Abstract
Highly oxygenated nortriterpenoids are the principle bioactive chemical constituents isolated from plants of the genera Schisandra and Kadsura. The stereochemistry of these molecules remains a highly challenging problem to solve. To establish the stereochemistry and develop a general nuclear magnetic resonance-electric circular dichroism-computational approach to solve these complex metabolites, we reviewed the published methods to solve the stereochemistry of these triterpenoids. A key goal of this review is to provide a protocol to solve the problems hampering the assignment of the relative absolute configurations of other members of this natural product class.
Nortriterpenoids are characteristically hepatoprotective active ingredients of Schisandra chinensis and Kadsura longipedunculata, known as a “liver tonic” in Asian herbal medicine, relieving symptoms of chronic liver disease and poor liver function. These nortriterpenoids have attracted considerable attention not only because of the intriguing structures but also due to their promising bioactivities, including cytotoxic, 1,2 antiviral, 3,4 and hepatoprotective activities. 5,6 Until now, there are more than 140 triterpenoids that have been isolated and identified, categorized into the following 16 groups based on their structural characteristics and biogenetic viewpoints: schiartanes, 18(13→14)- abeoschiartanes, 18-norschiartanes, 14(13→12):16(17→13)-diabeoschiartanes, wuweiziartanes, lancischiartanes, 2,3-dinorlancischiartanes, 1,2,3-trinorlancischiartanes, preschisanartanes, 16,17-secopreschisanartanes, lancifoartanes, 12,22-cyclopreschisanartanes, 14-nor-16,17-secopreschisanartanes, schisanartanes, 3-norschisanartanes, and 13,16-secopreschisanartanes. 7 Their planar structures and relative configuration have been successfully elucidated by modern nuclear magnetic resonance (NMR) techniques, but solving the absolute configuration remains a challenge due to the presence of noncontiguous stereogenic centers as well as conformationally flexible side chains. Assignment of their absolute configuration is critical because it influences their bioactivity and is essential for future efforts focused on synthesis and medicinal chemistry applications. This review aims to present an overall review of the methods used to solve the absolute stereochemistry of the nortriterpenoids to provide a protocol for the assignment of related natural products.
Methods to Assign the Absolute Stereochemistry
X-Ray for Crystallized Metabolites
Single-crystal X-ray diffraction remains the gold standard in the structural elucidation of novel and known molecules. The absolute configuration of a number of nortriterpenods was unambiguously assigned by single-crystal X-ray diffraction using Cu or Mo Kα radiation. These include schilancitrilactone A, B, and C, 8 wuweizidilactone P, 9 arisanlactone A, 7 propindilactone H, 10 micrandilactone I, 6 micrandilactone H, 5 lancolides A, B, C, and D, 11 preschisanartanin O, 11 lancifonin A, 12 schicagenin A, 13 rubrifloradilactone C, 14 and wilsonianadilactone A 15 (Figure 1). However, a crystal that diffracts well to solve absolute configuration directly is always challenging. The relative configuration was assigned by X-ray diffraction, and the absolute configuration of micrandilactone J was further solved combined with electric circular dichroism (ECD) and DP4+.

Nortriterpenoid absolute stereochemistry solved by X-ray diffraction.
Noncrystallized Samples
However, it has proven difficult to obtain a crystal that diffracts well enough for X-ray, especially for the small quantities of natural products. 16 For the assignment of the relative configuration of natural products that will not crystallize and that have high molecular complexity and signal ambiguity, NMR-based techniques were applied including nuclear Overhauser effect (NOE), heteronuclear single quantum coherence (HSQC) overlay, and J-based configuration analysis (JBCA) in combination with molecular and quantum mechanics calculations, including DP4, 17 as well as DP4+. 18 For the absolute configuration, ECD and Mosher reaction methods have often been used. The C-23 in 18(13→14)-abeoschiartane, 18-norschiartane, and schisanartane, with an α,β-unsaturated-γ-lactone moiety, was assigned by ECD spectrum. The absolute C-23 configuration could be confirmed by the positive Cotton effect (CE) at 210 nm for 23R, and negative CE around 210 nm for 23S (Figure 2). 19 The absolute configuration of C-20 in 2,3-dinorlancischiartanes with an α,β,γ,δ-unsaturated-γ-lactone moiety could be determined by the ECD method, with a negative CE at 268 nm indicating an R configuration and a positive CE at 271 nm suggesting an S configuration (Figure 2). 8
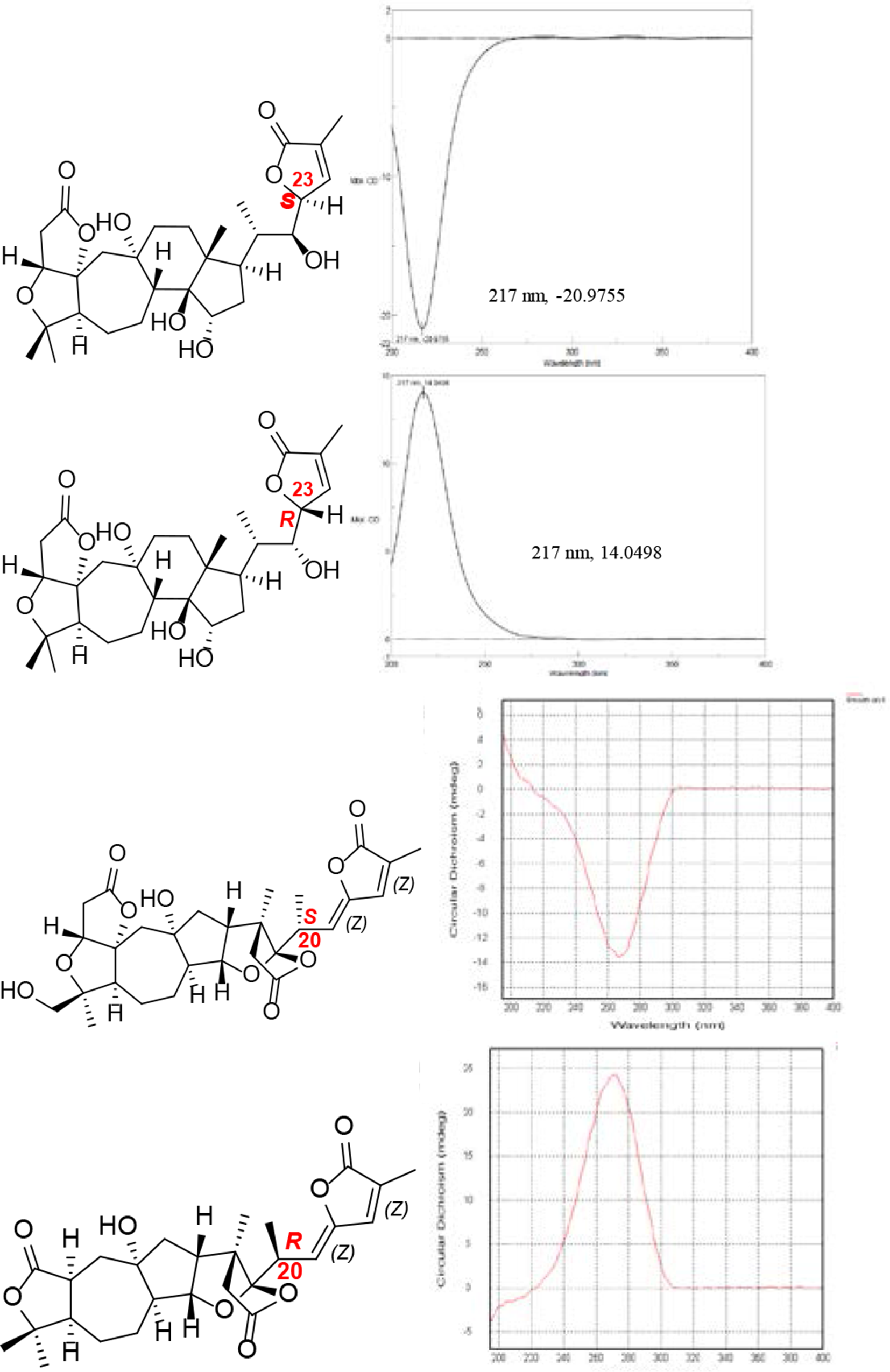
C-23 in 18(13→14)-abeoschiartane and C-20 in 2,3-dinorlancischiartanes.
JBCA data were utilized to determine the relative configurations of polyhydroxylated acyclic molecules by detailed analysis of the 3 J H, H and 2, 3 J C, H values, and thus can be applied to determine the relative configuration of the chiral carbons on the chain of 18(13→14)-abeoschiartane, preschisanartanes, and schisanartane. After the relative configuration was solved by JBCA (Figure 3), and combined with the absolute configuration of C-23 solved by ECD mentioned earlier, the absolute configuration of the chiral carbons on the chain would be solved.
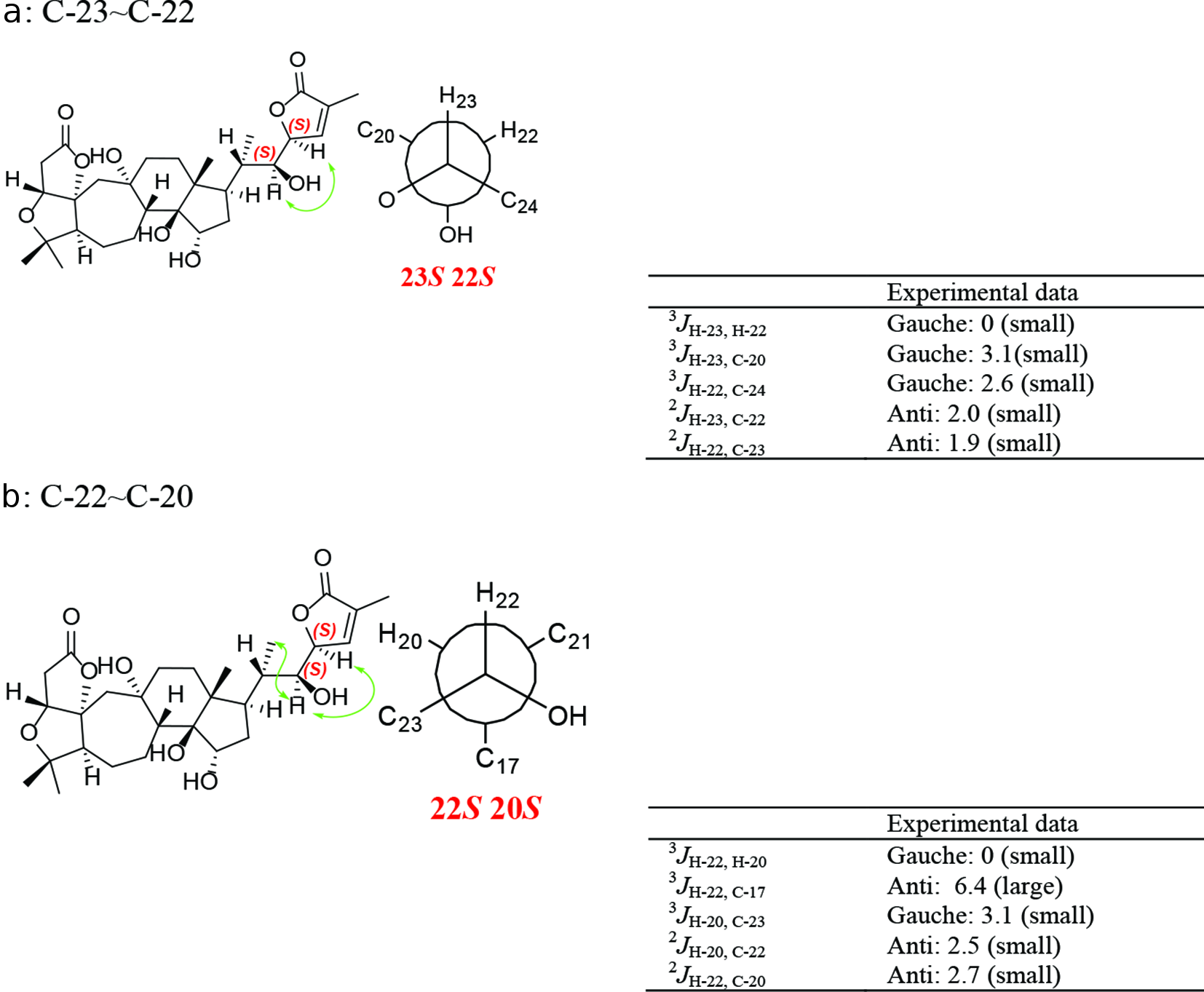
Rotamers and coupling constants for C-23 ~C-22 (a), C-22 ~C-20 (b) of micrandilactone J, key nuclear Overhauser effect spectroscopy correlation of micrandilactone J.
H-N HSQC overlay is frequently applied to monitor the binding and conformational changes of proteins based on the peak shift. 20 Also, H-C HSQC overlay was used to compare the configurations of isomers. In the overlaid HSQC spectrum of 2 isomers, the completely overlapped correlations indicate that they have the same relative configurations, which can also be confirmed by rotational Overhauser effect spectroscopy (ROESY) or NOE spectroscopy (NOESY) (Figure 4). For the nonprotonated stereogenic carbons, the signals of the adjacent carbons could be used to determine their relative configurations. Combined with the absolute configuration of chiral carbon shared by the chain and the ring, the absolute configuration of the chiral carbons on the ring will be solved.

Entire heteronuclear single quantum coherence (HSQC) overlay. The black parts of the structures are identical and the blue parts are different based on the HSQC data. Key nonoverlaid HSQC cross-peaks of 2 isomers are shown.
Conclusion
Single-crystal X-ray diffraction or X-ray diffraction combined provide the opportunity to solve stereochemistry unambiguously. However, for noncrystalline molecules, ECD was applied to determine the key chiral carbons’ absolute configuration; JBCA was applied to determine the relative configuration of chiral carbons on the chain; ROESY/NOESY or HSQC overlay helped to determine the relative configuration of chiral carbons on the ring. Combined, the absolute configuration solved by ECD and the relative configuration of chiral carbons solved by JBCA and HSQC overlay or ROESY/NOESY illustrate how the absolute configuration of all the chiral carbons can be solved for noncrystallized molecules (Figure 5).

Summary of the methods to solve the absolute stereochemistry.
Footnotes
Acknowledgments
This paper is dedicated to Dr N Rama Krishna on the occasion of his retirement, in appreciation of his career contributions to the field of NMR spectroscopy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by NCCIH (R01AT007318), China Scholarship Council, the Cooper Family, and the Abney Foundation.
