Abstract
Introduction
The Liver plays one of the most important human body organs due to its regulation of homeostasis, and neutralizing of toxins within the body, 1 it has multi-biological functions in carbohydrate, lipid, and protein metabolism. 2 Liver damage is caused by metabolic dysfunction, histopathological patterns, and associated diseases such as hepatitis, steatosis, neoplasms, and others. 3 Generally, the treatment of hepatic diseases causes various and serious long-term side effects;3,4 therefore, many studies showed interest in developing effective natural agents from medicinal plants in order to treat and prevent hepatic disorders. Treatment with medicinal plants has increased worldwide, as the use of herbs in an optimal way is harmless with no serious adverse reactions. 2
Cistus plants (rock rose) draw attention due to their active compounds such as phenolic compounds and terpenoids, well known for their different pharmacological activities. In Mediterranean traditional medicine, C. ladanifer have been used as herbal infusions or as decoction of the aerial parts for the prevention and the treatment of a variety of diseases and infections. It is commonly used as anti-inflammatory, anti-diarrhoeal, antiacid, antidiabetic, and antispasmodic anti-diarrhoeal, agents.4-6 C. ladanifer has been the subject of various studies as this plant presents significant pharmacological potential such as antimicrobial, antioxidant,7-10 anti-inflammatory, analgesic actions, 11 hypoglycemic and hypolipidemic,6,12 and cytotoxic effects.7,13,14 This plant is known for the presence of various compounds mainly Kaempferol, Punicalagingallate, Punicalin, and Punicalagin, Epigallocatechin, Ellagic acid, Gallic acid, Myricitrin and Quercetin.14-18 These compounds are known for their pharmacological effects and can be responsible for the plant therapeutic activities. Moreover, the whole plant, but mainly the leaves, secrete a very fragrant gum—labdanum—composed mainly of flavonoids and terpenes, in particular labdan-type diterpenes,19,20 it is traditionally used in low doses in sedative tea, coffee, and other infusions to prevent and to treat dysentery, diarrhea insomnia, catarrh, and anxiety.7,15,21,22
C. ladanifer is known for its pharmacological properties; however, it is little explored for its hepatoprotective activity. The aim of our work is to evaluate the hepatoprotective activity of C. ladanifer extracts in vivo and in silico and to identify their chemical profile.
Material and Methods
Plant Material
Leaves of C. ladanifer were harvested from the Taza region in May 2017 (006°28.382′ E; 004°49.405′ N). The authentication of the plant was effectuated by Dr Khabbach Abdelmajid. The leaves parts of C. ladanifer were dried at room temperature then powdered by a mechanical grinder. A quantity of 10 g was then macerated overnight with 100 mL of distilled water and methanol separately. The whole mixture was filtered and the filtrate was evaporated under pressure. The extracts were conserved at +4 °C until further use. The yield of both aqueous and methanolic extracts was calculated with a percentage of 6.64 ± 0.06% and 6.24 ± 0.08% respectively.
Hepatoprotective Activity of C. ladanifer Extracts
Animals
Adult male Swiss mice aged 2 months and weighing between 20 and 25 g were used to perform the current study. Animals were obtained from the animal house at the Department of Biology, Faculty of Sciences, Sidi Mohamed Ben Abdellah University. The animals were housed in controlled laboratory conditions with a set temperature (23 ± 2◦C) and light cycle (12 h light/dark cycles). The animals were allowed free access to food and water ad libitum. The Animal Ethics Review Committee at the faculty of Sciences, Fez, Morocco, approved this study under the ethical clearance number 02/2023/LBEAS. The use of laboratory animals in all experimental procedures was conducted according to ethical guidelines for the care and use of laboratory animals.
Paracetamol Dose Regimen
In order to evaluate the hepatoprotective effect of C. ladanifer extracts, the paracetamol tablets were used, each tablet contains 1000 mg of paracetamol. The tablets were obtained from a Bab Elghoul Pharmacy. The paracetamol was made into fine powder using a mortar and pestle. The powdered paracetamol was suspended in a saline solution and was administered according to the body weight of the mice. The dose administered to the mice was set as 1000 mg/kg. Grouping of mice and treatments. Thirty mice were randomly divided into three groups and each group consists of five mice. The first group received 1 mL/kg of saline (control). Group II was given paracetamol (1000 mg/kg) orally and group III received orally both 1000 mg/kg paracetamol and 100 mg/kg of the aqueous extract, respectively. Extract was administered 3 hours after the administration of paracetamol. Paracetamol 1000 mg/kg was given to mice to induce hepatotoxicity. The treatments were continued for 7 days and on the Eighth day of the experiment; all animals were anesthetized and dissected
Biochemical Parameters
At the end of the experimental period of the test, the animals were anesthetized with sodium pentobarbital at a dose of 30 mg/Kg before being sacrificed for blood collection. Blood samples were collected from the heart, in heparinized tubes (0.2 ml with 10 U/ml heparin) and then centrifuged at 1500 rpm for 10 min. Next, the recovered plasma was analyzed for aspartate aminotransferase (AST), alanine aminotransferase (ALT), creatinine, urea, cholesterol, and triglycerides (TG) parameters.
Histopathological Examination
Livers and kidney of mice were excised for histopathological examination. The tissues were washed and fixed in 10% formalin solution, dehydrated with alcohol, this step involves immersing the sample in increasing concentrations of alcohol to remove water and formalin from the tissues. Afterwards, an organic solvent such as toluene is used to remove the alcohol and allow infiltration of tissues with paraffin, and then enclosed in paraplast Afterward, tissues were embedded in paraffin (heated to 56 °C for 4 h in an drying oven), and sectioned using microtome. Sections of 5 µm thickness were cut and mounted on slides. The slides are then placed on a hot plate (15 min, 40 °C) for deparaffinization. Next, sections were stained with hematoxylin-eosin before being read using a light microscope. Hematoxylin binds to DNA, allowing it to stain the nucleus. Eosin binds to basic molecules, which allows the cytoplasm to be colored.
Chemical Composition
The identification of the compounds present in the extracts was carried out using the GC-MS technique according to the protocol detailed in our previous studies. 23 The mass spectra were matched to the NIST database for identification of compounds.
Computational Modeling
Our computational modeling investigations focused on the Human Cytochrome P450 2E1 protein and specific phytochemicals.24,25 To carry out these simulations, we utilized Maestro version 11.5 developed by Schrodinger, LLC. The procedural steps involved in our study encompassed the selection and preparation of the protein, preparation of ligands, creation of a receptor grid, and ligand docking.
Protein Preparation
The crystal structure of Human Cytochrome P450 2E1 (PDB ID: 3LC4) was obtained from the RCSB Protein Data Bank and underwent a series of steps, including examination, adjustments, and minimization through the use of the protein preparation wizard tool. The protein was subjected to preprocessing, which involved addressing missing loops and side chains using the Prime tool, with the PROPKA pH set at 7.4 for accurate ionization state predictions. Important residues were retained, followed by the addition of hydrogen atoms and the elimination of water molecules located more than 5 Å away. Subsequent energy minimization was conducted using the OPLS3e force field to achieve a protein structure in a low-energy state with an RMSD of 0.3 Å, which was then utilized in subsequent simulation investigations. 26
Ligand Preparation
To prepare ligands (phytochemicals extracted from our plant) for our molecular docking studies with the CYP2E1 protein, we employed the LigPrep tool. The ligands were subjected to preparation using the OPLS3e force field, and the pH was set at 7.4 ± 0.5 to generate potential ionization states. 27
Receptor Grid Generation
The receptor grid was generated by selecting atoms located within the binding site of the co crystallized ligand within the protein after it had been minimized. This specific region was earmarked for ligand docking and subsequent processing stages. The box has been identified under the following coordinates: X = -28.364, Y = -40.442, and Z = 30.193. 28
Molecular Docking
Using the receptor grid generated, the molecular docking studies were conducted in standard precision mode using the Glide tool. These investigations enabled us to uncover interactions between the ligands and the protein, along with associated docking scores. 28
Statistical Analysis
The data were presented as means and standard deviations. The statistical analysis was carried out by use of one-way analysis of variance, using Dunnett test along with GraphPad Prism 5 (GraphPad software, La Jolla, CA). When the P-value was less than .05, the values were statistically judged to be significant.
Results
Serum Biochemical Analysis
Effect of C. ladanifer Extracts on the Transaminases Levels and the Lipidic Profile
In order to evaluate the hepatoprotective activity of both aqueous and methanolic extracts, two doses were prepared 150 and 300 mg/kg, and the level of the AST (U/L), ALT (U/L) and lipidic profile (Total cholesterol and TG) was measured. As shown in Figure 1, the values of enzymatic activities of AST and ALT measured in the mice's serum treated with paracetamol alone were found to be significantly higher (P < .001) when compared to the group of mice treated with paracetamol along with C. ladanifer extracts. The levels of AST and ALT decreased when treated with the extracts, indicating the presence of an hepatoprotective activity from paracetamol-induced injury. However, the methanolic extract at the dose of 300 mg/kg showed some hepatic toxicity. Measurement of total cholesterol and TG levels is another indicator of the presence of an hepatic disorder. In Figure 2, decreased levels of cholesterol and TG were found in the positive control group compared to the negative presenting higher levels. When treated with paracetamol along with both C. ladanifer extracts, these levels were restored to normal in the mice's serum.
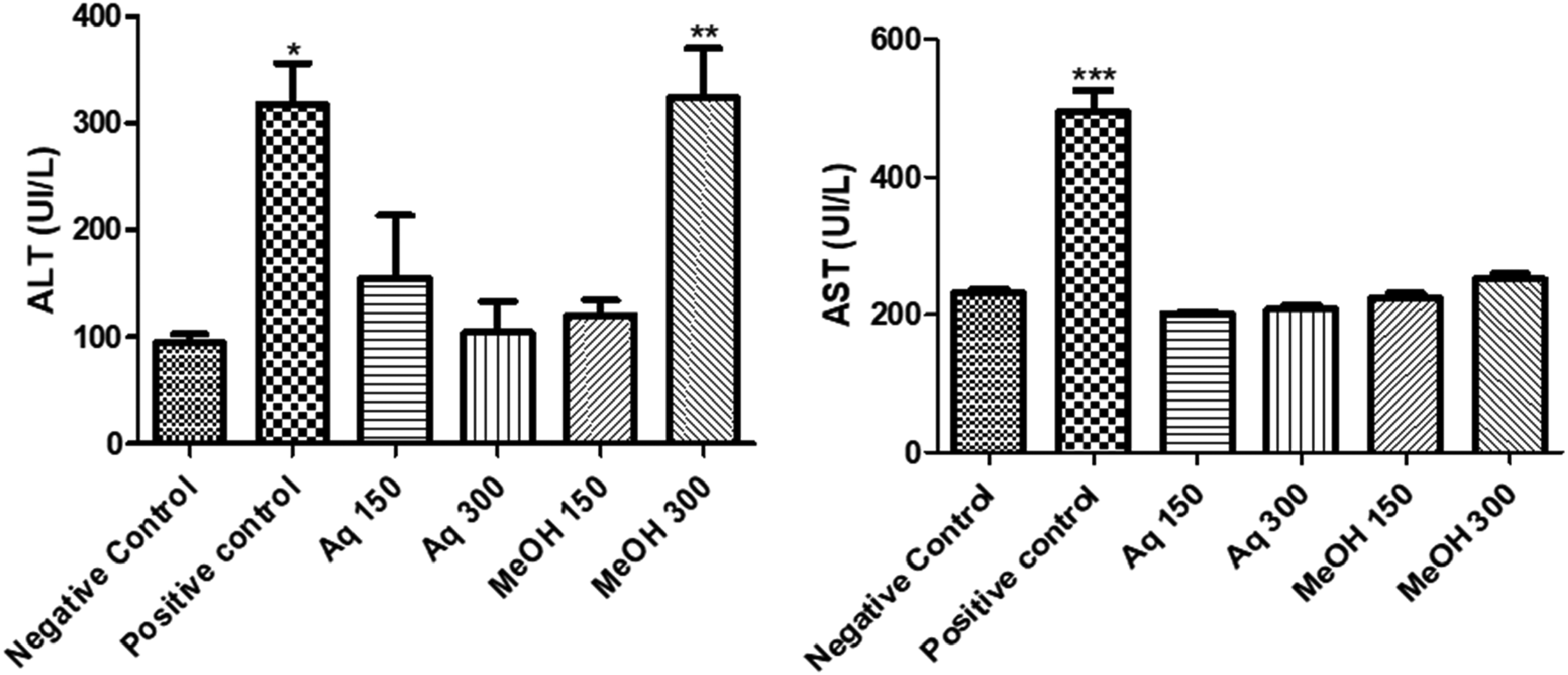
Effect of aqueous and methanolic extracts of C. ladanifer on Transaminsases (ALT and AST).Values significantly different compared to negative control: *P < .05; **:P < .01; ***P < .001. Aq: Aqueous extract; MeOH: Methanolic extract. AST, aspartate aminotransferase; ALT, alanine aminotransferase.
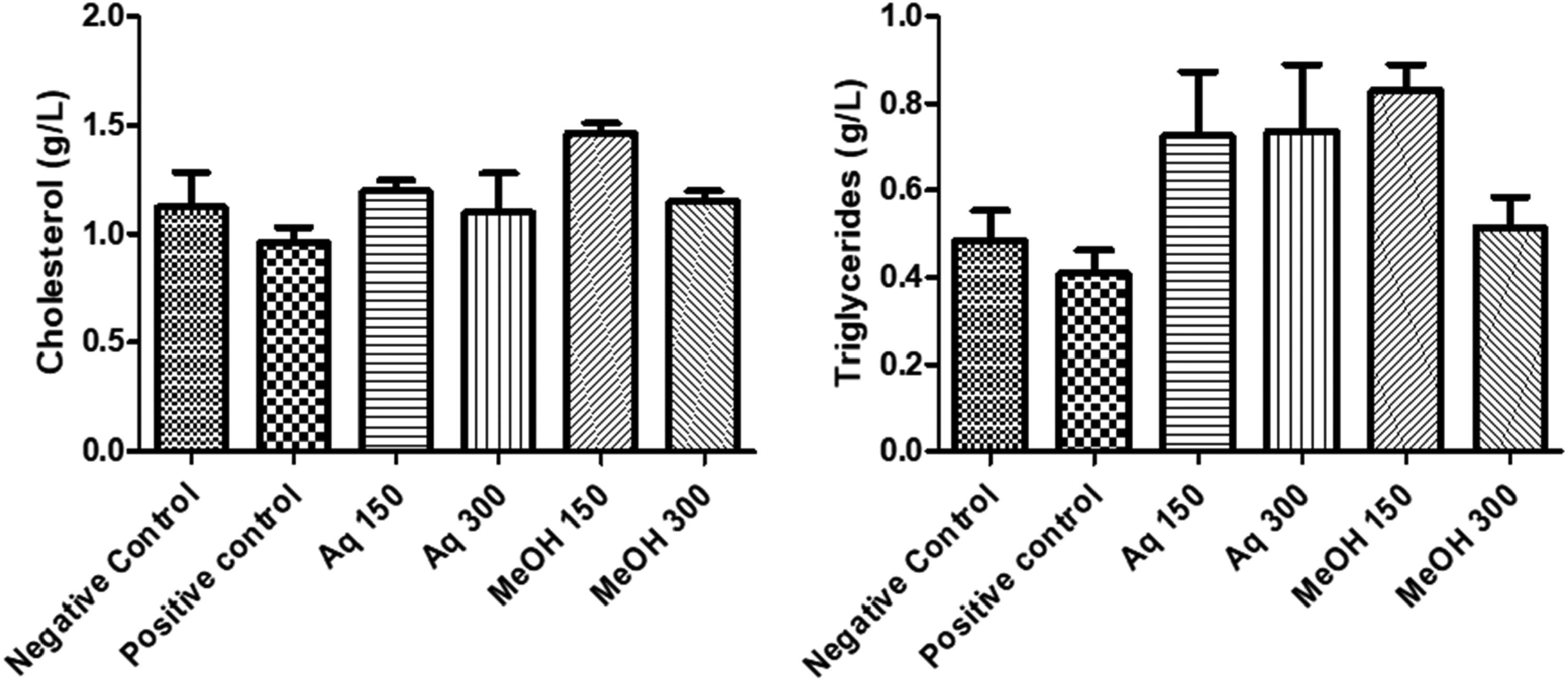
Effect of aqueous and methanolic extracts of C. ladanifer on Cholesterol and triglycerides. Aq: Aqueous extract; MeOH: Methanolic extract.
Effect of C. ladanifer Extracts on the Urea and Creatinine Levels
Urea and creatinine are measured as biomarkers of renal damage. Figure 3 shows that the negative group of mice presented high levels of urea and creatinine when compared to the positive control group. Treated mice with only paracetamol decreased the levels of the two biomarkers causing a renal damage. At tested concentrations, both aqueous and methanolic extracts increased the urea and creatinine levels in mice serum and were recovered to normal. However, mice treated with methanolic extract at 300 mg/kg showed a low concentration of urea compared to the positive control, indicating a toxicity.
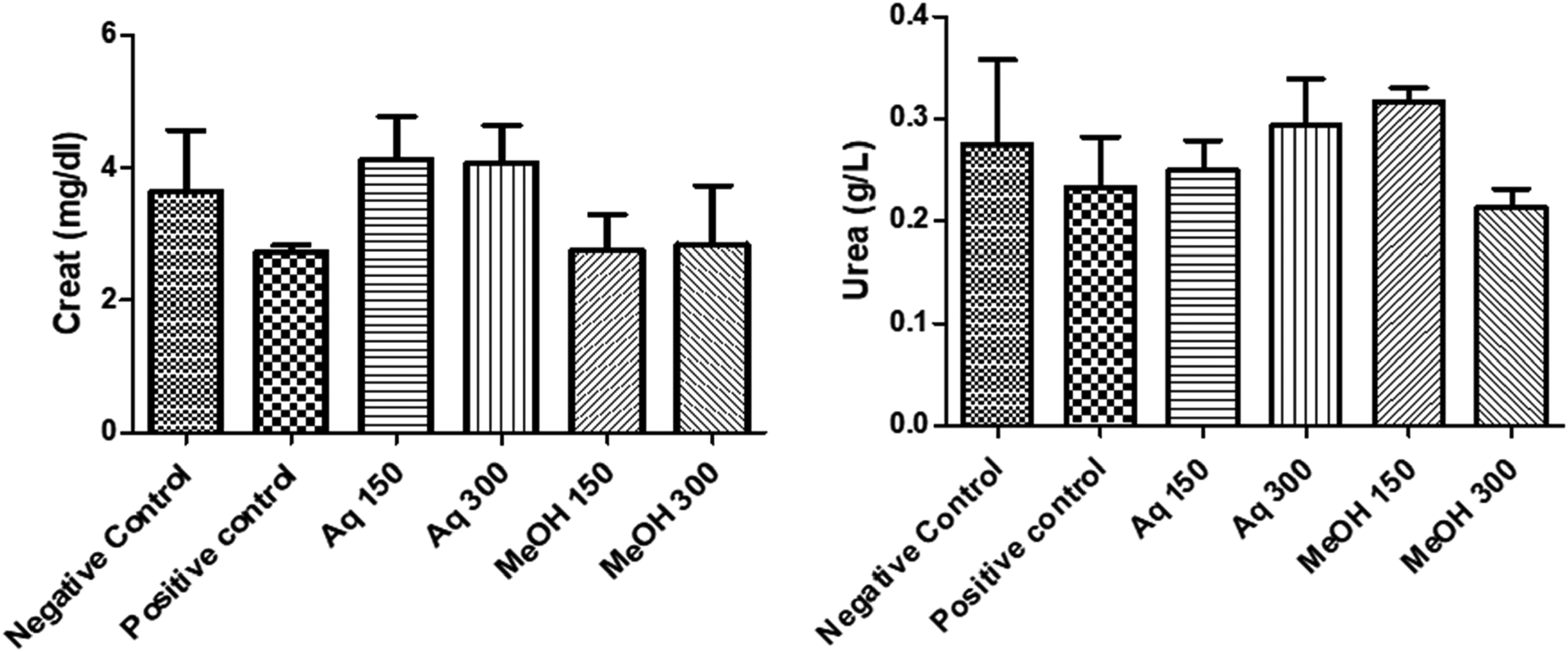
Effect of aqueous and methanolic extracts of C. ladanifer on urea and creatinine. Aq: Aqueous extract; MeOH: Methanolic extract.
Histopathological Alterations
The Histopathological analysis of the two organs Liver and kidney are shown in Figures 4 and 5. The histological analysis of animal kidneys didn’t reveal any pathological alterations that may be attributed to treatment with C. ladanifer aqueous extract at the doses of 150, and 300 mg/kg, and methanolic extract at 150 mg/kg respectively. The liver histology of rats treated with paracetamol at 1000 mg/kg, and the methanolic extract at the dose of 300 mg/kg demonstrated numerous pathological alterations, including sinus dilation, infiltration of inflammatory cells (lymphocytes), as well as necrosis. However, rats treated with aqueous extract at the doses of 150 and 300 mg/kg respectively and methanolic extract at the dose of 150 mg/kg didn’t show any signs of toxicity.

Histopathological photos of liver slices stained with hematoxylin-eosin (*40) after oral administration of paracetamol and the extracts. Liver section of groups treated with NaCl (A), paracetamol (B), aqueous extract (100 mg/kg) + paracetamol (C), aqueous extract (300 mg/kg) +paracetamol (D), methanolic extract (100 mg/kg) + paracetamol (E), methanolic extract (300 mg/kg) +paracetamol (F). H: Hepatocyte; CV: Central vein; L: Lymphocyte; N: Necrosis.
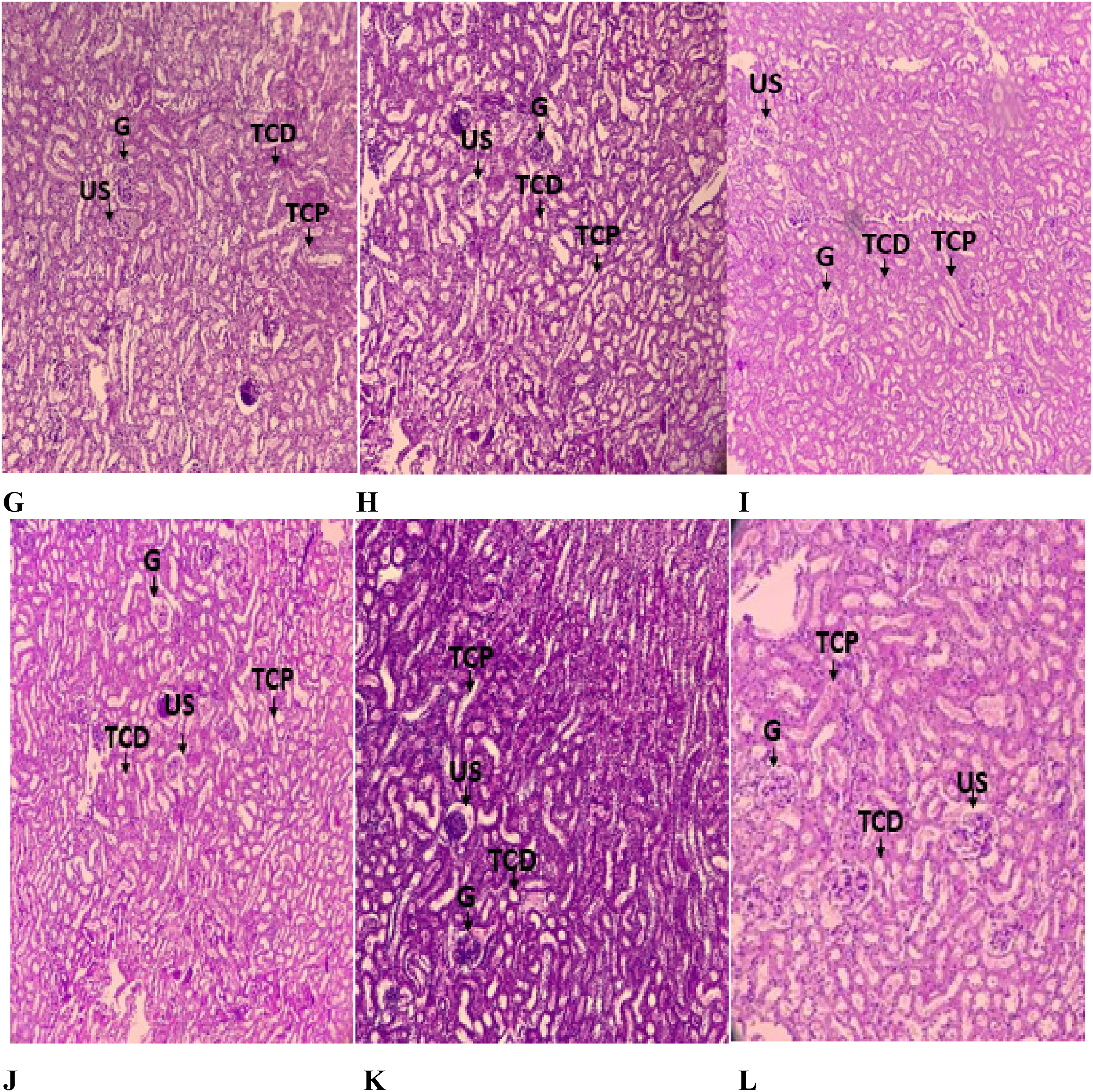
Kidney section of animal groups treated with NaCl (A), paracetamol (B), aqueous extract (100 mg/kg) + paracetamol (C), aqueous extract (300 mg/kg) +paracetamol (D), methanolic extract (100 mg/kg) + paracetamol (E), methanolic extract (300 mg/kg) + paracetamol (F). G: Glomerulus; TCP: Proximal Convoluted Tubule; TCD: Distal convoluted tubule; US: Urinary space.
Chemical Composition
The chemical composition of the methanolic and aqueous extracts showed the presence of various compounds known for their pharmacological activities. Tables 1 and 2 present the chemical composition of methanolic and aqueous extracts, respectively.
Chemical Composition of the Methanolic Extract of C. ladanifer.

Chemical Composition of the Aqueous Extract of C. ladanifer.

Docking Results in the Active Site.

The chemical composition of the studied extracts showed the presence of interesting compounds mainly Eicosanoic acid, Chrysin, 2-Methoxy-4-vinylphenol and 3-Methyl-2-(2-oxopropyl) furan. The compounds present in each extract act in synergy and are probably responsible of the obtained hepatoprotective activity
Molecular Docking
In our in silico study, Chrysin, 1H-Cycloprop[c]inden-7-ol, octahydro- Naphthalene, 1, 2, 3, 5, 6, 7, 8, 8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-,[1S-(1.alpha.,7.alpha.,8a.alpha.)]-, 1H-Cycloprop[c]inden-7-ol, octahydro-, and 3-Methyl-2-(2-oxopropyl)furan were the most active molecules in the active site of CYP2E1 with a glide score of −8.571, −7.005, −6.768, and −6.73 kcal/mol (Table 3). Figures 6 and 7 show the 2D and 3D viewers of different types of bonds established between the ligands and the active site.
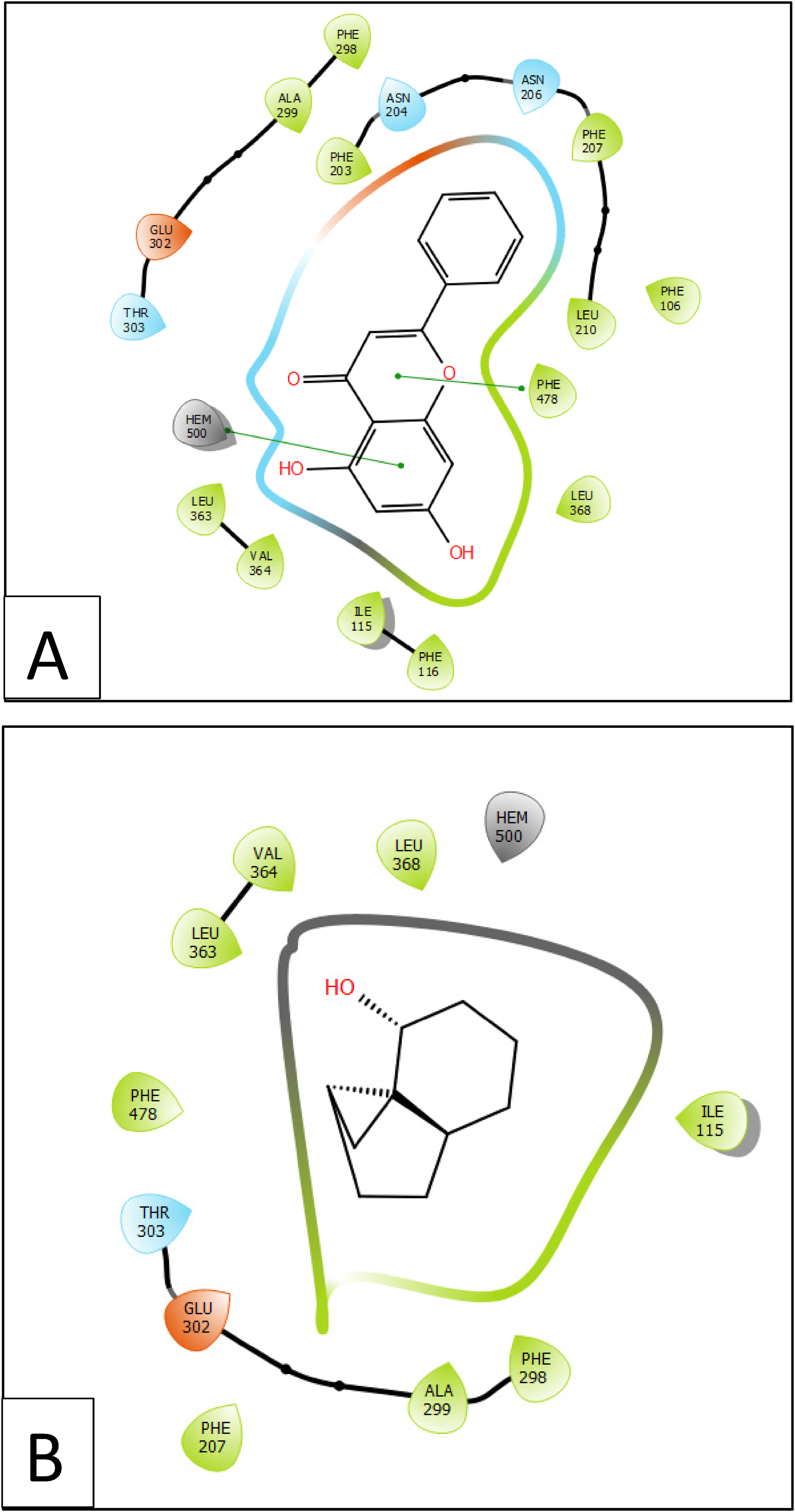
2D viewer interactivity of ligands with CYP2E1 (PDB :3LC4) active site; (A) chrysin interactivity between quercetin and vanillic acid and caspase-3 (PDB :3GJQ) active site; (B) 1H-cycloprop[c]inden-7-ol, octahydro-; (C) naphthalene, 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, [1S-(1.alpha.,7.alpha.,8a.alpha.)]-; (D) 3-Methyl-2-(2-oxopropyl)furan.

2D viewer interactivity of ligands with CYP2E1 (PDB :3LC4) active site; (A) chrysin interactivity between quercetin and vanillic acid and caspase-3 (PDB :3GJQ) active site; (B) 1H-cycloprop[c]inden-7-ol, octahydro-; (C) naphthalene, 1,2,3,5,6,7,8,8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-, [1S-(1.alpha.,7.alpha.,8a.alpha.)]-; (D) 3-Methyl-2-(2-oxopropyl)furan.
Chrysin docking established Pi-Pi stacking bonds with residues PHE 478 and HEM 500 inactive site of CYP2E1, when 3-Methyl-2-(2-oxopropyl) furan established one hydrogen bond with residue ASN 206 and one Pi-Pi stacking bond with residue PHE 298 in same active site (Figure 6A, 7A and Figure 6D, 7D).
Discussion
The normal function of the liver is evaluated by various serum biomarker enzymes. In our study, we evaluated the hepatoprotective activity of C. ladanifer aqueous and methanolic extracts at two doses of 150 and 300 mg/kg. According to EL Kabbaoui et al, these two doses don’t show any sign of acute toxicity. 6
In order to evaluate the activity of our extracts on liver damage, three groups of mice were prepared, group of negative control (Saline solution), group of positive control (paracetamol at 1000 mg/kg), and the third group (1000 mg/kg paracetamol additionally 150 and 300 mg/kg of both extracts), and different biochemical parameters were analyzed in the mice serum of different groups.
The transaminases levels of AST and ALT present a principal indication of liver dysfunction. In fact, increased levels of AST and ALT cause hypertrophy and injury in liver tissues.29,30 Our results showed that the level of AST and ALT in mice treated with the paracetamol and both C. ladanifer extracts decreased compared to positive control group; however, the methanolic extract showed at 300 mg/kg revealed some hepatic toxicity. The transaminases activities decrease and return to normal indicates the stabilization of plasma membrane, as well as repair of hepatic tissue damages caused by the paracetamol;1,31 this result suggests that C. ladanifer extracts protected the liver tissue from paracetamol-induced injury. Our findings are in agreement with El kabbaoui et al, 2017 that showed that aqueous extract of C. ladanifer may exhibit a potential hepatoprotective effect at 500 mg/kg, producing a decrease in the level of ALT. 6 The measurement of the total cholesterol and TG levels presented another biochemical parameter to investigate the hepatoprotective effect of C. ladanifer extracts. Low and decreased levels of TG and cholesterol usually indicate chronic liver diseases and a reduced liver biosynthesis capacity,. 32 Interestingly both aqueous and methanolic extracts at 150 and 300 mg/kg induced an elevation of total cholesterol and TG levels compared to the positive control; these two parameters were restored to normal after treatment with C. ladanifer extracts.
The kidneys are a vital organ, it filters large types of toxins due its high blood flows. 33 The examination urea and creatinine concentration are considered biomarkers of renal damage.6,34 In our case, urea and creatinine were recovered to normal levels in mice treated with C. ladanifer extracts. The different studied parameters were restored to normal levels in mice serum treated with both paracetamol and C. ladanifer extracts. All of our findings are in agreement with the study of El kabbaoui et al study
The histopathological analysis of liver and kidney recovered from the mice treated with paracetamol alone, and both extracts were identified. The histopathological analysis of liver sections excised from treated with paracetamol 1000 mg/kg alone and with paracetamol along with methanolic extract at 300 mg/kg showed the presence of various serious histopathological alterations including sinus dilation, infiltration of inflammatory cells as well as the necrosis. No significant histological modifications were noted for liver sections recovered from the mice treated with aqueous extract at both doses of 150 and 300 mg/kg and methanolic extract at 150 mg/kg. In the same study of Kabbaoui et al, aqueous extract of C. ladanifer at 500, 700 and 1000 mg/kg showed no remarkable pathological changes. Our findings revealed that aqueous extract at 150 and 300 mg/kg and methanolic extract at 150 mg/kg showed an interesting hepatoprotective activity. 6 However, the methanolic extract at 300 mg/kg showed a toxicity.
The GC-MS was used to identify the chemical composition of C. ladanifer both extracts, and the results showed the presence of various biomolecules. The compounds found in C. ladanifer extracts such as, chrysin, Vitamin E, Camphor, and borneol are well known for their therapeutic proprieties in alternative medicine and can probably be responsible of the hapatoprotective activity of the extract. It was demonstrated that chrysin exhibited hepatoprotective activity via various molecular and biochemical mechanisms, in liver-induced hepatotoxicity by chemical agents.35,36 Moreover, Vitamin E has shown in various studies its hepatoprotective effect, it decreased oxidative stress in the liver, tumor necrosis factor, liver fibrosis inflammation, and lipid peroxidation. 37 Camphor was also reported for its hepatoprotective effect. 38
Cytochrome P450 2E1 (CYP2E1) is an enzyme primarily found in the liver and plays a significant role in drug metabolism and the oxidation of various endogenous and exogenous compounds, including ethanol and certain drugs. Some studies have suggested that in certain circumstances, CYP2E1 might contribute to hepatoprotection. It has been proposed that CYP2E1 may play a role in the detoxification of certain chemicals and drugs that are harmful to the liver. The in silico study showed that some compounds present in C. ladanifer mainly Chrysin, 1H-Cycloprop[c]inden-7-ol, octahydro-, Naphthalene, 1, 2, 3, 5, 6, 7, 8, 8a-octahydro-1,8a-dimethyl-7-(1-methylethenyl)-,[1S-(1.alpha.,7.alpha.,8a.alpha.)]-,1H Cycloprop [c] inden-7-ol, octahydro-, and 3-Methyl-2-(2-oxopropyl)furan were the most active molecules in the active site of CYP2E1. CYP2E1 can be induced by various substances, including dietary compounds like polyphenols found in fruits and vegetables, which may have antioxidant properties and help counteract the harmful effects of CYP2E1-induced oxidative stress.24,25
The present study which aims to determine the activity of our extracts on hepatotoxicity induced by paracetamol in mice represents certain limitations. First, the duration of the test in vivo and the animal testing that raises ethical concerns. Second, the metabolism of paracetamol in animals differs from humans, which would affect the applicability of the results to humans. In other words, animal models may not accurately reflect the effect of these same extracts on humans.
Conclusion
C. ladanifer is a medicinal plant widely used in phytotherapy for its therapeutic and pharamcological activities. In our work, we evaluated the hepatoprotective activity of two C. ladanifer extract, the aqueous, and the methanolic. Our findings are encouraging, since this plant is little explored for its hepatoprotective activity. Both extracts showed an interesting hepatoprotective activity in vivo and in silico and in paracetamol liver toxicity-induced mice; however, the methanolic one showed a hepatic toxicity at higher dose. We reported that this plant is a source of various compounds that are probably responsible for the observed activity. Further investigations are needed to deepen the study.
Author's Contributions
Conceptualization, writing the original draft, reviewing and editing: Kaoutar Bouothmany, Fatima ez-zahra Amrati, Mohamed Chebaibi, Yousef A. Bin Jardan, and Hiba-Allah Nafidi. Formal analysis, investigations, funding acquisition, reviewing, and editing: Mohammed Bourhia, El mzibri Mohammed, and Fouad mellouki. Resources, data validation, data curation, and supervision: Dalila Bousta, Fakhreldeen Dabiellil, and Laila Benbacer.
Footnotes
Acknowledgement
The authors would like to extend their sincere appreciation to the Researchers Supporting Project, King Saud University, Riyadh, Saudi Arabia for funding this work through the project number (RSP2024R457).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Researchers Supporting Project, King Saud University, Riyadh, Saudi Arabia through the project number (RSP2024R457).
Ethical Approval
The Animal Ethics Review Committee at the faculty of Sciences, Fez, Morocco, approved this study under the ethical clearance number 02/2023/LBEAS.
Data Availability
Data will be available upon reasonable request from the authors.
