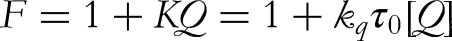Abstract
Keywords
Introduction
The contemporary global community faces significant challenges posed by recent diseases and disorders. Scientists are striving to develop improved therapeutic drugs with minimal adverse effects on human health to combat these conditions. 1 Diabetes has become a major health concern without a suitable drug for prevention. Increased sugar level impacts the structure and function of other biomolecules. The non-enzymatic interaction between proteins and sugars is a widely recognized process in cellular systems. It is associated with the production of harmful advanced glycation end-products (AGEs) and is thought to contribute to the development of secondary complications in various metabolic and cardiovascular disorders. 2 These products can contribute to increased aggregation and the generation of free radicals. Scientists have identified synthetic drugs and natural compounds to mitigate the formation of glycation-induced products (AGEs, aggregates, etc) and shield cells from their detrimental impact. 3
Albumin is crucial in transporting a wide range of metabolites, drugs, and dyes within the body and involves several indispensable physiological functions. Understanding the interactions between drugs and serum albumin is essential because administered drugs initially bind to serum proteins before they are distributed throughout the body to their target sites. 4 Research on how drugs interact with plasma proteins, including human serum albumin (HSA) and bovine serum albumin (BSA), holds significant importance in biological, biomedical, and pharmaceutical sciences, as strong binding can lower bioavailability and extend the half-life in vivo. 5 This binding process is instrumental in defining drugs’ absorption, distribution, metabolism, and excretion (ADME) characteristics, which is vital for the rational drug design process. 6 BSA is a valuable model for interaction studies due to its structural similarity to HSA. BSA and HSA share approximately 76% homology, with the primary distinction in their tryptophan amino acid residues (HSA has Trp-214, while BSA has Trp-134 and Trp-212). 7 Additionally, BSA possesses several advantages, including excellent ligand binding properties, cost-effectiveness, and accessibility. BSA has a molecular mass of 66,400 Da, constituting amino acid (583) residues. 4 These amino acid residues include 17 cysteine residues, which form a single chain through cross-linking. 8 This amino acid chain comprises three structurally distinct but homologous domains (I, II, and III). 9 BSA has features to bind with distinct sites with various specificities, with the most crucial sites referred to as Sudlow site-I and site-II. 10 The overall binding between ligands (drugs) and proteins is determined by various molecular forces at these binding sites, number of binding sites, and their affinity for the ligand. The presence of multiple binding sites highlights HSA's remarkable capacity to function as a primary storage and transport protein, enabling it to bind, carry, and deliver a wide variety of endogenous and exogenous compounds through the bloodstream to their target organs. 11 Achieving equilibrium constants between ligands and proteins is essential for understanding these interactions. 12
Vitamins play a crucial role in the defense system of the body. Vitamins, particularly ascorbic acid (Figure 1A), are potent antioxidants that can help in protecting the body from glycation. Ascorbic acid is the major water-soluble antioxidant in blood plasma which plays a primary defense against oxidants and free radical attacks. 13 Vitamins have been proven to safeguard cells and other bodily structures against harm arising from the actions of free radicals. 14 Pyridoxal-5-phosphate (PLP) (Figure 1B) can protect against diabetic complications by reducing the formation of AGEs in the body. PLP may also help reduce inflammation, a contributing factor in the development of diabetes and its complications. 15 Overall, vitamins B and C are used in day-to-day benefits for better physiological health. These vitamins are commercially marked by neutrapharmaceuticals. This scenario draws attention to checking their effect on the glycation process in diabetic conditions.
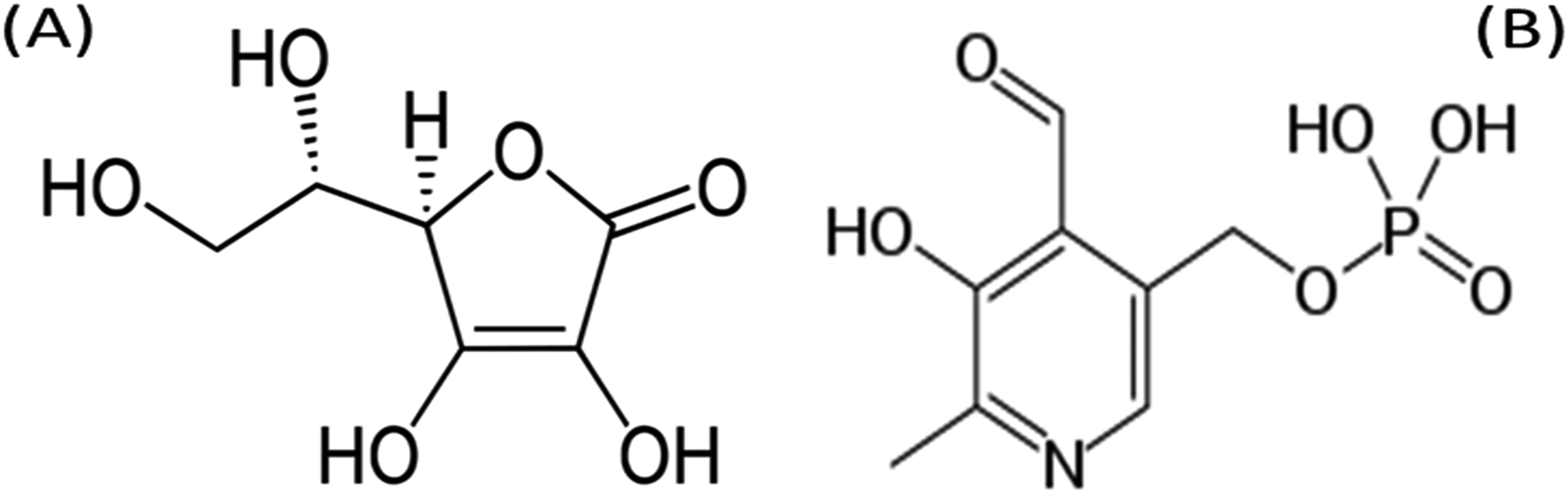
Molecular structure of (A) ascorbic acid and (B) pyridoxal-5-phosphate.
The present study is carried out to investigate the role of vitamins (Ascorbic acid and pyridoxal-5-phosphate) in the protein (BSA) glycation and aggregation of β-amyloid structures spectroscopically. The interaction between the AA/PLP-BSA complex provides insights into binding constant and affinity between the complex. It fulfils the research gaps by evaluating early, intermediate, and late-stage glycation products in the presence of ascorbic acid and pyridoxal-5-phosphate in 28 days. Moreover, aggregation of protein is assessed in the glucose-induced glycation. These vitamins are also evaluated for the conformational changes caused by glycation.
Material and Methods
Materials
Ascorbic acid, Bovine serum albumin, glucose (Glc), Pyridoxal-5-phosphate, and Thioflavin-T (ThT) were procured from Sigma-Aldrich. Nitro blue tetrazolium (NBT) and Di-nitrophenyl hydrazine (DNPH) were purchased from HiMedia. All other chemicals are of high-grade quality.
Fluorescence-based Interaction Studies
The fluorescence emission spectra for AA and PLP were measured within the range of 300 to 450 nm, employing a consistent excitation wavelength of 280 nm. Fluorescence titrations were conducted on BSA with a constant concentration of 0.5 mg/ml while varying the concentrations of AA and PLP from 0 to 7 µM at a temperature of 37 °C. All fluorescence emission spectra were captured using an Agilent Cary Eclipse spectrofluorometer. The excitation and emission slit widths were maintained at 5 nm.
4
The inner filter effect of AA and PLP fluorescence was eliminated with the following formula:
Glycation Sample Preparation
BSA (10 mg/ml) and glucose (100 mg/ml) were incubated in phosphate buffer (100 mM, pH 7.4) and sodium azide (3 mM) in the presence/absence of AA and PLP (both 25 mM) for 28 days at physiological temperature, 37 °C. The incubated protein samples were collected and stored at −20 °C after the completion of the incubation period for further analysis.16,17
Measurement of Browning
The generation of browning in the glycation process can be assessed to evaluate the extent of glycation products at 420 nm spectrophotometrically.
18
The inhibition percent was calculated with the formula:
Measurement of Amadori Products
The quantification of fructosamine or Amadori products was conducted following the NBT assay, as outlined by Meeprom et al (2013) with slight modifications. 19 Specifically, 100 µl of NBT solution (0.5 mmol/l) in carbonate buffer and 10 µl of the glycated sample were incubated at 37 °C for 15 min. Subsequently, the volume was adjusted to 1 ml using distilled water for spectrophotometric measurement at 530 nm.
Estimation of Carbonyl Content
The assessment of carbonyl content was carried out using the DNPH method described by Meeprom et al (2013). 19 The glycated samples and 400 µl of DNPH (10 mmol/l) were incubated at 37 °C in darkness for one hour. Further, 20% (w/v) TCA (500 µl) was added to the incubated mixtures and maintained on ice for 5 mins to facilitate protein precipitation. Subsequently, centrifugation was performed at 10,000 rpm for 10 min at 4 °C, and the resulting pellet was subjected to three washing using a mixture of ethanol and ethyl acetate (1:1). After evaporating the ethanol/ethyl acetate, the pellet was resuspended in 6 M guanidine HCl (250 µl). The final volume was adjusted to 1 ml with distilled water, and spectrophotometric measurement was conducted at 370 nm.
Estimation of Total Fluorescent AGEs
The quantification of total fluorescent AGEs was conducted by employing excitation and emission wavelengths set at 370 nm and 440 nm, respectively, using the Cary Eclipse Fluorescence spectrophotometer (Varian) following the method outlined by Ali et al 20
Glycation-Induced Aggregation of β-Amyloid Structures
The examination of β-amyloid structures was performed using the Congo red assay described by Balyan et al (2022). 17 In this procedure, 50 µl of Congo red dye (100 µM) was added to 50 µl of glycated samples and incubated for 20 mins. Subsequently, the absorbance was measured at 530 nm. Further, β-amyloid structures aggregates in the glycated sample (3 ml) were also measured by Thioflavin T (20 µM) dye using Cary Eclipse Fluorescence spectrophotometer (Varian) at the excitation and emission wavelengths of 440 nm and 490 nm, respectively.4,17
Agarose Gel Electrophoresis Analysis
A total of 0.25 μg pBR322 plasmid was subjected to incubation with MG (20 mM), lysine (20 mM), and FeCl3 (100 μM) with/without AA and PLP (both 25 mM) for 2 h at 37 °C. The subsequent agarose gel was electrophoresed, and analysis was carried out following the procedures outlined in Rubab et al (2021). 21
Statistical Analysis
The Microsoft Excel software and Origin v8.5 were employed to conduct the ANOVA test for statistical analysis. The result of analytical methods showed a p-value ≤ 0.05, which was considered statistically significant. The experimental measurements were performed thrice (n = 3) for quenching studies. And, glycation assays were repeated six times (n = 6).
Results
Fluorescence-Based Interaction Studies
BSA possesses intrinsic properties of fluorescence, which causes intense emission upon excitation at 280 nm. By exploiting this property of BSA, we can assess the interaction between AA/PLP and BSA by monitoring intrinsic fluorescence quenching. The fluorescence intensity of BSA showed a reduction due to its interaction with AA (Figure 2A) and PLP (Figure 2B). It was observed that the effect of increasing the concentration of AA and PLP quenched the fluorescence intensity of BSA, and a slight blue shift was observed in both vitamins.

Fluorescence quenching spectra of (A) AA-BSA and (B) PLP-BSA complex interaction. (BSA - bovine serum albumin; AA - ascorbic acid; PLP - pyridoxal-5-phosphate).
To ascertain the quenching mechanism, the recorded fluorescence was evaluated using the established Stern-Volmer mathematical equation.
22
F and F0 = Relative fluorescence intensity with and without a quencher, respectively. [Q] = Concentration of quencher. KSV = Stern Volmer quenching constant. Kq = Biomolecular quenching rate constant. τ0 = Average fluorophore lifetime, usually for a bio-macromolecule in the excited state.
Figure 3 represents the Stern-Volmer plot of F0/F against both [AA] and [PLP]. The value of Ksv was calculated by applying the Stern-Volmer equation through linear regression of the F0/F versus [AA] and [PLP] concentrations. The calculated value of KSV and Kq parameters is listed in Table 1. In both the vitamins, PLP was found to have more KSV and Kq values than AA. The higher Ksv value of PLP showed that it more efficiently reduced BSA's fluorescence intensity and binding rate, respectively, than AA.
The validation of the quenching process is additionally substantiated by the calculation of the Kq value, which was derived using the below equation:

Stern-Volmer plot of AA-BSA and PLP-BSA complex interactions. (BSA - bovine serum albumin; AA - ascorbic acid; PLP - pyridoxal-5-phosphate).
Stern-Volmer constant (Ksv), biomolecular rate constant (Kq), binding constant (Kb), and binding stoichiometry (n) of FA-BSA interaction.

Where
In this context, the maximum scatter collision quenching constant is 2.0 × 1010 M−1 s−1. Notably, the values of Kq for both AA and PLP are indicated in Table 1 and fall within the range of 10 16 M−1 s−1.
Another modified Stern-Volmer equation was employed to determine the binding constant and binding stoichiometry (n)
23
:

Modified Stern-Volmer Plot for AA-BSA and PLP-BSA complex interactions. (BSA - bovine serum albumin; AA - ascorbic acid; PLP - pyridoxal-5-phosphate).
Measurement of Browning
The browning intensity was negligible in all samples on the first day of incubation. Both ascorbic acid and pyridoxal-5-phosphate at concentrations of 25 mM suppressed browning intensity by 18% and 20% compared to the glycation system, respectively, after 28 days of incubation (Figure 5). In this study, both vitamins observed the inhibition of the development of brown colour in the glycation of BSA.
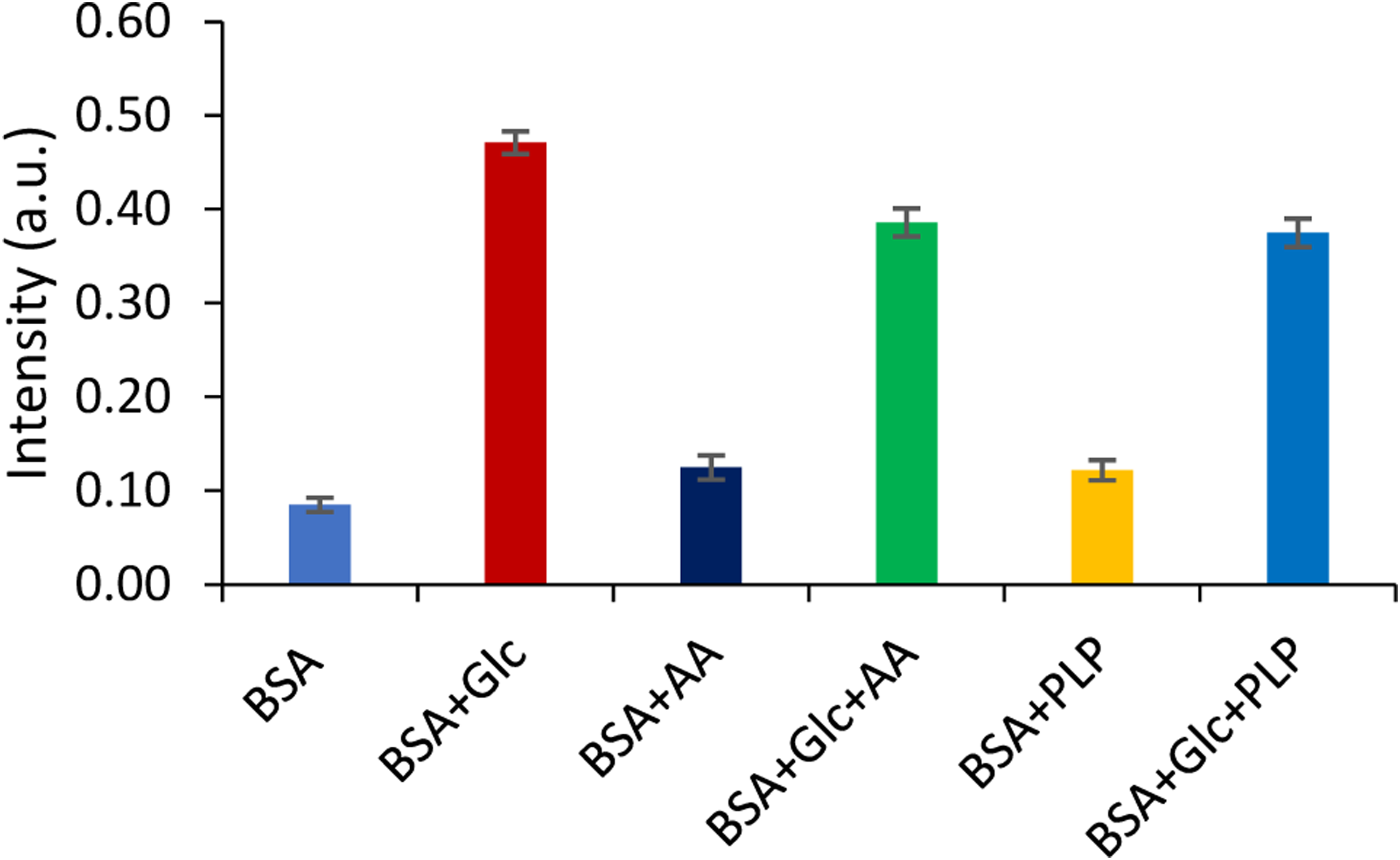
Measurement of browning in the presence of AA and PLP in glycated BSA (n = 6 and p-value < 0.5). (BSA - bovine serum albumin; Glc - glucose; AA - ascorbic acid 25 mM, PLP - pyridoxal-5-phosphate 25 mM).
Measurement of Amadori Products
The early glycation product, fructosamine content, was measured to be 31.31 µmol/mg protein in the glycated system on day 28. The fructosamine content in the presence of AA and PLP in the glycated system reduced to 20.36 and 17.24 µmol/mg protein, respectively (Figure 6). The formation of Amadori product attenuated by 45% in the presence of PLP. However, AA caused a 35% reduction of fructosamine content in the glycated protein on day 28.
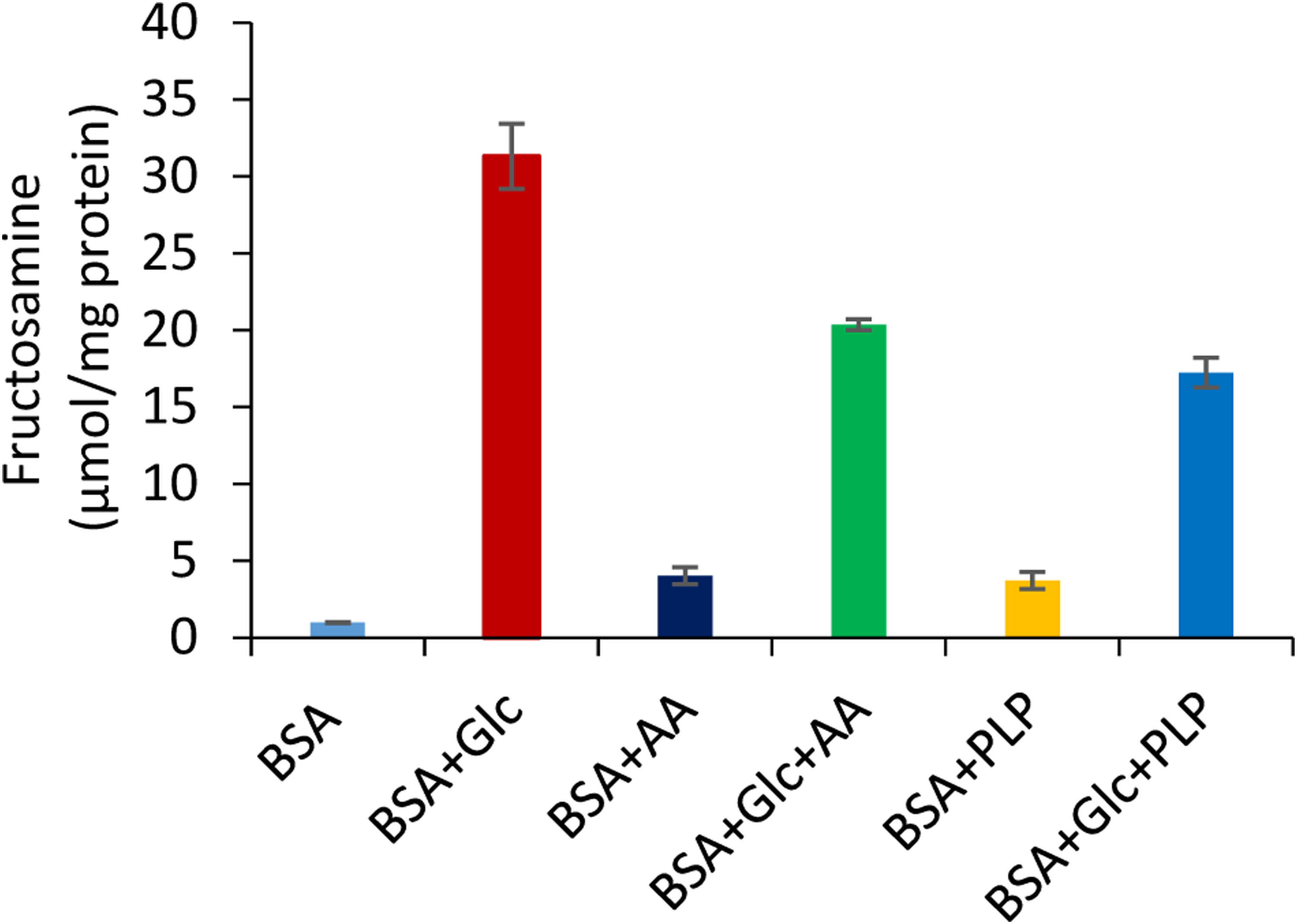
Measurement of fructosamine or Amadori products in the presence of AA and PLP in glycated BSA (n = 6 and p-value < 0.5). (BSA - bovine serum albumin; Glc - glucose; AA - ascorbic acid 25 mM, PLP - pyridoxal-5-phosphate 25 mM).
Measurement of Carbonyl Content
In glycated BSA, the carbonyl content was increased to 25.82 µmol/mg protein on day 28. The carbonyl content in the presence of AA and PLP was measured to be 20.64 and 17.16 µmol/mg protein, respectively, for the same period of incubation (Figure 7). It indicated the suppression of carbonyl content by 33.54% and 20.07% in the presence of PLP and AA, respectively.

Measurement of carbonyl content in the presence of AA and PLP in glycated BSA (n = 6 and p-value < 0.5). (BSA - bovine serum albumin; Glc - glucose; AA - ascorbic acid 25 mM, PLP - pyridoxal-5-phosphate 25 mM).
Measurement of Total Fluorescent AGEs
The formation of AGEs from the early and intermediate stage products occurs in the glycated system. Ascorbic acid and pyridoxal-5-phosphate (25 mM) attenuated the generation of total AGEs by 24.55% and 36.32%, respectively (Figure 8).

The spectrum of total AGEs formation in the presence of AA and PLP in glycated protein on day 28. (BSA - bovine serum albumin; Glc - glucose; AA - ascorbic acid 25 mM, PLP - pyridoxal-5-phosphate 25 mM, a.u.- arbitrary unit).
Glycation-Induced Aggregation of β-Amyloid Structures
In a glucose-induced glycation system, the aggregation of β-amyloid structures of BSA was considered 100%. In the Congo red assay, AA and PLP at 25 mM concentration reduced the protein aggregation by 21.58% and 30.32% in the glucose-induced glycation system on incubation of day 28 (Figure 9). Similarly, in the Thioflavin-T assay, the formation of β-amyloid structures was suppressed by 23.91% and 31.26% in glycated BSA by AA and PLP on day 28, respectively. BSA in the presence of AA and PLP showed almost similar aggregation of β-amyloid structures as native BSA (Table 2). In both assays, native BSA showed very insignificant aggregation.

Measurement of aggregation of β-amyloid structures in the presence of AA and PLP in glycated BSA by Congo red dye method (n = 6 and p-value < 0.5). (BSA - bovine serum albumin; Glc - glucose; AA - ascorbic acid 25 mM, PLP - pyridoxal-5-phosphate 25 mM).
Measurement of Aggregation of β-Amyloid Structures in Glycated BSA in the Presence of AA and PLP by Thioflavin-T (ThT) dye (Ex-440 nm, Em-490 nm). (n = 6 and p-Value < 0.5). (BSA - Bovine serum Albumin; Glc - Glucose; AA - Ascorbic Acid 25 mM, PLP - Pyridoxal-5-Phosphate 25 mM).

Agarose Gel Electrophoresis Analysis
pBR322 is a sensitive indicator for altering the conformation of single-strand breakages induced by oxidative stress, free radicals, and glycation. In Figure 10, Native DNA (L1) observed the supercoiled conformation band only, and the presence of AA (L2) and PLP (L3) alone showed the same band positions maintaining the conformation of DNA (Figure 10). Lysine, MG, and FeCl3 glycation systems caused DNA damage, indicating the conversion of the supercoiled conformation to the circular DNA conformation (Figure 10 – L4).
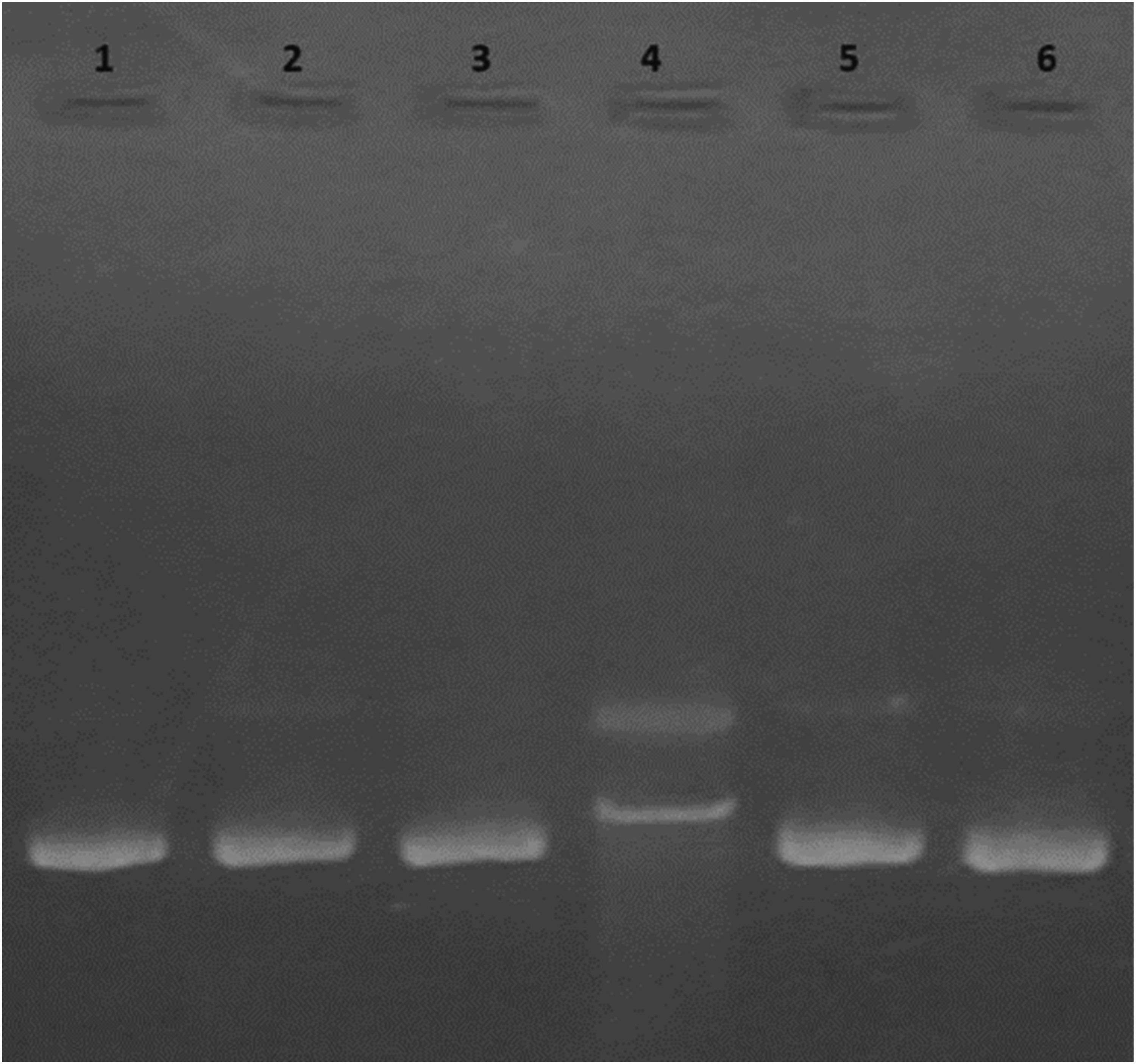
Effect of AA and PLP on glycated DNA (Lane description: L1 - DNA alone, L2 -DNA + AA [25 mM], L3 - DNA + PLP [25 mM], L4 - DNA + Lysine[20 mm] + MG[20 mM] + FeCl3 [100 µM], L5 - L2 + AA [25 mM], and L6 - L2 +PLP [25 mM].
Discussion
The fluorescence intensity of BSA exhibited a quenching effect with increasing concentrations of AA and PLP and caused a slight blue shift in both cases. The reports of Xu et al showed a reduction in quenching and blue shift by AA. 24 Similarly, the presence of PLP showed a blue shift in the peak of the spectrum of BSA. 25 From these findings, AA and PLP can interact with BSA and support the previously reported data. Nafisi et al (2011) reported the CD results stating the structure of BSA and vitamin C complex observed a slight variation in protein α-helix and β-sheet structures. 26 Ascorbic acid also interacts with BSA through hydrophobic interactions, resulting in the quenching of BSA's intrinsic fluorescence of involved fluorescent amino acids/ aromatic amino acids. 27 It observed that Kq is significantly higher than the maximum scatter collision value reported by Kumar et al 4 At the higher concentration of AA, the Stern-Volmer plot showed a slight upward curvature towards the y-axis which indicates the static and dynamic interaction with the protein molecule. PLP observed the linear relation with increasing quencher concentrations which represents the single static interaction. 13
It was noted that a high value of Kb suggested that PLP observed better binding affinity with serum protein than the ascorbic acid. Previous literature reports observed that PLP showed a static interaction and more stable formation of the PLP-BSA complex. 25 It was reported that ascorbic acid showed weak interaction or binding affinity with BSA. 7 These findings of quenching of fluorescence, Stern-Volmer constants and binding constants of both vitamins were found following previous reports. The interaction between pyridoxal-5-phosphate (PLP) and albumin involves the formation of a Schiff base with lysine residues, a process that is not instantaneous and exhibits specific kinetic characteristics. 28 Similarly, the interaction between vitamin C and albumin displays distinct kinetic behaviours, particularly under oxidative conditions. 29
The non-enzymatic interaction of BSA with AA and PLP was assessed at each stage of glycation. Ascorbic acid has been shown to reduce fructosamine content in the fructose-BSA system in 14 days. 30 Ramis et al reported that PLP suppressed the generation of free radicals and Amadori products in protein glycation. 31 Pyridoxamine (Vitamin B6) inhibits the formation of Amadori products by targeting Schiff bases and reactive intermediates, while Vitamin C reduces fructosamine and AGE formation by neutralizing dicarbonyl intermediates and mitigating oxidative stress.32,33 Tupe and Agte found that AA suppressed the carbonyl content caused by the glycation of BSA. 34 Adisakwattana et al also reported a similar pattern of reducing carbonyl content in the fructose-BSA system in 14 days. 30 The inhibition of post-Amadori products in the presence of PLP was reported in protein glycation. 31 Therefore, PLP suppressed the generation of fructosamine and carbonyl content followed by AA in the glycated system more significantly, and results were observed by published literature.
In the in-vitro study, the co-incubation of Zn and AA caused more inhibition of AGEs than the AA in a glycated BSA. 25 AA markedly decreased the formation of fluorescent AGEs in the fructose-BSA system in a concentration-dependent manner in two weeks. 30 In another in-vivo study, the pyridoxal-5-phosphate (PLP) significantly suppressed the formation of AGEs in STZ-induced diabetic rats. 3 PLP caused the suppression of AGEs formation in glycated protein, as reported by Ramis et al 31 Out of both vitamins, pyridoxal 5-phosphate caused more significant attenuation of the generation of Amadori products, intermediate glycation compounds, and AGEs in the glycated protein compared to ascorbic acid. The interaction of serum protein with vitamins and identification of specific AGEs in the in-vivo studies remain the limitations of this study due to ethical concerns.
Protein aggregation was analysed by both Congo red and Thioflavin-T assays. Tupe and Agte (2010) found that AA caused a slight reduction in the formation of protein aggregates in glycated BSA in the Congo red dye assay but not in the Thioflavin-T assay. 34 However, protein aggregation was inhibited by AA on the elevation of concentration in the fructose-BSA system in two weeks. 21 In the presence of both vitamins, PLP was found to be more potent in inhibiting the aggregation of β-amyloid structures in the glycation process with respect to AA. Thioflavin T binding assay using fluorescence spectroscopy is a vital, sensitive, and reliable method for β-amyloid structure aggregation analysis. Using consistent protocols and performing multiple replicates and statistical analyses reduced uncertainties. Native BSA observed a significant reduction of β-amyloid structure aggregation in the presence of both vitamins. Similarly, glycated BSA decreased aggregate formation in the involvement of vitamins. These findings also followed the results of the Congo red dye assay and supported the outcomes by Tupe and Agte (2010) in the literature. 34 It indicated that the Amadori products and AGEs of glycated protein contribution to the aggregation of β-amyloid structures was prevented in the presence of vitamins.
The exposure of any drugs, compounds, vitamins, etc to the BSA observed a change in its microenvironment which led to a slight change in secondary structure. Glycation of BSA by glucose often leads to the formation of aggregates in the size range of 100–500 nm or more, depending on reaction conditions like temperature, pH and concentrations.35,36 Considering the reduced formation of glycation product at each stage of glycation in the presence of both vitamins PLP and C. The outcomes of the Congo red dye assay and Thioflavin-T assay showed a reduced quantity of β-amyloid structures aggregates. Based on these results, vitamins PLP and C are expected to reduce the aggregate size, possibly limiting them to smaller glycated BSA.
Further, the generated Amadori and late-stage glycation products caused the damage to DNA. It was reported that free radicals and oxidative stress damaged the conformations of DNA strands, and the ferric ion (Fe3+) presence enhanced the damage. 23 This damage was reversed in the presence of AA and PLP vitamins in the model glycation system. It was noted that both the vitamins were observed to maintain the supercoiled conformation of DNA.
Conclusion
Natural products play a crucial role in several functions of the body and also help in the defence system. In the present study, pyridoxal-5-phosphate showed better interaction quenching of fluorescence intensity and binding affinity with serum protein compared to ascorbic acid. Pyridoxal-5-phosphate significantly attenuated the browning, Amadori products, and carbonyl content formation in the glycated BSA. The generation of early and intermediate-stage glycated products was also reduced by ascorbic acid in the glycated BSA. In the late stage of glycation, the extent of total AGEs generation was markedly decreased in the presence of pyridoxal-5-phosphate. Ascorbic acid also restricted the amount of total AGEs formation in the glucose-induced glycation system. However, ascorbic acid showed less inhibition of glycated products at all stages of glycation than pyridoxal-5-phosphate. Further, the presence of pyridoxal-5-phosphate caused a significant reduction in the aggregation index and formation of aggregates of β-amyloid in the glucose-induced glycation system in both assays. Ascorbic acid also suppressed the generation of protein aggregation but less than pyridoxal-5-phosphate. Ascorbic acid was a less potent inhibitor of the aggregation of β-amyloid in glycation. From these research findings, it can be concluded that pyridoxal-5-phosphate observed more binding affinity on interaction with BSA, the maximum inhibition of glycated product formation, β-amyloid structures aggregation on day 28 and maintained the structural conformational integrity of DNA.
Footnotes
Acknowledgements
The authors would like to extend their sincere appreciation to the Researchers Supporting Project number (RSP2025R154), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable to this article.
Funding
This work was supported by the Researchers Supporting Project number (RSP2025R154), King Saud University, Riyadh, Saudi Arabia.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.