Abstract
Objective
This study investigates how hydroalcoholic and ethanolic extracts from
Methodology
Using spectrofluorimetric and Thioflavin T fluorescence tests, the activity of plant extracts (ethanolic and hydroalcoholic) on albumin glycation and Aβ25-35 self-aggregation was examined. To comprehend the effects of plant extracts on inflammation, macrophages were subjected to plant extract treatment and, subsequently assessed for, cell viability, cytokine production (TNF-α, IL-6, and IL-10), and nitric oxide generation using XTT assay, ELISA, and nitric oxide estimation kit, respectively. Additionally, the DPPH and ABTS assays were used to evaluate the plant extracts capacity to scavenge radicals, and the total phenolic content was determined.
Results
Among the 12 plant extracts examined,
Conclusion
The results demonstrated that
Introduction
The biological phenomena of aging are characterized by a decline in function at both the cellular and molecular levels. This decline increases the threat of developing several chronic illnesses, such as Type 2 diabetes mellitus (T2DM) and Alzheimer's disease (AD). 1 According to recent research, AD may be a metabolic illness, and there may be a connection between T2DM and the associated mechanisms2‐5. The main factors associated with age-related diseases are glycation, amyloid beta (Aβ) aggregation, inflammation, and oxidative stress6‐8. Glycation, the non-enzymatic reaction between the carbonyl group of sugar and the free amino group of protein, eventually forms stable advanced glycation end products (AGEs). 9 These AGEs accumulate in different organs of the body, such as the kidney, liver, brain, and eyes, leading to chronological aging.10,11 Increased ROS production by AGEs activates the downstream pathways related to amyloid β (Aβ) production and progresses development of AD. 12
Aβ protein contains two self-aggregating fragments - Aβ25–35 and Aβ1–42. Among these two, Aβ25–35 has a higher tendency to self-aggregate. 13 Importantly, Aβ25–35 is physiologically present in older adults, and AD patients post-mortem have shown its presence in their brains. 14 Interestingly, microinjection of Aβ25–35 into the rat hippocampus induced neurotoxicity, potentially through the activation of the receptor for advanced glycation end products (RAGE). 15 Aβ binding to the RAGE can initiate pathways associated with decreased cerebral blood flow, blood vessel issues, and cell death. Additionally, Aβ deposits can cause an inflammatory response linked to several age-related diseases. 16 Activation of RAGE modulates signaling cascade eventually causing inflammation and oxidative stress. 17 Chronic low-level inflammation associated with aging is frequently called “inflammaging.” Numerous age-related illnesses, including diabetes, cancer, heart disease, and brain issues, are related to this inflammation. Our immune systems deteriorate with age, a phenomenon known as immunosenescence. 18 The study by Li et al, (2023) showed the increased serum levels of pro-inflammatory cytokines, such as interleukin 1, 6, 8 (IL-1, IL-6, IL-8) and, tumour necrosis factor α (TNF-α) in aged individuals. 18 Further studies also reported the positive correlation of TNF-α and IL-6 with ageing, indicating an interrelated activation of the entire inflammatory cascade. 18 In summary, glycation, amyloid formation, and inflammation are interconnected processes contributing to ageing and age-related diseases.
A small-molecule antagonist of the RAGE i.e. azeliragon extensively studied for its effect in mitigating the ageing associated diseases.
18
Despite promising early-stage clinical trials, the drug failed in third phase of clinical trials showed deleterious effect on cognitive behaviour, increased blood glucose level and increased metabolic risk in ageing population.
19
Therefore, a multipronged therapeutic approach with anti-glycation, anti-amyloid, and anti-inflammatory measures would be more effective in retarding the development of age-related disorders. Throughout India and in the Indian system of medicine, the value of ethnomedicine has been realized, and a majority of work is being done on these traditional plants to identify the potential bioactive natural products. Indian medicinal plants that are known in Ayurvedic system of Medicine for their antiaging properties including antioxidant, neuroprotective, anti-inflammatory and memory boosting capabilities. In addition, majority of these active medicinal plants have only been evaluated in preliminary stages till extract level and mechanistic study for specific activity are yet to be elucidated. Hence, the medicinal plants
Materials and Methods
Chemicals
The selection of reagents was based on their relevance and proven efficacy in the experimental setup. Aβ25-35 fragment (A4559), 1,1,1,3,3,3 - Hexafluoro - 2 - propanol (HFIP-H0424), Aminoguanidine (AG - 369494), Benzoquinone (12309), Methylglyoxal (MGO - M0252), Thioflavin T (3516), and 8-Anilinonaphthalene-1-sulfonic acid (ANS - A1028) were procured from Sigma Aldrich (St. Louis, USA). Bovine serum albumin (BSA - 97350), 2, 2′-diphenyl-1-picrylhydrazyl (DPPH - 29128), and 2, 2′ azinobis (3 ethylbenzothizoline-6-sulphonic acid (ABTS - 28042), Gallic acid (5995-86-8), Ascorbic acid (16544), Sodium hydroxide (NaOH - 15895), Dimethyl sulfoxide (DMSO – 30239), Alanine (64422) were purchased from Sisco research laboratories (India). EZcountTM XTT cell assay kit (CCK015), Trypsin (TCL031), Antibiotic antimycotic reagent (A001), and EZAssayTM nitric oxide estimation kit (CCK061), Glycine (MB013) were procured from Himedia (India). The ELISA kits, such as TNF-α (88−7324), IL-6 (88-7064-77), and IL-10 (88-7105) were purchased from Invitrogen (Waltham, US). Dulbecco's modified eagle medium (DMEM-12100046) was procured from Gibco (Waltham, USA).
Collection of Plant Materials and Extract Preparation
The selected medicinal plants were 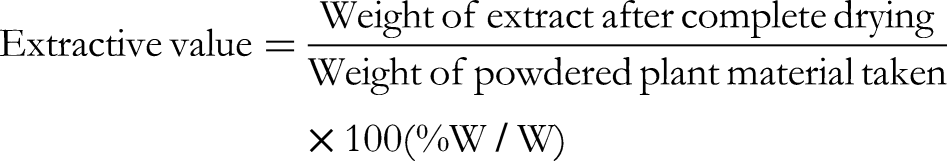

Schematic Representation of the Preparation of Plant Extracts.
Extractive Yield of the Selected Plant Materials.

For analysis, 100 mg of plant extract was first dissolved in 1 mL of DMSO to prepare a stock solution. A working solution of 100 µg/mL was then prepared in 1× PBS and used to evaluate the extract's anti-glycation, anti-amyloid, anti-inflammatory, and antioxidant properties.
In-Vitro Glycation of BSA
The BSA was glycated using a previously reported method with slight modifications. 30 Equal volumes of BSA (10 mg/mL) and glycating agent MGO (55 mM) were incubated simultaneously with or without plant extracts (100 µg/mL) for eight days at 37 °C. AG, a standard glycation inhibitor, was used as reference to evaluate anti-glycation activity. The experimental controls include: i) BSA (2 mL) + PBS (8 mL) as the negative control, ii) BSA (2 mL) + MGO (2 mL) + PBS (6 mL) as the positive control, and iii) BSA (2 mL) + MGO (2 mL) + AG (2 mL; 100 µg/mL) + PBS as the standard inhibitor controls. The All the reactions were performed in triplicates. After incubation, unbound MGO and plant extracts were removed by extensive dialysis for 48 h against PBS at 4 °C. The dialyzed samples were filtered through a membrane (0.22 µm) and stored at −20 °C till further analysis. The protein contents of all samples were assessed using the Bradford method.
Estimation of Anti-Glycation Activity of Plant Extracts
Estimation of AGEs Formation and Intrinsic Fluorescence
The formation of fluorescent AGEs such as malondialdehyde (MDA)-modified protein, pentosidine (P) like fluorescence, crossline, and intrinsic fluorescence of tryptophan were determined on a multimode microplate reader (BioTek Synergy H1, Vermont USA) using a previously reported method. 31 Samples (200 µg/ml) were excited at 370, 335, 485, and 280 nm, and emissions were collected at 440, 400, 530, and 310 nm (slit = 10 nm), respectively. The results were represented as Arbitrary Units (A.U.)/mg protein.
Free Amino Groups Modifications
The ρ-benzoquinone was used to estimate the free amino groups of protein in the sample. 32 Briefly, 100 µL of the glycated samples were incubated with 100 µL of PBS (pH 7.4) and 40 µL of benzoquinone; incubated at 37 °C for 30 min. The absorbance of the mixture was read at 480 nm. The amino group content was calculated with the help of standard curve of Alanine (0-100 nM) and results were represented as nM/mg protein.
Hydrophobicity Analysis by 8-Anilinonaphthalene-1-Sulfonic Acid (ANS)
The hydrophobicity of the treated BSA samples was analyzed using the ANS dye. 33 The ANS dye was used to analyse the monitor protein conformational changes through its affinity for hydrophobic protein surfaces. The 200 µg/mL of glycated BSA samples was incubated with 5 µL of ANS dye and mixed thoroughly. The fluorescence of the samples were measured at excitation wavelength 355 nm and emission wavelength of and 440 nm respectively and represented as Arbitrary Units (A.U.)/mg protein.
Anti-Amyloid Activity of Plant Extracts
To analyse the anti-amyloid activity, the Aβ25-35 fragment (1 mg/mL) was activated by dissolving in HFIP solution. The solution further sonicated for 15 min at room temperature. Further, the sonicated Aβ25-35 fragment was reconstituted in 50 mM phosphate buffer (pH 7.4) with a final peptide concentration of 200 μM. The 100 µL of activated fragment was incubated with 100 µL of plant extracts (100 µg/mL) or the standard inhibitor curcumin (100 µg/mL) and incubated at 37 °C for 100 h. 34 After incubation, the samples were analysed for the self-aggregation ability of Aβ25-35 fragment.
Thioflavin T Assay
The extent of Aβ25-35 aggregation in the presence of plant extracts, Thioflavin T was used due to its high sensitivity and specificity in detecting amyloid fibril formation through enhanced fluorescence. A volume of 75 µL of incubated samples was added to 675 µL of 25 µM Thioflavin T in a 50 mM glycine-NaOH buffer (pH 8.5), mixed thoroughly, incubated for 5 min at room temperature. The fluorescence was measured at 446 nm (excitation) and 490 nm (emission).
30
Percent inhibition of Aβ25-35 self-aggregation was calculated using the following formula:
Estimation of Antioxidant Capacity of Plant Extracts
2, 2’- Azino-bis (3-Ethyl Benzoline-6-Sulfonic Acid) (ABTS) Radical Scavenging Activity
The antioxidant capacity of plant extracts was determined using a standard method.
35
The ABTS solution was prepared by mixing an equal volume of ABTS (7 mM) and potassium Persulphate (2.45 mM). The solution was kept idle for 16 h without exposing it to light. The absorbance of the working solution was adjusted to 0.7 ± 0.005 at 734 nm. The radical scavenging activity of plant extracts was estimated by mixing 20 µL of the samples in 980 µL of the working ABTS solution, and absorbance of the sample was recorded at zero minutes and six minutes. The ascorbic acid (1 mg/mL) was used as a positive control.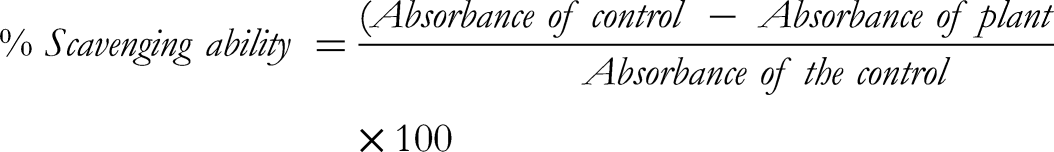
α-diphenyl-β-picrylhydrazyl (DPPH) Radical Scavenging Activity
The DPPH radical scavenging activity was estimated by measuring the decrease in the absorbance of the methanolic solution of DPPH.
36
100 µL of each plant extract was mixed with 500 µL of DPPH solution (3 mg/mL), and the mixture was incubated for 30 min in a dark environment. The sample's absorbance was measured at 517 nm. The control was ascorbic acid (1 mg/mL). The following formula was used to determine scavenging ability:
Total Phenolic Content
The phenolic content in the plant extracts was estimated using the reported method. 37 For estimation, 0.3 mL of plant extracts was added to 1.5 mL of freshly prepared 1N Folin–Ciocalteu reagent. After thorough mixing, 1.2 mL of 7.5% sodium carbonate solution was added, and the reaction mixture was incubated in the dark for 30 min at room temperature. The absorbance was then recorded at 765 nm using a spectrophotometer. Gallic acid (0.1-1 mg/mL) was used as the standard reference, and the total phenolic content was quantified as mg of gallic acid equivalents (GAE)/mL of the sample.
Estimation of Anti-Inflammatory Activity of the Plant Extracts
XTT Assay
To evaluate the cytotoxicity of plant extracts on the macrophages (J774A.1 cell line), XTT assay was performed. For this purpose, macrophages were pre-seeded in a 96-well plate at a density of 20,000 cells/well. They were exposed to varying concentrations (0.1 and 0.01 mg/mL) of selected plant extracts under the cell culture conditions (37 °C and 5% CO2). Cell viability was assessed after 24 and 48 h of treatment with plant extracts. Cells treated with LPS (2 μg/mL), or left untreated were considered positive, and negative controls. To check the cell viability after 24 and 48 h of incubation, 50 μL of freshly prepared XTT reagent (HiMedia) was added to each well containing cultured macrophages and kept in a CO2 incubator at 37 °C with 5% CO2 for 4 h. Post-incubation, absorbance was measured at 450 nm using a microplate reader. Percentage cell viability was calculated using the following formula:
Cytokine Assay
Enzyme-linked immunosorbent assay (ELISA) kits (Invitrogen) specific to IL-6, TNF-α, IL-10 were used to estimate the levels of these cytokines in the culture supernatants of variously treated cells. The assay was carried out following the manufacturer's manual.
Estimation of Reactive Nitrogen species (RNS)
RNS in the supernatants of the cultured cells was estimated by measuring the dissolved nitric oxide (NO) using a commercially available kit (HiMedia). The assay was carried out following the manufacturer's manual.
Statistical Analysis
The data were presented in the form of mean ± SD. Data was analyzed using GraphPad Prism (version 8.0.1). One-way ANOVA with post hoc Tukey's test was applied to determine the statistical significance if not stated otherwise. The values ****p < 0.0001, ***p < 0.001, **p < 0.01, and *p < 0.05 were considered significant in comparison to the negative control whereas, ####p < 0.0001, ###p < 0.001, ##p < 0.01 and #p < 0.05 was considered significant in comparison to the positive control. Correlation analyses were conducted in all study groups using Pearson's correlation to assess the associations between various parameters. The results were represented as the correlation coefficient (r). Significance levels were considered as follows: The levels of significance were denoted as: a(p < 0.001), b(p < 0.01), and c (p < 0.05).
Results
Extractive Yield of the Plant Extracts
The hydroalcoholic and ethanolic extractions of the selected six plants were performed, and their respective extractive yields were calculated. The yields ranged between 35.42% to 5.94%. For all the plants, hydroalcoholic yield was higher than the ethanolic yield (Table 1).
Anti-Glycation Activity of Plant Extracts
The fluorescent AGEs are heterocyclic aromatic amines that exhibit fluorescence at a specific wavelength and demonstrate characteristic peaks. The inhibition of AGEs formation was evaluated by measuring three different fluorescent AGEs such as MDA, pentosidine, and crossline (Figure 2a-c). As depicted in Figure 2a, native BSA showed a minimal MDA-modified protein formation compared to positive control. The presence of standard inhibitor (AG) depleted the fluorescence (p < 0.0001) as compared to glycated BSA. Interestingly, a reduction in MDA-modified protein was noted in the presence of plant extracts (ethanolic and hydroalcoholic), indicating their ability to inhibit MGO-induced conversion of BSA to AGEs. Among all the plant extracts,
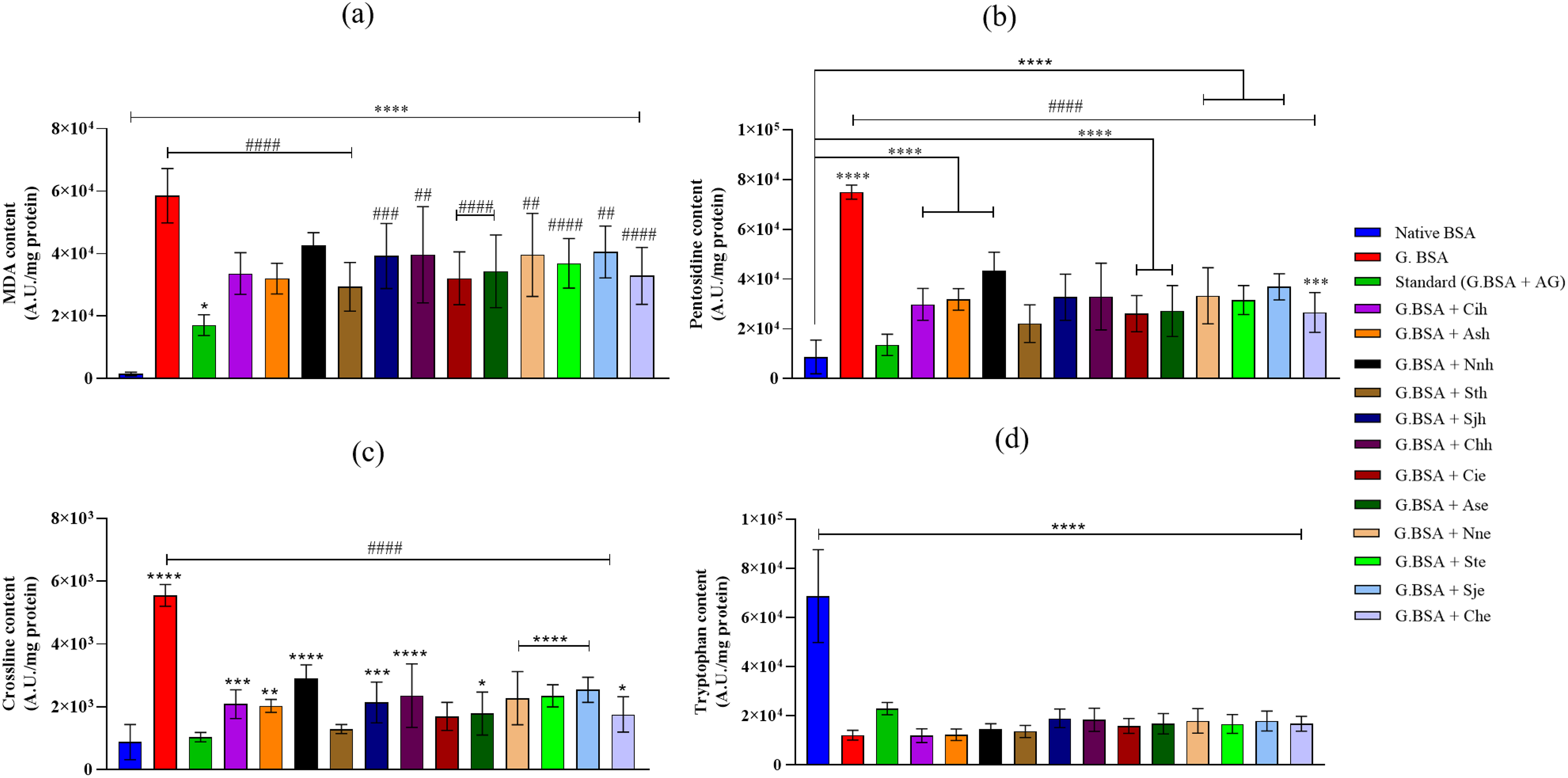
Presence of Hydroalcoholic and Ethanolic Plant Extracts (100 µg/mL) with Glycated BSA Inhibited the AGEs Content and Protected the Tryptophan Fluorescence - a) Malondialdehyde (MDA)-Modified Protein Fluorescence b) Pentosidine (P)-Like-Fluorescence c) Crossline d) Tryptophan. The data is Represented as Mean ± SD (n = 9). (Abbreviations: BSA-Bovin serum albumin, G.BSA-Glycated BSA, AG-aminoguanidine, Cih-
Effect of Plant Extracts on Free Amino Groups of BSA
The albumin has 59 lysine and 24 arginine, which makes it more prone to glycation. The process of glycation leads to the depletion of the free amino groups of protein. Native BSA showed the maximum free amino groups as compared to the treatments (Table 2). Presence of MGO alone with BSA showed depletion of the free amino groups of protein as compared to native BSA (p < 0.0001). Compared to glycated BSA, standard inhibitor AG, and plant extracts did not show any significant protection toward free amino groups of the protein.
Effect of Hydroalcoholic and Ethanolic Plant Extracts on Free Amino Groups and Hydrophobic Region of Glycated BSA.
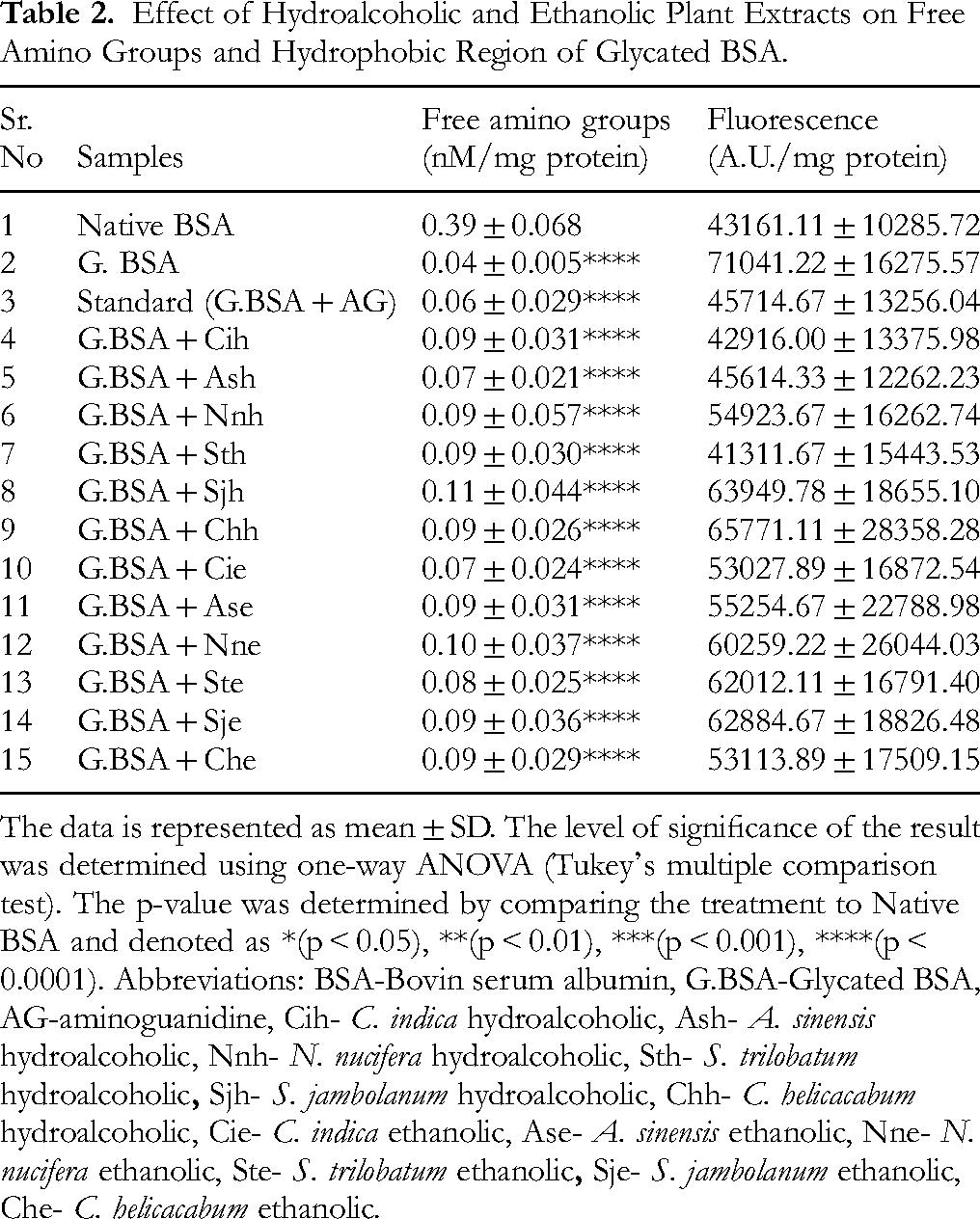
The data is represented as mean ± SD. The level of significance of the result was determined using one-way ANOVA (Tukey's multiple comparison test). The p-value was determined by comparing the treatment to Native BSA and denoted as *(p < 0.05), **(p < 0.01), ***(p < 0.001), ****(p < 0.0001). Abbreviations: BSA-Bovin serum albumin, G.BSA-Glycated BSA, AG-aminoguanidine, Cih-
Effect of Plant Extracts on the Hydrophobic Region of BSA
The impact of plant extracts on the hydrophobicity of BSA was assessed using ANS, which fluoresce upon binding to the exposed hydrophobic regions of the partially folded proteins. As seen in Table 2, glycated albumin exhibited increased ANS fluorescence as compared to native albumin (p < 0.05), whereas the presence of AG decreased the fluorescence as compared to glycated BSA. A significant decrease in fluorescence was observed for Cih and Sth extracts (p < 0.05) compared to glycated BSA.
Anti-Amyloid Activity of Plant Extracts
Extracellular aggregation of Aβ peptides is one of the major clinical hallmarks of AD. Aβ25−35 is produced in aged brains by proteolytic cleavage of soluble racemized Aβ1−40 peptides and forms aggregates. Therefore, we evaluated the effect of plant extracts on the self-induced aggregation of Aβ25−35. The plant extract showed more than 50% inhibition in amyloid aggregate formation. Among all extracts, Ash showed maximum inhibition (87.8%), followed by Cih (77.8%)

Plant Extracts (Hydroalcoholic and Ethanolic) Inhibited the Self-Aggregation of the Aβ25-35 Fragment (200 µM). the bar Graphs Represent Mean ± SD (n = 9). (Abbreviations: Cih-
Effect of Plant Extracts on Cell Viability of Macrophages
To evaluate the immunomodulatory properties of plant extracts, the murine macrophage cell line (J774A.1) was treated with various plant extracts, and first, their impact on cell viability was assessed. For this purpose, four plant extracts showing positive anti-glycation and ant-amyloid activities were chosen. Except

Relative Cell Viability of Cultured Macrophages After Treatment with Various Plant Extracts (0.1 mg/ml or 0.01 mg/ml) for a) 24 and b) 48 h as Determined by XTT assay. The Bar Graphs Represent Mean ± SD (n = 3). (Abbreviations: LPS-lipopolysaccharide, Chh-
Effect of Plant Extracts on Macrophage Activation
To test the immunomodulatory impact of plant extracts on macrophages, culture supernatants of treated cells were tested for the presence of secretory pro-inflammatory (TNF-α, and IL-6) and anti-inflammatory (IL-10) cytokines. To check the cytokine production, a lower concentration (0.01 mg/ml) of plant extracts was chosen for treatment as the cell viability at this concentration had not been affected significantly. With respect to TNF-α production, only

Production of Cytokines and Reactive Nitrogen species (RNS – NO) by Cultured Macrophages After Treatment with various Plant Extracts (0.01 mg/ml) as Determined by ELISA (Cytokines) or NO Estimation kit. a) TNF-α b) IL-6 c) IL-10 d) relative NO Levels. the bar Graphs Represent Mean ± SD (n = 3). (Abbreviations: TNF-α- Tumor Necrosis Factor-α, IL-Interleukin, NO-Nitric Oxide, LPS-Lipopolysaccharide, Chh-
Radical Scavenging Activity of Plant Extracts
The antioxidant potential of plant extracts was measured by ABTS and DPPH assay (Table 3) to understand the mode of action by which plant extracts inhibit the glycation process. Plant extracts’ ABTS radical scavenging activity ranged from 5% to 62%. Sjh and Cie extracts exhibited the maximum activity, whereas Che extract showed minimum activity (Table 3). Furthermore, the plant extracts showed elevated DPPH radical scavenging ability ranging from 20% to 88%. Maximum ABTS radical scavenging activity was observed for Sjh and Cie. Sje showed minimum radical scavenging activity.
Radical Scavenging Ability and Total Phenolic Content of the Plant Extracts.
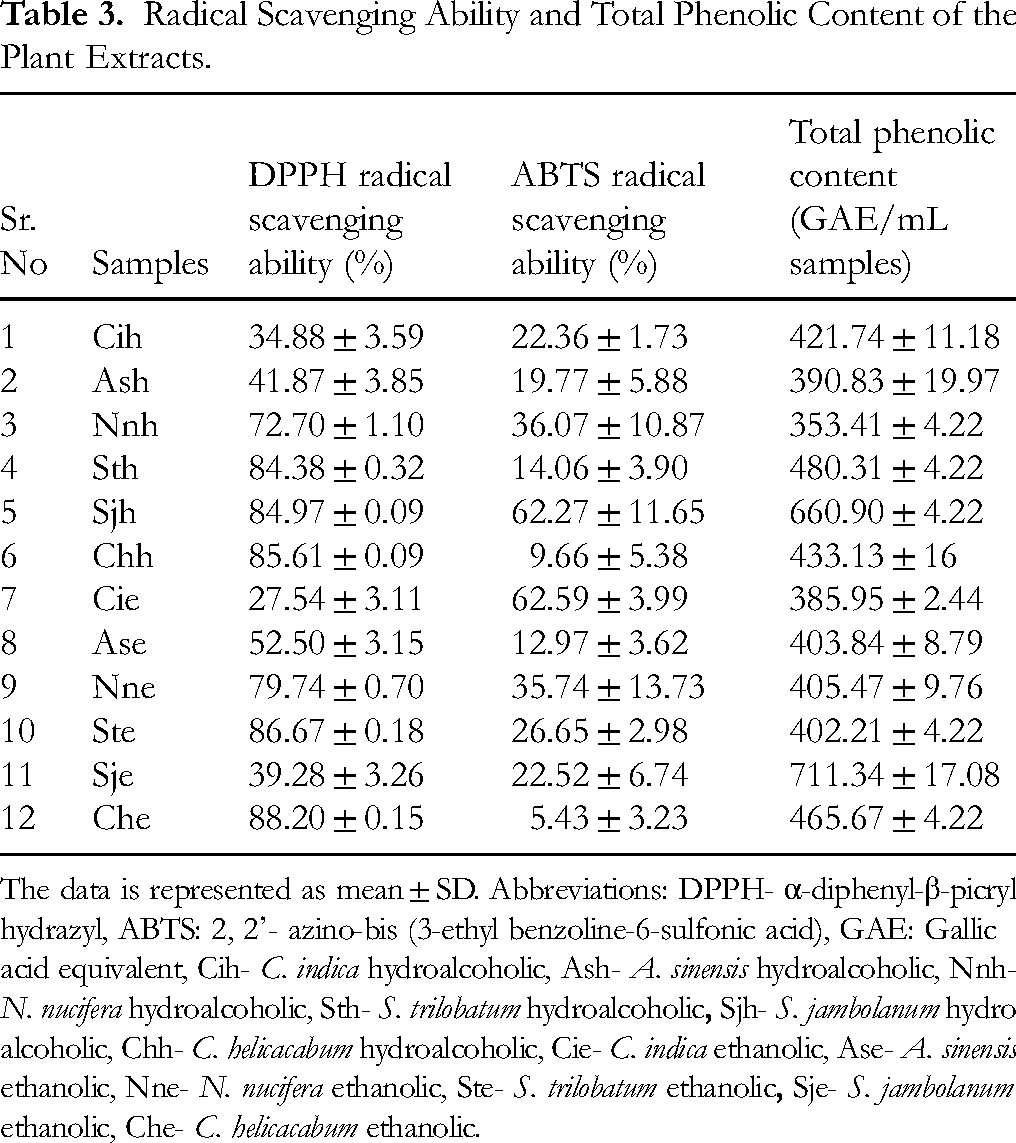
The data is represented as mean ± SD. Abbreviations: DPPH- α-diphenyl-β-picrylhydrazyl, ABTS: 2, 2’- azino-bis (3-ethyl benzoline-6-sulfonic acid), GAE: Gallic acid equivalent, Cih-
Total Phenolic Content of Plant Extracts
As observed in Table 3, all plant extracts showed a wide phenolic content ranging from 259 to 711 µg GAE/ml sample. The Cih and Sje fractions showed the maximum phenolic content compared to other plant extracts. At the same time,
Correlation Analysis
The Figure 6a illustrates the relationship between total phenolic content (TPC) expressed as GAE/mL (y-axis) and percent yield (size of the bubbles) for various plant extracts. Sjh shows the highest TPC (∼700 GAE/mL) with moderate yield, indicating phenolic richness. Ash demonstrates both high TPC and yield, showcasing efficient phenolic extraction. Sth and Nnh show moderate TPC with varying yields, while Che has the lowest TPC (∼300 GAE/mL). Variations emphasize the importance of solvent and method optimization for maximizing phenolic recovery and yield. Further, to explore the mode of action, DPPH radical scavenging ability with respect to glycation, hydrophobicity, and anti-amyloid activities were analysed (Figure 6b). The hydroalcoholic extracts, particularly Sth and Sjh, stand out with high DPPH scavenging activity and Aβ-inhibition, coupled with lower MDA levels, showcasing their antioxidant and anti-amyloid potential. Che also shows promising results with high ANS hydrophobicity and moderate antioxidant properties, indicating its relevance in modulating protein stability and reducing oxidative stress. These findings highlight the therapeutic potential of these extracts in managing oxidative stress and amyloid-associated disorders (Figure 6b). The Table 4 highlights the correlation between DPPH radical scavenging ability and the levels of cytokine TNF-α and NO for various plant extracts. The Cih displayed the strongest negative correlation with TNF-α production and DPPH scavenging ability (p < 0.001), indicating its potential to modulate pro-inflammatory cytokines. Similarly, Chh exhibited a significant negative correlation with NO levels (p < 0.001) and a strong positive correlation with TNF-α (p < 0.001), highlighting its dual role in reducing oxidative stress and influencing immune responses. Conversely, Sth showed significant positive correlations with both TNF-α and NO levels, suggesting its potential to enhance these parameters. Other extracts, such as Sjh, Cie, and Sje, displayed weaker correlations, indicating a lesser dependence on these markers. These findings showed the differential abilities of the plant extracts in balancing antioxidant activity with the regulation of pro-inflammatory cytokines and NO production, with Cih, Chh, and Sth standing out for their promising therapeutic potential.

Relation of Plant Extracts with Different Activities. a) Bubble Plot with Total Phenolic Content and Extractive Yield. Colours were Attributed for Different Plant Extracts and the Size of the Bubble Represents Extractive Yield b) DPPH Radical Scavenging Activity, Aβ-Inhibition, MDA Content, and ANS Hydrophobicity of Different Plant Extracts. (Abbreviations: Chh-
Correlation Between DPPH Radical Scavenging Ability with TNF-α and NO Production.

The levels of significance were denoted as: a(p < 0.001), b(p < 0.01), and c (p < 0.05). Abbreviations: TNF-α-Tumor necrosis factor alpha, NO- Nitric oxide, : DPPH- α-diphenyl-β-picrylhydrazyl, Cih-
Discussion
One of the major contributors to the aging process is protein glycation, complex reactions where the sugar moiety non-covalently binds to free amino groups of protein and forms stable Amadori products. 38 MGO, a highly reactive dicarbonyl compound generated through glucose metabolism, is the primary precursor of AGEs formation. It is known to glycate protein by altering the lysine and arginine residues, thus affecting the secondary structure of the protein.39,40 These AGEs disrupt protein structure and function, promote the aggregation of amyloid fibrils, and exacerbate chronic inflammation, all of which are hallmarks of aging.5,8 The phytoconstituents such as quercetin, tannin, gallic, and ellagic acid hinder the early and middle stages of glycation by scavenging ROS and dicarbonyl compounds.41,42 These phytoconstituents exhibit the anti-glycation, anti-aggregation, and antioxidant activities due to the presence of the hydroxyl groups, making them effective in slowing the progression of aging and protecting against cellular damage. 43 The plant extracts with high phenolic and flavonoid content inhibit protein glycation modifications and further prevent cell damage particularly promising for combating glycation-related aging processes and associated pathologies.44,45
One of the main ingredients in senile plaques, a pathogenic feature of AD, is Aβ fragments. Both
The antioxidant potential of the plant extracts is well correlated to their phenolic content.73‐75 Hence, the antioxidant activity observed for the plant extracts in this study strongly suggests the presence of phenolic compounds. These molecular constituents, known for their electron-donating property, might be responsible for the observed decolorization of the DPPH and ABTS reagents. Our findings with respect to the antioxidant activity of plant extracts are consistent with an earlier study where the methanolic extract of
Plant-derived natural products have gained strong research attention because of the high content of anti-inflammatory and antioxidant compounds and their safety and affordability compared to synthetic alternatives.
53
For these reasons, extracts of some plants have selectively gained research focus for ethnobotanical studies and to explore their curative potentials. Innate immune cells, such as macrophages, play a significant role in inflammatory responses. Chronic and unregulated inflammatory responses are associated with immune-pathological conditions such as diabetes and AD. The downregulation of inflammatory mediators is essential to reduce the risk of escalation of auto-immune diseases. In this study, ethanolic and hydroethanolic extracts of four plants (
When tested for their effect on the growth of macrophages, in contrast to other plant extracts,
Cytokines play a key role in activating the immune cells and regulating their function. When tested for its impact on the production of inflammatory mediators by macrophages, plant extracts did cause measurable changes in the production of TNF-α and IL-6. Ethanolic extracts of all the tested plants increased the production of TNF-α and IL-6 considerably. Interestingly, both - hydroethanolic and ethanolic extracts of
IL-10 plays an essential role in the regulation of inflammatory responses. When tested for its impact on IL-10 production, plant extracts did not deliver significant changes at the tested concentration of 0.01 mg/ml. However, treatment with hydroethanolic and ethanolic extracts of
The phytochemical composition of medicinal plants is influenced by geographic origin, climate, and environmental conditions. Soil nutrient composition affects plant growth, while higher altitudes increase UV exposure, enhancing secondary metabolite production (eg, flavonoids, phenolics). Water availability influences stress-induced metabolite synthesis, such as alkaloids and terpenoids. Climatic factors (temperature, humidity, rainfall, sunlight) regulate enzymatic and metabolic processes, impacting phytochemical yield. 86 Additionally, plant varieties, cultivars, and agricultural practices (fertilizers, pesticides) can further modify phytochemical content. 87 Hydroalcoholic solvents, typically a combination of water and ethanol, can dissolve both hydrophilic compounds (such as polyphenols, flavonoids, glycosides, and alkaloids) and lipophilic compounds (such as terpenoids, steroids, and essential oils). 88 This dual solubility makes hydroalcoholic mixtures ideal for extracting a wide array of phytochemicals with varying polarity. Ethanol, a key component in this mixture, is particularly efficient in extracting bioactive compounds that possess varying degrees of lipophilicity. 89 Ethanol's ability to dissolve both polar and non-polar substances enhances its versatility compared to water alone, which mainly dissolves hydrophilic compounds. As a result, ethanol is commonly used for extracting compounds like flavonoids, terpenoids, and alkaloids that are often found in medicinal plants. 90 Additionally, ethanol's low boiling point makes it easy to remove, leaving behind a clean extract. Moreover, the combination of hydroalcoholic and ethanol extraction methods has proven to be efficient in yielding high-quality extracts. 90
Different solvent extraction techniques may result in diverse phytochemical compounds in plant materials.
91
The results showed hydroalcoholic extracts of
Despite the promising findings, this study has several limitations that warrant consideration. Firstly, the experiments were conducted in vitro, which may not fully capture the complexity of in vivo systems. Secondly, while the study focused on the anti-glycation, anti-inflammatory, and anti-amyloid properties of specific plant extracts, the active compounds responsible for these effects were not identified. Furthermore, cellular studies and animal models have not yet been explored, which are crucial for a more comprehensive understanding of the observed effects and their potential therapeutic implications.
Conclusion
All the plant extracts showed significant anti-glycation, anti-amyloid, anti-inflammatory, and anti-oxidant activity. Among all extracts, the
Footnotes
Acknowledgment
We would like to acknowledge Dr P. Santhan, PhD (Botany), Taxonomist, Chennai, for the authentication and identification of the collected plants. The authors would like to extend their sincere appreciation to the Ongoing Research Funding Program, (ORF-2025-694), King Saud University, Riyadh, Saudi Arabia.
ORCID iDs
Ethical Approval
Ethical approval is not applicable to this article.
CRediT authorship contribution statement
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Symbiosis International Deemed University Intramural Grant -Major Research Project [Grant no - SIU/SCRI/MJRP-Approval/2021-22/4180]. This work was supported by the Ongoing Research Funding Program, (ORF-2025-694), King Saud University, Riyadh, Saudi Arabia. The study was further supported by the Ministry of Science and Higher Education of the Russian Federation (Project No. 122042600086-7).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be available on request.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
