Abstract
Objectives
Liver injury is a major global public health concern. Despite advancements in modern medicine, there remains a pressing need to identify more effective and less toxic therapeutic options for managing liver diseases. This study aimed to assess the hepatoprotective potential of Erythroxylum daphnites Mart., Erythroxylum suberosum A.St.-Hil., Eugenia dysenteria (Mart.) DC., Pouteria torta (Mart.) Radlk., and Pouteria ramiflora (Mart.) Radlk. against acetaminophen (APAP)-induced injury in human hepatocarcinoma (HepG2) cells.
Methods
The hepatoprotective potential of the extracts was evaluated in HepG2 cells using APAP as the injury-inducing agent. Additionally, the chemical composition of the extracts was analyzed using high-performance liquid chromatography coupled with diode-array detection (HPLC-DAD).
Results
Chemical analysis of the plant extracts revealed the presence of polyphenolic compounds, including flavonoids and phenolic acids. All tested extracts exhibited a significant protective effect against APAP-induced cell death in both pre-treatment and post-treatment protocols.
Conclusion
This study highlights the potential of natural products from the Cerrado biome as hepatoprotective agents. The observed protective effects are likely attributed to the diverse chemical compounds present in these extracts.
Introduction
Drug-induced liver injury (DILI) represents the most prevalent cause of acute liver failure in numerous countries worldwide. 1 According to a systematic review and meta-analysis by Li et al (2023), the overall incidence of DILI in the general population was estimated at 4.94 per 100 000 person-years across 14 studies. 2 Regional variations in DILI incidence have been reported, with rates of 2.70 per 100 000 inhabitants in the United States (US), 3 and a significantly higher annual incidence of 23.80 per 100 000 persons in mainland China. 4 N-Acetyl-p-aminophenol or paracetamol (APAP) is the leading cause of acute liver failure in the US, certain regions of Europe, and Australia, accounting for over 50% of such cases.5,6 In addition to conventional pharmaceutical drugs, the increasing use of herbal and dietary supplements has emerged as a significant public health concern in the US, contributing to approximately 20% of acute DILI cases. 7 Similarly, in China, traditional Chinese medicines and herbal and dietary supplements stood out as the primary cause of DILI, responsible for 26.8%. 4 Geographical differences in the etiology of DILI have also been observed. In Western countries, antibiotics are the most common drugs associated with DILI, whereas in Eastern countries, traditional Chinese medicines predominate as the leading cause. 8 These findings underscore the global burden of DILI and highlight the need for continued research and public health initiatives to address this critical issue.
APAP is an analgesic and antipyretic drug, one of the most commonly used drugs in the world, and is generally well tolerated at therapeutic doses. However, an overdose (starting at doses between 7.5 g and 14 g in healthy adults or above 150 mg/kg in children younger than 6 years) can lead to extensive liver injury and even acute liver failure (plasmatic concentrations over 200 µg/mL 4 h after the ingestion are predictive for hepatic toxicity).9,10
The Consumer Healthcare Products Association (CHPA) reported that approximately 23% of US adults (52 million consumers) use an APAP-containing medicine each week. 11 Global APAP sales are estimated to be approximately USD 11 000 million in 2025 and are projected to reach more than USD 16 000 million by 2035, driven in part by the high incidence of infections, cold and chronic pain management, as well as increased access to nonprescription medication in specific world regions. 12 High consumption and indiscriminate use contribute to cases of intoxication by APAP.
Currently, DILI treatment depends on early diagnosis and withdrawal of the suspected medication, which is essential to prevent or minimize progressive damage. Some substances can be administered in specific situations, such as N-acetylcysteine for APAP intoxication or carnitine in case of damage by valproate. Ursodeoxycholic acid has been used in cases of cholestasis. In other situations, such as severe liver disease, without improvement after the drug is discontinued, cholestasis with hypersensitivity, or autoimmunity phenomena, corticosteroids have been recommended. 13
Even with the significant advances in modern medicine, no completely effective drugs provide adequate stimulation of liver function, offer total protection to the liver, or help regenerate liver cells. In the case of N-acetylcysteine for APAP intoxication, the treatment is effective only in instances of acute poisoning. 14 Therefore, it becomes necessary to identify more efficient and less toxic therapeutic options for treating liver diseases.
Researchers have explored the safety and efficacy of plant extracts and compounds derived from plants as standalone treatments or in conjunction with conventional drugs, aiming to mitigate the toxicity associated with APAP. 15
In a recent study, we explored the potential hepatoprotective effects of four Cerrado plant species, Cheiloclinium cognatum (Miers) A.C.Sm, Guazuma ulmifolia Lam., Hancornia speciosa Gomes, and Hymenaea stigonocarpa Mart. ex Hayne, against APAP toxicity. Utilizing an in vitro assay with human hepatocarcinoma (HepG2) cells, the extracts exhibited a protective impact, shielding HepG2 cells from potential APAP toxicity. These findings suggest that these plant extracts hold promise as potential supplements to prevent liver damage. 16
Building upon the information mentioned above, the primary aim of this manuscript was to assess the hepatoprotective capabilities against APAP-induced injury of Erythroxylum daphnites Mart., Erythroxylum suberosum A.St.-Hil., Eugenia dysenteria (Mart.) DC., Pouteria torta (Mart.) Radlk., and Pouteria ramiflora (Mart.) Radlk. using a HepG2 cell line. Furthermore, the hepatotoxic activity was also evaluated.
These five Brazilian species are native plants from the Cerrado biome. Eugenia dysenterica contains phenolic compounds such as tannins and flavonoids, along with volatile components including essential oils like β-caryophyllene, α-humulene, limonene, α-thujene, α-terpineol, and β-caryophyllene oxide. Its leaves have been used in folk medicine for their antidiarrheal properties and in the treatment of heart conditions. Studies have reported various pharmacological potentials, including enzyme inhibition, antioxidant, antifungal, antimicrobial, antidiarrheal, antiulcer, antitumor, and antiviral activities. 17 Erythroxylum daphnites hexane extract has demonstrated strong cytotoxic activity and an apoptosis-inducing effect in an oral cancer cell line. 18 Flavonoids such as kaempferol, quercetin, ombuin, as well as mono- and diglycosides, have been identified in E. daphnites. 19 Erythroxylum. suberosum has been used in Brazilian folk medicine as an anti-rheumatic, anesthetic, and remedy for indigestion. Compounds such as isoquercitrin, rutin, quercetin, and the isomers catechin and epicatechin have been identified, 20 along with tricyclic diterpenes. 21 Antitumoral activity has been described.18,22 Extracts from various parts of P. torta (leaves, stem, bark, or fruits) exhibit various biological activities, including cytotoxicity18,23,24 and antifungal 25 enzyme inhibition in vitro.26-28 Among the chemical compounds present in this plant are α- and β-amyrin, lupeol, taraxasterol, pseudotaraxasterol, cycloartenol, lanosterol, α- and β-friedelinol, and betulinic and ursolic acids, as well as their derivatives. 29 The presence of miricetin-3-O-β-D-galactopyranoside, miricetin-3-O-α-L-arabinopyranoside, and miricetin-3-O-α-L-rhamnopyranoside has also been reported. 30 Pouteria ramiflora is traditionally used as an antihyperlipidemic, vermifuge, antifungal, and antioxidant agent for the treatment of dysentery, pain, and inflammation. Studies showed their antitumoral, antimutagenic, antifungal, and neuroprotective activity, as well as in vitro α-amylase and α-glucosidase inhibition. Chemical compounds described in this plant were polyphenols as myricetin-3-O-β-D-galactopyranoside, myricetin-3-O-α-L-ramnopyrananoside and catechins. 31
Methods
Plant Material and Extraction Process
Leaves of the species described in Table 1 were collected in Brasília and its surroundings, Brazil, and identified by botanist Christopher William Fagg. An exsiccate was deposited in the Herbarium of the University of Brasília (UB).
Collection Data and Identification of Plant Material.

Leaves were dried at room temperature, then powdered in a knife mill, and subjected to extraction by infusion at a ratio of 1:10. 32 The aqueous extract was then lyophilized (VirTis SP Scientific Advantage Plus XL-70 Benchtop Freeze Dryer) and stored at −20 °C.
The yield of the aqueous extracts was calculated using the weight (g) of the powdered dry leaves of each species, as 100% of the material. After obtaining the dried extracts, the material was weighed, and a relationship between the weight of the final product and the initial material was established.
This project was registered at SisGen (National System for the Management of Genetic Heritage and Associated Traditional Knowledge) under the code A215A9A.
Chromatographic Profile Evaluation
All extracts were analyzed using high-performance liquid chromatography coupled with diode-array detection (HPLC-DAD) LaChrom Elite (Hitachi, Tokyo, Japan), equipped with L2130 pump, L2200 automatic sampler, and L2455 DAD detector as described by Leite et al (2014). 33
All samples were solubilized to a concentration of 1 mg/mL in methanol and subsequently filtered through a disposable syringe filter with a 0.45 µm Polyvinylidene fluoride membrane prior to analysis. Compounds present in the extracts were characterized and identified by comparing the similarity of their ultraviolet and visible spectra and by determining their retention times with commercially available standards. The phytochemical marker content in the sample was calculated using linear regression analysis based on the standard. The following commercial standards were used for comparison: chlorogenic acid (Sigma®), caffeic acid (Cromadex®), ellagic acid (Cromadex®), ferulic acid (Sigma®), gallic acid (Sigma®), rosmarinic acid (Cromadex®), catechin (Sigma®), epicatechin (Sigma®), isoquercitrin (Cromadex®), hesperidin (Sigma®), hyperoside (Sigma®), myricetin (Sigma®), quercetin (Cromadex®) and rutin (Sigma®). The analyses were performed in triplicate.
Cell Culture
Human hepatocarcinoma cells (HepG2) code 0291, obtained from the cell bank of Rio de Janeiro, were used. Cells were grown in sterile Dulbecco's Modified Eagle Medium (DMEM) with pH 7.0, supplemented with 10% fetal bovine serum (Thermo Fisher Scientific®), 100 IU/mL penicillin, 100 μg/mL streptomycin, and 3.7 g/L sodium bicarbonate. The cells were incubated with 5% carbon dioxide (CO2) at 37 °C.
Cell Viability Assay
Cytotoxicity assays for HepG2 cells were performed using an adaptation of the colorimetric method with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma-Aldrich) as described by Mosmann 34 and Hansen et al. 35 A cell density of 2.5 × 104 cells per well was used in 96-well plates. The cells were treated for 24 h with extracts diluted in DMEM at concentrations ranging from 25 to 1000 µg/mL. Cell cytotoxicity was calculated by comparing the absorbance obtained after treatment with plant extracts with that of the untreated control (DMEM) and was expressed as the percentage of viable cells after treatment in relation to the control. The half maximal effective concentration (EC50) values were determined by averaging at least three independent assays.
Studies have shown that polyphenols can interfere with the MTT assay.4,36 To evaluate the interference of the extracts in the MTT assay, a test was conducted using the same protocol described above, but without HepG2 cells.
Hepatoprotective Assay
Hepatoprotective assays using HepG2 cells were performed with adaptations as described by Ribeiro et al (2023). 16
The induction of liver cell damage was achieved through APAP overdose in HepG2 cells. To determine the concentration of APAP required to reduce cell viability by 50%, cytotoxicity assays were conducted using APAP concentrations ranging from 5 to 50 mM over a 24-h exposure period. APAP was diluted in DMEM without fetal bovine serum, supplemented with 1% dimethyl sulfoxide (DMSO) as solvent. This experimental setup was designed to establish the dose-response relationship and identify the optimal concentration for subsequent studies on hepatotoxicity.
After determining the APAP concentration required for 50% viable cells, the extracts were tested to assess their potential hepatoprotective effect on HepG2 cells. Only extract concentrations that maintained cell viability above 80% after 24 h of treatment were selected for further evaluation. HepG2 cells were seeded at a density of 2.5 × 104 cells per well in a 96-well plate and allowed to adhere for 24 h. After this incubation period, the DMEM was removed, and the cells were exposed to 25 mM APAP for 24 h to induce hepatotoxicity. Subsequently, the APAP-containing medium was removed, and the cells were treated with the extract solutions for an additional 24 h. Cell viability was then assessed using the same methodology described in the cytotoxicity assay. In addition to the post-treatment protocol, the hepatoprotective potential of the extracts was also evaluated using a pre-treatment approach. In this setup, the cells were exposed to extracts for 24 h prior to APAP treatment, aiming to determine whether prior exposure to the extracts could confer protective effects against APAP-induced hepatotoxicity.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism version 8 (GraphPad Software, San Diego, CA, USA) and Microsoft Office Excel® version 2504. Data normality was assessed using the Shapiro–Wilk test. The one-way analysis of variance (ANOVA) was employed to determine cell viability, followed by Tukey's multiple comparison test. A significance criterion of P < .05 was applied to all analyses.
Results
Chromatographic Profile Evaluation
In HPLC-DAD analysis, some compounds in the plant extracts were identified, as described in Table 2 and Figures 1–5. Chlorogenic acid, rutin, and hyperoside were identified in the aqueous extract of E. daphnites (EDAE), with contents of 0.77, 0.60, and 1.82 µg/mL, respectively (Figure 1). The aqueous extract of E. suberosum (ESAE) contained rutin, catechin, and epicatechin. The concentration of these compounds was quantified as 0.86 µg/mL for rutin, 0.58 µg/mL for catechin, and 0.26 µg/mL for epicatechin (Figure 2).

Chromatographic Profile of Erythroxylum daphnites Mart. Aqueous Extract (EDAE). Analysis Conditions: LiChroCART 150-4.6 Purospher STAR RP-18 Column (5 μm) with LichroCART 4-4.6 Purospher STAR RP 18, pre-Column (5 μm), Maintained at 25 °C. Eluent: Aqueous Phosphoric Acid (1%) (Solvent A) and Acetonitrile (Solvent B). Analysis Time: 55 min, at a 0.6 mL/min Flow Rate. Data Extracted at 354 nm. 33

Chromatographic Profile of Erythroxylum suberosum A.St.-Hil. Aqueous Extract (ESAE). Analysis Conditions: LiChroCART 150-4.6 Purospher STAR RP-18e Column (5 μm) with LichroCART 4-4.6 Purospher STAR RP 18e pre-Column (5 μm), Maintained at 25 °C. Eluents: Aqueous Phosphoric Acid (1%) (Solvent A) and Acetonitrile (Solvent B). Analysis Time: 55 min, at a 0.6 mL/min Flow Rate. Data Extracted at 280 nm. 33
Compounds Identified in the Plant Extracts.
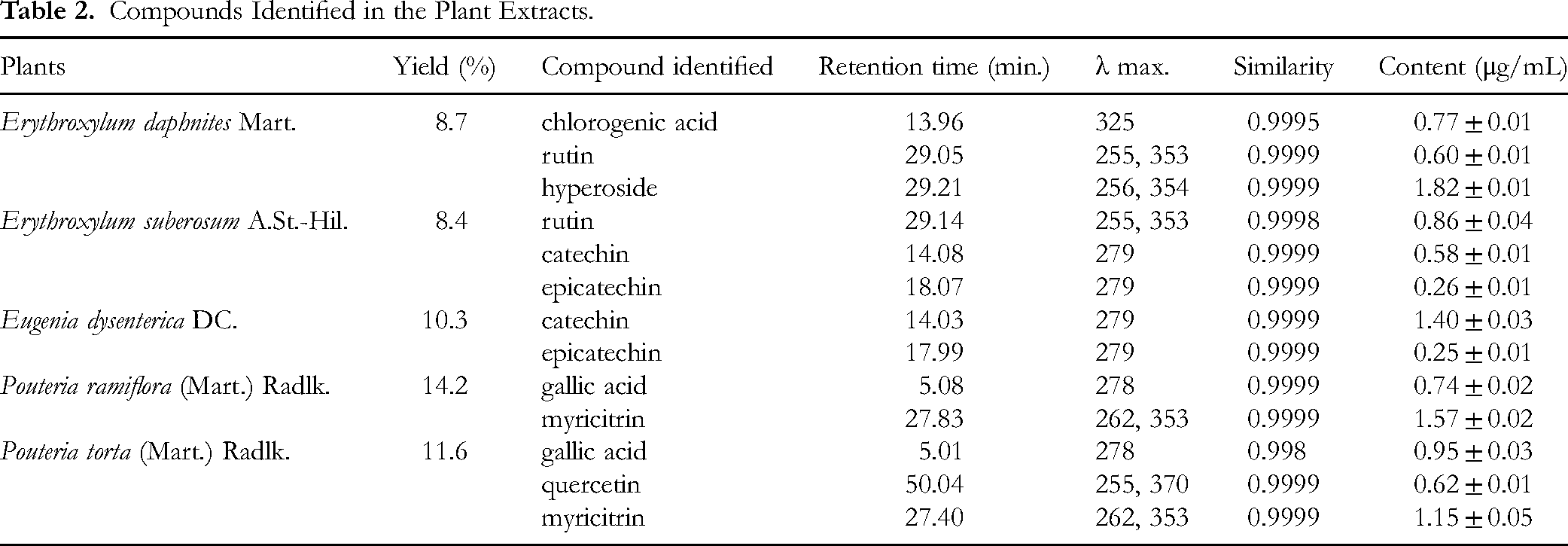
Catechin and epicatechin were also identified in the E. dysenterica aqueous extract (EUAE), with contents of 1.40 and 0.25 µg/mL, respectively (Figure 3). For P. ramiflora aqueous extract (PRAE), the HPLC-DAD analysis revealed the presence of gallic acid (0.74 µg/mL) and myricitrin (1.57 µg/mL). The chromatographic profile demonstrating the presence of these compounds is illustrated in Figure 4.

Chromatographic Profile of Eugenia dysenterica (Mart.) DC. Aqueous Extract (EUAE). Analysis Conditions: LiChroCART 150-4.6 Purospher STAR RP-18e Column (5 μm) with LichroCART 4-4.6 Purospher STAR RP 18e pre-Column (5 μm), Maintained at 25 °C. Eluents: Aqueous Phosphoric Acid (1%) (Solvent A) and Acetonitrile (Solvent B). Analysis Time: 55 min, at a 0.6 mL/min Flow Rate. Data Extracted at 280 nm. 33

Chromatographic Profile of Pouteria ramiflora (Mart.) Radlk. Aqueous Extract (PRAE). Analysis Conditions: LiChroCART 150-4.6 Purospher STAR RP-18e Column (5 μm) with LichroCART 4-4.6 Purospher STAR RP 18e pre-Column (5 μm), Maintained at 25 °C. Eluents: Aqueous Phosphoric Acid (1%) (Solvent A) and Acetonitrile (Solvent B). Analysis Time: 55 min, at a 0.6 mL/min Flow Rate. Data Extracted at 354 nm. 33
The aqueous extract of P. torta (PTAE) chromatographic profile also showed the presence of gallic acid and myricitrin, as well as quercetin. The concentrations of these compounds were quantified as 0.95 µg/mL for gallic acid, 1.15 µg/mL for myricitrin, and 0.62 µg/mL for quercetin (Figure 5).

Chromatographic Profile of Pouteria torta (Mart.) Radlk. Aqueous Extract (PTAE). Analysis Conditions: LiChroCART 150-4.6 Purospher STAR RP-18e Column (5 μm) with LichroCART 4-4.6 Purospher STAR RP 18e pre-Column (5 μm), Maintained at 25 °C. Eluents: Aqueous Phosphoric Acid (1%) (Solvent A) and Acetonitrile (Solvent B). Analysis Time: 55 min, at a 0.6 mL/min Flow Rate. Data Extracted at 354 nm. 33
The compounds identified in the plant extracts were analyzed based on their distinctive properties, including ultraviolet and visible spectra, retention time, and comparison with commercially available standards. These findings provide valuable insights into the chemical composition of the extracts, thereby deepening our understanding of their potential applications and benefits.
Cell Viability Assay
The cell viability curves for HepG2 treated with the plant extracts are depicted in Figure 6. The EC50 values determined for each extract were as follows: 465.9 µg/mL for EDAE, 642.9 µg/mL for ESAE, 466.8 µg/mL for EUAE, and 652.6 µg/mL for PTAE, respectively. In contrast, PRAE exhibited cytotoxicity in HepG2 cells only at the lowest tested concentration (25 µg/mL), resulting in an 80.8% reduction in cell viability. Notably, PRAE promoted a significant improvement in cell viability within the 200-1000 µg/mL concentration range, suggesting a potential protective or proliferative effect at higher concentrations.

Cell Viability Curves in HepG2 Cells from Different Extracts. EDAE: Erythroxylum daphnites Mart. Aqueous Extract. ESAE: Erythroxylum suberosum A.St.-Hil. Aqueous Extract. EUAE: Eugenia dysenterica DC. Aqueous Extract. PRAE: Pouteria ramiflora (Mart.) Radlk. Aqueous Extract. PTAE: Pouteria torta (Mart.) Radlk. Aqueous Extract. the Values Were Determined by Averaging at Least Three Independent Assays.
An interference test was conducted to assess the potential impact of the extracts on the MTT assay. The results indicated that EDAE, PTAE, and PRAE exhibited minimal interference (absorbance readings below 0.1) at concentrations exceeding 500 µg/mL. The extracts ESAE and EUAE showed no interference at any concentration tested.
To ensure significant cell viability and biological relevance, extract concentrations that maintained cell viability above 80% were selected for use in the hepatoprotective assay. The chosen concentrations were 100 µg/mL for EDAE and PTAE, 200 µg/mL for ESAE, 50 µg/mL for EUAE, and 25 µg/mL for PRAE. These concentrations were optimal for evaluating the hepatoprotective potential of the samples while minimizing interference and maintaining cellular integrity.
Hepatoprotective Assay
To determine the EC50 of APAP, cytotoxicity assays were conducted using concentrations ranging from 5 to 50 mM over a 24-h exposure period. The EC50 of APAP was approximately 25 mM, which was subsequently used in the hepatoprotective assay to induce liver cell damage.
The hepatoprotective effect of the extracts on HepG2 cells is illustrated in Figure 7. The results from the pre-treatment of HepG2 cells with different extract solutions, followed by exposure to APAP, are shown in Figure 7A. All tested extracts demonstrated a significant protective effect against APAP-induced cell death. Compared to APAP treatment alone, cell viability improvement was quantified as follows: 20.1% for EDAE, 23.1% for ESAE, 18.5% for EUAE, 28.4% for PRAE, and 25.3% for PTAE.
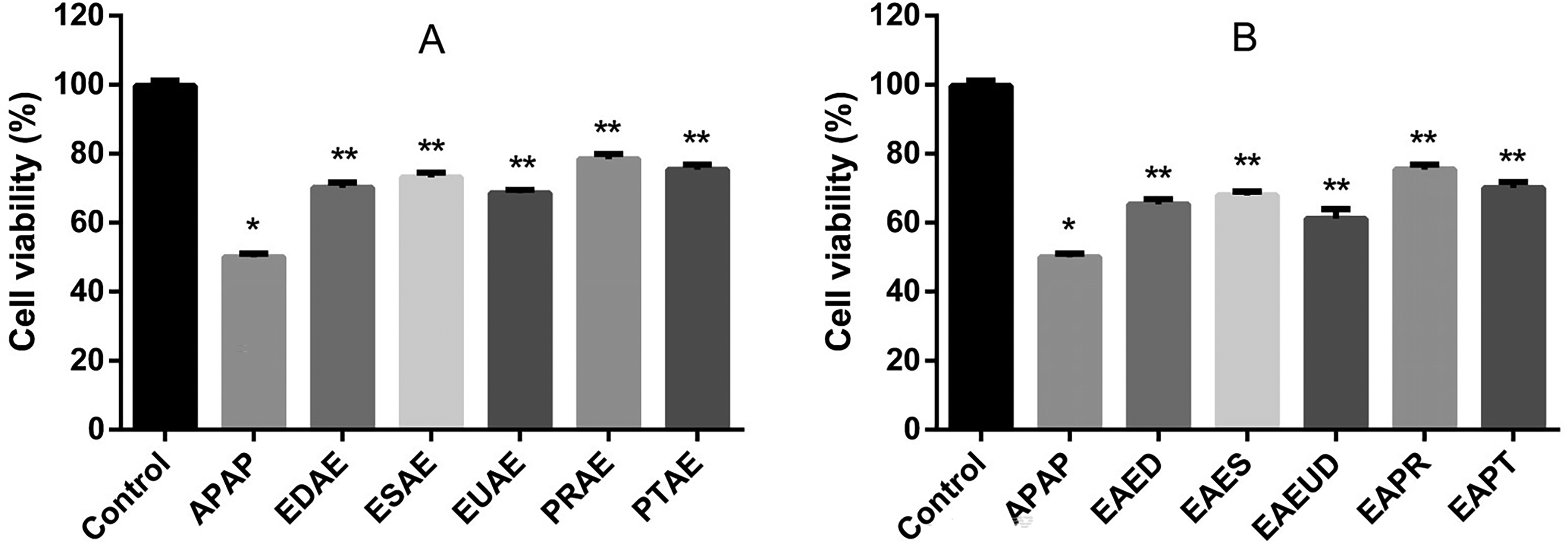
Hepatoprotective effect in HepG2 cells from different extracts. EDAE: Erythroxylum daphnites Mart. aqueous extract (100 µg/mL), ESAE: Erythroxylum suberosum A.St.-Hil. aqueous extract (200 µg/mL), EUAE: Eugenia dysenterica DC. aqueous extract (50 µg/mL), PRAE: Pouteria ramiflora (Mart.) Radlk. aqueous extract (25 µg/mL), PTAE: Pouteria torta (Mart.) Radlk. aqueous extract (100 µg/mL), APAP: N-acetyl-p-aminophenol (25 mM). A: pre-treatment and B: post-treatment containing extract solutions. The values were determined by averaging at least three independent assays. *P < .05 in relation to control and **P < .05 in relation to APAP group using one-way Anova followed by Tukey's multiple comparisons.
The results from the post-treatment of HepG2 cells with different extract solutions after exposure to APAP are presented in Figure 7B. Like the pre-treatment results, all extracts tested in the post-treatment protocol demonstrated a significant protective effect against APAP-induced cell death. The improvement in cell viability, compared to APAP treatment alone, was quantified as follows: 15.3% improvement for EDAE, 17.8% for ESAE, 11.1% for EUAE, 25.4% for PRAE, and 19.9% for PTAE, demonstrating their ability to mitigate, although partially, the APAP-induced cytotoxicity, even when administered after the onset of liver cell damage.
Discussion
In order to identify more efficient and less toxic therapeutic options for the treatment of liver diseases, the present study evaluated the hepatoprotective potential against APAP injury of aqueous extracts obtained from two Erythroxylaceae species (E. daphnites and E. suberosum), two Sapotaceae (P. torta and P. ramiflora), and one Myrtaceae
Initially, the cytotoxic potential of the extracts was evaluated on HepG2 cells to determine the concentration that maintained cell viability above 80%. These concentrations were subsequently used in the hepatoprotective assay. Based on the results, the cytotoxicity of the aqueous extracts can be ranked in descending order as follows: EDAE exhibited the highest cytotoxicity, followed by EUAE, ESAE, and PTAE, which showed the lowest cytotoxicity. At the same time, PRAE promoted a significant improvement in cell viability.
According to Nelson (1990) and Jaeschke et al (2020), APAP-induced liver damage initiates with the metabolism of the drug by cytochrome P450 enzymes, resulting in the formation of the reactive metabolite N-acetyl-p-benzoquinone imine (NAPQI).37,38 This metabolite depletes cellular glutathione and creates protein adducts, particularly affecting mitochondrial proteins. The depletion of glutathione leads to oxidative stress within the mitochondria, which activates the mitogen-activated protein c-Jun N-terminal kinase (JNK) and facilitates its translocation to the mitochondria. Once there, phosphorylated JNK binds to the outer membrane protein Sab (SH3 domain-binding protein that preferentially associates with Btk), disrupting the electron transport chain. This disruption causes the release of superoxide from respiratory complexes I and III. Superoxide subsequently reacts with nitric oxide in the mitochondria, producing the highly reactive nitrogen species peroxynitrite, which nitrates tyrosine residues on proteins, further worsening mitochondrial dysfunction. This series of events ultimately triggers the mitochondrial permeability transition, releasing pro-apoptotic factors such as endonuclease G and apoptosis-inducing factors into the cytosol. These factors then translocate to the nucleus, inducing deoxyribonucleic acid fragmentation and ultimately resulting in hepatocyte necrosis. These molecular mechanisms driving the hepatotoxic effects of APAP could offer potential targets for preventing and treating acute liver damage. Du et al (2016) suggested that strategies aimed at reducing or neutralizing mitochondrial reactive oxygen species (ROS) and peroxynitrite represent some of the most promising therapeutic approaches for mitigating APAP-induced liver toxicity in patients. 39
Natural products have been widely studied for their potential to prevent APAP-induced toxicity. In a previous study, we demonstrated the protective effects of extracts from C. cognatum, G. ulmifolia, H. speciosa, and H. stigonocarpa against APAP-induced toxicity in an in vitro HepG2 cell model. The protection observed in cell viability was associated with a decrease in reactive oxygen species levels. The hepatoprotective effects of these extracts are likely attributed to their antioxidant compounds, such as rutin and mangiferin. 16
Badr et al (2023) reviewed the potential of traditional hepatoprotective plants as effective and safe alternatives to protect against APAP overdose. The authors highlighted the protective potential of various plants, including Aloe vera (L.) Burm. f., Ocimum basilicum L., Morus nigra L., Nigella sativa L., Elettaria cardamomum (L.) Maton, Coffea arabica L., Saussurea lappa Decne. C.B. Clarke, Curcuma longa L., Taraxacum officinale F.H.Wigg., Phoenix dactylifera L., Zingiber officinale Roscoe, Camellia sinensis (L.) Kuntze, Gynura procumbens (Lour.) Merr., Simmondsia chinensis (Link) C.K. Schneid., Cymbopogon citratus (D.C.) Stapf, Glycyrrhiza glabra L., Silybum marianum (L.) Gaertn., Salvadora persica L., Punica granatum L., Hibiscus sabdariffa Linn, Salvia rosmarinus Spenn., Suaeda vermiculata Forssk. ex J.F.Gmel., Rhus coriaria L., Helianthus annuus L., and Citrus x aurantium L., and concluded that natural products have demonstrated potential as protective agents against APAP toxicity by alleviating oxidative stress, inflammation, and cellular damage. 40
Silymarin, derived from milk thistle seeds, is a remedy with a centuries-old history of treating hepatic conditions. Known for its antioxidant properties, silymarin functions as a free radical scavenger and regulates enzymes linked to the progression of hepatic damage. 41
In the present study, chemical analysis of five Cerrado's plant extracts revealed the presence of polyphenolic compounds, including flavonoids and phenolic acids. Chromatographic analysis of E. daphnites identified chlorogenic acid, rutin, and hyperoside; in E. suberosum, rutin, catechin, and epicatechin were found; E. dysenterica contained catechin and epicatechin; P. ramiflora showed gallic acid and myricitrin; and P. torta presented gallic acid, quercetin, and myricitrin.
Flavonoids and phenolic acids have been known due to their pharmacological properties, mainly antioxidant 42 and anti-inflammatory activities. In vitro and cellular studies have shown that flavonoids exert anti-inflammatory effects by suppressing the production and activity of various pro-inflammatory mediators, including eicosanoids, cytokines, adhesion molecules, and C-reactive protein. At the molecular level, these compounds modulate key signaling pathways by inhibiting transcription factors such as nuclear factor kappa-B (NF-κB) and activator protein 1 (AP-1), while also promoting the activation of nuclear factor-erythroid 2-related factor 2(Nrf2). 43
In the present study, all the tested extracts demonstrated a significant protective effect against cell death induced by APAP in both treatment protocols. This protective effect is likely attributed to the chemical compounds present in these extracts, mainly flavonoids and phenolic acids. Some of these compounds have been previously investigated for this purpose and have demonstrated efficacy, including quercetin, catechin, chlorogenic acid, and gallic acid.
Quercetin is a prominent flavonoid known for its widespread distribution throughout the plant kingdom. Research indicates that quercetin exhibits a protective effect against liver toxicity induced by APAP, demonstrating efficacy comparable to that of N-acetylcysteine, a well-established antidote for APAP poisoning. 44 El-Shafey et al (2015) demonstrated that quercetin may have a protective function against APAP-induced hepatorenal toxicity in rat models. This protective effect is associated with a decrease in oxidative and nitrosative stress in both hepatic and renal tissues, alongside its capacity to enhance mitochondrial energy production. 45 Pingili et al (2019) showed that quercetin can potentially inhibit the metabolism of APAP mediated by cytochrome P 2E1 (CYP2E1). This inhibition may lead to a decrease in the formation of NAPQI, thereby offering protective benefits for both the liver and kidneys. 46
Catechin and epicatechin are phenolic compounds predominantly found in green tea (Camellia sinensis L.). Lv et al (2020) showed that these green tea polyphenols offer protection against liver injury induced by APAP in mice. This protective effect may be attributed to their role in regulating drug-metabolizing enzymes and transporters following an APAP overdose. 47 Another study suggested that catechin may also inhibit the production of NAPQI, thereby preventing adduct formation and reducing the depletion of microsomal thiol content. 48
In addition, Rasool et al (2010) showed that gallic acid possesses a potent hepatoprotective effect in APAP-intoxicated mice due to its antioxidant activity. 49 Moreover, chlorogenic acid appears to mitigate liver injury induced by APAP, likely through a modest inhibition of the enzymatic activities of CYP2E1 and cytochrome 1A2 (CYP1A2). Furthermore, key cellular antioxidant signals, including the peroxiredoxin (Prx) isoforms 1, 2, 3, 5, and 6, the enzyme Soluble Epoxide Hydrolase-2 (Ephx2), RNA Polymerase II subunit k (Polr2k), and Nrf2, play a significant role in enhancing the protective effects of chlorogenic acid against oxidative stress caused by APAP. 50 Zheng et al (2015) demonstrated that the protective effects of chlorogenic acid against APAP-induced hepatic inflammation are mediated through the involvement of Toll-Like Receptors 3 and 4 (TLR3/4) and Fator Nuclear Kappa B (NFκB) signaling pathways. 51
One limitation of this study is that it was conducted in vitro using the HepG2 cell line, which may not fully replicate in vivo conditions. To confirm the protective effects of these extracts against APAP-induced cytotoxicity, additional in vivo studies are required.
Conclusion
All tested extracts, obtained from Cerrado plants E. daphnites, E. suberosum, E. dysenteria, P. torta, and P. ramiflora, exhibited a significant protective effect against cell death induced by APAP in the HepG2 cell line, both during pre-treatment and post-treatment protocols. This protective effect is likely due to the presence of bioactive flavonoids and phenolic acids in the plant extracts. According to the literature, the main compounds present in these extracts appear to exert their effects primarily by reducing mitochondrial oxidative stress and inhibiting cytochrome P450 enzymatic activity, thereby disrupting APAP metabolism and preventing NAPQI formation. Since these promising findings are based on in vitro experiments, preclinical and clinical studies should be considered to test the efficacy and safety of humans suffering from liver injury.
Footnotes
Acknowledgement
The authors are grateful to the University of Brasília (UnB), National Council for Scientific and Technological Development (CNPq), and Federal District Research Support Foundation (FAPDF).
Author Contributions
All authors listed have made substantial, direct, and intellectual contributions to the work and have approved it for publication.
M C C Leão, P O Magalhães, D Silveira and Y M Fonseca-Bazzo: conceptualization of the overarching research goals and aims; development and design of the methodology; visualization and interpretation data; formal analysis of study data; preparation, creation and/or presentation of the published work; critical review and revision of the manuscript.
M Homem-de-Mello, C W Fagg, and L A Simeoni: data/evidence collection, data curation, formal analysis of study data, critical review and revision of the manuscript.
P O Magalhães, D Silveira and Y M Fonseca-Bazzo: acquisition of funding to support the research.
Y M Fonseca-Bazzo: study supervision, approval of the final version for publication; conceptualization of overarching research goals and aims, data curation, coordination of the overall research activity, and planning.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available on request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
