Abstract
Introduction
The genus Aralia is a large genus in the family Araliaceae. Up to now, scientists have discovered about 30 different species, popularly distributed in Asia and America. 1 Aralia armata, a common herb in China and Vietnam, is known as a folk medicine reported to show anti-inflammatory (arthritis, hepatitis), immunostimulating, and antimalarial activities. 2 Previous studies have shown that major constituents of the Aralia genus include saponins (triterpenoid glycosides), flavonoids, steroids, and acetylenic lipids,3,4 among which saponins are characteristic of A. armata in particular and the Aralia genus in general.5,6 The saponin compounds in A. armata have exhibited various activities, including antibacterial,7–10 antifungal,11–14 antioxidant,15–17 anti-inflammatory,18–22 in vitro inhibition of cancer cells,23–27 and anti-HIV activities.28–30 Moreover, saponins are also utilized as treatments against mollusks that caused crop damage, namely the species Pomacea canaliculata, Pomacea maculata, Gyraulus convexiusculus, and Tarebia granifera.31–35
Currently, diseases related to parasites are a concern in many countries around the world.36,37 The freshwater snail P. canaliculata (also known as the golden apple snail) is known to be an intermediate host for several parasites that are pathogenic to humans. For example, the parasitic worm Angiostrongylus cantonensis is the cause of filariasis in human beings, which is characterized by eosinophilic meningitis.38–40 Gnathostoma spinigerum causes gnathostomiasis, an infection that human acquires by ingesting the third-stage larvae of this worm,41,42 which can lead to blindness, paralysis, and even death. 43 Notably, P. canaliculata also seriously affects agricultural production, especially rice,44–46 and it is one of the most invasive species in the world. 47 A significant portion of agricultural land in Asia has been invaded and damaged by the golden apple snail. 44 The large-scale increase in the number of golden apple snails has affected rice cultivation throughout tropical and subtropical regions as well as southern Europe and the United States of America.48,49 One of the common solutions employed is pesticide application, which contains many toxic chemicals that can seriously affect human health, the ecosystem, and the environment. Alternatively, natural biological agents that could control the snail population effectively and reduce the risk of development of resistance to chemical pesticides would be welcome. To the best of our knowledge, no studies on the molluscicidal activity and chemical composition of A. armata grown in Vietnam have been published. Driven by those challenges, the chemical composition from the roots of A. armata and their activity against P. canaliculata is reported for the first time, simultaneously exploring the relationship between the chemical structures and molluscicidal activity. This study also illustrates the safety of components from A. armata for the aquatic environment in general and nontarget aquatic animals in particular.
Results and Discussion
Isolation
Eight oleanolic acid saponins, pseudoginsenoside RT1 (
Molluscicidal Activity
The molluscicidal activity of fractions and compounds isolated from A. armata roots against golden apple snail (P. canaliculata) after 24 h is represented by LC50 and LC90 values (µg/mL). A comparison of the molluscicidal activity of the fractions, the active compounds from A. armata, and the positive control (tea saponin) is illustrated in Table 1. The experimental results indicate that the molluscicidal activity is directly proportional to the concentration of the active ingredients. It can be seen that the molluscicidal activity of the fractions is particularly noteworthy, especially the aqueous fraction (LC50 18.59 µg/mL), which is the basis for choosing the aqueous fraction to further separate and search for active compounds with molluscicidal activity from the roots of A. armata. Indeed, the compounds isolated from A. armata roots exhibit remarkably robust activity against the P. canaliculata snail, the LC50 values of the compounds are in the range of 7.90 to 17.50 µg/mL. Furthermore, the molluscicidal capacities of several compounds from A. armata are higher than that of tea saponin (LC50 = 11.02 µg/mL), an agent known to have powerful molluscicidal capacity. 59 Thus, the contents of saponin compounds separated from A. armata roots demonstrated significant molluscicidal activity.
The Molluscicidal Activity of Compounds Isolated from Aralia armata.

AChE Enzyme Inhibitory Activity
Several isolated compounds from A. armata were screened for inhibitory activity against acetylcholinesterase (AChE, see Table 2).
AChE Inhibitory Activity of Some Isolated Compounds.

*Galantamine was tested in the concentration range of 10, 2, 0.4, and 0.08 µg/mL.
Acute Toxicity for Brine Shrimp (Artemia sp.)
To evaluate the impact of active ingredients from A. armata on the aquatic environment, acute toxicity experiments on adult brine shrimp (Artemia sp.) were conducted. The fractions closest to the pure compounds were put through trials. Experimental data indicate that the higher the sample concentrations, the higher the toxicity to brine shrimp (Figure 1). At concentrations of 25 to 300 µg/mL, most of the fractions were strongly toxic for Artemia sp., with mortality rates higher than 80%. The fraction AA2A2 shows the highest toxicity with 100% of mortality at 250 µg/mL concentration.
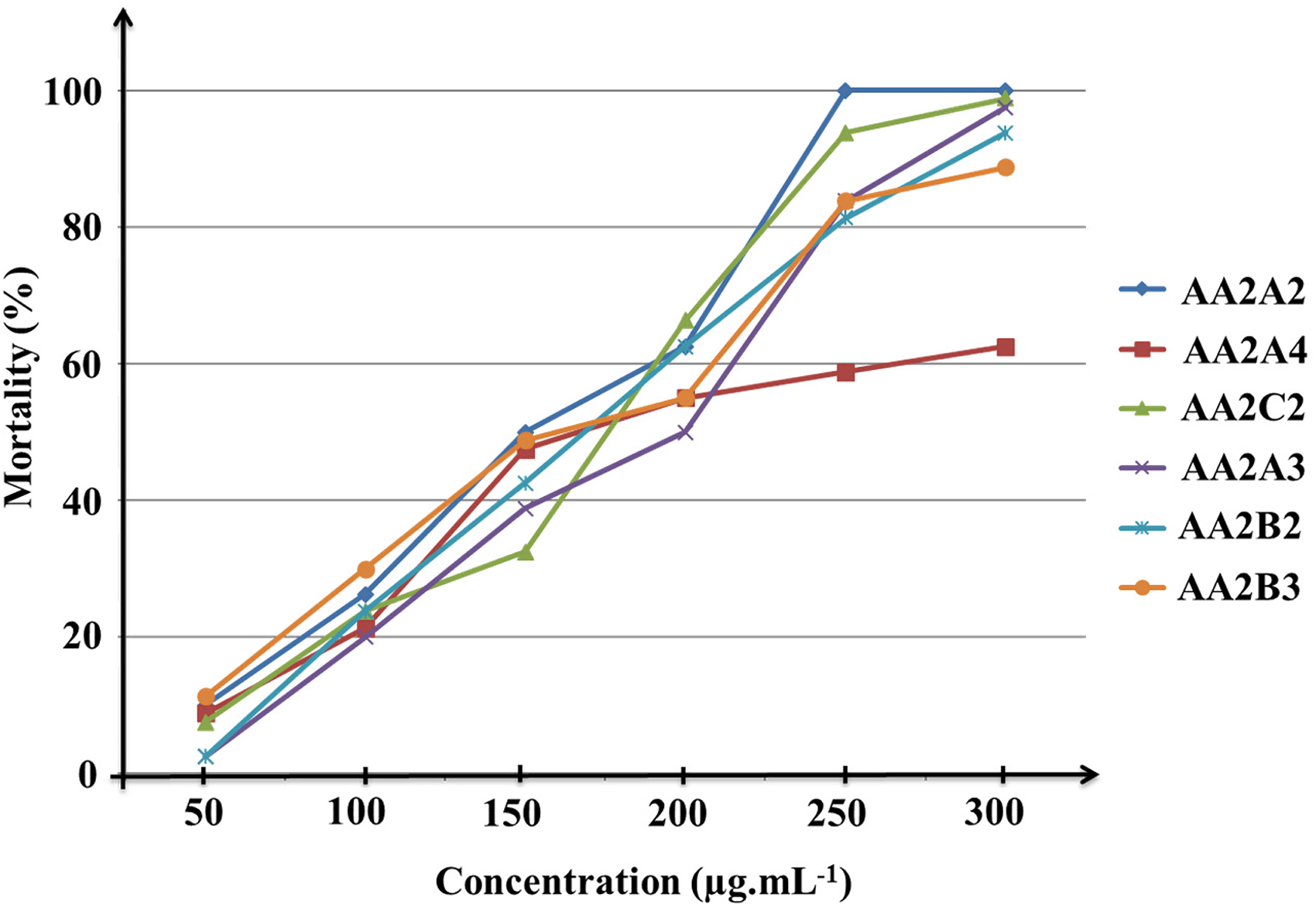
Schematic demonstration of the mortality of Artemia sp. depending on the concentration of fractions.
The LC50 values (µg/mL) for brine shrimp ranged from 148.55 to 193.22 µg/mL after 24 h of exposure. Compared with the toxicity to P. canaliculata, which has LC50 values in the range of 11.54 to 17.88 µg/mL, the toxicity of the fractions from A. armata to the golden apple snail is much higher than that of the brine shrimp (10–15 times) under the same experimental conditions (Table 3). Over the LC90 concentration range of the fractions CAA2A2, CAA4A3, CAA2A4, CAA4B2, CAA4B3, AA2C2 (40.71–52.42 µg/mL) killed <8% of Artemia sp. Thus, it can be seen that the molluscicidal components of A. armata roots are virtually safe for aquatic animals at the median lethal concentrations.
Comparison of Acute Toxicity to Artemia sp. and Molluscicidal Activity of Fractions Isolated from Aralia armata.

*SI = selectivity index = LC50 (Artemia sp.)/LC50 (P. canaliculata).
Phytotoxic Activity of Aqueous Fraction
This study aimed to evaluate the control effect against P. canaliculata of the water-soluble fraction of the roots of A. armata. Additionally, the phytotoxicity of the aqueous fraction was evaluated on rice (Oryza sativa L.), the results of which are presented in Table 4. Compared with the negative control (water), the aqueous fraction did not exhibit detrimental effects on the germination or growth of rice.
Phytotoxic Activity Results of the Aqueous Fraction.

In silico Evaluation of A. armata Saponins
There are apparently no P. canaliculata protein structures available in the Protein Data Bank. 60 There are, however, several P. canaliculata protein sequences available from the Protein Database. 61 Homology models for several P. canaliculata proteins, acetylcholinesterase (PcAChE), alanine aminotransferase (PcALT), aspartate aminotransferase (PcAST), heat-shock protein 70 (PcHsp70), phenylalanine-4-hydroxylase (PcPheOH), and actin-binding protein villin-1 (PcVillin), were constructed from crystal structure templates available in the Protein Data Bank using the SWISS MODEL server (https://swissmodel.expasy.org/).
Molecular docking was carried out using Molegro Virtual Docker v 6.0.1 (Molegro ApS). From the docking scores (Table 5), it is apparent that the A. armata saponins preferentially target P. canaliculata acetylcholinesterase (PcAChE) and actin-binding protein (PcVillin).
Normalized Docking Scores (DSnorm, kJ/mol) of Aralia armata Saponins with Pomacea canaliculata Target Proteins.

Lowest-energy docking scores for each ligand are highlighted in
Acetylcholinesterase catalyzes the hydrolysis of the neurotransmitter acetylcholine, which serves to terminate synaptic transmission. Inhibition of AChE leads to paralysis and possibly death of the organism. Thus, PcAChE inhibition by A. armata saponins may account for the observed toxicity of these compounds to P. canaliculata. The lowest-energy docking scores for ligands against PcAChE were
Key Intermolecular Contacts Between the Aralia armata Ligands With the Best Docking Scores and Their Pomacea canaliculata Protein Targets.

H-bond = hydrogen-bonding interaction; vdW = van der Waals (hydrophobic) interaction.
Villin is a member of the gelsolin family of actin-modulating proteins.
62
These proteins block the ends of actin causing the depolymerization of actin filaments, but also catalyzing the bundling activity of actin, thereby influencing the polymeric state of actin. As important components of the cytoskeleton, actin-binding proteins play a central role in cell locomotion and motility, and inhibition of actin-binding proteins would likely have serious consequences. The compounds with the best docking scores with PcVillin were
Experimental
General
The mass spectra were recorded on an Agilent 6530 accurate-mass Quadrupole Time-of-Flight liquid chromatography-mass spectrometry system. NMR spectroscopic analyses were carried out by the Bruker 500 MHz spectrometer. The Jasco P2000 polarimeter is utilized for measuring the optical rotation of pure compounds. Silica gel (43–60 μm), reversed-phase C18 gel, and diaion HP-20 resins were used in the column chromatography. In addition, the Agilent 1100 HPLC system was utilized for HPLC analysis. The compounds were examined by thin-layer chromatography using the precoated silica gel 60 F254, RP-18 F254S sheets. The spots were visualized by dipping thin-layer chromatography sheets into 5% H2SO4 solution in ethanol followed by heating at 110°C for 10 min.
Plant Material
The roots of A. armata were collected in Hoa Vang district, Danang city (16°00′55″N 108°07′8″E) in October 2020. Plant samples were authenticated by Nguyen The Cuong (The Institute of Marine Biochemistry, VAST, Hanoi, Vietnam). The voucher specimen (Code: NCCT-P71R) was deposited in the Institute of Marine Biochemistry, VAST, Hanoi, Vietnam.
Snails (P. canaliculata)
Eggs of wild P. canaliculata were collected from a field in Dien Ban District, Quang Nam province, Vietnam (15°55′58″N 108°11′46″E) in November 2021, the time to suspend rice seeding from the rice fields in the central provinces of Vietnam. Snail eggs after harvest were incubated in the laboratory at 25 ± 2°C, 70 ± 5% humidity. After hatching, snails were fed water spinach leaves (Ipomoea aquatica) and raised to a size of 3 to 5 mm (length) in about 2 to 3 weeks. P. canaliculata snail was identified by Dr Nguyen Huy Hung.
Brine Shrimp (Artemia sp.)
Juvenile brine shrimps (3–4 mm) were collected at Danang city, Vietnam. Brine shrimps after collection were cultured at salinity 3.5%, pH 7.5 to 8.0, temperature 25 ± 2°C, humidity 70 ± 5%, cycle 12 h dark: 12 h light, fed algae during growth. Adult brine shrimps were 9 to 10 mm long (10–12 days from the collection date).
Rice
The original 13/2 rice provided by Quang Nam Agricultural Seed Joint Stock Company in Vietnam was used for germination assay and seedling toxicity test. The soil used for the seedling rice toxicity test was collected in Dien Ban District, Quang Nam province, Vietnam (15°55′58″N 108°11′46″E), in the rice field where golden apple snails were collected. After collection, the soil was loosened before testing.
Extraction and Isolation
Fresh roots of A. armata (5 kg) were dried and pulverized and then extracted with methanol (MeOH) in an ultrasonic bath 4 times (30 min each). The combined extracts were evaporated to obtain MeOH extracts (135 g). The MeOH extracts were partitioned by the liquid–liquid extraction with dichloromethane and ethyl acetate successively. The afforded aqueous fraction was chromatographed on a diaion column (HP-20) with an aqueous solvent to remove salts and oligosaccharides. Next, the collected sample was eluted on a chromatographic column with MeOH/water (m/w) solvents (25%, 50%, 75%, and 100% volume of MeOH), obtaining four major fractions (AA1–AA4).
Fraction AA2 (12.0 g) was chromatographed on a silica gel column, eluting with dichloromethane/methanol (D/M) (1/0–0/1, v/v) to obtain four fractions, AA2A–AA2D. The AA2A fraction (6.3 g) was eluted by the reversed-phase C18 gel chromatographic column using the solvent system MeOH/water (3/4, v/v) to obtain five subfractions, AA2A1–AA2A5. Subfraction AA2A2 (521 mg) was purified on an HPLC (J'sphere ODS-H-80 column 250 mm × 20 mm, eluting with 20% acetonitrile in water, a flow rate of 3 mL/min) to obtain three compounds
Fraction AA4 (15.0 g) was chromatographed on a silica gel column, eluting with dichloromethane/methanol (1/0–0/1, v/v) to give four fractions AA4A–AA4D. Fraction AA4A (3.5 g) was chromatographed on a reversed-phase C18 column, eluting with methanol/water (3/3, v/v) to give four subfractions AA4A1–AA2A4. Subfraction AA4A3 (890 mg) was further purified by HPLC (J'sphere H-80 column, 250 mm length × 20 mm ID, eluting with 20% acetonitrile in water, a flow rate of 2 mL/min) to give compounds
Molluscicidal Activity
Molluscicidal activity tests of pure compounds were performed according to the procedure of Ding et al 63 with slight modifications. For the assay, the prepared samples were dissolved in DMSO (1% stock solution), placed in 150 mL distilled water, and added 20 snails into each beaker. The sets of controls using tea saponin and DMSO were treated as positive and negative controls, respectively. Tests were performed with concentrations of 100, 50, 25, 12.5, 6.0, 3.0, and 1.5 μg/mL; and concentrations of 50, 25, 12.5, 6.0, 3.0, and 1.5 μg/mL for the positive control (tea saponin), repeating the experiment 4 times at each concentration. The experiment was conducted at a temperature of 25 ± 2 °C with a photoperiod of 12 h:12 h (light/dark). After 24 h, the treated snails were transferred to beakers containing 150 mL of distilled water for recovery. After the next 24 h, the snails were assessed for mortality (immobility, does not attach to the wall of the beaker, and retraction inability of the head inside the shell in response after being lightly pushed by a blunt object). The number of dead/live snails was recorded.
Acute 48 h Toxicity Test for Artemia sp.
The experimental method with Artemia sp. was referenced based on the procedure of Cong et al. 64 Test samples were prepared by dissolving in DMSO (1% stock solution). The prepared sample was then dissolved in a distilled water solution containing 3.5% NaCl (solution 1). Twenty adult brine shrimps were placed in a beaker containing 150 mL of solution 1 with a certain concentration, and the same was done for the blank control (only brine shrimps with 3.5% NaCl solution and 1% DMSO). The individuals were not fed during the experiment. The experiment was carried out at concentrations of 300, 250, 200, 150, 100, 50, and 25 μg/mL, repeated 4 times at each concentration with the condition of temperature 25 ± 2 °C, humidity 70 ± 5%, cycle 12 h dark: 12 h light. After 24 and 48 h, the number of dead/living individuals was recorded. The individuals that stopped moving within 10 s were determined to be dead.
Germination Assay
Germination assay was performed based on the method of Abedin and Meharg 65 with some modifications. The test was performed using the aqueous fraction of A. armata root extract, near the mean lethal concentration of the aqueous fraction and tea saponin for the golden apple snail, of 23 µg/mL and at the concentration 10 times higher than that value of 230 μg/mL. Twenty-five rice seeds were placed in a petri dish 4 cm in diameter; the experiment was repeated 4 times at each concentration. Rice seeds were washed 3 times with distilled water before soaking with 2 mL of distilled water for 24 h. Next, filter papers (9 cm in diameter) moistened with distilled water were used to wrap the rice seeds and placed in a petri dish with a lid. The experiments were conducted in the condition of temperature 25 ± 2 °C, humidity 70 ± 5%, cycle 12 h dark:12 h light. After 2 days, the percentage of germinated and nongerminated rice seeds was recorded.
Seedling Toxicity Test
Toxicity to rice at the seedling stage was carried out based on the method of Khan et al 66 with a few modifications. Rice seeds in the germination assay continued to be used for this trial. The trial was performed on the aqueous fraction of A. armata root as above (23 and 230 µg/mL). The blank sample considered as a negative control was also performed using water without any agent. The experiment was conducted at the Medicinal Garden, Duy Tan University, Vietnam. The experiment was carried out in conditions of temperature 28 °C to 33 °C, humidity 65% to 85%, and cycle 12 h dark:12 h light. The pots (20 cm high, 10 cm diameter) were filled with soil, and 300 mL of the test solution was added to soften the soil, which is favorable for sowing. Five seeds were sown in each pot, and the experiment was repeated 10 times at each concentration. The heights of the rice plants were measured after 7, 14, 21, and 28 days of growth. On day 28, the roots of rice plants were measured. The height of the rice plant was measured from the culm base to the tip of the longest leaf, and the root length was measured from the junction between the stem and the base of the plant to the tip of the longest root. Mean values of rice plant height and root length were recorded.
AChE Enzyme Inhibitory Test
Acetylcholinesterase (AChE) inhibitory activity of essential oil was performed according to the method described by Ellman et al 67 and our previous study. 35 The stock solution was obtained by dissolving the essential oil in DMSO (Merck), which was then diluted with H2O (deionized distilled water) to obtain different experimental concentrations. Each solution mixture consisted of 140 μL of phosphate buffer solution (pH 8), 20 μL of essential oil at concentrations of 500, 100, 20, and 4 μg/mL) and 20 μL of the enzyme AChE (0.25 IU/mL). The reaction mixtures were transferred to the test wells of a 96-well microtiter plate and incubated at 25 °C for 15 min. Then, solutions of 10 μL dithiobisnitrobenzoic acid (DTNB; 2.5 mM) and 10 μL acetylthiocholine iodide (ACTI; 2.5 mM) were added to each of the test wells and incubation continued for 10 min at 25 °C. At the end of the experiment, the absorbance of each solution was measured at 405 nm. Galantamine was used as a positive control. The negative control well did not contain the test sample. Each test was carried out in triplicate.
Data Analysis
Median lethal concentration (LC50) values were computed via log-probit analysis 68 using Minitab® version 19.2020.1 (Minitab, LLC) with 95% confidence limits. The differences between treatments were controlled by the Euclidean distance.
Homology Modeling
Homology models for several P. canaliculata proteins, available from the Protein Database, 61 were constructed from crystal structure templates available in the Protein Data Bank 60 using the SWISS MODEL server. 69 Three-dimensional structural models of P. canaliculata acetylcholinesterase (PcAChE), P. canaliculata alanine aminotransferase (PcALT), P. canaliculata aspartate aminotransferase (PcAST), P. canaliculata heat-shock protein 70 (PcHsp70), P. canaliculata tyrosine hydroxylase (PcPhOH), and P. canaliculata actin-binding protein (PcVillin) were generated based on multiple-threading alignments; the global model quality estimation was used to rank models (Table 7).
Pomacea canaliculata Target Protein Structures from Homology Modeling.

Molecular Docking
Molecular docking of the A. armata saponin ligands (
Conclusions
The molluscicidal activity of 15 purified saponins separated from A. armata roots grown in Vietnam against golden apple snail P. canaliculata has been investigated in this study. The structures of the compounds are determined by spectroscopic and spectrometric analyses as well as comparisons with the previously published data. The correlation between the saponins and their molluscicidal activity is also unveiled. The robust molluscicidal activity of these isolated saponins exhibited LC50 values ranging from 7.90 to 17.50 µg/mL. Experiments show that at the median lethal concentration for P canaliculata, the active ingredients have no significant impact on the aquatic environment. Specifically, the LC50 values of the compounds for brine shrimp (Artemia sp.) range from 148.55 to 193.22 μg/mL. In addition, the data obtained from the experiment also demonstrate the safety of the A. armata root extract. Based on molecular docking, P. canaliculata acetylcholinesterase and villin may be considered molecular targets of the A. armata saponins isolated and characterized in this work. There is much that is still unknown about the proteomics of P. canaliculata, however, and there may be other, yet uncharacterized, protein targets accounting for the molluscicidal activity of A. armata components. The obtained results illustrate that the saponin compounds from A. armata roots in Vietnam turn out to be potential compounds with excellent efficacy and safety for agricultural applications.
Footnotes
Author Contributions
THCN, GTKL, PHY, T-TH, DTTV, PVK, NHH, P-CK, and WNS contributed to conceptualization; THCN and NHH contributed to formal analysis; THCN and PVK contributed to writing‒original draft preparation; THCN, NHH, P-CK, and WNS contributed to visualization; GTKL, PHY, T-TH, and DTTV contributed to writing‒review and editing. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or vertebrate animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
