Abstract
Aedes aegypti is the main transmitter of several arboviruses, mainly dengue. It occurs, recently, in more than 100 countries and majority of the world population lives in areas of mosquito incidence, marking its control relevant and necessary. Presently, the main form of vector control is the use of synthetic insecticides; however, its continuous application has led to inefficiency due to resistance development. Based on this fact, the insecticides from natural sources appear as a friendly alternative for man and the environment. This study provides an overview of the larvicidal compounds isolated from plant extracts while controlling A. aegypti, in the previous 6 years (2013-2018), and aims to impart more knowledge regarding the described metabolites and to encourage the search for new bioactive compounds. In addition, the proposals for mechanisms of action and structure-activity relationships that may justify the larvicidal potential are also discussed.
Aedes aegypti is the main transmitter of the viruses that cause dengue, urban yellow fever, chikungunya, and zica. In the past 10 years, there was an increase in studies exploring about A. aegypti control, since a majority of the world population live in areas with incidence of the mosquito and are vulnerable to the aforementioned diseases. The dispersion of this vector is characterized as a political, social, and economic concern. 1
Present control measures adopted by Mosquito Control Programs and indicated by the World Health Organization (WHO) involve the use of synthetic insecticides; however, their continuous employment resulted in resistance development. In addition, such insecticides are toxic to humans and also to the environment when used indiscriminately. 2,3
Based upon these measures, the search for new compounds with potential insecticidal action and low toxicity toward humans and environment emphasized on the natural sources. Plants are well known to produce secondary metabolites, which act as defense mechanisms against predators. This characteristic reveals that the natural insecticides play a pivotal role in vector control, and their use represents an excellent alternative to synthetic insecticides. 4
Vector control is preferably performed at the larval stage, due to its larger vulnerability. 5 The search for natural larvicidal compounds is recent and the initial studies were published in the 1980s. Until 2013, certain plant families, such as Fabaceae, Rutaceae, Piperaceae, and Boraginaceae, 6 were highlighted as producers of compounds with larvicidal activity against the A. aegypti mosquito.
The increase in publications and the high records of outbreaks of mosquito-borne diseases in the past few years in several countries worldwide reveal an urgent interest in secondary metabolites that may have potential larvicidal activity. 7 Moreover, the mechanism of action and the structure-activity relationship of these compounds have been the subject of research.
Aedes aegypti and Dengue
Aedes aegypti (Linnaeus; Family: Culicidae) is a mosquito originated in Egypt, which began to spread from Africa, via slaveships toward the tropical and subtropical regions of the world. Presently, it is considered as a “cosmopolitan species” in numerous countries. It was first described in 1762 but its scientific name was only defined in 1818: Aedes is the Greek word for “unpleasant/odious” and aegypti comes from the Latin vocabulary meaning “of Egypt”. 8
This vector has a three-phase life cycle: egg, aquatic, and adult. The complete cycle for mosquito development lasts from 7 to 10 days, and after reaching adulthood, the mosquito can survive till 30 days. The female mosquito is able to deposit up to 1000 eggs in this period, which have considerable resistance and can sustain for 1 year in the dry environment, waiting for water to hatch. 8,9
The female A. aegypti mosquito is the main vector for different viral diseases, with dengue being the most relevant. The dengue-causing pathogen is an arbovirus belonging to the genus Flavivirus (Flaviviridae). There are 390 million annual dengue infections worldwide. About 3.9 billion people in 128 countries are at a risk of infection by the virus. 10 Brazil is among the South American countries with the highest record of dengue cases, attaining 52% of the total cases of dengue fever in 2012. 11,12
Forms of A. aegypti Control
Presently, no effective vaccines are available to prevent dengue, chikungunya, and zica. The best way to control the vector is to eliminate the larvae foci multiplication. In addition, the oviposition of A. aegypti occurs in clean and still water, facilitating the larval population control. 13
Vectors can be controlled environmentally, biologically, and chemically. Environmental control occurs via the disposal of breeding sites. Biological control uses natural predators to eliminate the vector population. Chemical control requires insecticides, including larvicides for controlling larvae and adulticide for controlling adult mosquitoes. 14
The use of insecticides is the predominant form of controlling the A. aegypti vector. The main classes recommended by WHO are organochlorines, organophosphates, carbamates, and pyrethroids. Juvenile hormone analogs such as pyriproxyfen, and insect growth regulators such as diflubenzuron and novaluron are also employed. 14 -16
In Brazil, the National Dengue Control Program adopts the organophosphate temephos as larvicide and pyrethroids as adulticide. Although they have significant results, its continuous use has caused the development of resistance in mosquitoes. 17 Moreover, several of these insecticides remain in the environment for long time periods, causing contamination, imbalance, and damage to the human health. 18
The harmful effects caused by the use of synthetic insecticides led to the search of more selective and less aggressive compounds. Within this context, larvicides from the natural sources emerged. 19 Among the plant compounds known for their insecticidal potential, nicotine (present in plants of the genus Nicotiana), pyrethrins (extracted from chrysanthemum flowers), and rotenoids (existing in some species in Fabaceae) can be mentioned. 20
The use of natural insecticides is safer to man and the environment, as they are rapidly degraded, more selective to the target insect, less harmful to the nontarget organisms, and lessen the possibility of resistance. To date, more than 2000 species of plants have already been described to have insecticidal action. 21
Compounds of Plants With Larvicidal Activity
“Web of Science” and “Scopus” databases were used as search engines. Keywords employed were A. aegypti, larvicidal activity, natural, and plant. The search emphasized on articles published between 2013 and 2018.
About 37 publications describing compounds with larvicidal action isolated from plants for controlling A. aegypti mosquito were found. Several studies exhibit an increase in the search for these compounds in the past 6 years, in contrast to the 38 publications published from 1986 to 2012. 6
Table 1 presents all the compounds found from this survey, organized in an increasing order of larvicidal activity, based on the lethal concentration (LC) of the compounds against the larvae. Noteworthily, a protocol is used for performing larvicidal tests created by the WHO, with standardizing procedures 22 ; however, most of the methods found in the literature do not use WHO protocol, making this comparison very difficult. Another complication is the variation in LC value, which differs for the same compound.
Compounds With Larvicidal Potential Described in the Literature During 2013 to 2018.
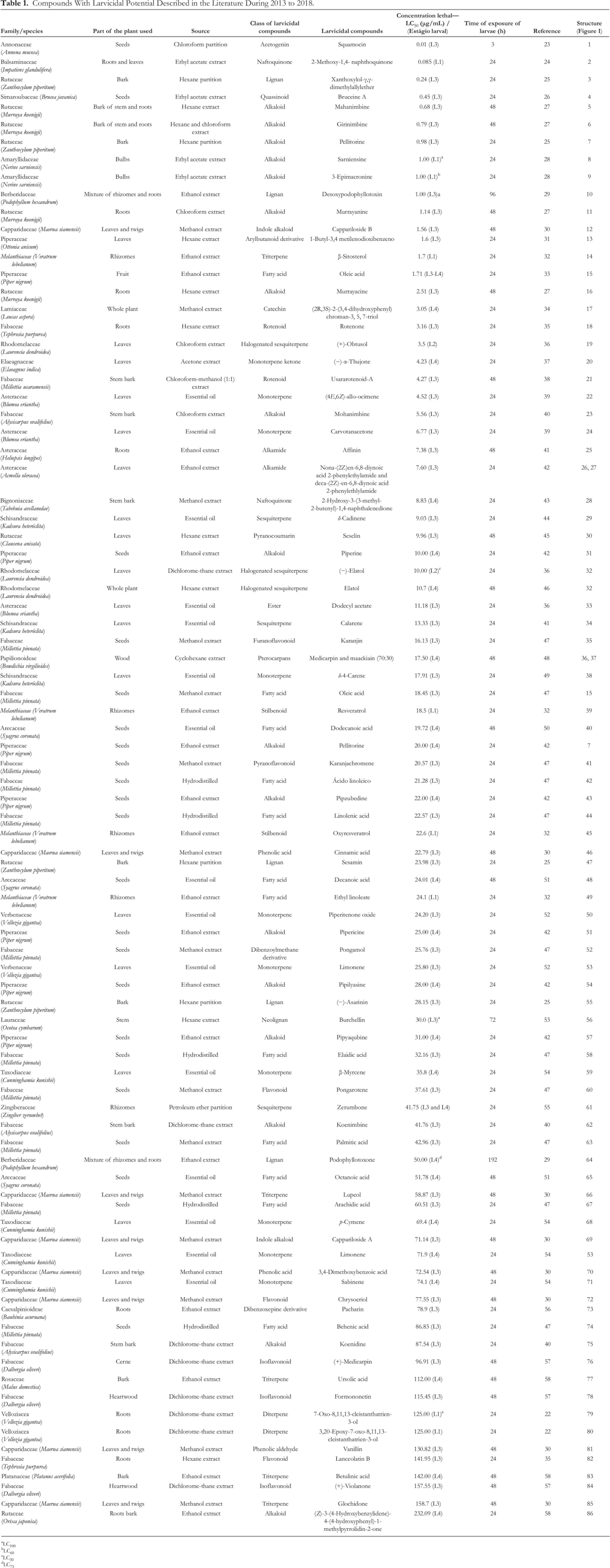
aLC100
bLC60
cLC30
dLC73
This search registered 86 larvicidal compounds (Figure 1), distributed through 26 families and 36 plant species. Families with the highest number of species presenting active compounds were Fabaceae, Rutaceae, Piperaceae, and Asteraceae, with respectively 20, 10, 8, and 5 secondary metabolites recorded. Among the classes of compounds that presented major potential for larvicidal activity, acetogenins, naphthoquinones, lignans, quassinoid, and alkaloids stand out. The families Fabaceae, Rutaceae, and Piperaceae were also highlighted in the previously published review, and the compound classes were quite similar. The metabolites piperine, pelitorin, and rotenone have been described in the previous studies for their larvicidal activity 6 ; however, in this study, they were isolated from different families and/or parts of the plants.

Structure of the compounds that present larvicidal activity against Aedes aegypti isolated from plants in the past 6 years.
Although acetogenin squamocine (1) presented the highest larvicidal potential (LC50 = 0.01 µg/mL) among the compounds found, alkaloids class revealed the highest number of active compounds: 19 secondary metabolites with larvicidal action, followed by fatty acids, monoterpenes, flavonoids, sesquiterpenes, and lignans, with, respectively, 12, 8, 8, 7, and 6 secondary metabolites. In the alkaloid class, carbazols mahanimbine (4) and the girinimbine (5) presented highest potential as larvicides.
Mechanism of Action
The mechanism of action of secondary plant metabolites against A. aegypti larvae is still poorly understood, in particular at the molecular level. Most of these secondary metabolites demonstrated certain interference in the central nervous system via cutaneous or respiratory absorption, leading to death by intoxication, via the inhibition of acetylcholinesterase (AChE), which is similar to organophosphates and carbamate insecticides. 59
Some other mechanisms of action observed for the insecticidal compounds when in contact with the predators involve action on the GABA system, leading to seizures and death; inhibition of mitochondrial activity; action as repellent preventing oviposition; action in the digestive system inhibiting the appetite, among others. 60
Recent studies have revealed that the acetogenin squamocin (1) affects the digestive cells of the midgut from the larvae of A. aegypi causing cell death by autophagy. Additionally, mortality was observed also due to the structural damages in the anal papillae of the larvae. 61,62 Neolignan burchellin (49) was tested in the third instar larvae of A. aegypi and caused death by histomorphological changes in the midgut. 63
The karanjin (32) and karanjachromene (37) flavonoids were reported as strong inhibitors of AChE from mosquito larvae indicating it to be the presumable site of larvicidal action. The same results were observed for oleic (12), linoleic (38), linolenic (40), and palmitic (55) fatty acids. The fatty acids linoleic (38), linolenic (40), elaidic (51), arachidic (58), and behenic (62) presented to have an effect on the octopaminergic system. It has also been observed that the karanjin (32) flavonoid can inhibit feed and act as a growth regulator. 46
Alkaloids have an influence in the central nervous system of the insects, acting on the receptors of several neurotransmitters, provoking uncontrolled muscular movements, paralysis, seizures, and death. They can also affect the sodium channels of the nerve cell membrane, preventing the transmission of nerve impulses. 64 Studies with synthetic analog alkaloids have revealed to have a greater degree of inhibition of AChE. 65,66
Understanding the mechanism of action of secondary metabolites with larvicidal action can help in reducing the resistance of insecticides and aid the production of analogs with more pronounced activity with specific or multiple sites of action.
Structure-Activity Relationship
Studies on the relationship between chemical structure and their larvicidal activity are based on the comparisons between structurally similar compounds. The influence of functional groups, degree of unsaturation, and lipophilicity are used to differentiate the analogs indicating how they may affect the potential of the biological activity. Molecular modeling techniques were also applied to establish a quantitative structure-activity relationship (QSAR). 57,67-70
Structure-activity relationship studies were performed for acetogenins emphasizing the inhibition of the mitochondrial I complex. Data revealed that the presence of 1 or 2 tetrahydrofuran (THF) rings would have similar inhibitory effect. Moreover, the existence of THF and tetrahydropyran rings reveal the same degree of activity. It was further observed that structurally similar C-35 acetogenins are more active than the C-37 ones. 67
In addition, a decreased activity was observed when a keto group is present instead of a hydroxyl group and when the double bond of the α, β-unsaturated lactone is reduced. The most active acetogenins are bis-THF with adjacent rings, followed by nonadjacent bis-THF, mono-THF, and those without THF rings. 67
The monoterpenes eugenol, geraniol, linalool, L-menthol, and terpineol, and their respective acetylated derivatives were studied. It was observed that the acetylation of the hydroxyl groups increases the larvicidal activity against A. aegypti larvae. Additionally, eugenyl acetate exhibited more pronounced activity among all monoterpenes, presumably due to the presence of an aromatic ring and an unsaturated side chain. 68
Fourteen different cyclic monoterpenes were evaluated for their structural characteristics. A decreased activity was observed when the heteroatoms were present in the main skeleton, hydroxyl groups were present in cyclic structures, and double bonds were epoxidated. The presence of conjugated or exo double bonds increases the larvicidal activity. Another important factor that has a strong influence on the activity of monoterpenes is related to the lipophilicity of its chemical structure, since the presence of lipophilic groups revealed an increase in the larvicidal activity. 69
Larvicidal activity observed for both betulinic and ursolic triterpenic acids revealed that the presence of the hydroxyl group in C-3 is fundamental, as the esterification of hydroxyl decreases this activity. 57
By using QSAR method, a study was recently carried out, specifically for 61 plant-derived compounds known to have A. aegypti larvicidal activity. Compounds of the terpene class, phenylpropanoids, ketones, and oxygenate compounds were selected for the study. Some parameters that influenced the increase of the activity have been defined: the presence of atoms involved in hydrogen bonds, presence of multiple bonds, heteroatoms, electronegative atoms, and smaller polar surface area. 70
Effects on Nontarget Organisms
Studies related to toxicity to humans and other nontarget organisms have been scarce in the publications used in this research. These studies are extremely important since the compounds of Table 1 present a great potential for larvicidal activity against the A. aegypti mosquito and the effective use of these substances requires research that establishes safety profiles regarding the stabilization of the ecosystem and undesirable effects in the nontarget organisms and human health. Among all the substances present in Table 1, toxicity tests were performed for 4 monoterpenes (4E, 6Z), -allo-ocimene (22), and carvotanacetone (24) using 4 nontarget species: Anisops bouvieri, Diplonychus indicus, Poecilia reticulata, and Gambusia affinig, where it was observed that there was no damage to the survival and swimming activity of these nontarget predators. 38 Acetogenin squamocin (1) was tested in the species of Culex bigoti and Toxorhynchites theobaldi, revealing no toxic effect against these predators. In addition, the tests were performed on human leukocytes, and the results suggest that squamocin is nontoxic to mammals. 71
Final Remarks
This compilation revealed the advances in the past 6 years in search for compounds of plant with activity against A. aegypti larvae. This study gained immense interest due to the high number of publications. About 86 compounds were settled as potentially larvicidal, and Fabaceae had the highest number of records. A wide variety of compounds have been found, such as acetogenins, alkaloids, naphthoquinones, lignans, quassinoids, flavonoids, fatty acids, monoterpenes, sesquiterpenes, and others. Among the aforementioned compounds, acetogenin squamocin presented the highest larvicidal potential, killing 50% of the larvae of A. aegypti in the concentration of 0.01 µg/mL. Alkaloids presented the highest number of larvicidal metabolites. It is interesting to observe that the plant families that presented the highest number of active substances and the classes of secondary metabolites described in this study were similar to those presented in the previous review. This provides evidence regarding numerous studies carried out for specific families and the importance of the structural characteristics of some classes of substances for larvicidal activity.
The mechanism of action and the structure-activity relationship for larvicidal compounds remain unclear, and few studies are observed in the literature. Most of the compounds with larvicidal activity presented the inhibition of AChE as the main mechanism of action.
A major understanding of the relationship between chemical structure and larvicidal activity would facilitate the search for new metabolites or the synthesis of compounds with more pronounced activity.
Footnotes
Acknowledgments
We would like to express our gratitude to the Research Institute of Natural Products and CAPES for their financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared no financial support for the research, authorship, and/or publication of this article.
