Abstract
Background
Wound healing progresses through phases of homeostasis, inflammation, proliferation, and remodeling, regulated by signaling pathways. The root extract of Onosma microcarpum (OM), traditionally used in Western Iran for wound care, contains bioactive compounds like alkannin, shikonin, and polyphenols, potentially enhancing healing. This study evaluates OM's effects on wound healing through the signaling pathways in each phase.
Methods
Dried OM root was formulated into ointments, and male Wistar rats with excisional wounds were treated with OM (0.2% or 1%) or vehicle. Wound closure rate, histological and gene expression analyses were conducted on days 3, 8, and 14, measuring inflammation, granulation, angiogenesis, and collagen formation. The mRNA expression of cytokines (Interleukin-1β (IL-1β), IL-6, tumor necrosis factor-α (TNF-α)), growth factors (Transforming Growth Factor-β1 (TGF-β1), platelet-derived growth factor (PDGF)), and cell regulators (extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinases (JNK), p38 mitogen-activated protein kinase (p38 MAPK)) were analyzed.
Results
OM at 0.2%, significantly modulated key pathways in wound healing. By day 3, OM reduced TNF-α (P = 0.003) and IL-1β (P = 0.008), decreasing inflammation and JNK activity (P = 0.009). On day 8, OM further reduced IL-6 (P = 0.002) and p38 MAPK (P = 0.005) while increasing TGF-β1 (P = 0.008) and PDGF (P = 0.001), promoting proliferation. By day 14, increased ERK (P = 0.008) and p38 MAPK (P = 0.002) activity facilitated tissue repair. Wounds treated with 0.2% OM showed better outcomes than 1%, with more organized collagen (mean score 3.0 vs 2.3, P = 0.021), improved angiogenesis (3.4 vs 2.5, P = 0.038), and higher wound closure (87% vs 69%, P = 0.023).
Conclusion
OM enhances wound healing by targeting each phase: reducing early inflammation, promoting mid-phase fibroblast proliferation, and supporting collagen organization in remodeling. These targeted effects suggest OM's therapeutic potential for wound repair.
Keywords
Introduction
Wound healing follows a coordinated sequence of activations and inhibitions through inflammation, proliferation, and remodeling. 1 Effective regulation is essential, as recent research has shown that disrupted signaling pathways leads to chronic non-healing wounds.2,3
Certain medicinal plants, including those from the Boraginaceae family, have shown wound-healing potential by modulating key pathways involved in healing.4,5 The genus Onosma, comprising 150 species across the Mediterranean and Asia, includes species with demonstrated antibacterial6,7 and wound-healing properties.8,9 Onosma species contain a wide range of bioactive substances, with alkaloids and naphthoquinones predominantly concentrated in the roots, while polyphenols, phytosterols, and terpenoids are primarily found in the aerial parts, and fatty acids are abundant in the flowers and fruits,10,11 while extracts containing these shared compounds have demonstrated a broad safety profile and wound-healing efficacy in both in vivo and in vitro studies.9,10 The roots of Onosma microcarpum (OM) have been traditionally used in Western Iran for treating infectious wounds, inflammation, and episiotomy pain, and have demonstrated promising wound-healing effects in a diabetic rat model. The various healing patterns observed at different time points, depending on the formulation, suggest the presence of multiple bioactive compounds, each distinctly influencing the wound healing process. However, the underlying mechanisms and involved signaling pathways remain unexplored. 12
OM extracts are rich in polyphenols, including rosmarinic acid, caffeic acid, and luteolin, which possess strong antioxidant and anti-inflammatory properties, as well as terpenoids such as carvacrol, thymol, pulegone, and spathulenol, known for their antimicrobial and anti-inflammatory effects.13,14 These phytochemicals contribute to different phases of healing, with alkaloids primarily exhibiting anti-inflammatory effects, naphthoquinones promoting collagen formation, polyphenols contributing to both anti-inflammatory and angiogenic processes, and terpenoids facilitating tissue regeneration. These effects are mediated through key signaling pathways that coordinate each phase of healing.15–18
The Janus kinase/signal transducer and activator of transcription (JAK/STAT) pathway activation is key for immune gene transcription and recruiting macrophages and neutrophils to clear debris particularly during the inflammatory phase.19–21 Simultaneously, mitogen-activated protein kinase (MAPK) pathways amplify this response: ERK aids immune cell migration and promotes cell proliferation, while JNK and p38 regulate cytokines like IL-1β, IL-6, and tumor necrosis factor (TNF-α), angiogenesis, fibroblast function, and collagen production for extracellular matrix (ECM) synthesis.21–28 The phosphoinositide 3-kinases (PI3 K)/Akt pathway, activated by growth factors like Transforming growth factor-β1 (TGF-β1) and platelet-derived growth factor (PDGF), enhances fibroblast and endothelial activity, supporting granulation and angiogenesis.29–31
Understanding the mechanisms driving each phase of healing provides a foundation for developing targeted therapeutic strategies to enhance wound healing. We hypothesized that Onosma microcarpum extract would enhance wound healing by modulating the three primary upstream pathways -MAPK, JAK/STAT3, and PI3 K/Akt- which are critical regulators of this process. To gain mechanistic insight, we analyzed gene expression profiles of key inflammatory, proliferative, and regenerative biomarkers.
Materials and Methods
Plant Material and Preparation of Extract
Fully matured (flowered) Onosma microcarpum specimens were collected from Kermanshah province, Iran, in June 2022, with roots dried in the shade to preserve their phytochemical integrity. Species identification was confirmed by the Department of Plant Biology at the University of Tehran using an established herbarium reference (Herbarium No. 162). Dried specimens were ground to a fine powder, and 100 g of this powder was extracted with an initial 500 mL of petroleum ether in a Soxhlet apparatus for 18 h at ∼22 °C. After extraction, solvent removal was done with a rotary evaporator at 40 °C under reduced pressure, yielding a thick residue. This was dried in a vacuum oven at 45 °C until constant weight (∼18 h) and stored at −20 °C in airtight containers for later use in ointment formulation.
Ointment Formulation
Two ointment concentrations were prepared for topical application based on previous studies of Onosma genus showing anti-inflammatory effects, 32 and a preliminary dose-selection study using 0.04%, 0.2%, 1%, and 5% concentrations to identify effective doses. 0.2% and 1% were selected as the lowest concentrations with substantial effects on wound healing. The 0.2% and 1% (w/w) ointments were prepared by dissolving 2 g and 10 g of OM extract, respectively, in the ointment base (Vaseline, beeswax, and sesame oil in a 5:3:2 volume ratio), adjusting to 1000 g total weight. A vehicle control ointment, identical in base composition but without OM extract, was also prepared. All formulations were homogenized under sterile conditions and gamma-irradiated (25 kGy) to eliminate microbial contaminants, including bacterial spores. To assess the stability of the extract after gamma irradiation and further characterize its composition, we measured total antioxidant activity using the phosphomolybdenum method and total phenolic content using the Folin–Ciocalteu method before and after sterilization in three technical replicates, as previously described.33,34
Chemicals and Reagents
Anesthesia was induced via intraperitoneal injection of Ketamine (10% solution, Alfasan pharmaceuticals, The Netherlands) and Xylazine (2% solution, Bremer Pharma, Germany). Surgical site, skin, and tools were treated with 2% Chlorhexidine gluconate (C9394, Sigma-Aldrich, The USA) as antiseptic preparation. Laboratory-grade 70% ethanol (107017, Supelco, The USA), 10% neutral-buffered formalin (HT501128, Sigma-Aldrich, The USA), and ready-to-use Trizol reagent for RNA extraction (15596026, Invitrogen, The USA) were also sourced.
Animals
Seventy-two male Wistar rats (8-10 weeks old, weighing 200-300 g) were obtained from the School of Pharmacy, Tehran University of Medical Sciences. Rats were housed in groups of three per cage under controlled conditions (22 ± 1 °C, 55 ± 5% humidity) with a 12-h light/dark cycle, ensuring that each cage contained rats assigned to different experimental groups. Water and standard rodent chow were provided ad libitum. The study was approved by the Animal Research Ethics Committee of Tehran University of Medical Sciences (approval number: IR.TUMS.IKHC.REC. No.1399.341) and conducted in accordance with the NIH Guide for the Care and Use of Laboratory Animals, eighth edition (2011), to ensure compliance with animal welfare standards. Each rat was considered an experimental unit in all experiments. Exclusion criteria included instances of improper wound induction during the procedure.
Excisional Wound Model
The excisional wound model involved creating 2 mm deep full-thickness wounds on the dorsum of anesthetized rats, ensuring skin removal to the subcutaneous layer while sparing the muscle, with dimensions precisely measured using a Digital Caliper. Anesthesia was induced via intraperitoneal injection of Ketamine (100 mg/kg) and Xylazine (10 mg/kg). The surgical area was shaved, disinfected with Chlorhexidine gluconate, and a 25 × 16 mm rectangle (approx. 400 mm²) was marked, starting just caudal to the scapulae and extending 2.5 cm in length and 8 mm laterally from the midline vertebra on each side. 35 Immediately after wounding, the site was cleansed with saline, and carprofen (5 mg/kg) was administered subcutaneously for pain management before consciousness returned. Wounds were left undressed to ensure natural healing and assess treatment effects without external interference.
Experiment Design
Seventy-two rats were divided into three groups, each containing three subgroups assigned to different timepoints, via block randomization using a computer based random generator (n = 8 for each subgroup). Groups (I) vehicle, as the control group, (II) 0.2% OM, and (III) 1% OM received respective ointments just after the wound induction and every 12 h until the tissue collection. Each group contains three subgroups euthanized on days 3, 8, and 14, with tissues collected. Wound closure was the primary outcome, while signaling biomarkers assessments and histopathological evaluations were conducted at three phases: inflammatory (day 3), proliferative (day 8), and regenerative (day 14). 36 The sample size was determined based on previous studies using this model. 35 After wound induction, rats were individually housed to minimize infection risk and confounding factors. Since OM exhibits anti-inflammatory, pro-regenerative, and antimicrobial properties, it may influence multiple phases of wound healing. Given that standard therapeutic options, such as epidermal growth factor and silver sulfadiazine, primarily target single aspects of healing, this study did not include a positive control group. To ensure blinding, a single investigator (BA) handled drug administration and was the only person aware of treatment allocations.
Histological Analysis
Histological analysis was performed on skin samples collected from eight rats in each group on days 3, 8, and 14 after wound induction. Samples from the 3 mm margin of the wounds were excised and fixed in 10% neutral buffered formalin for 24 h, followed by dehydration, clearing, and embedding in paraffin. Sections of 5 µm thickness were stained with hematoxylin-eosin (H&E) for general morphology and Masson's trichrome to evaluate collagen deposition considering morphological appearance and architectural arrangement, as previously described. 37
Slides were examined using a light microscope under ×400 magnification by a blinded pathologist, assessing collagen formation, inflammation, angiogenesis (by qualitative assessment of capillary density), and granulation tissue formation on a scale from 0 (none) to 4 (severe), with intermediate values of 1, 2, and 3 indicating minimal, mild, and moderate severity, respectively. Representative images were captured to provide qualitative and quantitative assessments of healing process.
Quantitative Polymerase Chain Reaction (qPCR)
qPCR was used to assess pathway involvement, providing a stable and reproducible measure of gene expression throughout the dynamic phases of healing. Given the continuous progression of this process, qPCR allows for the evaluation of key signaling molecules over time. Total RNA was extracted from individual skin samples collected from eight rats in each group on days 3, 8, and 14 after the wound induction, from the 3-mm margin of the healing wounds using TRIzol reagent according to the manufacturer's instructions. The RNA concentration and purity were assessed using a NanoDrop spectrophotometer (NDLPLUSGL, Thermo Scientific, The USA), with samples showing an A260/A280 ratio greater than 1.8 deemed acceptable. RNA integrity was further confirmed via agarose gel electrophoresis. High-quality RNA was reverse-transcribed into complementary DNA (cDNA) using the High-Capacity cDNA Reverse Transcription Kit (4368814, Applied Biosystems, The USA).
Quantitative PCR was performed using SYBR Green PCR Master Mix (4309155, Applied Biosystems, The USA) on a StepOnePlus Real-Time PCR System (4376600, Applied Biosystems, The USA). Specific primers for target genes were designed and validated for efficiency, as listed in Table 1. The PCR conditions were set as follows: initial denaturation at 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s and 60 °C for 1 min. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) served as the internal control for normalization. Relative gene expression levels were calculated using the 2−ΔΔCT formula.
Forward and Reverse Sequences of Primers Used in qPCR.

Wound Area and Closure Measurement
Wound healing was assessed by measuring the initial wound area and ulcerated area on days 3, 8, and 14 using SketchAndCalc (an online planimeter tool for precise measurement of irregular shapes), with wound photographs taken at each time point including a scale ruler for reference. The percentage of wound closure was determined using the formula:
Statistical Analysis
Data were analyzed using SPSS (version 26, IBM, The USA) and are presented as mean ± standard error of the mean (SEM). Normality was assessed using the Shapiro-Wilk test. Normally distributed data were compared using ANOVA, followed by Tukey's post hoc test, while non-normally distributed data were analyzed using the Kruskal-Wallis test, with Mann-Whitney U test for post hoc comparisons. For all statistical tests, a P-value less than 0.05 was considered statistically significant.
Customized Generative Pre-trained Transformer 4 (GPT-4) Configurations
Our approach utilized tailored GPT-4 settings, adapted to meet specific research goals such as conducting literature reviews and refining manuscript language. Each configuration was designed to improve data accuracy, research synthesis, and clarity in academic presentation.
Results
Anti-oxidant Activity and Phenolic Content
The total antioxidant activity of the extract was 411.5 (mg of trolox equivalents/g of extract) before and 423.8 (P = 0.48) after gamma irradiation, while the total phenolic content was 27.34 (mg of gallic acid equivalents/g of extract) before, and 26.97 (P = 0.65) after sterilization, indicating no significant changes in these parameters due to irradiation.
Inflammatory Phase
Figures 1–3, illustrate the temporal progression of wound healing from the inflammatory phase through the proliferative to the regenerative phase. At day 3 post-wounding, histological analysis revealed that the 0.2% OM group had significantly reduced inflammation scores (mean ± SEM: 2.2 ± 0.13) compared to the vehicle group (3.0 ± 0.15, P = 0.016). In contrast, the 1% OM group did not show a significant reduction in inflammation (3.2 ± 0.13, P = 0.405). Collagen and granulation formation, angiogenesis, and wound closure were minimal and not significantly different across all groups (Figures 1–3). The expression levels of the evaluated genes at Day 3 are shown in Figure 4. Compared to the vehicle group, the 0.2% OM concentration significantly reduced the relative expression of TNF-α (P = 0.003) and IL-1β (P = 0.008). The expression of JNK was also significantly lower in the 0.2% OM group (P = 0.009). p38 and collagen type I expression levels were significantly higher in the 1% OM group (P = 0.038, and 0.022) compared to the vehicle group.
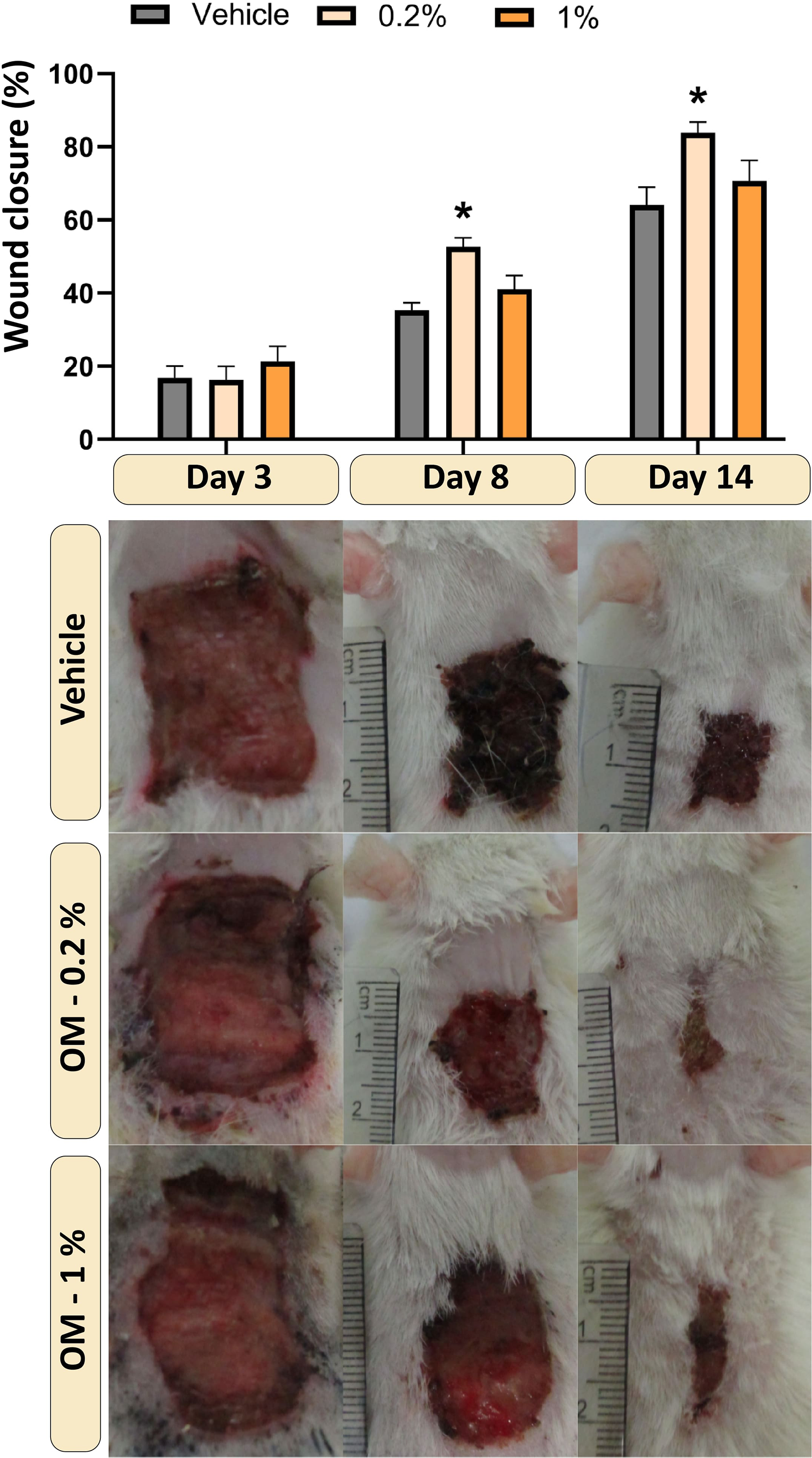
Wound Closure Progression. The Bar Graph Represents Wound Closure Percentages at Days 3, 8, and 14 Across Different Treatment Groups (Mean ± SEM). Representative Wound Images for Each Group at the Respective Time Points Illustrate Healing Progression. Images Shown in Each Row are from the Same Individual Rat Per Group Across all Time Points. * Indicates P < 0.05 Compared to the Vehicle Group. Statistical Analysis was Performed Using One-way ANOVA Followed by Dunnett's Multiple Comparison Post-hoc Test (n = 8 Rats per Group).

Histological Evaluation of Wound Healing. (A-D) Show Histopathological Scores of Inflammation (A), Granulation Tissue Formation (B), Angiogenesis (C), and Collagen Formation (D) at Days 3, 8, and 14 Across Different Treatment Groups. Individual Data Points are Presented, with the Line Indicating the Median Score (n = 8 rats per group). * Indicates P < 0.05 Compared to the Vehicle Group. Statistical Analysis was Performed Using the Kruskal-Wallis Test Followed by Dunn's Post-hoc test. (E & H & K) Inflammatory Phase at Day 3: Vehicle (E) and 1% OM (K) Groups Show Dense Inflammatory Cell Infiltration, Indicative of an Early Immune Response. 0.2% OM-Treated Group (H) Displays Reduced Inflammation and Early Tissue Organization. (F & I & L) Proliferative Phase at Day 8: Vehicle (F) and 1% OM (L) Groups Depict Minimal Fibroblast Activity and Early Collagen Deposition, Indicating Slower Tissue Remodeling. 0.2% OM-Treated Group (I) Demonstrates Enhanced Fibroblast Proliferation and Pronounced Collagen Synthesis. (G & J & M) Regenerative Phase at Day 14: Vehicle Group (G) Shows the Natural Progression of Healing, with Sparse Cellularity and the Formation of Early Acellular Zones. 0.2% OM Group (J) Exhibits Structured Fibrous Tissue with Dense Collagen Deposition. 1% OM group (M) Reveals Disorganized Collagen Deposition. Sections were Stained with Hematoxylin-eosin and Examined Under a Light Microscope at 400× Magnification. Open Arrows Indicate Inflammatory Cells, Solid Arrows Denote Blood Vessels, and Arrowheads Highlight New Tissue Formation.

Histological Evaluation of Collagen Deposition Using Masson's Trichrome Staining. Representative Sections from the Inflammatory Phase (Day 3) Show Minimal Collagen Deposition in the Vehicle (A), 0.2% OM-Treated (D), and 1% OM-Treated (G) Groups. During the Proliferative Phase (day 8), Mild Collagen Deposition is Observed in the Vehicle (B), 0.2% OM-Treated (E), and 1% OM-Treated (H) Groups. by the Regenerative Phase (day 14), the Vehicle Group (C) Exhibits Moderate Collagen Formation Consistent with Natural Healing Progression, the 0.2% OM Group (F) Demonstrates Dense and Well-Organized Collagen Deposition, and the 1% OM Group (I) Shows Moderately Increased But Disorganized Collagen Structure. All Sections Were Stained with Masson's Trichrome and Examined Under a Light Microscope at 200× Magnification.

Comparative Analysis of Relative Gene Expression Levels During Inflammatory Phase at Day 3. Each Panel Represents the Relative Expression Levels of Key Genes Involved in Inflammation (A), Growth Factor Signaling (B), Intracellular Pathways (C), and Extracellular Matrix Remodeling (D). Expression Levels are Normalized to the Vehicle Control Group (set at 100), With Bars Representing Vehicle (gray), 0.2% OM (Light Orange), and 1% OM (Orange) Treatments. Data are Expressed as Mean ± Standard Error of the Mean (SEM). n = 8 Rats for Each Measurement in Each Group. * and ** Denote P < 0.05 and 0.01 Compared to the Vehicle Group. Statistical Analysis was Performed Using the Ordinary One-Way Followed by Dunnett's Multiple Comparison Post-Hoc Test.
Proliferative Phase
By day 8, granulation tissue formation in the 0.2% OM group was significantly higher (mean ± SEM: 2.3 ± 0.13) compared to the vehicle group (1.5 ± 0.13, P = 0.038), and angiogenesis was also significantly increased (2.7 ± 0.23 vs 1.8 ± 0.16, P = 0.023). The 1% OM group did not show significant differences in granulation tissue formation (2.0 ± 0.31, P = 0.105) or angiogenesis (2.2 ± 0.13, P = 0.059) compared to the vehicle group. Wound closure was significantly higher in the 0.2% OM group (57 ± 3.9%) compared to the vehicle group (39 ± 3.2%, P = 0.014). The 0.2% OM concentration significantly decreased the expression of p38 MAPK (P = 0.005) compared to the vehicle group (Figure 5). This reduction was accompanied by a significant decrease in the expression of IL-1β (P = 0.001) and IL-6 (P = 0.002), consistent with reduced inflammatory signaling. Notably, the levels of TGF-β1 and PDGF were significantly elevated in the 0.2% OM group (P = 0.008 and 0.001). MMP9 levels were significantly reduced in the 0.2% OM group (P = 0.003). In 1% OM groups, TGF-β1 and AKT1 expression levels has shown significant changes (P = 0.018, and 0.041) compared to the vehicle group.

Comparative Analysis of Relative Gene Expression Levels During Proliferative Phase at Day 8. Each Panel Represents the Relative Expression Levels of Key Genes Involved in Inflammation (A), Growth Factor Signaling (B), Intracellular Pathways (C), and Extracellular Matrix Remodeling (D). Expression Levels are Normalized to the Vehicle Control Group (Set at 100), with Bars Representing Vehicle (Gray), 0.2% OM (Light Orange), and 1% OM (Orange) Treatments. Data are Expressed as Mean ± Standard Error of the Mean (SEM). n = 8 Rats for Each Measurement in Each Group. * and ** Denote P < 0.05 and 0.01 Compared to the Vehicle Group. Statistical Analysis was Performed Using the Ordinary One-way Followed by Dunnett's Multiple Comparison Post-hoc Test.
Regenerative Phase
By day 14, the 0.2% OM group demonstrated significantly higher collagen formation scores (mean ± SEM: 3.0 ± 0.30) compared to the vehicle group (2.3 ± 0.13, P = 0.021), as evidenced by Masson's trichrome staining (Figure 3). Angiogenesis scores were also significantly higher in the 0.2% OM group (3.5 ± 0.09) compared to the vehicle group (2.5 ± 0.38, P = 0.038). The 1% OM group did not show significant changes in collagen formation (2.5 ± 0.16, P = 0.476) or angiogenesis (2.8 ± 0.38, P = 0.357) compared to the vehicle group. The 0.2% OM group, also exhibited the highest wound closure percentage (87 ± 2.8%) compared to both the vehicle group (69 ± 3.9%, P = 0.01) and the 1% OM group (73 ± 3.2%, P = 0.045). The 0.2% OM concentration significantly increased the expression of ERK and p38 MAPK (P = 0.008 and 0.002) compared to the vehicle group (Figure 6). In the 0.2% OM group, the levels of TGF-β1 and PDGF remained elevated (P = 0.001 and 0.003), and α-SMA exhibited a significant expression surge (P < 0.001). Additionally, collagen type I expression was significantly higher in the 0.2% OM group (P = 0.007).
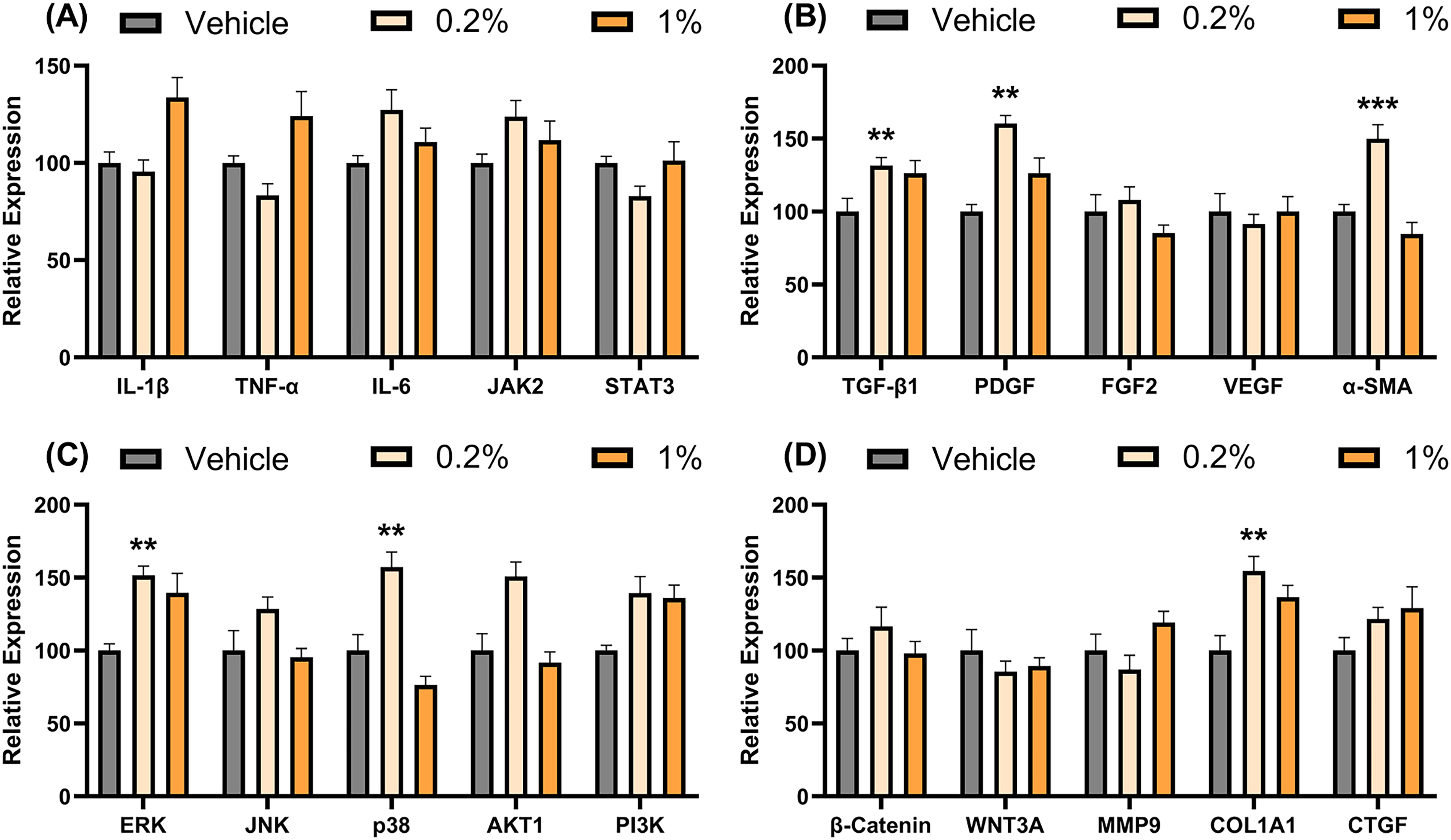
Comparative Analysis of Relative Gene Expression Levels During Regenerative Phase at Day 14. Each Panel Represents the Relative Expression Levels of Key Genes Involved in Inflammation (A), Growth Factor Signaling (B), Intracellular Pathways (C), and Extracellular Matrix Remodeling (D). Expression Levels are Normalized to the Vehicle Control Group (Set at 100), with Bars Representing Vehicle (gray), 0.2% OM (light orange), and 1% OM (orange) Treatments. Data are Expressed as Mean ± Standard Error of the Mean (SEM). n = 8 Rats for Each Measurement in Each Group. *, **, and *** Denote P < 0.05, 0.01 and 0.001 Compared to the Vehicle Group. Statistical Analysis was Performed Using the Ordinary One-Way Followed by Dunnett's Multiple Comparison Post-hoc Test.
Discussion
Our study demonstrates that 0.2% OM extract significantly enhances wound healing by modulating key signaling pathways, particularly MAPKs, while reducing inflammation, promoting fibroblast proliferation, and supporting collagen organization during the regenerative phase. These findings highlight OM's potential as a promising therapeutic agent for wound care and tissue regeneration. However, the 1% OM extract did not yield comparable benefits, likely due to the complex interactions of bioactive compounds at higher concentrations. The reduced efficacy may result from potential cytotoxicity, differential peak activity of specific compounds, or an imbalance in bioactive interactions. This underscores the importance of dose optimization to maximize OM's therapeutic potential.
Pro-inflammatory cytokines including IL-1b, IL-6, and TNF-a, exhibit persistently high levels during both acute and chronic wound phases. 38 Reducing these cytokines can accelerate wound repair, as their excessive activation dysregulates healing by triggering immune-mediated pathways. Among these, IL-1β is particularly disruptive, as IL-1 receptor knockout has been shown to improve wound closure across different healing phases. 39 In contrast, IL-6 plays a dual role, acting as either detrimental or beneficial depending on timing and context. While excessive IL-6 can prolong inflammation, it also promotes fibroblast-derived growth factors and enhances myofibroblast survival, contributing to wound closure. 40 At the signaling level, MAPK pathways are essential for key wound-healing mechanisms such as migration and proliferation. JNK, ERK, and AKT inhibition impairs keratinocyte migration, and JNK knockout disrupts fibroblast motility, delaying repair.41,42 Additionally, p38 MAPK stimulates growth factor secretion, promoting epithelial migration, while ERK activation drives cell proliferation, highlighting the coordinated role of these pathways in effective healing. 43 However, excessive activation of these pathways can hinder the healing process. For instance, IL-1β-mediated p38 MAPK activation has been shown to inhibit fibroblast proliferation and migration by upregulating MMP9 and MMP2, leading to delayed wound closure. 44
During the inflammatory phase (peaks around Day 3), 36 0.2% OM treatment led to a reduction in TNF-α, IL-1β, and JNK expression. In contrast, 1% OM treatment increased p38 expression and collagen production. Activation of AKT and ERK through the p38 phosphorylation enhances the collagen production. 45 While p38 phosphorylation is known to enhance collagen synthesis via AKT and ERK activation, neither AKT nor ERK expression increased in our study, suggesting that collagen production occurred independently of these pathways. Consistent with our findings, p38 inhibition has been shown to reduce collagen I expression and limit hypertrophic scarring in wound healing models, reinforcing p38 role in collagen I regulation. 46 However, while collagen I deposition is essential for tissue repair, excessive early production may contribute to scarring and delayed wound healing.
Anti-inflammatory effects of phenolic, terpenoid, and alkaloid compounds found in Onosma microcarpum are well-documented in wound healing models. 47 Consistently, our findings demonstrate OM's anti-inflammatory properties, particularly during the inflammatory phase, by reducing IL-1β and TNF-α levels. In line with our results, berberine, a phytochemical present in some Onosma species, and Zizhu extract have been shown to enhance wound healing by inhibiting JNK activity.48,49 However, most OM-derived compounds and epidermal growth factor are associated with increased JNK activity during healing, 50 suggesting that the reduced JNK expression observed in our study is likely an indirect effect resulting from modulation of other pathways or reduced tissue damage, rather than a direct inhibition of JNK signaling. Additionally, lupeol, deoxyshikonin, and shikonin, bioactive compounds present in the Onosma genus, have been shown to accelerate healing by activating the p38 pathway.51,52 In agreement with this, we observed p38 activation following 1% OM treatment on day 3 and 0.2% OM treatment on day 14. However, unlike 0.2% OM, early p38 activation in the 1% group was accompanied by increased collagen production, which may promote scarring rather than efficient wound healing.
During the proliferative phase (peaks around day 8), 0.2% OM administration continued to suppress inflammation, as indicated by lower IL-1β and IL-6 expression. This anti-inflammatory effect was accompanied by increased TGF-β and PDGF expression, which are crucial for establishing a foundation for wound repair. Additionally, 0.2% OM treatment led to reduced p38 and MMP9 expression, aligning with previous findings that IL-1β inhibits fibroblast proliferation and migration by upregulating MMP9 and MMP2 via p38 activation. 44 This regulatory pattern is consistent with the effects of activated protein C, which improves wound healing through a dynamic p38 phosphorylation pattern, where p38 activity increases at Day 3 but declines by Day 6. 53 In contrast, 1% OM administration was associated with a reduction in AKT and TGF-β expression. Since the AKT pathway is a key upstream regulator of TGF-β, this suggests that 1% OM may exert detrimental effects by suppressing TGF-β production via AKT inhibition, a pathway known to be essential for efficient wound healing. 54 In addition to the potential dose-dependent biphasic effects of OM's active compounds, several natural constituents found in OM extract have been reported to exert differential effects on the AKT pathway. This may explain the inverse regulation observed in our study, where compounds with inhibitory effects on AKT become more prominent at higher concentrations. For instance, resveratrol and coumaric acid, compounds commonly present in the Onosma genus, have demonstrated dose- and time-dependent biphasic effects on AKT phosphorylation, 55 aligning closely with our findings. Moreover, other OM-derived constituents such as apigenin and quercetin exhibit opposing effects on AKT signaling: apigenin enhances AKT phosphorylation and expression, while quercetin inhibits its activation. 56 These complex interactions may contribute to the reduced therapeutic efficacy observed with the 1% OM formulation.
Thymol, the most abundant compound in the essential oil of OM extract, has been shown to enhance wound healing and epithelialization by exerting anti-inflammatory effects, reducing IL-1, IL-17, and TNF-α levels.57,58 Similarly, carvacrol, another major compound in OM extract, has demonstrated protective effects against burn damage by modulating inflammation, angiogenesis, and apoptosis. 59 It has also been reported to promote collagen type I synthesis60,61 and improve healing by regulating IL-1β, TNF-α, and TGF-β expression. 62 Luteolin, a polyphenolic compound in OM extract, has been shown to accelerate wound healing in diabetic rats, particularly on Day 14, by downregulating IL-1β, TNF-α, IL-6, and MMP9, findings that are consistent with our results. 63 Additionally, rosmarinic acid contributes to wound repair through its antioxidant activity, 64 while apigenin improves healing by inhibiting collagenase and hyaluronidase on Day 12. 65 Apigenin also suppresses TNF-α and IL-1β, while increasing IL-10 and TGF-β through the promotion of macrophage polarization to the M2 phenotype, further supporting its role in tissue regeneration. 66 These findings collectively reinforce our data, highlighting the therapeutic potential of OM extract in wound healing.
During the regenerative phase (peaks around day 14), pro-inflammatory cytokines and pathways play a minimal role, and as expected, OM extract did not alter inflammatory markers. However, 0.2% OM treatment led to increased p38 and ERK expression, accompanied by elevated TGF-β, PDGF, α-SMA, and collagen type I production, fully supporting tissue regeneration. The coordinated activation of p38 and ERK pathways is essential for growth factor-induced migration and proliferation, where inhibition of either pathway can reduce healing, and their simultaneous inhibition results in a complete loss of growth factor effects. Additionally, blocking one pathway can trigger a compensatory increase in the other, highlighting their interdependent role in the healing process. 43 Beyond their direct role in promoting healing, p38 and ERK also mutually enhance growth factor production, reinforcing their pro-regenerative effects. Moreover, p38 activation of ERK further supports collagen synthesis, contributing to effective tissue repair. 45
Despite its modulatory effects on MAPKs, OM extract had minimal impact on the JAK/STAT and AKT/PI3 K pathways, as well as VEGF expression, suggesting that combining OM extract with other therapeutic agents targeting these pathways may yield more robust wound-healing outcomes. Although OM extract exhibited a general trend toward improved healing, the inferior results observed with 1% OM treatment, along with its inconsistent modulation of pathways and factors, suggest either potential toxicity or an imbalance in bioactive compounds. It is possible that healing-promoting compounds are more effective at lower doses, while at higher concentrations, some components may counteract these benefits, emphasizing the importance of dose optimization to maximize therapeutic efficacy.
Extracts from Onosma species with comparable phytochemical compositions have not demonstrated systemic or in vitro toxicity, even at concentrations similar to our 1% formulation.67,68 However, at extreme doses, some OM-derived compounds may exert cytotoxic effects through mechanisms such as oxidative stress induction, as seen with thymol/carvacrol mixtures and apigenin, which exhibit EC50 values of 50–100 µM.69,70 Nevertheless, key OM phytochemicals—including thymol, luteolin, rosmarinic acid, carvacrol, docosene, and apigenin—have demonstrated acceptable safety profiles for fibroblasts, keratinocytes, and topical skin application,71–76 suggesting that the inferior performance of 1% OM is more likely due to differential pathway modulation rather than direct cytotoxicity to skin or dermal cells. Furthermore, formulation factors can influence compound composition and concentration, as demonstrated by the distinct phytochemical profiles of methanolic versus oil-based OM extracts.13,14 Additionally, the ointment base itself may modify compound efficacy by altering the wound environment, affecting oxygen availability, redox potential, or pH, which could influence the stability and activity of bioactive compounds.
This study has several limitations that may affect the generalizability and reproducibility of the findings. The sample size was limited to male Wistar rats, which may not fully represent biological responses in other species or females. Additionally, reliance on a single plant source and extraction method may introduce variability, potentially affecting the consistency of the extract's pharmacological properties. 12 The use of gamma irradiation for sterilization, while effective, may alter the chemical composition of both the ointment base and extract, influencing the results. Another limitation is the lack of a comprehensive analysis of the extract's bioactive compounds. While the extraction and formulation methods are described, the study does not quantify or identify specific phytochemicals, limiting the ability to standardize and optimize OM extract for therapeutic use. Additionally, pathway involvement was assessed only at the transcriptional level, preventing direct evaluation of protein phosphorylation. While qPCR provides a stable measure of pathway regulation over time, a more comprehensive approach including phosphorylation assessments would clarify pathway activation. Furthermore, since gene expression changes may result from indirect pathway interactions, confirming a causal relationship between OM extract and MAPK modulation would require selective inhibitors or genetic models. Another limitation is the absence of a positive control for benchmarking efficacy. However, due to Onosma microcarpum anti-inflammatory, regenerative, and antimicrobial properties, no single standard treatment would comprehensively represent its multifunctional effects. Future studies should include comparative analyses with established wound-healing agents to evaluate specific aspects of OM treatment. Finally, this study focused on a specific set of molecular markers and mechanical properties, potentially overlooking other biological factors critical to wound healing.
Conclusion
Administration of OM on excisional wounds in rats demonstrated significant therapeutic potential in optimizing wound healing. The bioactive compounds, likely mediate this effect by modulating key signaling pathways specific to each phase of wound healing. These compounds support a balanced inflammatory response, controlled cell proliferation, and effective tissue regeneration. Future research should focus on detailed studies of the concentration-dependent effects of these compounds, their synergistic interactions with other wound healing agents, and clinical trials to validate their efficacy and safety in human wound healing.
Footnotes
Acknowledgements
The authors declare no competing interests or financial relationships that could potentially influence this work. All animal procedures were conducted in accordance with the guidelines approved by the Animal Ethics Committee of Tehran University of Medical Sciences, and we adhered to the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines throughout the study. We extend our gratitude to the Department of Plant Biology at the University of Tehran for their valuable contribution to species identification.
ORCID iDs
Ethical Approval
Ethical approval for reporting this case was granted by the Animal Ethics Committee of Tehran University of Medical Sciences (approval number IR.TUMS.IKHC.REC. No.1399.341).
Funding
The authors report no financial or personal relationships with individuals or organizations that could have influenced this work. There are no professional or personal interests in any product, service, or company. This study was supported by Tehran University of Medical Sciences (IR.TUMS.VCR.REC. No.02-5-161-74903)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Statement of Human and Animal Rights
All animal procedures in this study were conducted following the guidelines approved by the Animal Ethics Committee of Tehran University of Medical Sciences (approval number IR.TUMS.IKHC.REC. No.1399.341) and in compliance with the NIH Guide for the Care and Use of Laboratory Animals (eighth edition, 2011).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
