in kidney yin, which subsequently affects liver yin. 15 Over the past few years, there has been a notable rise in the number of animal and clinical studies investigating the efficacy of TCM and CHM for the management of CPP. 16 Several studies found the therapeutic benefits of CHM as an adjuvant therapy along with GnRH analogs for the treatment of CPP. However, there is currently a shortage of large-scale studies on CHM interventions for CPP. This systematic review and meta-analysis aims to assess the effectiveness and safety of CHM in treating CPP, with a focus on exploring the mechanisms of CHM interventions.
Materials and Methods
Search Strategy
We searched Six main databases including Web of Science, PubMed, and Scopus, China National Knowledge Infrastructure (CNKI), Wan-fang Database, and Chinese Biomedical Literature Database (CBM) using the related search keywords. The detailed search strategy is available in Supplementary File 1. All reviews and full-text articles of included studies were manually searched for additional studies.
Study Selection
To ensure the relevance and appropriateness of the articles, we conducted a meticulous screening process based on their titles and abstracts. Any studies that were deemed irrelevant to our research were excluded. Based on this examination, we further classified them as either included or excluded. All full-text manuscripts were thoroughly evaluated to determine their eligibility, considering factors such as being written in English or Chinese and involving human subjects. Studies that utilized animal models, reviews, and congress abstracts, or were written in languages other than English or Chinese were deliberately excluded from our analysis.
Inclusion criteria for this study encompassed various aspects. All studies that focused on CHM therapy for CPP and were published in both Chinese and English were considered. The inclusion of participants was not restricted based on factors such as nationality, sex, race, education, or medical units. Moving on to the types of intervention, the intervention group in this study received a combination of CHM therapy and GnRH analogs. In contrast, the control group was treated solely with GnRH analogs.
Outcomes encompassed measurements of primary sexual characteristics such as ovarian volume (OV), and uterine volume (UV), as well as serum sex hormone levels (LH, FSH, E2), bone age index (BAI), and adverse events (AEs) were also considered as secondary outcomes. The bone age index (BAI) is defined as the bone age difference/chronological age difference value (ΔBA/ΔCA).
Exclusion Criteria for This Study
The exclusion criteria for this research encompass comments, reviews, case reports, as well as cell line or animal experiments. Any studies with duplicate publications or incomplete data are not considered for inclusion in this study. Central Precocious Puberty (CPP) caused by organic lesions in the CNS is also a criterion for exclusion. Additionally, studies on peripheral precocious puberty, include those involving adrenal disorders, gonadal tumors, or hormone-containing food/drugs.
This study evaluates the effect of CHM as an adjuvant therapy in girls, and only data concerning the efficacy of CHM in alleviating symptoms in girls were added.
Data Extraction
The data from all “included” studies was extracted by two researchers. We gathered information from all the studies that were identified, including the publication year, first author's name, and details about the participants such as demographic, clinical, laboratory, and molecular data. In cases where patients appeared in multiple articles, their overlapping data was removed. To ensure the accuracy of the selection process, two reviewers independently performed the task. Any disagreements that arose between the two reviewers were resolved by a second review.
Statistical Analysis
The studies retrieved from the database were imported into Endnote X for deduplication. Following this, two reviewers independently selected relevant studies based on the abstracts, titles, and keywords. The remaining articles underwent full-text screening. Any discrepancies between the two reviewers were resolved by reviewing a discussion between the two authors. The data extraction process involved capturing key information about the studies, including author names, publication year, intervention details, sample size, mean age, intervention duration, and outcomes. We conducted the statistical analysis using Revman 5.1 software. To account for potential variability across studies, we employed a random-effects model to calculate Standardized Mean Difference (SMD). The I² statistic was used to measure the heterogeneity between studies, with values of 25%, 50%, and 75% indicating low, moderate, and high heterogeneity, respectively. Subgroup analyses were performed to find the source of heterogeneity and compare the efficacy of various herbal formulas.
Quality Assessment
Evaluation of included studies was conducted using the Cochrane risk-of-bias tool for randomized trials (RoB 2) to validate and address the potential risk of bias in included studies. Discrepancies between the two researchers were resolved through discussion and consultation between reviewers.
Results
Study Characteristics
The literature screening process yielded a total of 857 studies, which were then subjected to further analysis. After the removal of duplicate studies, 545 studies were retained for evaluation. Following a thorough review of titles and abstracts and the main texts, 110 studies were deemed suitable for inclusion. Subsequently, 17 studies17–33 were selected after a detailed examination of the full text. The selection section is outlined in Figure 1. These studies were published between 1998 and 2023. In total, there were 1590 patients included in the studies, with 780 in the control group and 811 in the experimental group. The intervention duration ranged from 3 months to 2.7 years on average. The most prescribed formula among included studies was Zhibai dihuang, followed by Dabuyin, Zhibai jianghuo, and Ziyin xiehuo formula, and thecombination of Zhibai dihuang and Dabuyin pill

PRISMA flow diagram.
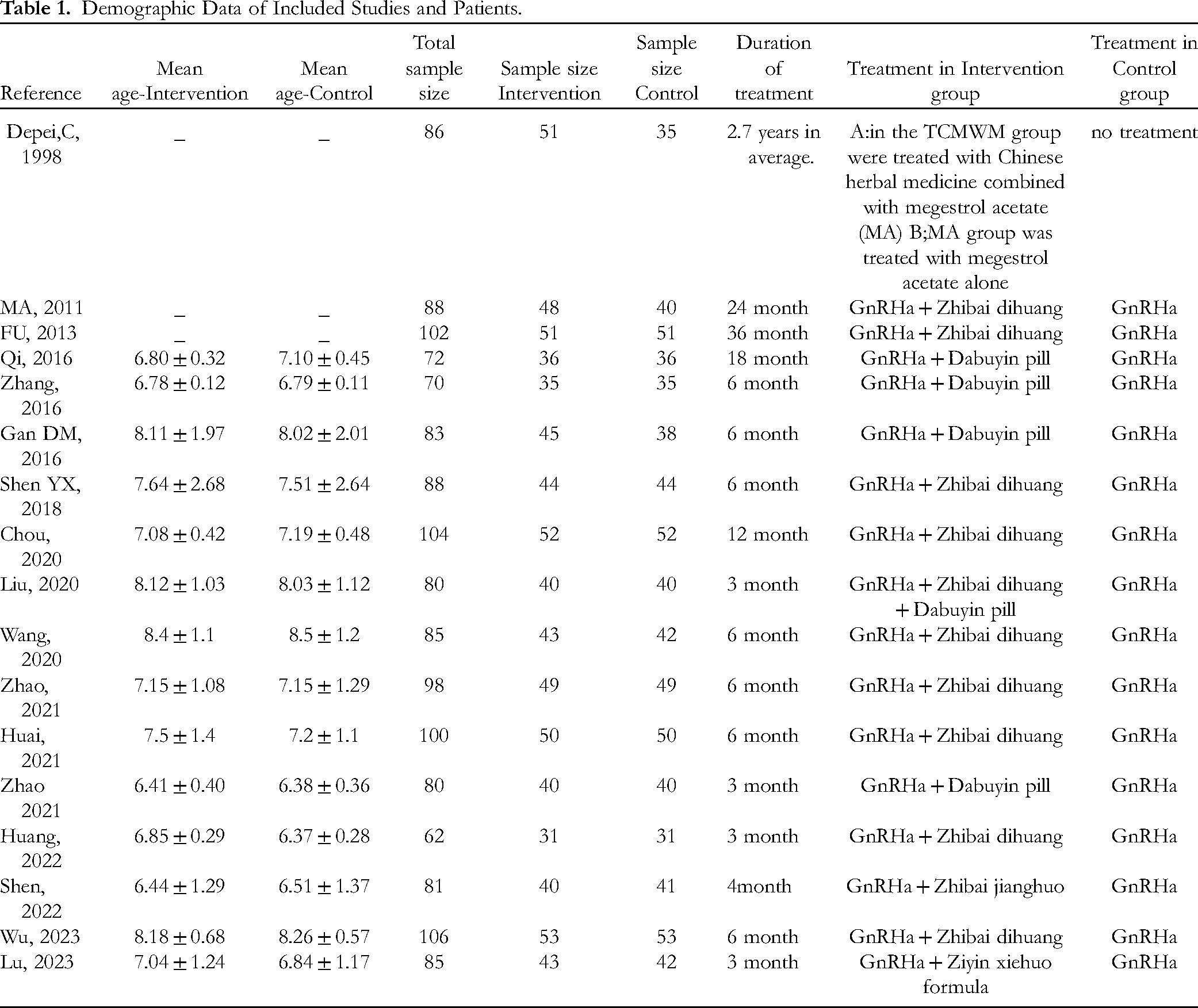
The basic characteristics of the included studies are summarized in Table 1.
Demographic Data of Included Studies and Patients.
Primary Sexual Characteristics
Fourteen studies assessed the efficacy of CHM on primary sexual traits. The results of the meta-analysis showed that the experimental group displayed significant improvements in uterine volume (UV) (SMD = −0.97, 95% CI [−1.34, −0.61], I2 = 89%) when compared to the control group. Subgroup analysis showed a significant decrease in UV (SMD = −1.08, 95% CI [−1.61, −0.56], I2 = 92%) when GnRHa + Zhibai dihuang compared to the control group but there were no significant improvements in UV (SMD = −0.54, 95% CI [−1.21, 0.12], I2 = 83%) when GnRHa + Dabuyin pill compared to the control group. In the results for median ovarian volume (OV), a significant decrease in the size was observed (SMD = −0.79, 95% CI [−1.15, −0.42], I² = 90%). Subgroup analysis showed a significant decrease in the size of OV for GnRHa + Zhibai dihuang but not significant for GnRHa + Dabuyin pill-treated patients compared to control group (SMD = −0.69, 95% CI [−0.97, −0.40], I2 = 77%, and SMD = −0.54, 95% CI [−1.66, 0.58], I2 = 95%, respectively). Data regarding primary sexual characteristics are shown in Figure 2.

The forest plot of primary sexual traits. A: improvements in uterine volume (UV), B: subgroup-analysis for changes in uterine volume, C: improvements in median ovarian volume, D: subgroup-analysis for changes in median ovarian volume.
Serum sex Hormone Levels
Fifteen RCTs investigated the level of serum sex hormones. The experimental group was composed of 691 patients, while the control group had 667 patients. The meta-analysis demonstrated a significant decrease in serum FSH (SMD = −1.84, 95% CI [−2.33, −1.36], I2 = 93%), LH (SMD = −2.07, 95% CI [−2.78, −1.37], I2 = 96%), and E2 (SMD = −1.49, 95% CI [−1.89, −0.94], I2 = 92), in the CHM receiving group compared to the control group. The subgroup analysis revealed that both Zhibai dihuang and Dabuyin significantly reduced FSH levels (SMD = −1.61, 95% CI [−2.09, −1.13], I2 = 89%, and SMD = −2.13, 95% CI [−3.79, 0.47], I2 = 97%, respectively), level of LH (SMD = −2.53, 95% CI [−3.30, −1.76], I2 = 93%, and SMD = −1.77, 95% CI [−3.14, − 0.39], I2 = 96%, respectively) and E2 (SMD = −1.61, 95% CI [−2.09, −1.13], I2 = 89%, and SMD = −2.13, 95% CI [−3.79, −0.47], I2 = 97%, respectively). Data regarding Serum sex hormone levels are shown in Figure 3.

The forest plot of serum sex hormone. A: decrease in serum FSH B: decrease in serum LH C: decrease in serum E2 D; subgroup-analysis for the decrease in serum FSH E: subgroup-analysis for the decrease in serum LH F: subgroup-analysis for the decrease in serum E2.
Bone age index
Ten studies included in the analysis assessed the Bone Age Index (BAI), with a total of 408 individuals in the control group and 439 individuals in the experimental group. The meta-analysis results showed a significant decrease in BAI for the experimental group when compared to the control group (SMD = −1.69, 95% CI [−2.58, −0.80], I2 = 97%). In contrast, the subgroup meta-analysis showed that Zhibai dihuang and Dabuyin were not significantly efficient for BAI improvement (SMD = −0.75, 95% CI [−1.59, −0.09], I2 = 95%, and SMD = −0.98, 95% CI [−2.20, − 0.24], I2 = 94%, respectively). Data regarding bone age index are shown in Figure 4.

The forest plot of bone age index (BAI). A: changes in BAI B:subgroup-analysis for the changes in BAI.
Safety of CHM
A total of 10 studies mentioned adverse effects during treatment with CHM as neoadjuvant therapy. Nausea and vomiting were the most prevalent adverse effects, which were reported in 8 studies followed by rash (reported in 3 studies). One study reported a transient vaginal bleeding in the GnRH-receiving group (but not CHM + GnRH group). In total, no serious adverse effect was reported among the included studies.
Quality Assessment
The outcomes of the risk-of-bias assessment conducted on the included studies are indicated in Figure 5.

Summary of bias assessment of included studies.
Sensitivity Analysis
We conducted the sensitivity analysis using a Leave-one-out meta-analysis to find the source of heterogeneity. For Uterine volume, studies by Huai 2021, Qi 2016, Shen 2022, and Zhao 2021 were the most possible cause of heterogenicity. By excluding these studies from the meta-analysis, the SMD was −0.78, with 95% CI [−1.02, −0.54], and the I2 dropped to 62% (from 89%). For median ovarian volume, studies by Huai 2021, Gan DM 2016, Liu 2020, Lu 2023, Wu 2023, Qi 2016, Shen 2022, and Zhao 2021 were the most possible cause of heterogenicity. By excluding these studies from the meta-analysis, the SMD was −0.64, with 95% CI [−0.83, −0.46], and the I2 dropped to 27% (from 90%). For the FSH level, studies by Huang 2022, Zhang 2016, Qi 2016, Shen 2022, and Zhao 2021 were the possible causes of heterogenicity. By excluding these studies from the meta-analysis, the SMD was −1.74, with 95% CI [−2.08, −1.40], and the I2 was dropped to 81% (from 93%). For the LH level, studies by Huang 2022, Huai 2021, Qi 2016, Depei 1988, and Chou 2022 were the possible causes of heterogenicity. By excluding these studies from the meta-analysis, the SMD was −2.34, with 95% CI [−2.68, −1.99], and the I2 dropped to 73% (from 96%). For the LH level, the study by Huang 2022 was the possible cause of heterogenicity. . By excluding this study from the meta-analysis, the SMD was −1.07, with 95% CI [−1.36, −0.78], and the I2 dropped to 78% (from 92%). For the BAI, the study by Depi 1988 was the possible cause of heterogenicity. By excluding this study from the meta-analysis, the SMD was −1.07, with 95% CI [−1.36, −0.78], and the I2 dropped to 93% (from 97%). We also conducted a meta-regression based on the duration of treatment in each study. The result for meta-regression was R-squared = 0, P value = .095 for uterine volume, R-squared = 0, P value = .128 for median ovarian volume, R-squared = 0, P value = .031 for FSH, R-squared = 0, P value = .196 for LH, R-squared = 0, P value = .099 for E2, R-squared = 20.62, P value = .019 for BAI. This suggests that the duration of treatment was one of the sources of heterogeneity among studies only in BAI but not in other variables. The graphs of sensitivity analysis and the bubble plot of meta-regression for BAI are available in supplementary file 1.
Discussion
The increasing prevalence of Central Precocious Puberty (CPP) has become a major concern, with contributing factors such as dietary patterns, changes in lifestyle, and the social environment. In China, Traditional Chinese Medicine (TCM) and Chinese Herbal Medicine (CHM) are commonly used in the treatment of CPP and other complications. 34
We performed a meta-analysis to investigate the effectiveness of CHM in the treatment of CPP in the available literature. The study included 17 studies with a total of 1590 patients. The results revealed that the combination of CHM and Western medicine (GnRH analogs) compared to WM significantly reduced the uterine and ovarian volume, suggesting that CHM could be an adjuvant therapy in treating patients with CPP. The experimental group also demonstrated a significant reduction in serum sex hormone levels (FSH, LH, and E2). Furthermore, the combination of CHM and Western medicine (WM) compared to WM significantly reduced patients’ bone age index. Our subgroup analysis showed that the combination GnRHa + Zhibai dihuang but not significant for GnRHa + Dabuyin pill was effective for reducing serum sexual hormones. While the combination of Zhibai dihuang and GnRHa was effective in reducing UV and OV, the combination of the Dabuyin pill and GnRHa was not effective. However, no significant improvements were observed in bone age index (BAI) for either formula, indicating that CHM may have limited effects on delaying skeletal maturation.
According to TCM principles, the fundamental pathogenesis of central precocious puberty aries from deficiencies in kidney yin, yin-yang imbalance, yin depletion, excessive fire, and the premature activation of “Tiangui.” 35 Rehmannia Pill, Zhibai dihuang, also known as Phellodendron, and Anemar-reemerged as the most prescribed herbal remedy for idiopathic precocious puberty in the articles reviewed. 36 Traditional Chinese medicine principles suggest that Zhibai dihuang can nourish yin and eliminate fire, similar to the effects of Dabuyin. The latter has been demonstrated to diminish the axis of hypothalamic-pituitary-gonadal by decreasing Kiss-1/GPR54 expression and inhibiting the production of GnRH in animal models’ hypothalamus. 37 Moreover, TCM herbal combinations were observed to impact hypothalamic kisspeptin expression, down-regulate elevated GnRH levels, and notably delay sexual development in animal experiments.38,39 Several Chinese studies have also suggested that ‘nourishing yin-purging fire’ herbal remedies can influence the axis of hypothalamic-pituitary-ovarian postpone skeletal maturity in patients with idiopathic precocious puberty.40,41 Although current research on Zhibai dihuang is lacking, our investigation proposes that this herbal formula holds promise for treating ICP and should be further explored in research and clinical trials. The XinYiSan, Zhibai dihuang, XiaoQingLongTang, GeGenTang, and CangErSan XinYiQingFeiTang, were identified as the top most frequently prescribed formulae for idiopathic precocious puberty. Research in network pharmacology has identified kaempferol, quercetin, β-sitosterol, and other compounds as the primary constituents of “Zhimu-Huangbai” that can modulate central precocious puberty. Quercetin, known for its diverse antioxidant and anti-inflammatory properties, is a flavonoid abundant in herbs, teas, and vegetables. 42 β-Sitosterol, a naturally occurring phytosterol found in various plant-based medicines, vegetable oils, and nuts has been shown to possess antioxidative, anti-inflammatory, and anticancer effects. 43 Huangbai, a medicinal herb, contains both quercetin and β-sitosterol as its active components. Research has indicated that quercetin can impede the proliferation of ovarian cells and the release of ovarian steroids and peptide hormones. Kaempferol, a natural compound commonly found in vegetables and herbs, exhibits numerous health benefits including antioxidant, anti-inflammatory, neuroprotective, cardioprotective, and antidiabetic activities. 44 The active constituent of kaempferol, Zhimu, has been found to inhibit the secretion of E2 when its levels become elevated in the body. 45 TCM theory suggests that the Dabuyin formula possesses the ability to nourish yin and remove fire (NYRF), similar to the effects of Zhibai dihuang. Chinese herbal mixtures with NYRF action decreased the expression of hypothalamic NKB/NK3R in the hypothalamus as well as kisspeptin protein in the arcuate and periventricular nucleus, and preoptic area. This down-regulation resulted in reduced GnRH expression and significantly delayed the development of primary sexual characteristics in animal studies. 46
Our finding is concordant with a previous study on 3390 patients with CPP. A current study showed that patients treated with TCM agents had higher predictive height and lower bone maturation rates compared to the control group. 47 However, this study used more than 20 herbal remedies or formulas and the effect of each formula on the outcomes of patients was not determined. Our study showed that although CHM is effective for BAI, the Zhibai dihuang and Dabuyin pill were not efficient, and other CHM might improve BAI in CPP patients. Another randomized control trial demonstrated the sole usage (without combination with GnRH analogs) of ZiYin Xiehuo and Zishen Qinggan granules on primary sexual traits such as mammary nuclei and the size of the uterus and ovaries. 48 It is of note to say that other TCM methods such as acupuncture had a regulatory effect on the serum sex hormone levels and early development process and, subsequently could be an alternative in CPP management. 49
Other herbal formulas such as Mai-Ya, which is the most singly prescribed remedy for CPP based on the Gung Memorial Hospital registry are believed to harmonize the stomach and aid in digestion data and also alleviate CPP symptoms in traditional Chinese medicine theory. 47 However, it is important to note that a high dosage of Mai-Ya can have the side effects of stopping lactation and reducing breast swelling.50,51 Breast growth is typically the initial indicator of sexual maturation in girls during puberty.52,53 Therefore, the use of Mai-Ya is thought to halt the onset of puberty, specifically in terms of breast development. Cortex Phellodendri (Huang-Bai) has been found to delay puberty by suppressing GnRH synthesis in the hypothalamus, while also promoting growth through increased GH synthesis and secretion in the pituitary. 54 Although our study showed that Chinese herbal medicine and formulas are effective as adjuvant therapy in treating patients with CPP, other studies and meta-analyses are warranted to show the efficacy of other CHM formulas in the treatment of CPP patients. Totally there was no serious reported adverse effect among included studies, suggesting that CHM might be a safe therapeutic strategy. Nevertheless, more investigation is warranted in to find potential adverse effect of CHM.
Limitations
Our review had some limitations. Although we attempted to obtain complete texts for all pertinent research, data from some studies remained inaccessible and were therefore excluded from our analysis.
Our analysis revealed substantial heterogeneity in some of the parameters we examined. To investigate this variability, we conducted subgroup meta-analyses, but heterogeneity persisted in certain subgroups. The unfound source of heterogenicity could be the difference in the methodology of included studies. For instance, included studies did not mention in which the stage (luteal or follicular) or level (basal or peak) of the serum sex hormones were measured. This could be the source of heterogeneity and future investigations should consider the methodology of studies.
Moreover, many of the studies did not report long-term outcomes, such as the impact on adult height or future reproductive health.
Conclusion
In conclusion, Chinese Herbal Medicine is a promising adjuvant therapy in combination with Western medicine for treating Central Precocious Puberty (CPP). CHM offers potential efficacy in managing primary sexual characteristics, serum sex hormone levels, and bone age index. However, further large-scale, high-quality clinical trials are needed to confirm the long-term effectiveness and safety of CHM for CPP treatment.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251323772 - Supplemental material for Efficacy of Chinese Herbal Medicine as an Adjuvant Therapy in Treating Central Precocious Puberty: A Systematic Review and Meta-Analysis of Randomized Control Trials
Supplemental material, sj-docx-1-npx-10.1177_1934578X251323772 for Efficacy of Chinese Herbal Medicine as an Adjuvant Therapy in Treating Central Precocious Puberty: A Systematic Review and Meta-Analysis of Randomized Control Trials by Huiming Cheng and Xin Wan in Natural Product Communications
Footnotes
Authors contribution
HM: Conceptualization, Supervision, Writing, Review and Editing, Methodology
XW: Writing, Review and Editing, Methodology
Data Availability
All data are available from the corresponding author
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
