Abstract
Background and Purpose
Platycodon grandiflorum is a traditional Chinese herbal medicine, which can disperse lung, promote pharynx, dispel phlegm and expel pus. Its processing history was first seen in the “Zhou Hou Bei Ji Fang” of the Jin Dynasty in China. Through processing, effective substances in plants can be enriched and negative substances can be reduced. Therefore, this study explored the effects of different processing methods on the antitussive and expectorant effects of Platycodon grandiflorum through metabolomics and pharmacology, aiming to provide a theoretical basis for clinical application.
Experimental Approach
The changes of metabolites before and after processing of Platycodon grandiflorum were analyzed by Nexera UHPLC LC-30A-HILIC-Orbitrap-Q Exactive HF-X platform. The antitussive and expectorant effects of processed Platycodon grandiflorum were verified by mouse concentrated ammonia induced cough method and tracheal phenol red drainage method.
Key Results
After processing, some metabolites of Platycodon grandiflorum changed, among which 24 kinds of terpenoid metabolites changed significantly. Through the experiment of ammonia-induced cough in mice and phenol red expectorant experiment, it was found that fresh JZI 40%, fresh CZ 80%, dry JZZ 40% and dry JZI 80% had obvious antitussive effect, and dry JZZ 40% had obvious expectorant effect.
Conclusions and Implications
Some processing methods can indeed further improve the antitussive and expectorant effects of Platycodon grandiflorum, which may be due to the effect of increasing the content of triterpenoids after processing, but the mechanism of processing to improve the effect of relieving cough and expectorant needs further study.
Introduction
Platycodon grandiflorum is the dried root of Platycodon grandiflorum (Jacq.) A.DC., which is a traditional Chinese herbal medicine. It was first recorded in Shennong herbal Scripture. 1 It can disperse lung, relieve sore throat, eliminate phlegm and expel pus. Platycodon grandiflorum is mainly distributed in northeast China, and is also planted in Korea and Japan, which can be used as medicine and food. Platycodon grandiflorum contains a variety of active ingredients, including saponins, flavonoids, polyphenols, polysaccharides, etc. 2 Triterpenoid saponins are one of the main chemical components of Platycodon grandiflorum. Among more than 80 different Platycodon grandiflorum saponins, 3 Platycodon grandiflorum saponins A, Platycodon grandiflorum saponins D and Platycodon grandiflorum saponins D3 are three compounds with higher activity in Platycodon grandiflorum saponins. 4 Platycodin D is an index component for the identification of Platycodon grandiflorum medicinal materials and decoction pieces recorded in the 2020 edition of “Chinese Pharmacopoeia” (Part I). 5 It has antitussive and expectorant,6,7 anti-tumor,8,9 anti-inflammatory, 10 anti-oxidation, 11 immune regulation 12 and other effects.
The processing of traditional Chinese medicine is a unique traditional pharmaceutical technology in China. 13 It can enrich the effective substances in plants and reduce the negative substances. 14 Platycodon grandiflorum processed products have a long history. It was first seen in the ‘Zhouhou Beiji Fang’ in the Jin Dynasty, including “Jing Zhi” (JZ, impurities removing), “Qie Zhi”(QZ, cut into a certain specification of pieces, silk, blocks, segments, etc), “Chao Zhi”(CZ,put into a pan and stir fry), “Bai He Zhi”(BHZ,fry with fresh lily juice), “Jiang Zhi Zhi”(JZZ, fry with ginger juice), “Mi Zhi”(MZ,stir-fry with honey), “Mi Gan Shui Zhi”(MGZ, fry with rice swill), “Fu Chao”(FC, fry with wheat bran), “Jiu Zhi”(JZI, fry with yellow wine) and “Cu Zhi”(CZ, fry with rice vinegar) and other processing methods, among which JZ, QZ and MZ are still used in modern times.
The results of the study on the effect of different processing methods on the content of platycodin D in the early stage of this project showed that 15 different processing methods and different excipient concentrations would affect the content of platycodin D. In addition, references show that platycodin D can have a good intervention effect on cough and expectoration through a rich regulatory mechanism.16,17 However, the effects of different processing methods on the metabolites of Platycodon grandiflorum are not clear. Therefore, in this study, Nexera UHPLC LC-30A-HILIC-Orbitrap-Q Exactive HF-X platform was used to preliminarily analyze the changes of metabolites before and after processing of Platycodon grandiflorum, and the differential metabolites between groups were screened and analyzed. In addition, the effects of different processing methods of Platycodon grandiflorumon on the antitussive and expectorant were further verified by pharmacological experiments. This can not only broaden the basic research on the components of Platycodon grandiflorum processed products, but also provide a theoretical basis for the development and utilization of Platycodon grandiflorum processed products.
Results
The Changes of Metabolites Before and After Processing of Platycodon grandiflorum
Based on Nexera UHPLC LC-30A-HILIC-Orbitrap-Q Exactive HF-X platform, the changes of metabolic components of Platycodon grandiflorum before and after processing were analyzed. In the positive and negative ion modes, the differences between the six groups of G1 and G4, G2 and G4, G3 and G4, X1 and X4, X2 and X4, X3 and X4 were analyzed.
As shown in Table 1, compared with G1 and G4, the positive ion mode (POS) was up-regulated by 526, down-regulated by 468, and there was no difference of 2983 ; negative ion mode (NEG) was up-regulated by 321, down-regulated by 298, and there was no difference of 2068. Compared with G2 and G4, POS was up-regulated by 562, down-regulated by 398, and there was no difference of 3017 ; NEG was up-regulated by 342, down-regulated by 254, and there was no difference in 2094. Compared with G3 and G4, POS was up-regulated by 415, down-regulated by 363, and there was no difference of 3199 ; NEG was up-regulated by 250, down-regulated by 254, and there was no difference in 2184. Compared with X1 and X4, POS was up-regulated by 688 and down-regulated by 582, with no difference of 2677 ; NEG was up-regulated by 512, down-regulated by 319, and there was no difference in 1848. Compared with X2 and X4, POS was up-regulated by 776, down-regulated by 739, and there was no difference of 2448 ; NEG was up-regulated by 538, down-regulated by 417, and there was no difference in 1702. Compared with X3 and X4, POS was up-regulated by 671, down-regulated by 529, and there was no difference of 2737 ; NEG was up-regulated by 511, down-regulated by 325, and there was no difference in 1840.
Comparison Analysis and Statistics of Differences Between Groups.

Changes in the Content of Metabolites Between Groups
According to the classification level of natural products for metabolites (Pathway, super class, class), the percentage of metabolites in the samples was counted, and the stacking histogram was used for visualization to visualize the difference in metabolite composition results. As shown in Figure 1A, The metabolites of different groups mainly included Alkaloids, Amino acids and Peptides, Carbohydrates, Fatty acids, Polyketides, Shikimates and carbohydrates Phenylpropanoids, Terpenoids, etc In order to more directly observe the expression trend of different metabolites, a chord analysis was performed for different metabolites in each group (Figure 1B). In the diagram, the outermost circle is the expression calorimetric map of differential metabolites, the second circle is the classification of differential metabolites, and the line colors represent the correlation coefficients of metabolites at corresponding positions. The results showed that there were significant differences in metabolites among all groups.

Changes in the content of metabolites between groups.
Because the terpenoids in the metabolites are the main active components of Platycodon grandiflorum and have good antitussive and expectorant effects, the changes of terpenoids were mainly analyzed. Through the analysis of differential metabolites between G1 and G4 groups, it was found that there were 481 terpenoid metabolites with significant differences, including 63 triterpenoid metabolites, 52 significantly increased and 11 significantly decreased. Through the analysis of differential metabolites between G2 and G4 groups, it was found that there were 444 terpenoid metabolites with significant differences, including 83 triterpenoid metabolites, 76 significantly increased and 7 significantly decreased. Through the analysis of differential metabolites between G3 and G4 groups, it was found that there were 330 terpenoid metabolites with significant differences, including 46 triterpenoid metabolites, 43 significantly increased and 3 significantly decreased. Through the analysis of differential metabolites between X1 and X4 groups, it was found that there were 512 terpenoid metabolites with significant differences, including 76 triterpenoid metabolites, 47 significantly increased and 29 significantly decreased. Through the analysis of differential metabolites between X2 and X4 groups, it was found that there were 608 terpenoids metabolites with significant differences, of which 99 triterpenoid metabolites were found, with a significant increase of 64 and a significant decrease of 35. Through the analysis of differential metabolites between X3 and X4 groups, it was found that there were 474 terpenoid metabolites with significant differences, including 86 triterpenoid metabolites, which increased significantly by 63 and decreased significantly by 23.
PCA
Principal Component Analysis (PCA) is an unsupervised data analysis method. 18 In data analysis, PCA method is generally used to observe the overall distribution trend of samples between groups and the difference between samples between groups. In Figure 2, PCA1 represents principal component 1, PCA2 represents principal component 2, and ellipse represents 95% confidence interval. The points of the same color represent the biological repetitions in the group, and the distribution of the points reflects the degree of difference between groups and within groups.
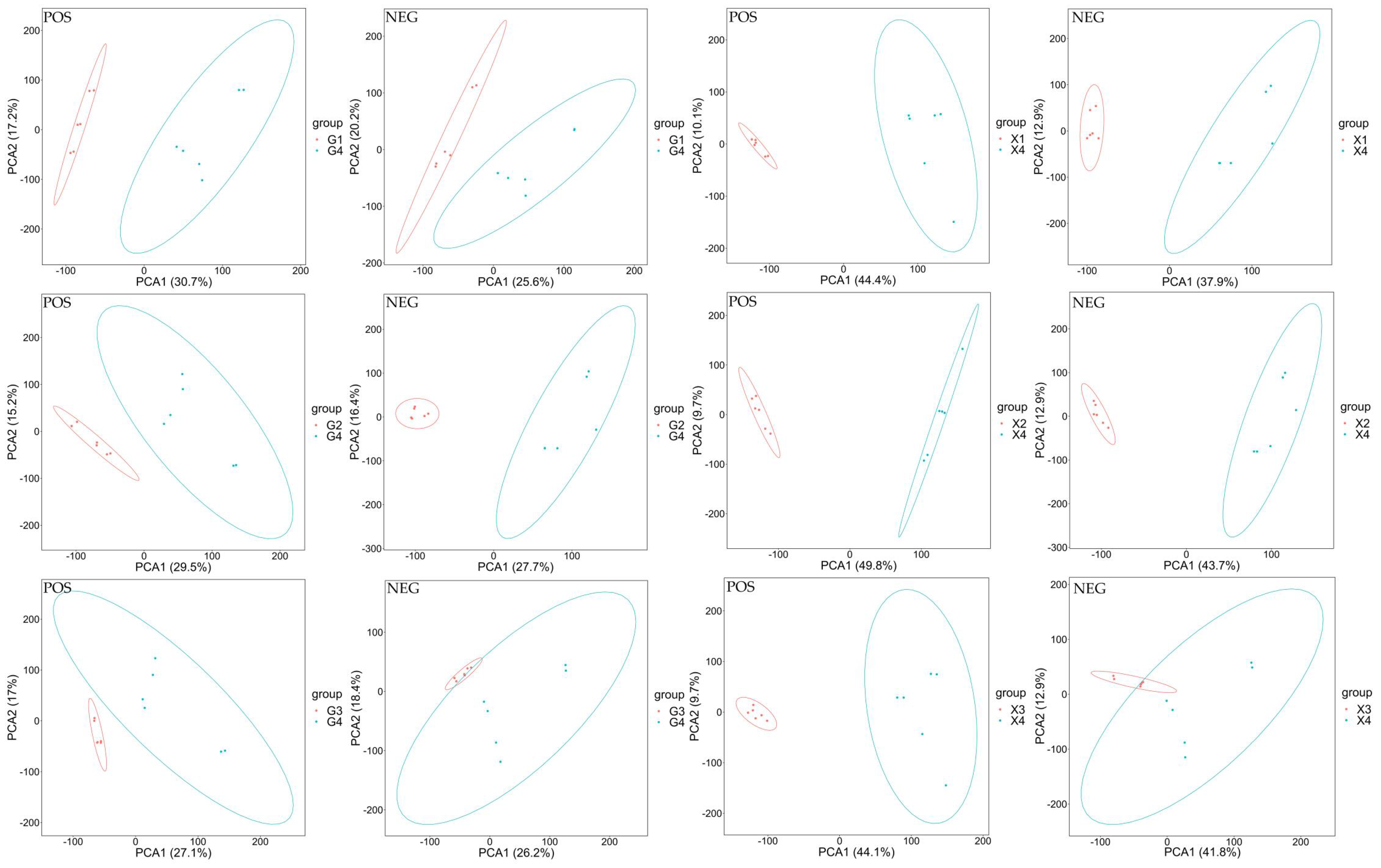
PCA results among groups. Note: In the figure, PCA1 represents principal component 1, PCA2 represents principal component 2, and the ellipse represents the 95% confidence interval. The dots of the same color represent each biological duplication within the group, and the distribution of the dots reflects the degree of difference between and within the group.
From Figure 2, it can be seen that in the comparison of metabolites in each group, the separation effect between G1 and G4, X1 and X4, X2 and X4, X3 and X4 experimental groups is better, and the separation effect of POS samples in G2 and G4, G3 and G4 experimental groups is better. It shows that different processing techniques do have an impact on metabolites.
PLS-DA
Partial Least Squares Discrimination Analysis (PLS-DA) is a supervised pattern recognition method based on partial least squares method. PLS-DA projects the predictive variables and the observed variables into a new space by projection. In the PLS-DA score map, if the point clouds of samples in different groups are distributed in different regions, it shows that the PLS-DA model has a better discriminant effect, and there should be metabolites with significant differences between groups.
Cross Validation is used to obtain the model evaluation parameters (R2Y, Q2), where R2Y is the model’s interpretation of the categorical variable Y, and Q2 is the model’s predictability. Generally, when Q2 is greater than 0.5, the model is stable and reliable, 0.3 < Q2 ≤ 0.5, indicating that the model is stable, and Q2 < 0.3, indicating that the model is less reliable. 19 The Q2 of this experiment is greater than 0.5, indicating that the model is stable and reliable.
From Figure 3, it can be seen that the point clouds of samples in different groups are distributed in different regions, indicating that the PLS-DA model has a better discriminant effect. There are significant differences in metabolites between groups, and the results of metabolite data are reliable.

PLS-DA results and cross validation results between groups.
OPLS-DA
PLS-DA uses all components of metabolomics data for prediction, which often leads to serious over-fitting. Orthogonal Partial Least Squares Discriminant Analysis (OPLS-DA) is an analysis method that modifies PLS-DA, 20 which can filter out irrelevant information, improve the analytical ability of the model, and further improve the effectiveness of the model. In the OPLS-DA score plot, t1 reflects the difference between groups, and to1 reflects the variation within the group.
In order to avoid overfitting during the modeling process, Permutation Test is used to test the model. 21 The permutation test establishes the corresponding OPLS-DA model several times by randomly changing the order of the categorical variable Y to obtain the R2Y and Q2 values of the random model, which plays an important role in avoiding the over-fitting of the test model and evaluating the statistical significance of the model. 22 As the replacement retention gradually decreases, the proportion of the Y variable of the replacement increases, and the Q2 of the random model gradually decreases. It can be explained that the model has good robustness and there is no over-fitting phenomenon.
It can be seen from Figure 4 that the point clouds of the samples in different groups are distributed in different regions, indicating that the OPLS-DA model has a good discriminant effect, and there are significant differences in metabolites between the groups.

OPLS-DA results and permutation test results between groups. Note: Horizontal coordinates indicate the replacement retention of the replacement test (the proportion of the Y variable in the same order as the original model, the point where the replacement retention is equal to 1 is the R2Y and Q2 value of the original model), and vertical coordinates indicate the value of R2Y or Q2, the green dots indicate the R2Y value obtained from the replacement test, and the blue square dots indicate the Q2 value obtained from the replacement test.
Pharmacological Experiment
Analysis of Antitussive Effect of Different Processed Products of Platycodon grandiflorum on Mice
The results of Table 2 showed that the cough latency of fresh JZI 40% group was (22.90 ± 11.704) s, which was significantly longer than that of fresh SP group (14.60 ± 4.600) s (P < .05). The number of coughs in the fresh CZ 80% group was (40.30 ± 16.667) times in 2 min, which was less than that in the fresh SP group (48.50 ± 8.923) times (P < .05), with significant difference.
Comparative Analysis of Cough Incubation Period and Cough Frequency in Each Group.
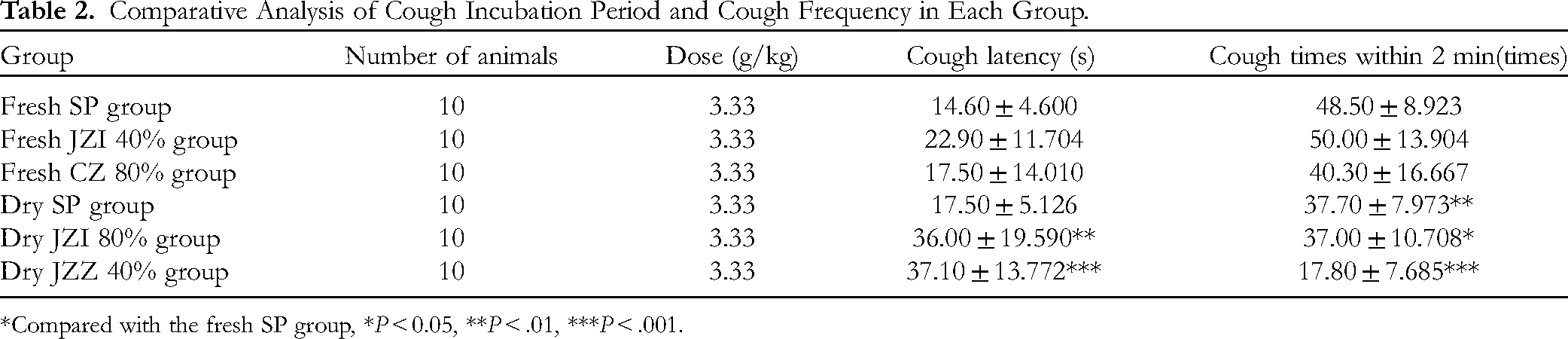
*Compared with the fresh SP group, *P < 0.05, **P < .01, ***P < .001.
The cough latency of mice in the dry JZI 80% group was (36.00 ± 19.590) s, and the cough latency of mice in the dry JZZ 40% group was (37.10 ± 13.772) s. There was no significant difference between the two groups and both were significantly longer than the cough latency of mice in the dry SP group (17.50 ± 5.126) s (P < .05). The number of coughs in the dry JZZ 40% group was (17.80 ± 7.685) times within 2 min, which was significantly less than that in the dry SP group (37.70 ± 7.973) times (P < .05).
It can be seen that fresh Platycodon grandiflorum group, fresh JZI 40% group and fresh CZ 80% group have a certain effect on relieving cough in mice ; in the dry Platycodon grandiflorum group, the dry JZZ 40% group had the best antitussive effect, and the dry JZI 80% group had the second best antitussive effect.
Analysis of Expectorant Effect of Different Processed Products of Platycodon grandiflorum on Mice
The results in Table 3 showed that the tracheal phenol red excretion in the fresh JZI 40% group and the fresh CZ 80% group was higher than that in the fresh SP group, but there was no significant difference between the fresh JZI 40% group and the fresh CZ 80% group. The secretion of phenol red in the trachea of mice in the JZZ 40% group was significantly higher than that in the SP group (P < .05), with significant difference.
Comparative Analysis of Phenol red Excretion in trachea of Mice in Each Group.

*Compared with the fresh SP group, *P < 0.05, **P < .01, ***P < .001.
It can be seen that in the fresh Platycodon grandiflorum group, there was no significant difference between the fresh JZI 40% group and the fresh CZ 80% group and the fresh SP group. In the dry Platycodon grandiflorum group, the dry JZZ 40% group had the most obvious expectorant effect.
Materials and Methods
Sample Treatment
Fresh platycodon grandiflorum was produced in the Chinese herbal medicine market in Tonghua area, Jilin Province, China. Dried platycodon was purchased from Hongjian Pharmacy Co., LD, Jilin Province, China. The Platycodon grandiflorum was divided into dry Platycodon grandiflorum group and fresh Platycodon grandiflorum group. Each group was processed by four processing techniques : JZZ, JZI, CZ and BHZ. The excipients of each processing technology were prepared with distilled water into a solution containing 20%, 40%, 60% and 80% of the excipients. Add an appropriate amount of Platycodon grandiflorum tablets, fully stirred, moistened for 5 h, dried at 80 °C for 5 h, and taken out to cool. Through the experimental results of the previous study of the subject, it was found that the dry JZZ40% group, the dry JZI80% group, the dry BHZ80% group, the fresh JZI40% group, the fresh CZ80% group and the fresh JZZ40% group had a significant effect on the content of platycodon saponin D.Therefore, this study selected these 6 groups and used “Sheng Pin” (SP, unprocessed platycodon grandiflorum) as a blank control group for experiments. Dry JZZ 40% group, dry JZI 80% group, dry BHZ 80% group, dry blank group, fresh JZI 40% group, fresh CZ80% group, fresh JZZ 40% group and fresh blank group were named G1, G2, G3, G4, X1, X2, X3, X4, respectively. Six biological replicates in each group were named 1-1, 1-2, 2-1, 2-2, 3-1, 3-2.
Non-target Metabolome Analysis
Sample Information
The powder of platycodon platycodon was crushed and stored at −20 °C. After thawing at 4 °C, a certain amount of sample was weighed and added with 4 times the volume of pre-cooled methanol / acetonitrile / water solution (2 : 2 : 1), vortexed for 30 s, placed at-20 °C for 1 h to precipitate protein, 14000 g, centrifuged at 4 °C for 10 min, the supernatant was taken, dried at room temperature, and stored at-80 °C.
Chromatographic Condition
Positive ion mode (POS) : mobile phase A : 10 mM ammonium acetate, acetonitrile : water 95 : 5, .1% formic acid ; mobile phase B : 10 mM ammonium acetate, acetonitrile : water 50 : 50, 0.1% formic acid ; negative ion mode (NEG) : mobile phase A : 10 mM ammonium acetate, acetonitrile : water 95 : 5 ; adjust the pH to 8.0 with ammonia. Mobile phase B : 10 mM ammonium acetate, acetonitrile : water 50 : 50 ; adjust the pH to 8.0 with ammonia. The samples were separated by Nexera UHPLC LC-30A ultra-high performance liquid phase system with micro-liter flow rate. The chromatographic column was first balanced with 98% mobile phase A, and then the sample was transported by an automatic sampler to the HILIC column (Waters, ACQUITY UPLC BEH Amide 1.7 μm, 2.1 × 100 mm) at a flow rate of 0.3 mL/min.
Mass Spectrometer Conditions
The samples were separated by analytical column and analyzed by Q Exactive HF-X mass spectrometer. POS: parent ion scanning range of 70-1050 m/z, MS resolution of 120,000, AGC (Automatic gain control) target of 3 × 106, Maximum IT of 100 ms. The resolution of secondary mass spectrometry was 7,500, AGC (Automatic gain control) target was 2 × 105, Maximum IT was 50 ms, HCD fragmentation mode was used, Normalized Collision Energy was 20,40,60, Isolation window was 1.5 m/z, and ion scanning range was 200-2000 m/z. NEG : precursor ion scanning range 70-1050 m/z, MS resolution of 120,000, AGC (Automatic gain control) target of 3 × 106, Maximum IT of 200 ms. The resolution of secondary mass spectrometry was 7,500, AGC (Automatic gain control) target was 2 × 105, Maximum IT was 50 ms, HCD fragmentation mode was used, Normalized Collision Energy was 20,40,60, Isolation window was 1.5 m/z, and ion scanning range was 200-2000 m/z.
Pharmacological Experiment
Experimental Animal
Sixty healthy SPF ICR mice, half male and half female, weighing 18.0-20.0 g, were provided by China Liaoning Changsheng Biotechnology Co., Ltd [ Production License No. : SCXK- (Liaoning) 2020-0001 ]. Routine feeding, 12 / 12 h of light and dark cycle, free access to food and drinking water, feeding environment temperature is 22 ∼ 25 °C, relative humidity is (55 ± 5)%.
Administration Dose
According to the 2020 edition of Chinese Pharmacopoeia, the daily human dosage of platycodon platycodon is 3∼10 g/d. In this study, the dosage of mice was 20 times of the recommended human dosage, that is, 10 g crude drug /60 kg body weight ×20 times =3.33 g/kg, which was prepared into 16.65 g/100 mL and administered by gavage at 0.2 mL/10 g body weight.
Antitussive Experiment
Sixty mice were divided into fresh SP group, fresh JZI 40% group, fresh CZ 80% group, dry SP group, dry JZI 80% group and dry JZZ 40% group according to body weight, with 10 mice in each group. The mice in each group were given intragastric administration once a day for 14 days. One hour after the last administration, the mice were placed in an inverted transparent glass beaker, placed in a cotton ball of concentrated ammonia water and quickly began to time. The cough latency of the mice after the cotton ball was placed and the number of coughs within 2 min after the cotton ball was taken out were observed and recorded. When the test is carried out, the room temperature should be above 20 °C, and the beaker should be odoured with ammonia if it is reused.
Expectorant Experiment
The standard phenol red curve was drawn : A certain amount of phenol red was accurately weighed and dissolved in 5% NaHCO3 solution to prepare a solution with a concentration of 100 ug/mL, and then diluted to 1, 2, 3, 4, 5, 6, 7 ug per milliliter. The absorbance at 546 nm was measured to be 0.0878,0.2043,0.3208,0.4373,0.5538,0.6703,0.7868, respectively. The standard curve was made with phenol red dose (C) as abscissa and OD value as ordinate. The regression equation was obtained : y = 0.1165x-0.0287(R2 = 0.9989).
Sixty mice were divided into fresh SP group, fresh JZI 40% group, fresh CZ 80% group, dry SP group, dry JZI 80% group and dry JZZ 40% group according to body weight, with 10 mice in each group. The mice in each group were given intragastric administration once a day for 14 days. Using tracheal phenol red excretion method, 30 min after the last administration, intraperitoneal injection of 5% phenol red saline solution 0.1 mL/10 g body weight. Half an hour after the injection of phenol red, the mice were sacrificed by cervical dislocation, the tissue around the trachea was removed, and a section of the trachea from the thyroid cartilage to the tracheal branch was cut. Each tracheal segment was placed in a test tube containing 1.5 mL of 5% NaHCO3 solution in advance, and then placed on a vortex mixer for 2 min to shake, so that the phenol red in the tracheal segment was completely released. Before the test, 0.1 mL of 1 mol/L NaOH solution was added to the test tube, centrifuged (3000 rpm, 5 min), and then the solution in the test tube was measured at 546 nm of the spectrophotometer OD value, and the phenol red secretion was calculated according to the standard curve.
Data Processing
The raw data (RAW files) collected by mass spectrometry analysis were subjected to database search by Progenesis_QI software to finally obtain the identification information of the samples. The quantitative information of the target metabolite set is normalized (z-score). Then, using the pheatmap R package, both sample and protein expression dimensions were categorized (distance algorithm: Euclidean, linkage: Average linkage) and a hierarchical clustering heatmap was generated.
Limitations of the Study
This study mainly discussed the effect of different processing techniques of platycodon grandiflorum on the antitussive and expectorant effects of platycodon grandiflorum. In order to identify the chemical component with the highest antitussive and antisputum activity and clarify its possible mechanism of action, we can further explore its molecular mechanism by combining network pharmacology and molecular docking, so as to better guide the development and utilization of orange products.
Discussion
Platycodon grandiflorum is one of the commonly used antitussive, 23 expectorant and antiasthmatic traditional Chinese medicines in clinic. Platycodon deapioside D can exert antitussive and expectorant activity by regulating the same metabolic pathway, and has a good intervention effect on cough and sputum with complex etiology. 24 Through processing, it can not only enrich the effective substances, but also reduce the toxicity and side effects, and further enhance its efficacy while ensuring the safety and effectiveness of clinical medication. In this study, the effects of different processing techniques on the antitussive and expectorant effects of Platycodon grandiflorum were preliminarily explored through metabolomics and pharmacological design.
The terpenoids in the metabolites are the main active components of Platycodon grandiflorum, and the marker component Platycodin D is a triterpenoid component. Therefore, the changes of terpenoids were analyzed. The results of metabonomics showed that the triterpenoids in each group had obvious changes. Through PCA analysis, it was found that G3 and X3 did not show good differences in negative ion mode, and in the comparison of triterpene components between groups, the log2FC value was smaller than that of other groups, indicating that the difference between groups was small. Therefore, in the subsequent pharmacological experiments, only the other four processing methods were studied.
Through pharmacological experiments, it was found that the antitussive effect of fresh JZI 40%, fresh CZ 80%, dry JZZ 40% and dry JZI 80% was significantly improved, and the expectorant effect of dry JZZ 40% was significantly improved. Triterpenoids are the main active ingredients of Platycodon grandiflorum for relieving cough and relieving sputum. We have previously done relevant research on the effect of different processing methods on the content of total saponins in Platycodon grandiflorum, and found that processing can change the content of total saponins in Platycodon grandiflorum. Combined with the results of this study, it may be due to the fact that the four processing methods of fresh JZI 40%, fresh CZ 80%, dry JZZ 40% and dry JZI 80% increase the content of triterpenoids, thus promoting the effect of antitussive and expectorant. In addition, the excipients used in this study, such as yellow wine, ginger juice and rice vinegar, also had certain anti-inflammatory, analgesic and antitussive effects, which further enhanced the pharmacological activity of Platycodon grandiflorum.
Conclusions
In this study, different metabolites of platycodon grandiflorum under different processing processes were identified by non-target metabolomics. Pharmacological experiments verified that there were significant differences in some active components of platycodon grandiflorum after processing. Triterpenoids, as the main active components of platycodon grandiflorum, showed significant differences through different processing processes. It can be processed to promote the antitussive and expectorant effect of platycodon grandiflorum. This provides a theoretical basis for the development and utilization of platycodon grandiflorum.
Footnotes
Author Contributions
SZ: writing-original draft, writing-reviewing and editing, formal analysis, software, visualization; JX: methodology, conceptualization; MT: software, visualization; WH: methodology, conceptualization, supervision.
Data Availability
The experimental data can be seen in the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Experimental Animal Management and Welfare Ethics Committee of Institute of Specialty, Chinese Academy of Agricultural Sciences.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Jilin Provincial Scientific and Technological Development Program, grant number 20210204059YY.
Informed Consent Statement
Not applicable.
Statement of Human and Animal Rights
All animal-related experiments in the present study were performed in accordance with the guidelines of the Animal Care and Use Committee of Institute of Special Economic Animal and Plant Sciences (Permit Number: ISAPASAEC-2022-60B). The animals were kept in the Animal Room (SPF grade) of Jilin Academy of Chinese Medicine. The experimental animal use license number is SYXK (Ji) 2022-0004.
Statement of Informed Consent
There are no human subjects in this article and informed consent isnot applicable.
