Abstract
This review systematically explores the rapidly developing green nanotechnology field of herbal mediated synthesis of nanomaterials, with a focus on the latest research progress, current key challenges, and future development directions. The synthesis of nanomaterials using plant extracts provides a sustainable alternative to traditional high-energy consumption and environmentally harmful physical and chemical methods, simplifying process conditions and endowing products with good biocompatibility and potential functionality. The article elaborates on the basic principles of herbal synthesis, deeply analyzes the mechanisms of phytochemicals such as polyphenols, flavonoids, and terpenes in metal reduction and nanoparticle stabilization, and summarizes the synthesis strategies and characteristics of different types of nanomaterials. However, herbal mediated synthesis still faces several core challenges, including reproducibility differences caused by the complex composition of plant extracts, difficulties in scaling up the synthesis process, and limitations in controlling the size and morphology of nanoparticles. Future research should focus on the following directions: developing new plant sources with controllable components, analyzing the molecular mechanisms of plant chemical reactions with metal ions, improving the structural consistency and functional stability of products, and establishing standardized and industrializable synthesis systems.
Introduction
Nanomaterials have emerged as a cornerstone of modern scientific and technological advancements, revolutionizing fields ranging from electronics to medicine. These materials, typically defined as having at least one dimension under 100 nanometers, exhibit unique physicochemical properties that differ significantly from their bulk counterparts. 1 The distinctive characteristics of nanomaterials, including their high surface area-to-volume ratio, quantum confinement effects, and enhanced reactivity, have opened up a myriad of possibilities for novel applications and improvements in existing technologies. The importance of nanomaterials in contemporary research and industrial applications cannot be overstated. They have found uses in diverse areas such as drug delivery systems, advanced sensors, catalysts, energy storage devices, and environmental remediation technologies. 2 Nanomaterials have the potential to address some of the most pressing challenges facing humanity, including healthcare, energy efficiency, and environmental sustainability. 3 As such, the development of efficient and sustainable methods for nanomaterial synthesis has become a critical area of research. Nanomaterials have become an important cornerstone of modern technological progress, widely and profoundly changing the technological landscape in multiple fields from electronic engineering to biomedical science. Traditionally, nanomaterials are defined as materials with at least one dimension less than 100 nanometers. However, with the continuous deepening of research and the expansion of application scope, many systems with sizes between 200 and 300 nanometers have also been widely included in the field of nanomaterials due to their exhibited nanoscale effects, such as surface effects and quantum confinement behavior. Therefore, the definition of nanomaterials has gradually expanded from strict geometric scale standards to composite identification that combines size and functional characteristics.
Conventional methods for nanomaterial synthesis, while effective, often come with significant drawbacks. Physical and chemical approaches typically rely on high-energy inputs, extreme conditions, or toxic chemicals. These methods can be energy-intensive, environmentally harmful, and potentially dangerous to human health. 4 Moreover, the nanomaterials produced through these conventional routes may have limited biocompatibility, restricting their use in biological and medical applications. The need for safer, more sustainable, and biocompatible synthesis methods has driven researchers to explore alternative approaches. 5 In recent years, green synthesis methods have gained considerable attention as a promising alternative to conventional nanomaterial synthesis techniques. These approaches utilize biological entities or their products to facilitate the formation of nanomaterials under mild conditions. 6 Among the various green synthesis strategies, herbal-mediated synthesis has emerged as a particularly attractive option. 7 This method harnesses the power of plant extracts to reduce metal ions and stabilize the resulting nanoparticles, offering a simple, cost-effective, and environmentally friendly route to nanomaterial production. Figure 1 shows the typical route of synthesis of nanoparticles from plant source. 8

Synthesis of Nanoparticles from Plant Source. 8
With the rise of green and sustainable preparation concepts, herbal mediated nano material synthesis technology has become a research hotspot due to its advantages of environmental friendliness, low cost, and simple process. This method utilizes natural phytochemicals (such as polyphenols, flavonoids, terpenes, etc) in plant extracts to achieve metal ion reduction and nanoparticle stabilization under mild conditions. It has good biocompatibility and potential functionality, and is suitable for multiple fields such as medicine, sensing, and environment. 9
Although herbal mediated synthesis has shown promising prospects, there are still many unresolved issues with this technology: the complex and diverse composition of different plant extracts leads to a lack of standardization and reproducibility in the synthesis process; The mechanism of action of plant chemical components in synthetic systems is not yet clear, and there is a lack of molecular level analysis of reduction and nucleation processes; The ability to control the morphology of synthesized products is insufficient, especially in terms of particle size, uniformity, and functional regulation; In practical applications such as biomedical and drug delivery, there is insufficient systematic research on their drug loading capacity, biological stability, and toxicological evaluation. Therefore, there is an urgent need for a systematic review and in-depth analysis of the principles of herbal mediated synthesis of nanomaterials, the mechanisms of action of phytochemicals, synthesis control parameters, structural characterization methods, and application performance, in order to lay a theoretical and practical foundation for standardization, controllability, and functional expansion in this field. This article aims to comprehensively review the research progress of herbal mediated nano synthesis, explore its basic mechanisms, synthesis processes, characterization methods, and application scenarios, and propose future research directions based on the current problems, in order to promote the sustainable development of green nanotechnology in practical fields. 10
Principles of Herbal-Mediated Nanomaterial Synthesis
The herbal-mediated synthesis of nanomaterials represents a fascinating intersection of plant biology and materials science. This approach harnesses the rich biochemical repertoire of plants to facilitate the formation and stabilization of nanostructures. At the heart of this process are the diverse phytochemicals present in plant extracts, which play crucial roles in the reduction of metal ions and the subsequent stabilization of the formed nanoparticles. Among the key phytochemicals involved in herbal-mediated nanomaterial synthesis, polyphenols stand out as particularly important. 11 These compounds, characterized by multiple phenol structural units, are ubiquitous in the plant kingdom and possess strong antioxidant properties. In the context of nanoparticle synthesis, polyphenols act as powerful reducing agents, capable of converting metal ions to their elemental form. 12 Common polyphenols such as gallic acid, tannic acid, and catechins have been extensively studied for their roles in the green synthesis of various metal nanoparticles.
Although herbal mediated green synthesis methods have received widespread attention in the past decade, the systematic comparative analysis of various green synthesis pathways in current literature is still insufficient. Most studies focus on experimental reports from a single plant source, lacking an analysis of the advantages and disadvantages between methods and questioning the repeatability and consistency of experiments and mechanisms. Table 1 provides an overview of the skeletal structures and functional roles of different plant chemicals:
Overview of the Skeletal Structure and Functional Roles of Different Plant Chemicals.

Table 2 is a comparative analysis of herbal-mediated synthesis and traditional nanomaterials synthesis methods:
Comparative Analysis of Herbal Mediated Synthesis and Traditional Nanomaterial Synthesis Methods.

Flavonoids, a subclass of polyphenolic compounds, also play a significant role in herbal-mediated nanomaterial synthesis. These compounds, which include quercetin, rutin, and kaempferol, are known for their ability to chelate metal ions and subsequently reduce them to form nanoparticles. 13 The unique molecular structure of flavonoids, featuring multiple hydroxyl groups, enables them to interact effectively with metal ions and stabilize the resulting nanostructures. 14
Terpenoids represent another important class of phytochemicals involved in the green synthesis of nanomaterials. These naturally occurring organic compounds, derived from isoprene units, exhibit a wide range of structures and functions in plants. In the context of nanoparticle synthesis, terpenoids can act as both reducing and capping agents (Figure 2). 15 Their diverse structures allow for varied interactions with metal ions and nanoparticle surfaces, contributing to the formation of stable nanostructures with different morphologies. 16
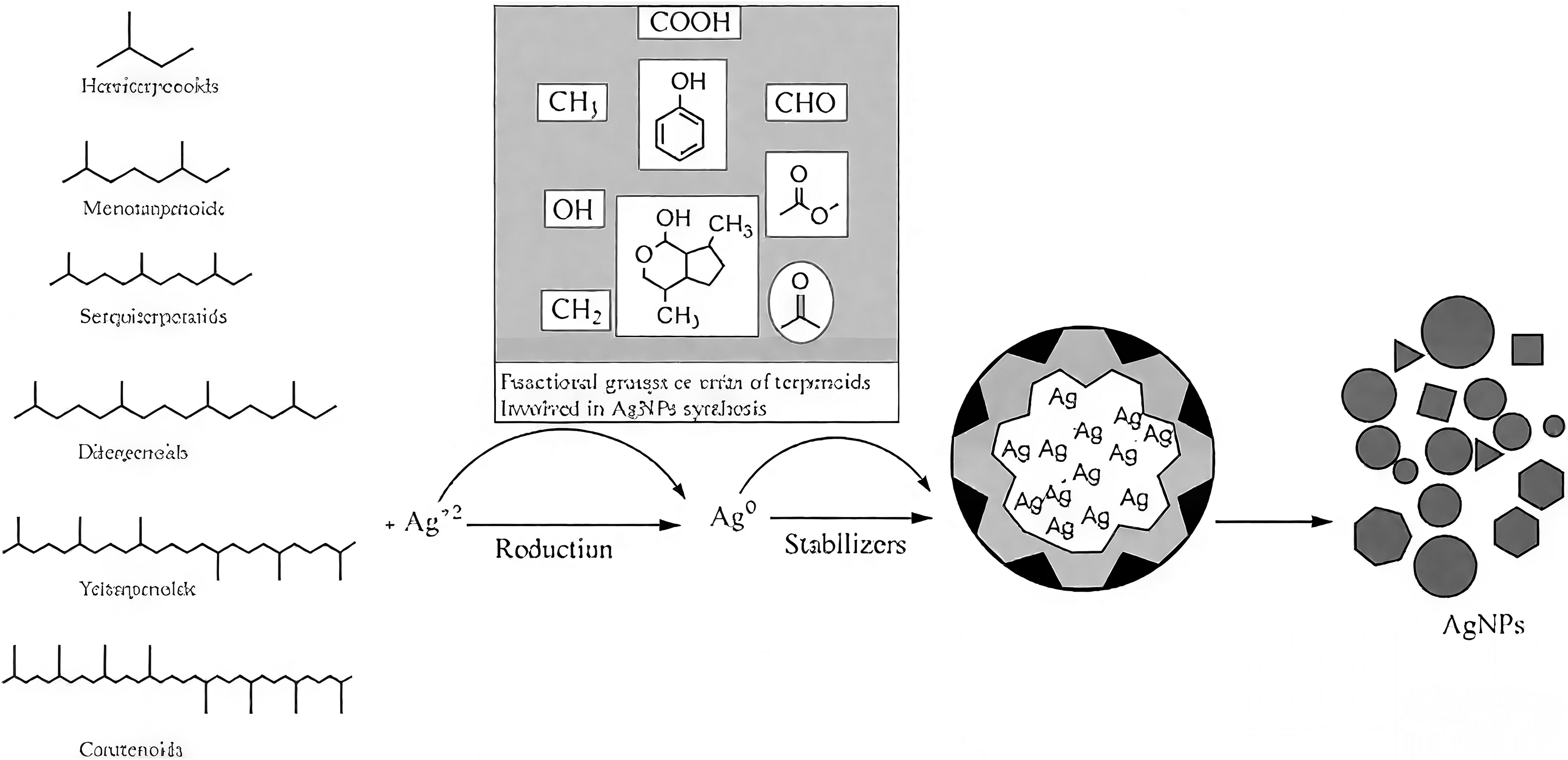
Functions of Terpenoids as Reducing and Capping Agents in the Synthesis of AgNPs. 16
The mechanisms of bioreduction and stabilization in herbal-mediated nanomaterial synthesis are complex and multifaceted. The process typically begins with the interaction between metal ions and the reducing phytochemicals present in the plant extract. 17 This interaction leads to the transfer of electrons from the phytochemicals to the metal ions, resulting in the reduction of the ions to their elemental state. 8 As this reduction occurs, the metal atoms begin to nucleate and grow into nanoparticles. Simultaneously with the reduction process, other plant biomolecules play a crucial role in stabilizing the forming nanoparticles. 18 Proteins and polysaccharides present in the plant extract can adsorb onto the surface of the growing nanoparticles, providing steric stabilization and preventing agglomeration. 19 This stabilization mechanism is critical for controlling the size and shape of the resulting nanostructures and ensuring their long-term stability in solution.
The bioreduction and stabilization processes in herbal-mediated synthesis are influenced by a variety of factors, which can significantly affect the characteristics of the resulting nanomaterials. pH plays a crucial role in the synthesis process, as it can influence the ionization state of the phytochemicals and thus their reducing capacity. 20 Generally, a slightly alkaline pH is favorable for the reduction of metal ions, but the optimal pH can vary depending on the specific plant extract and metal precursor used. Temperature is another critical factor affecting nanoparticle formation in herbal-mediated synthesis. Higher temperatures typically accelerate the reduction process and can lead to the formation of smaller nanoparticles. 21 However, excessively high temperatures may cause degradation of the plant biomolecules, potentially compromising their stabilizing effects. 22 Therefore, finding the optimal temperature range is essential for achieving controlled nanoparticle synthesis. The concentration of the plant extract used in the synthesis process also significantly influences nanoparticle formation. Higher extract concentrations generally lead to faster reduction rates and can result in the formation of smaller nanoparticles. 23 However, an excess of plant extract may lead to oversaturation of stabilizing agents on the nanoparticle surface, potentially affecting their properties and applications.
The metal precursor concentration is equally important in controlling the synthesis process. Higher metal ion concentrations can lead to the formation of larger nanoparticles or even aggregates if the amount of reducing and stabilizing agents in the plant extract is insufficient. 24 Balancing the ratio of metal precursor to plant extract is crucial for achieving the desired nanoparticle size and morphology. 25 Reaction time is another factor that can influence the characteristics of the synthesized nanomaterials. Longer reaction times may allow for more complete reduction of metal ions and growth of nanoparticles, but can also lead to agglomeration if not properly controlled. 26 Monitoring the reaction progress and optimizing the synthesis duration is essential for obtaining nanoparticles with the desired properties.
Herbal-mediated synthesis offers several advantages over other green synthesis methods and conventional approaches. One of the primary benefits is the inherent biocompatibility of the synthesized nanomaterials. The use of plant-derived compounds as reducing and stabilizing agents often results in nanoparticles that are less toxic and more suitable for biological applications compared to those produced using chemical methods. 27 Furthermore, herbal-mediated synthesis is typically a one-pot process that occurs under mild conditions. This simplicity reduces energy consumption and eliminates the need for complex equipment or harsh chemicals, making it an environmentally friendly and cost-effective approach. 28 The process is also highly versatile, allowing for the synthesis of a wide range of nanomaterials, including metal nanoparticles, metal oxides, and even more complex nanostructures. Another significant advantage of herbal-mediated synthesis is the potential for obtaining multifunctional nanomaterials. The diverse array of biomolecules present in plant extracts can impart additional functionalities to the synthesized nanoparticles, such as antimicrobial or antioxidant properties. 29 This intrinsic multifunctionality can be particularly beneficial in biomedical applications, where it may enhance the therapeutic efficacy of the nanomaterials.
Herbal mediated synthesis of nanomaterials is based on the reduction and capping effects of various natural active molecules in plant extracts. These active ingredients, such as polyphenols, flavonoids, terpenes, etc, can reduce metal ions under mild conditions while stably generating nanoparticles. The synthesis process usually includes three key steps: metal ion reduction, nanoparticle nucleation and growth, and surface capping.
Key Factors Affecting Synthesis
(1) The influence of temperature
Temperature is an important parameter that affects the formation rate and morphology of nanoparticles. Generally speaking, higher temperatures can accelerate the reduction rate of metal ions by active ingredients in plant extracts, promote the nucleation and growth process of nanoparticles, and thus shorten the reaction time. For example, raising the temperature from room temperature to 60-80°C typically significantly increases the formation rate of nanoparticles.
However, excessively high temperatures may lead to thermal degradation of plant active ingredients, weakening their reduction and stabilization functions, thereby affecting the morphological uniformity and stability of nanoparticles. The temperature tolerance of different plant extracts varies, and the optimal temperature range needs to be determined through experiments.
Temperature also affects the size and distribution of nanoparticles. Generally, an increase in temperature is beneficial for the formation of smaller and more evenly distributed nanoparticles, but exceeding the optimal temperature may cause particle aggregation.
(2) The impact of pH
The pH value regulates the ionization state of plant chemicals, affecting their reducing ability and interaction with metal ions.
Acidic conditions (pH < 6) typically inhibit the reduction process, resulting in a slow generation rate of nanoparticles and the possible formation of larger particles or irregular morphology.
Neutral to weakly alkaline conditions (pH 7-9) typically facilitate the reduction reaction, promote the formation of uniform nanoparticles, and enhance their dispersibility and stability.
Strong alkaline conditions (pH > 9) may sometimes promote faster reduction, but they can also cause excessive aggregation of nanoparticles, affecting their dispersion state.
Therefore, different plant extracts and metal precursor systems need to optimize pH to achieve optimal particle size and morphology.
(3) The influence of plant extract concentration
The concentration of plant extracts directly affects the supply of reducing agents and end capping agents.
When the concentration is low, the reducing ability is insufficient, the reduction of metal ions is incomplete, and the generation of nanoparticles is slow and the particle size is large.
Moderate concentration is beneficial for rapid reduction and formation of uniform and stable nanoparticles.
Excessive concentration may cause excessive capping agent to cover the surface of particles, generating strong surface charges that help prevent particle aggregation. However, it may also increase the viscosity of the colloidal system, affecting dispersibility.
The appropriate concentration range varies depending on the plant source and reaction system, and needs to be adjusted through experiments.
Synthesis of Different Nanomaterials Using Plant Extracts
The versatility of herbal-mediated synthesis is exemplified by its ability to produce a wide array of nanomaterials, ranging from simple metallic nanoparticles to complex nanostructures. This section explores the synthesis of various nanomaterials using plant extracts, highlighting the unique aspects and challenges associated with each category.
Metal nanoparticles have been at the forefront of herbal-mediated synthesis research, with silver (Ag) and gold (Au) nanoparticles being the most extensively studied. The synthesis of AgNPs using plant extracts has garnered significant attention due to their potential applications in antimicrobial treatments and biosensing. Typically, the process involves mixing silver nitrate solution with a plant extract, which results in the rapid reduction of silver ions to form nanoparticles. The phytochemicals present in the extract, such as flavonoids and polyphenols, act as both reducing and stabilizing agents. The size and shape of the resulting AgNPs can be tuned by adjusting factors such as the concentration of silver ions, the type of plant extract used, and the reaction conditions. For example, Chen et al 30 explored the green synthesis of AuNPs (Figure 3A) using the aqueous extract of Curcumae kwangsiensis leaves and evaluated their cytotoxicity, antioxidant, and anti-human ovarian cancer activities. The use of Curcumae kwangsiensis in synthesizing these nanoparticles highlights its potential as a novel chemotherapeutic supplement due to its bioactive properties and environmentally friendly synthesis process. Varghese et al 31 explored the green synthesis of AuNPs (Figure 3B) using the rhizome extract of Kaempferia parviflora, commonly known as black ginger. This eco-friendly method was a sustainable alternative to synthetic processes, leveraging the plant's natural compounds to create nanoparticles with significant biological and environmental applications. The study highlighted the benefits of using Kaempferia parviflora in nanoparticle synthesis, emphasizing its role in enhancing the stability and performance of the resulting AuNPs. AuNPs synthesized through herbal-mediated methods have shown promise in various fields, including catalysis and biomedical applications. The synthesis process is similar to that of AgNPs, often utilizing chloroauric acid as the gold precursor. Plant extracts rich in antioxidants, such as those from tea leaves or citrus fruits, have been particularly effective in reducing gold ions to form stable nanoparticles.32,33 The resulting AuNPs often exhibit unique optical properties, including surface plasmon resonance, which can be exploited for sensing and imaging applications. 34

Platinum (Pt) and palladium (Pd) nanoparticles, while less commonly studied than silver and gold, have also been successfully synthesized using plant extracts. These noble metal nanoparticles are particularly valued for their catalytic properties. The synthesis typically involves the reduction of chloroplatinic acid or palladium chloride by plant extracts. The process often results in smaller nanoparticles compared to silver or gold, due to the higher reduction potential of platinum and palladium. This characteristic can be advantageous for catalytic applications where a high surface area is desirable. For example, Alinaghi et al 35 synthesized Pt and Pd nanoparticles using rosemary and ginseng extracts to explore their potential as anticancer agents. The eco-friendly approach utilized the natural reducing agents in these herbal extracts to produce nanoparticles. The study highlighted the enhanced anticancer effects of these nanoparticles compared to the plant extracts alone, emphasizing the potential of using rosemary and ginseng in nanoparticle synthesis for medical applications. This research contributes to the growing field of green synthesis of nanoparticles, aiming to create biocompatible and effective therapeutic agents with minimal environmental impact.
Moving beyond noble metals, the herbal-mediated synthesis of metal oxide nanoparticles has gained traction due to their diverse applications in areas such as photocatalysis, gas sensing, and energy storage. ZnONPs are among the most commonly synthesized metal oxide nanomaterials using plant extracts. The process typically involves the reaction of a zinc salt, such as zinc nitrate or zinc acetate, with a plant extract. 36 The phytochemicals in the extract not only reduce the zinc ions but also play a crucial role in controlling the morphology of the resulting nanostructures. ZnONPs synthesized through this method have shown promising antibacterial and photocatalytic properties. Karam and Abdulrahman 37 explored the green synthesis of ZnONPs using thyme plant leaf extract, focusing on the impact of different calcination temperatures on the nanoparticles’ properties. The thyme leaf extract served as a natural reducing and stabilizing agent, enabling the environmentally friendly production of ZnONPs (Figure 4). Various calcination temperatures (150 °C, 250 °C, 350 °C, and 450 °C) were tested to determine their effects on the nanoparticles’ size, morphology, and optical properties. Results indicated that ZnONPs synthesized at 450 °C exhibited superior quality, with a high degree of purity and improved characteristics compared to those synthesized at lower temperatures. The average particle size increased from 39.4 yn to 51.86 nm as the calcination temperature rose. Additionally, the energy band gap decreased from 2.7 o to 2.645 eV, indicating enhanced optical properties. This study demonstrated that thyme leaf extract is an effective and sustainable choice for synthesizing ZnONPs, which have potential applications in photocatalysis, antibacterial treatments, and solar cell development.

Schematic Representation of Green Synthesis Process of ZnO NPs by Using the Leaf Extract of Thyme Plant and Zinc Nitrate Hexahydrate. 37
TiO2NPs, known for their photocatalytic and UV-blocking properties, have also been successfully synthesized using plant extracts. The process often involves the hydrolysis of titanium precursors, such as titanium isopropoxide, in the presence of plant extracts. 38 The phytochemicals in the extract can influence the crystal phase of the resulting TiO2NPs, potentially affecting their photocatalytic activity. This green synthesis approach has opened up new possibilities for the eco-friendly production of TiO2NPs for applications in sunscreens, self-cleaning surfaces, and water purification systems. 39 For example, Rao et al 40 focused on the green synthesis of TiO2NPs using Aloe Vera extract. This eco-friendly approach utilized the natural compounds in Aloe Vera, known for its rich content of vitamins, amino acids, and fatty acids, to synthesize nanoparticles without the need for hazardous chemicals. The synthesis process involved boiling Aloe Vera leaves to extract their juice, which was then mixed with titanium chloride to form TiO2NPs. SEM images showed the particles had an irregular structure ranging from 60 to 80SE (Figure 5). The use of Aloe Vera not only provided a sustainable synthesis method but also resulted in nanoparticles with desirable properties for applications in environmental remediation and other industrial uses.

SEM Image of TiO2NPs Synthesized Using Aloe Vera. 40
Iron oxide nanoparticles, particularly magnetite (Fe3O4), have attracted attention for their magnetic properties and potential applications in biomedicine and environmental remediation. The herbal-mediated synthesis of these nanoparticles typically involves the reduction of iron salts by plant extracts rich in polyphenols. The resulting nanoparticles often exhibit superparamagnetic behavior, making them suitable for magnetic separation processes and as contrast agents in magnetic resonance imaging. 41 The use of plant extracts in the synthesis process can enhance the biocompatibility of the iron oxide nanoparticles, making them particularly attractive for biomedical applications. For example, Ramesh et al 42 synthesized superparamagnetic Fe3O4NPs using the aqueous leaf extract of Zanthoxylum armatum DC. This green synthesis approach utilized the plant extract as both a reducing and capping agent, which facilitated the formation of nanoparticles with an average size of 17 C. The study aimed to develop an environmentally friendly method to create nanoparticles for the adsorption of methylene blue, an organic pollutant.
The field of herbal-mediated synthesis has expanded to include the production of bimetallic and alloy nanoparticles, which often exhibit enhanced properties compared to their monometallic counterparts. These nanoparticles can be synthesized by simultaneously reducing two different metal precursors in the presence of a plant extract. For example, Malik et al 43 explored the green synthesis of Ag-Fe bimetallic nanoparticles using an aqueous extract of Salvia officinalis. The synthesis process utilized the phytochemicals present in the plant extract as reducing and stabilizing agents, leading to the formation of nearly spherical nanoparticles with an average size of 30Th (Figure 6). The study demonstrated the potential of using plant extracts in nanoparticle synthesis to achieve high catalytic activity, stability, and reusability, providing a promising alternative to conventional chemical methods. More et al 44 explored the synthesis and antimicrobial applications of bimetallic nanoparticles using Ocimum basilicum (basil) leaf extract. The researchers aimed to develop an eco-friendly and effective antibacterial strategy to combat antibiotic-resistant pathogens. By combining Ag-PtNPs with basil extract, they achieved a novel synthesis method that resulted in nanoparticles approximately 59 it in size with a polydisperse index of 0.159. The AgPtNPs demonstrated significant antibacterial activity, showing zones of inhibition ranging from 9 to 25 mm against various strains of bacteria, including multidrug-resistant Staphylococcus aureus and Escherichia coli. The study highlighted the potential of using basil as a natural reducing agent in nanoparticle synthesis, offering a sustainable and efficient approach to developing new antibacterial agents.

Schematic Illustration of the Green Synthesis Process of Ag–Fe Bimetallic Nanoparticles. 43
The synthesis of different nanomaterials using plant extracts demonstrates the versatility and potential of this green approach. From simple metallic nanoparticles to complex bimetallic and alloy structures, the herbal-mediated method offers a sustainable route to a wide range of nanomaterials. Each type of nanomaterial presents unique challenges and opportunities in terms of synthesis control and potential applications. The ability to synthesize such a diverse array of nanomaterials using plant extracts underscores the richness of phytochemicals present in plants and their capacity to act as reducing and stabilizing agents. As research in this field progresses, it is likely that new types of nanomaterials will be added to the repertoire of herbal-mediated synthesis. This ongoing exploration not only expands our understanding of green synthesis methods but also opens up new avenues for the development of sustainable nanomaterials with tailored properties for specific applications.
Characterization and Evaluation of Herbal-Synthesized Nanomaterials
In the research of synthesizing herbal nanomaterials for drug delivery systems, the characterization of drug loading capacity and release behavior is the core link in evaluating their actual functionality. Drug Loading Content (DLC) and Encapsulation Efficiency (EE) are two commonly used indicators to describe the drug mass carried by a unit mass of nanocarrier and the proportion of successfully loaded nanomaterials in the input drug. Common characterization methods include:
UV Vis spectrophotometry: By measuring the absorption peak intensity of free drugs, the amount of drug adsorbed or encapsulated by nanoparticles is calculated;
High performance liquid chromatography (HPLC): It can quantitatively analyze multiple drugs with high sensitivity and accuracy, especially suitable for precise determination of drug content in complex samples;
Thermogravimetric analysis (TGA): used to estimate the thermogravimetric difference before and after drug loading on nanomaterials, in order to indirectly calculate the drug loading amount.
The study of drug release behavior is usually conducted through in vitro release experiments, monitoring the release rate of drugs from nanomaterials at different pH values (such as simulated blood pH 7.4, tumor microenvironment pH 5.5) or enzyme environments. The release kinetics can be analyzed through the following methods:
Cumulative release curve drawing: By regularly sampling and measuring drug concentration, calculate the cumulative release ratio;
Release model fitting: Fit the release data to zero order, first-order, Higuchi model, or Korsmeyer Peppas model to analyze the release mechanism (such as diffusion control, dissolution control, or both);
PH responsiveness testing: Evaluating whether nanomaterials have pH sensitive release ability, which is an important function of many tumor targeted drug systems.
These characterizations are of great significance for understanding the effectiveness, controlled release ability, and targeted release potential of herbal synthesized nanomaterials in drug delivery applications, and also provide experimental basis for designing more functional herbal derived nanosystems.
The characterization and evaluation of herbal-synthesized nanomaterials are crucial steps in understanding their properties, assessing their quality, and determining their potential applications. A comprehensive analysis typically involves a combination of techniques that provide insights into the morphological, structural, compositional, and functional aspects of the nanomaterials.
Morphological characterization is often the first step in evaluating herbal-synthesized nanomaterials. TEM is widely used for this purpose, offering high-resolution imaging that allows for precise measurement of particle size and shape. TEM can reveal the internal structure of nanoparticles and provide information about their crystallinity. For instance, in the case of metallic nanoparticles, TEM can show the characteristic lattice fringes, confirming their crystalline nature (Figure 7A). SEM complements TEM by providing detailed information about the surface topography and texture of nanomaterials. SEM is particularly useful for examining larger nanostructures or aggregates of nanoparticles (Figure 7B). AFM offers another dimension to morphological characterization by providing three-dimensional surface profiles. AFM is especially valuable for analyzing the thickness of two-dimensional nanomaterials or the roughness of nanoparticle coatings (Figure 7C).

Structural and compositional analysis of herbal-synthesized nanomaterials is typically performed using XRD, FTIR, and EDX. XRD provides information about the crystalline structure of the nanomaterials, allowing for the identification of specific crystal phases and the calculation of crystallite size. This technique is particularly useful for confirming the formation of pure metallic or metal oxide nanoparticles (Figure 8A). FTIR spectroscopy is employed to identify the functional groups present on the surface of the nanomaterials. In the context of herbal-synthesized nanomaterials, FTIR can reveal the presence of plant-derived biomolecules that may be acting as capping agents, providing insights into the stabilization mechanism (Figure 8B). EDX, often coupled with SEM or TEM, offers elemental analysis of the nanomaterials, confirming their composition and purity (Figure 8C).

The optical properties of herbal-synthesized nanomaterials, particularly for metallic nanoparticles, are commonly evaluated using UV-Visible spectroscopy. This technique exploits the surface plasmon resonance phenomenon exhibited by many metallic nanoparticles. The position and intensity of the absorption peak can provide information about the size, shape, and concentration of the nanoparticles. For instance, AgNPs typically show an absorption peak around 400-450 yp, while gold nanoparticles exhibit a peak around 520-550 nm. 45 Shifts in these peak positions can indicate changes in particle size or shape, making UV-Vis spectroscopy a valuable tool for monitoring the synthesis process and assessing the stability of the nanoparticles over time. Stability and dispersibility are critical parameters for many applications of nanomaterials. Zeta potential measurements are commonly used to assess the stability of nanoparticle suspensions. A high absolute value of zeta potential (typically above 30 re) indicates good stability, as the particles have sufficient electrical charge to repel each other and resist aggregation. 46 Dynamic Light Scattering (DLS) is another technique used to evaluate the dispersibility of nanoparticles in solution. DLS provides information about the hydrodynamic size of the particles, which includes the core of the nanoparticle and any associated capping agents or solvation shell. This technique is particularly useful for detecting the presence of aggregates or monitoring changes in particle size over time. 47
Biocompatibility and toxicity studies are essential for evaluating the potential of herbal-synthesized nanomaterials in biomedical applications. These studies typically involve in vitro assays using various cell lines to assess cytotoxicity, cellular uptake, and potential genotoxicity. Common techniques include MTT assays for cell viability, flow cytometry for cellular uptake studies, and comet assays for assessing DNA damage. 48 In vivo studies in animal models may also be conducted to evaluate the biodistribution, pharmacokinetics, and long-term effects of the nanomaterials. It's worth noting that herbal-synthesized nanomaterials often show enhanced biocompatibility compared to their chemically synthesized counterparts, due to the presence of natural capping agents derived from plant extracts. 49
Characterization methods and advantages and disadvantages analysis of herbal mediated nanomaterials
(1) Transmission Electron Microscope (TEM)
Advantages: TEM can provide high-resolution morphological information of nanoparticles, including direct observation of particle size, shape, and crystal structure. It can display the lattice patterns of nanoparticles and verify the crystal structure.
Disadvantages: Sample preparation is complex and may introduce sample preparation deviations; The instruments and equipment are expensive and the operation is complex; Not suitable for large-scale sample statistics, and has strict requirements for sample thickness and conductivity.
(2) Scanning Electron Microscope (SEM)
Advantages: SEM is suitable for observing the surface morphology and aggregation state of nanomaterials, with relatively simple sample preparation and a wide imaging range.
Disadvantages: The resolution is lower than TEM, making it difficult to observe the internal structure of nanoparticles; Metal film treatment is required for non-conductive samples, which may affect the true surface characteristics.
(3) Atomic Force Microscopy (AFM)
Advantages: AFM can provide three-dimensional surface profiles of nanomaterials, suitable for non-conductive samples, especially for analyzing the thickness and surface roughness of two-dimensional nanomaterials.
Disadvantages: Slow scanning speed, limited imaging area, and complex measurement for soft samples or liquid environments.
Applications of Herbal-Synthesized Nanomaterials
Herbal-synthesized nanomaterials have garnered significant attention due to their unique properties and potential applications across various fields. The integration of plant-derived compounds in the synthesis process often imparts additional functionalities to these nanomaterials, enhancing their performance in diverse applications. This section explores the wide-ranging applications of herbal-synthesized nanomaterials, highlighting their impact in biomedical sciences, environmental remediation, catalysis, sensing technologies, and emerging fields.
Efficiency of Pollutant Removal in Environmental Applications
Herbal synthesis nanomaterials are widely used for the removal of harmful substances in water, such as heavy metal ions, organic dyes, and pathogenic microorganisms, due to their green synthesis process and high surface area. In practical applications, these nanomaterials not only exhibit excellent adsorption performance, but also possess efficient catalytic degradation ability.
(1) Organic dye degradation
Taking silver nanoparticles (AgNPs) as an example, the degradation rate of methyl orange (MO) dye by AgNPs synthesized using Azadirachta indica leaf extract under photocatalytic conditions reached over 95% within 90 min (pH 7, AgNPs concentration 0.1 mM).
Similarly, the photocatalytic degradation rate of Rhodamine B (RhB) dye by ZnO nanoparticles prepared using Cymbopogon citratus extract reached 98.3% within 120 min (under UV irradiation at a concentration of 10 mg/L).
(2) Heavy metal removal
Iron oxide nanoparticles synthesized from herbal medicine, such as Fe ∝ O4, have significant adsorption properties for heavy metal ions in water. The Fe-O4 nanoparticles synthesized from Moringa oleifera extract showed a maximum adsorption capacity of 145.6 mg/g for lead ions (Pb2+) in batch adsorption experiments, with a removal efficiency exceeding 96% (initial concentration of 50 mg/L, pH 6).
(3) Antibacterial purification
AgNPs synthesized from herbs also exhibit broad-spectrum antibacterial properties in water purification. For example, AgNPs synthesized using Ocimum sanctum extract showed a 99.9% killing rate against Escherichia coli (E. coli) in experiments (concentration 10 µ g/mL, contact time 30 min).
In the realm of biomedical applications, herbal-synthesized nanomaterials have shown remarkable potential, particularly in antimicrobial treatments, cancer therapy, and drug delivery systems. The antimicrobial properties of these nanomaterials, especially silver and zinc oxide nanoparticles, have been extensively studied. These nanoparticles often exhibit enhanced antimicrobial activity compared to their conventionally synthesized counterparts, likely due to the synergistic effects of the plant-derived capping agents. For example, Verma et al 50 reported a green synthesis of AgNPs using the aqueous extract of Salvinia molesta. The synthesized AgNPs were characterized to be spherical with an average size of 12.46 nm and demonstrated significant antimicrobial efficacy. Specifically, the AgNPs exhibited strong antibacterial activity against both Gram-positive (Staphylococcus aureus) and Gram-negative (Escherichia coli) bacteria. The zone of inhibition was measured at 21 mm for E. coli and 16 mm for S. aureus (Figure 9), indicating a higher susceptibility in Gram-negative bacteria. The minimum inhibitory concentration (MIC) was determined to be 10.50 μg/mL for E. coli and 13.0 μg/mL for S. aureus. This study not only highlighted the potential of using plant extracts for nanoparticle synthesis but also emphasized the effective antimicrobial properties of the resulting AgNPs, making it a promising approach for developing antibacterial agents.

Antimicrobial Efficacy of Synthesized AgNPs Against; (a) S. aureus (Gram + ve) (b) E. coli (Gram − ve). Disk (1) Buffer Solution, (2) AES (3) AgNO3 and (4) AgNPs. 48
In cancer therapy, herbal-synthesized nanomaterials have shown potential in both diagnostic and therapeutic applications. AuNPs, in particular, have been explored for their ability to enhance contrast in imaging techniques and for targeted drug delivery to cancer cells. The plant-derived capping agents on these nanoparticles can provide additional functionalities, such as improved cellular uptake or intrinsic anticancer properties. For example, Vemuri et al 51 synthesized and evaluated biosynthesized AuNPs as potential anti-cancer agents against breast cancer. The nanoparticles were created using phytochemicals such as Curcumin, Turmeric, Quercetin, and Paclitaxel, and were tested for their cytotoxic effects on breast cancer cell lines MCF-7 and MDA-MB 231 (Figure 10). The study found that combinations of these b-AuNPs were significantly more effective in inhibiting cell proliferation, inducing apoptosis, and reducing angiogenesis compared to individual treatments. Notably, the combination of all four types of b-AuNPs demonstrated the highest therapeutic activity, showing a synergistic effect. Importantly, these combinations did not exhibit cytotoxicity towards normal human embryonic kidney cells (HEK 293), suggesting their biocompatibility. The research highlighted the potential of these b-AuNPs in cancer nanomedicine, offering a promising approach due to their high efficacy and low toxicity. Drug delivery is another area where herbal-synthesized nanomaterials have made significant strides. The biocompatible nature of these nanomaterials, coupled with their ability to be functionalized with various ligands, makes them excellent carriers for drug molecules. Fahimmunisha et al 52 explored the green synthesis of ZnONPs using Aloe socotrina leaf extract, aiming to develop a novel drug delivery approach with antibacterial properties. The study concluded that Aloe socotrina-capped ZnO NPs are promising for managing drug-resistant bacterial infections, particularly in urinary tract infections, offering a sustainable and effective solution for drug delivery in biomedical applications.

(A–B): Spheroid Migration Assay-Effect of AuNPs-Cur, AuNPs-Tur, AuNPs-Qu, AuNPs-Pacli, AuNPs-CTQ and AuNPs-PCTQ on Spheroid Forming and Migration Ability of MCF-7 and MDA-MB 231 Cell Lines. 53
Environmental remediation is another field where herbal-synthesized nanomaterials have shown great promise. The unique properties of these nanomaterials, such as high surface area and enhanced reactivity, make them effective agents for removing pollutants from water and soil. Karimi et al 54 focused on the removal of arsenic from mining effluents using iron nanoparticles synthesized through a green method involving plant extracts from Prangos ferulacea and Teucrium polium. These nanoparticles, characterized by techniques like UV-visible spectrometry and x-ray powder diffraction, had a particle size of 42 nm. The adsorption experiments demonstrated a 93.8% arsenic removal efficiency under optimal conditions: an agitation rate of 200 rpm, a contact time of 20 min, a pH of 6, and an adsorbent dosage of 2 g/L. The maximum uptake capacity was about 61.7 a /g. The adsorption process followed a pseudo-second-order kinetic model with a high correlation coefficient (R²=0.99), indicating a spontaneous and exothermic process. Thermodynamic studies showed negative values for Gibbs free energy and enthalpy, confirming the physical nature of the adsorption. Nikić et al 55 study on arsenic removal from groundwater, researchers developed magnetic nanoparticles using green synthesis methods with onion peel and corn silk extracts. These nanoparticles, named MNp-OP and MNp-CS, demonstrated high specific surface areas of 243 m²/g and 261 m²/g, respectively. The adsorption capacities for arsenic were 1.86 mg/g for MNp-OP and 2.79 mg/g for MNp-CS, compared to 1.30 mg/g for conventionally synthesized nanoparticles (MNp-CO). Kinetic experiments indicated that the adsorption followed a pseudo-second-order model, suggesting chemisorption as the primary mechanism.
In the field of catalysis, herbal-synthesized nanomaterials have emerged as efficient and environmentally friendly alternatives to traditional catalysts. Noble metal nanoparticles, such as palladium and platinum, synthesized using plant extracts have shown excellent catalytic activity in various organic transformations. The plant-derived capping agents can play a dual role in these systems, not only stabilizing the nanoparticles but also potentially acting as co-catalysts or selectivity modifiers. For example, Veisi et al 56 developed PdNPs using an aqueous extract of Stachys lavandulifolia, a herbal tea. The extract acted as a reducing, stabilizing, and capping agent, eliminating the need for additional surfactants or capping agents. The synthesized palladium nanoparticles, which were spherical and measured between 5 and 7 nm, were used as a homogeneous catalyst for Suzuki coupling reactions conducted in water under mild conditions. The reactions yielded high product outputs, with the catalyst demonstrating excellent reusability, maintaining its activity for at least eight cycles without significant palladium leaching. The study highlighted the eco-friendly and cost-effective nature of using natural plant extracts in nanoparticle synthesis, providing an environmentally benign alternative for industrial applications. The catalytic system was effective across various aryl halides, achieving yields of up to 98% in some cases. Dobrucka 53 synthesized PtNPs using Fumariae herba extract, demonstrating their potential as eco-friendly catalysts. The study highlighted the nanoparticles’ catalytic efficiency in degrading organic dyes, such as methylene blue and crystal violet. Specifically, the absorbance intensity peak of methylene blue significantly decreased within 15 min, indicating rapid catalytic activity. This research underscored the potential of plant-based synthesis methods for producing high-purity platinum nanoparticles with effective catalytic properties, offering an environmentally friendly alternative to conventional chemical processes. The findings suggest promising applications in industries where organic dye degradation is critical, such as textiles, by utilizing the enhanced surface-to-volume ratios of metal nanoparticles.
Nano materials synthesized from herbs have shown extensive potential in catalysis, especially in the degradation of organic pollutants, organic synthesis reactions, and energy conversion. Compared with traditional physical or chemical methods for synthesizing nanocatalysts, the herbal synthesis method is not only more environmentally friendly, but also endows the catalyst with unique surface activity and multifunctionality. Taking silver nanoparticles (AgNPs) as an example, herbal synthesized AgNPs exhibit catalytic performance comparable to or superior to chemically synthesized AgNPs when treating organic dyes such as methyl orange (MO) or Congo red (CR).
The field of sensing and biosensing has also benefited from the unique properties of herbal-synthesized nanomaterials. The optical and electronic properties of these nanomaterials, combined with their biocompatibility, make them excellent candidates for developing sensitive and selective sensors. Uddin et al 57 synthesized AgNPs using the plant extract of Matricaria recutita (Babunah) to create a highly selective sensor for mercury ions (Hg2+). AgNPs, when mixed with Hg2+ ions, exhibited a decrease in UV–Vis absorption intensity, which facilitated the colorimetric detection of mercury. The sensor demonstrated high selectivity for Hg2+ over other metal ions such as Co2+, Zn2+, and Ni2+. It was capable of detecting Hg2+ concentrations as low as 10 ppm, evidenced by a color change from yellowish-brown to colorless, and further confirmed through UV–Visible spectroscopy. This approach offers an eco-friendly, cost-effective method for mercury detection, emphasizing its potential application in environmental monitoring and water safety. Qian et al 58 explored the application of AgNPs synthesized using Origanum majorana leaf extract as both a hydrazine sensor. The AgNPs showed high sensitivity in detecting hydrazine, a potentially carcinogenic compound, with a detection limit of 0.25 μM. This sensor exhibited excellent catalytic performance and a simple preparation process, making it promising for environmental monitoring.
Although herbal synthesized nanomaterials have shown great potential in biomedical fields such as antibacterial, anticancer, drug delivery, and tissue repair, their clinical translation still faces a series of challenges:
(1) Lack of standardization in the synthesis process
The components of herbal extracts are complex and diverse, and the types and concentrations of active compounds in different plants or batches of extracts may vary significantly, leading to poor reproducibility of the synthesis process and unstable product quality. This severely limits the large-scale preparation and clinical application of nanomaterials.
(2) Lack of systematic toxicological evaluation
Although many studies have shown that nanomaterials synthesized from herbs have good biocompatibility, their long-term toxicity, metabolic behavior, and tissue accumulation still lack systematic evaluation. Safety data is still limited, especially under different doses, times, and individual backgrounds.
(3) Lack of regulatory recognition and clinical certification pathways
At present, most herbal synthesis nanomaterials are still in the stage of in vitro experiments or small animal models, and feasible clinical trial plans have not yet been established. There is also a lack of relevant drug registration standards and quality control specifications. This poses a high barrier for it to move from the laboratory to the pharmaceutical market.
Conclusion
Herbal mediated synthesis of nanomaterials provides a green, sustainable, and functionally diverse alternative synthetic pathway for the field of nanotechnology. This method fully utilizes the natural plant chemical components such as polyphenols, flavonoids, terpenes, etc in plant extracts to achieve the reduction of metal ions and stable nucleation of nanostructures under mild conditions. It not only avoids the problems of toxic reagents and high-energy consumption in traditional physical and chemical methods, but also endows the synthesized nanomaterials with good biocompatibility and potential biological activity functions. By selecting plant materials and regulating reaction conditions, metal or metal oxide nanoparticles with diverse forms and specific functions can be synthesized, which are widely used in various fields such as biomedical, environmental remediation, catalysis, and sensing. Although herbal mediated synthesis of nanomaterials has shown many advantages under laboratory conditions, such as green environmental protection, low cost, and strong biocompatibility, its scalability still faces a series of technical and process challenges in practical industrial applications. The composition of plant extracts is highly dependent on species, collection time, geographical environment, and extraction methods, resulting in significant fluctuations in the types and concentrations of their active ingredients. The differences between batches make it difficult to maintain product consistency during large-scale synthesis, thereby affecting the particle size distribution, morphology, and functional properties of nanomaterials. Establish a standardized system for plant extract components, introduce quantitative analysis methods (such as HPLC, LC-MS) to monitor key reducing/blocking components, and combine with quality control protocols.
Current Challenges and Future Perspectives
The field of herbal-mediated synthesis of nanomaterials, while promising, faces several challenges that need to be addressed to fully realize its potential. These challenges span from fundamental scientific understanding to practical implementation issues, and addressing them will be crucial for the advancement of this green nanotechnology approach. One of the primary challenges in herbal-mediated synthesis is the issue of reproducibility and scalability. The complex and variable nature of plant extracts can lead to inconsistencies in the synthesized nanomaterials from batch to batch. Factors such as the plant's growth conditions, harvesting time, and extraction methods can significantly influence the composition of the extract and, consequently, the characteristics of the resulting nanomaterials. This variability poses a significant hurdle for large-scale production and industrial adoption. Future research should focus on developing robust protocols that can accommodate these inherent variations while still producing consistent nanomaterials.
A deeper mechanistic understanding of the herbal-mediated synthesis process is another critical area that requires attention. While the general principles of bioreduction and stabilization by plant-derived compounds are well-established, the specific roles of individual phytochemicals and their interactions in the synthesis process are not fully understood. Elucidating these mechanisms will be crucial for optimizing the synthesis process and tailoring the properties of the resulting nanomaterials. Advanced analytical techniques and in-situ characterization methods will be invaluable in unraveling these complex interactions.
The standardization of protocols for herbal-mediated synthesis is closely linked to the issues of reproducibility and mechanistic understanding. Developing standardized methods for plant extract preparation, reaction conditions, and purification processes will be essential for ensuring consistency across different research groups and facilitating the comparison of results. This standardization will also be crucial for any future regulatory approvals, particularly for biomedical applications of herbal-synthesized nanomaterials. The exploration of new plant sources for nanomaterial synthesis presents both a challenge and an opportunity. While many common plants have been investigated, there remains a vast untapped potential in the plant kingdom. Future research should focus on systematically evaluating diverse plant species, particularly those with known medicinal properties, for their potential in nanomaterial synthesis. This exploration could lead to the discovery of novel nanomaterials with unique properties and functionalities.
Enhancing the stability and functionality of herbal-synthesized nanomaterials is another important area for future research. While the plant-derived capping agents often provide good stability, there is room for improvement, particularly for long-term storage and in complex biological environments. Strategies such as cross-linking of the capping agents or incorporation of additional functional molecules could enhance both stability and functionality. Furthermore, developing methods to precisely control the surface chemistry of these nanomaterials will be crucial for tailoring their properties for specific applications.
With the continuous development of green nanotechnology, the synthesis of herbal mediated nanomaterials in the future will increasingly rely on interdisciplinary collaboration to overcome key bottlenecks such as repeatability, standardization, and scalability. Especially with the support of artificial intelligence (AI) and machine learning (ML) technologies, the process of herbal synthesis is expected to achieve a higher degree of automation and intelligent optimization.
Footnotes
Author Contribution Statement
Zhirong Gu: methodology; software; validation; formal analysis; investigation; data curation; writing—original draft preparation,
Yan Guo: conceptualization; methodology; validation; writing—original draft preparation,
Xiaowen Mao: methodology; software; validation; formal analysis; data curation; writing—original draft preparation,
Mei Qi: methodology; formal analysis; investigation; data curation,
Bin Ge: conceptualization; formal analysis; investigation; data curation; writing—review and editing; project administration; supervision
Funding
Scientific Research Project on Drug Administration of Gansu Provincial Drug Administration (No. 2023GSMPA046). High-level Key Project of Gansu Provincial Administration of Traditional Chinese Medicine (GZKZ-2024-16).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
