Abstract
Introduction
The most common causes of cirrhosis which contributes greatly to liver-related deaths accounting for 4% of all deaths worldwide are related to viral hepatitis, alcohol, and non-alcoholic fatty liver disease (NAFLD). NAFLD is characterized by pathological intrahepatic triglycerides (TGs) excess represents a burgeoning epidemic around the world. 1 The mechanism underlying the pathogenesis is supposed to be related to the imbalance between lipid uptake/anabolism and output/catabolism in chronic states of over-nutrition. Due to its complex etiology reflecting multiple interactions between environmental and genetic factors, there is no official approved pharmacological therapy available for NAFLD currently except for weight reduction to meet the great demand. 2 Traditional Chinese medicine (TCM) has undergone thousands of years of development and has also formed a unique theoretical system that acknowledged NALFD is related to deficiency and depression of Qi. The practice of TCM has been guided by the principle of “observing its pulse patterns, knowing where to go wrong, and treating according to the symptoms”. TCM and Western medicine have different perspectives on observing and solving problems, innovative concepts, and unique strengths. 3 In addition, many dietary strategies include the medicinal and edible homologous herb medicines such as some green tea have shown its preventive effects on the development of liver steatosis or its progression. 4 Therefore, exploring and developing the therapeutic candidates from these plant medicines including TCM is a big concern in the phytomedicine community.
In recent years, with functional ingredients as the core to seek, construction and application of a technology system for discovery, enrichment, and quality control of active substances in TCM based on integration strategies is a hot topic.
18
Among these state-of-the-art technologies, quadrupole time-of-flight mass spectrometry technology (Q-TOF-MS) generating accurate measurements of mass, elemental composition and isotopic abundance to identify the possible molecular formula of each analyte with the mosaic library database, equipped with ultra-high-performance liquid chromatography (UPLC) systems separating solitary constituents rapidly with a high resolution, has become a powerful method for the rapid separation and identification of active ingredients in the complex traditional herb medicine as shown in our previous work.
19
In addition,
Materials and Methods
Materials Preparation
The root of KC was purchased from Guanyang County, Guilin City, Guangxi province and identified
Reagents
The reagents for cell culture and treatments are as follows: Triglycerides (TGs) kits (Biosino Biotechnology & Science Inc., Beijing, China), Oil red O (Kanglang Biological Technology Co. Ltd, Shanghai, China), Dulbecco's Modified Eagle Medium (Invitrogen Co. Ltd, New York, USA), Fetal bovine serum (Hyclone Co. Ltd, Utah, USA), Trypsin (Amresco Co. Ltd, Ohio, USA), Sodium oleate and Penicillin-Streptomycin(Tokyo chemical industry Co. Ltd, Tokyo, Japan), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (Thiazolyl blue tetrazolium bromide, MTT)(Sigma Co. Ltd, San Francisco, USA). In addition, acetonitrile (UPLC grade, Merck), Formic acid (UPLC grade), Ammonium formate (Merck) were purchased, phosphate-buffered saline was bought from Guangzhou Chemical Agent Factory (Guangzhou, China); Lockmass enkephalin (Sigma Company, USA, batch number: L9133-50MG) was obtained; standards of Gomisin J (Lot No. zzs17022206), Gomisin D (Lot No. zzs17022204), Gomisin E (Lot No. zzs17022205), Kadsulignan L (Lot No. zzs17022207) were purchased from Shanghai ZZBIO Co, LTD,. Other reagents were obtained from Guangzhou Chemical Agent Factory (Guangzhou, China).
Cell Culture
HepG2 was provided by the Institute of Chinese Medical Science of Guangdong Pharmaceutical University, which is bought from the American Type Culture Collection (Mannassas, VA, USA). Cell culture is similar to the previous report of our work. 21 In short, after preparation and thawing frozen HepG2cells, the operator carefully removed the supernatant and resuspend the cell pellet in fresh, pre-warmed DMEM with FBS, count the cells using a hemocytometer and adjust the cell density to the desired concentration 2 × 105 cells/mL. Then cells were cultured in a 96-well plate with 2 × 105 cells/mL (100 µl each well) in culturing medium including 13.5 g/L DMEM containing 10% FBS, double-antibiotic solution and high glucose. Place the culture plate in a cell culture incubator set to 37 °C with 5% CO2 and check the cells daily and replace the culture medium every 2-3 days depending on the cell growth rate. Use the cultured HepG2 cells for the following viability assay and oil-red staining experiments.
Cell Viability Assay
The effect of KCE with different concentration on the viability of HepG2cells was evaluated using the MTT Detection Assay System. The MTT assay is a commonly used colorimetric assay to assess cell viability. It measures the metabolic activity of cells, which is an indirect indicator of cell viability. It involves the conversion of the yellow MTT dye into a purple formazan product by mitochondrial enzymes in metabolically active cells. The intensity of the purple color is proportional to the number of viable cells. For performing the MTT assay, HepG2 cells were inoculated with 1.0
Oil Red O Staining and Determination of the Intracellular TG
Oil red O staining is a commonly used method to visualize and quantify intracellular TGs or lipid droplets in cultured cells or tissues. The procedure we adopted in this study is similar to the previous report of our colleagues.
22
Briefly, HepG2 cells were plated in 6-well plates (2.5
In addition, measuring TGs content in HepG2 cell lines is commonly done by commercial TG determination kits (Biosino Biotechnology & Science Inc., Beijing, China). The kits are typically based on enzymatic methods, where specific enzymes are used to catalyze reactions with TGs, producing a measurable signal that is directly proportional to the TGs concentration. The enzymatic method derived kit for TGs determination involves hydrolysis of TGs into glycerol, phosphorylation by glycerol kinase, oxidation induced production of hydrogen peroxide which reacts with a chromogenic substrate to produce a colored product. The color intensity is directly proportional to the concentration of TGs in the Cells. Similar to the reported procedures,
23
the total protein was quantified by the BCA protein concentration determination kit method, and the intracellular concentration of TGs was measured by the TGs determination kits strictly. Then, the TG
UPLC-Q-TOF-MS Analysis for the Chemical Composition in KCE
Similar to our previous work,19,21 UPLC-Q-TOF-MS was used to separate and analyze KCE constituents. Briefly, a 50 mm × 2.1 mm (Acquity 1.7 µm) UPLC BEH C-18 column (Waters, Milford, MA) was adopted for ingredients separation. The flow rate of the mobile phase was 0.3 mL/min at temperature 40°C with a gradient of MeCN-H2O mixtures ranging from 2/98 to 55/45(v/v) containing 0.2% formic acid in a 12.5 min run. The injection volume was 10 μL each time. The full mass scanning range from m/z 100 to 1000 Da with TOFMS was operated in positive mode and negative mode with electrospray ionization. High purity nitrogen was used as the nebulizer and auxiliary gas; argon was used as the collision gas. The structure of each chemical was elucidated by tandem mass spectrometry fragmentation with collision energy ramp ranging from 10 to 30 eV. The other parameters were set as follows: capillary voltage: 3.2 kV, cone gas flow: 50 L/h, desolvation gas flow:500L/h, The source temperature and desolvation temperature were at 120 °C and 350 °C, respectively. Mass accuracy was maintained by using a lock spray with leucine enkephalin 500 pg/μL ([M + H]+m/z 556.2771, [M-H]−m/z 554.2615)) as reference. All the solvents for liquid chromatography and mass spectrometry in this study were of the highest grade commercially available.
1 mL of KCE was centrifuged for 5 min (12,000 r/min), the supernatant was taken for analysis. The repeatability was assessed by analyzing six independently prepared samples. The method precision was evaluated by analyzing the same sample for six consecutive times. The sample stability test was determined with one sample within one day. In this period, the solution was stored at room temperature and analyzed at 0, 1,2,4,12, 24 h, respectively.
Data Analysis
All experimental values are expressed in the figures as mean ± SD, and data were analyzed by two-tailed Student's t test.
Results
Toxicological Evaluation of KCE to HepG2 Cells
We preliminarily tested the effects of KCE in different concentrations (1-1000 μg/mL) on HepG2 cell survival with the classic MTT method. Among the tested concentrations of KCE (1-1000 μg/mL), KCE exhibits some cytotoxicity with less than 95% viability only at high concentrations more than 80 μg/mL. To seek the non-toxic KCE concentration for the lipid droplets imaging and quantification, less than 50 μg/mL as the criteria was determined. Among the chosen concentrations of KCE (5, 10, 20 μg/mL) for lipid staining and quantification of the intracellular TGs, no significant cytotoxicity was noted.
Effect of KCE Treatment on in Vitro Oleic Acid Induced Model TGs
Oil red O is highly soluble in fats and can specifically stain TGs in tissues. As shown in Figure 1B model group, the cell lipid aggregation model to mimic the NAFLD manifestation induced by oleic acid has been successfully constructed. Compared to oleic acid induced model group, the 10 μmol/L Berberine treatment (Figure 1C) can slightly reduce lipid content as previous researcher reported. Interestingly, 5-20 μg/mL KCE can reduce the number and volume of lipid droplets in the oleic acid induced HepG2cells in a dose-dependent manner. The effective potency triggered by the positive control berberine is between low and medium doses of KCE, but it is significantly lower than that of the high-dose 20 μg/mL KCE group. In another parallel experiment, the TG content of each group after treatment was determined by the quantitative reagent kit of TGs. As demonstrated in Figure 2, the results of TGs quantification were almost consistent with the results of the above oil red O staining experiment. In addition, the decrease of TGs content in the HepG2 cells is also a dose-dependent manner in the low, middle and high dose KCE groups.

Oil red O staining maps of HepG2 cells (10

Effect of KCE on the content of TG in oleic acid-induced HepG2 cells. Note: KCE groups with low (5 μg/mL), middle (10 μg/mL) and high (20 μg/mL) concentrations are indicated as KCE-L,KCE-M, KCE-H for short respectively. ###
Constituents Identification of KCE by UPLC-Q-TOF-MS
UPLC-Q-TOF-MS was used to analyze the chemical constituents of KCE. The method validation demonstrated that the relative standard deviation (R.SD) of the main peaks retention time in the sample was no more than 0.3%.The total ion current chromatograms in both positive and negative ion modes were shown in Figure 3. By UPLC-Q-TOF-MS, the mass spectrum data of each compound in KCE were obtained. The chemical constituents were analyzed and identified by searching in database and matching, and comparing the references and the standards.

The profile for base peak ion (BPI)chromatogramof KCE in positive(A) andnegative(B) ion mode with electrospray ionizationby UPLC/Q-TOF-MS.
The identification results of the compounds are detailed in Table 1 with the corresponding references.24–40 Among them, there were 16 lignans (peaks 3, 4, 6, 7, 10, 11, 14, 15, 19, 20-26), 2 triterpenoids, 1 glycosides, and 7 others compounds. In this study, most of the identified lignans in KCE are dibenzocyclooctadiene lignans except for peak 30.The structural skeleton of these lignans was shown in Figure 4. Some special lignans of biphenyl cyclooctadiene, such as 6, 9-oxo-bridged biphenyl cyclooctadiene (peak 25) were found.

The structural skeleton of dibenzocyclooctadiene lignans.
Constituents Identification of KCE by UPLC/Q-TOF-MS.

Note:“Det.” means detected ion mass; “Cal.” means calculated ion mass; “RE” means relative error between detected ion mass and calculated ion mass. RE = [Mass Det.-Mass Cal.]/Mass Cal.×106; “–” means not identified yet.
In this study, it was found that the fragmentation ion produced by loss of C7-C8 and its substituent in the eight-membered ring of dibenzocyclooctadiene lignans (Figure 4) was characteristic ion in ESI source mass spectrometry. In addition, the ring opening and ring shrinking cracking of the eight-membered ring in the parent structure of dibenzocyclooctadiene lignans took place.
Take peak 23 as an example to describe the process of identification and analysis. Peak 23(tR = 10.90 min) with a molecular ion of [M + H]+at
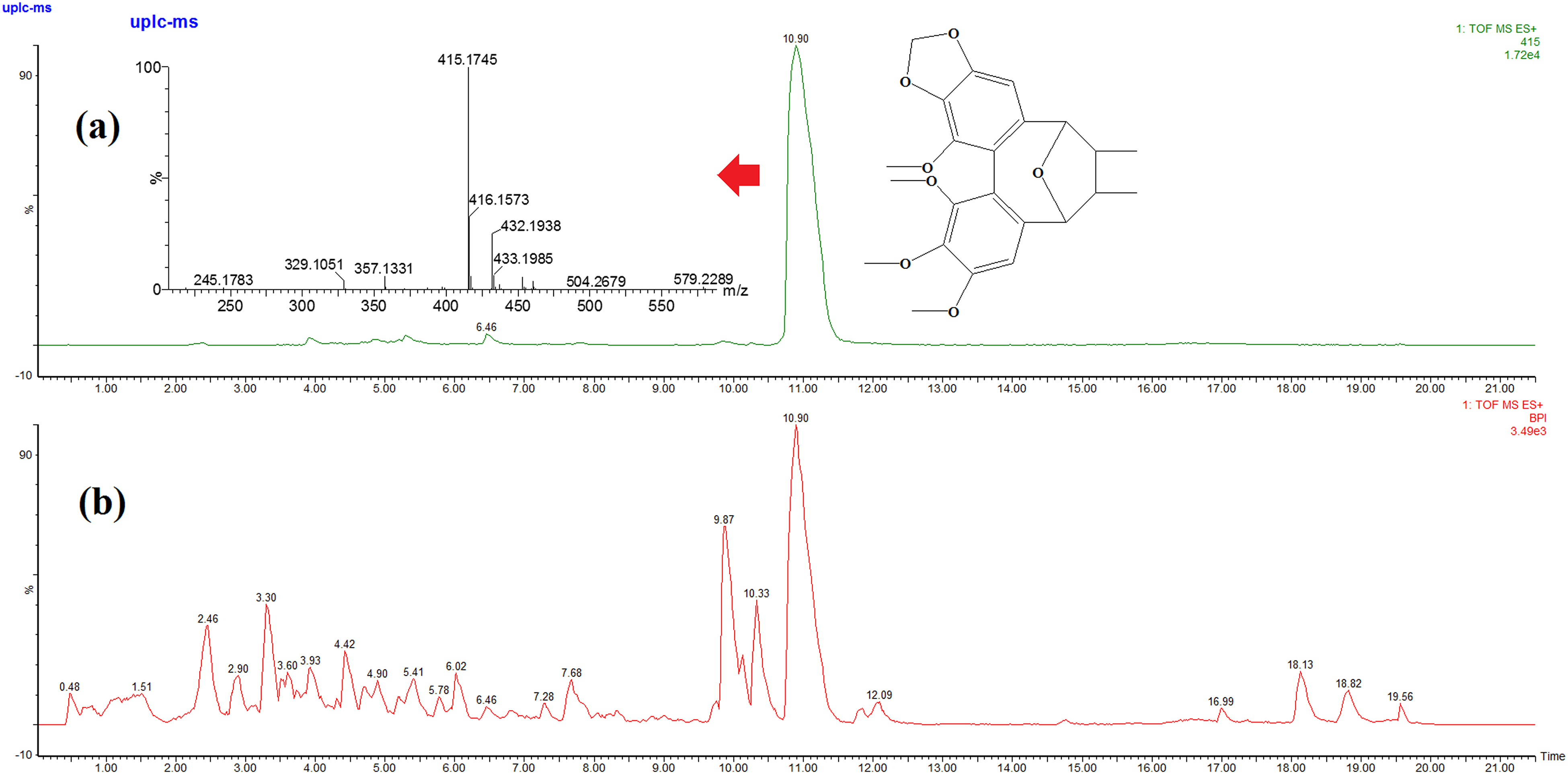
The base peak ion (BPI) chromatogram of standard Kadsulignan L (a) and KCE(b), MS spectrum of Kadsulignan L in positive ion mode by UPLC/Q-TOF-MS.

The MS/MS spectrum and possible Mass fragmentation pathway of Peak 23 represented as Kadsulignan L.
As the result, peak 23 was identified and confirmed as Kadsulignan L39,40 based on comparison of the retention time, and mass spectral data with those of the reference standard. Its possible fragmentation pathway was shown in Figure 6. Other lignans were identified by the similar way.
The standards of Gomisin J, Gosimin D, Gomisin E as samples were used for verifying the results of the putative identification. Take the Gomisin J for an example, specifically, according to MS spectra information, it was obviously observed that ions of [M + H]+ and [M-H]− at m/z 389.1975 and 387.1815 of peak 21 in KCE sample (Figure 7), and corresponding to the ions in standard were 389.1958 and 387.1819 under positive and negative respectively; as the results, the ions had the same chemical formulas of C22H29O6 (
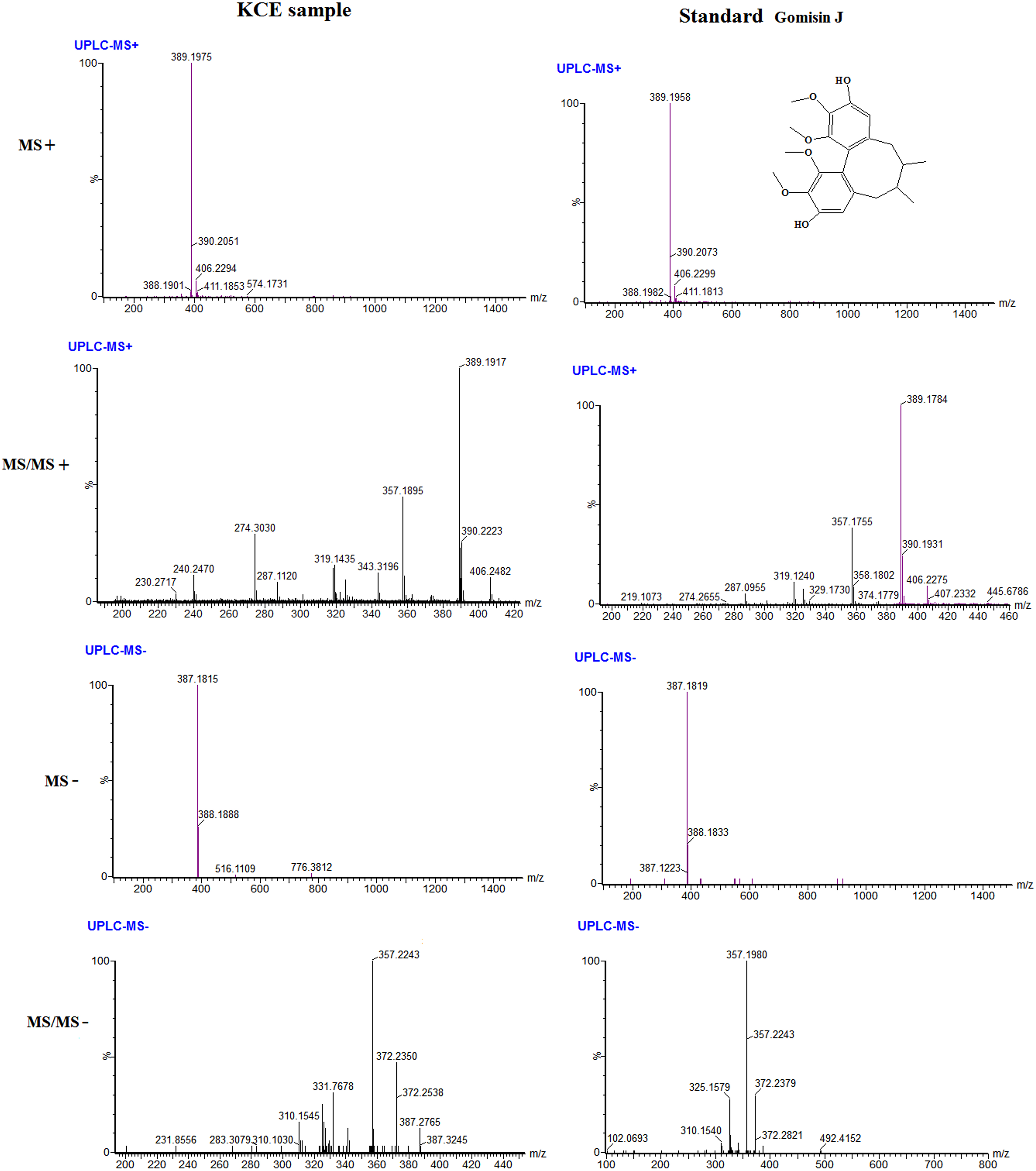
The comparison of MS, MS/MS spectra of Gomisin J identified in KCE to the standards both in positive and negative ion modes.
Discussions
Some researchers have evaluated the cytotoxicity of lignans Heilaohulignan C, kadsuralignan I, longipedunin B, kadusurain A from KC ethyl acetate fraction on HepG2 Cells,5,13 all the IC50 values of pure compound are more than 10 μg/mL. In our EtOH extract, HepG2 cells showed less than 95% viability at concentrations more than 80 μg/mL. Therefore, we choose the concentrations of KCE 5, 10, 20 μg/mL for the later oil red O staining and determination of the intracellular TG. The accurate lignan content calculation in the KC roots and its extract depends on the standard reference available needed to be developed.
Oleic-acid is frequently used to induce the accumulation of lipid droplets in HepG2. 22 In the current study, oil red O imaging demonstrated that KCE in gradient concentrations (5, 10, 20 μg/mL) processing significantly mitigated lipid agglomeration and fusion in HepG2cells induced by oleic acid. These data are consistent with the intracellular TG content determination, which also showed a dose-dependent manner. These results suggest that KCE could improve abnormal lipid metabolism in the liver characterized in NAFLD initiation and progression. However, the detailed mechanism involved in the improvement needs to be investigated in further.
The putative active components of KCE related to this effect were investigated by UPLC-Q-TOF-MS analysis. As the results, 26 compounds from KCE were identified, dibenzocyclooctadiene lignans were the main supposed components contained in KCE according to the literature.13–16,40,42,43 The follow-up separation and purification of these supposed compounds are warranted to confirm its structure by other technologies such as nuclear magnetic resonance.
It is noteworthy that lignans were frequently reported that have the potentiality to treat NAFLD. Schisandrin B, are presentative DCL isolated from the fruit of
Conclusions
In this study, the effects of KCE on lipid accumulation were assessed, and chemical profiles of KCE were analyzed by UPLC/Q-TOF-MS. The KCE has ameliorative effect on lipid accumulation in the model
Footnotes
Acknowledgements
This work was financially funded from the Natural Science Foundation of China (82274120 and 81673689) and Guangdong Natural Science Foundation Project (No.2023A1515010405).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 81673689 and 82274120).
Ethical Approval
This study including the animal experiments was approved by the Administration Committee of Experimental Animals, Guangdong Pharmaceutical University.
Statement of Human and Animal Rights
All of the previous and subsequent experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Guangdong Pharmaceutical University, China and approved by the Administration Committee of Experimental Animals, Guangdong Pharmaceutical University.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
