Abstract
Objectives
Diabetes involves abnormalities in glucolipid metabolism, with oxidative stress serving as a major contributing factor. The Hengtang capsule is a dietary supplement that helps maintain healthy blood glucose (BG) and lipid levels as well as healthy body weight. Given the antioxidative stress effects of various components in the Hengtang capsule, the Hengtang capsule may offer a novel therapeutic approach for treating type 2 diabetes mellitus (T2DM). In this study, we investigated the ability of the Hengtang capsule to treat T2DM through a combination of network pharmacology and animal experiments.
Methods
The underlying mechanisms of the Hengtang capsule in treating T2DM were predicted via network pharmacology. After conducting an experiment for 4 weeks, the oral glucose tolerance test (OGTT), fasting blood glucose (FBG), liver function, lipid profile, and oxidative stress markers of the db/db mice were measured. Additionally, the effectiveness of the Hengtang capsule as an antidiabetic agent was assessed via hematoxylin and eosin (HE) staining and immunofluorescence assays.
Results
Five core targets, including TP53, AKT1, CTNNB1, STAT3, and EGFR, were identified. The targets were associated mainly with the PI3K/Akt, lipid and atherosclerosis, RAGE/AGE, and FoxO signaling pathways. Additionally, treatment with the Hengtang capsule resulted in significant reductions in the area under the curve (AUC), FBG, total cholesterol (TC), triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), aspartate transaminase (AST), alanine transaminase (ALT), and malondialdehyde (MDA) compared to those of the negative control group. In contrast, the concentrations of high-density lipoprotein cholesterol (HDL-C), superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH), and the area occupied by insulin-positive cells increased significantly. Moreover, improvements in liver histopathology were observed.
Conclusion
The results showed that the Hengtang capsule may exert therapeutic effects on diabetes by modulating glycolipid metabolism and oxidative stress.
Introduction
The increase in the incidence and mortality of diabetes has made it a significant global health burden. According to the latest study published in The Lancet (2024), around 828 million adults (aged 18 years and older) worldwide are diagnosed with diabetes, representing an overall prevalence of 14%, an increase of 630 million compared to 1990. 1 Diabetes mellitus is a chronic metabolic disorder characterized by impaired glucose and lipid metabolism due to insufficient or dysfunctional secretion of insulin. 2 It has long been a central focus of contemporary medical research. Glycolipid metabolism has been strongly implicated in the development of T2DM.3,4 The dysregulation of glucose and lipid metabolism is associated with the vascular complications associated with T2DM. 5 Oxidative stress plays a key role in the onset and progression of diabetes and significantly influences its pathophysiology. 6 It facilitates the initiation of diabetes and further aggravates disease progression and the development of related complications by disrupting the secretion and function of insulin. 7 An increase in the levels of reactive oxygen species (ROS) during oxidative stress leads to an increase in MDA concentrations. Consequently, MDA serves as a widely used biomarker to assess lipid peroxidation along with small-molecule antioxidants, such as reduced GSH, which also serves as a key indicator of oxidative damage. 8 SOD and CAT also serve as key indicators of oxidative stress in the liver. 9
Although some drugs can control blood sugar levels, they are single-target and have some adverse effects, such as weight gain, hypoglycemia, gastrointestinal side effects, etc 10 Therefore, other effective drugs need to be identified to treat T2DM. Among natural products, many plant extracts have been identified as potential drugs for improving diabetes and its complications. These extracts play important roles in delaying or preventing diabetes and its complications by regulating glucose-lipid metabolism and improving oxidative stress.11,12
The Hengtang capsule is composed of berberine hydrochloride, gamma-oryzanol, and a specialized gastrointestinal support cream. The gastrointestinal support cream consists of dihydromyricetin, sesame oil, and beeswax. Berberine hydrochloride is extracted from the rhizome of Phellodendron amurense. Gamma-oryzanol is extracted from the endosperm of Oryza sativa. Dihydromyricetin is extracted from the petiole of Ampelopsis grossedentata. Sesame oil is extracted from the seeds of Sesamum indicum. Beeswax is extracted from Apis cerana or Apis mellifera. Sesame oil and beeswax were used as excipients of the Hengtang capsule to protect the gastrointestinal mucosa, reduce gastrointestinal adverse reactions, and increase the solubility of the active ingredients. 13 Berberine hydrochloride, an alkaloid derived from P. amurense serves as the principal active ingredient in the Hengtang capsule. It has various biological activities, including therapeutic effects on gastrointestinal disorders, hypertension, diabetes, and other metabolic conditions. 14 Berberine hydrochloride modulates the GnRH-GLP-1 and MAPK signaling pathways in the intestine, thereby influencing glucose metabolic. 15 It also upregulates the insulin receptor and insulin receptor substrate, facilitating the uptake of glucose in adipose tissue and skeletal muscle and ultimately lowering FBG levels and improving insulin sensitivity. 16 Additionally, the activation of AMPK in adipose tissue and skeletal muscle regulates the expression of metabolic genes, suppresses adipogenesis, and directly activates the ERK signaling pathway to stabilize low-density lipoprotein receptor mRNA. This process decreases serum cholesterol, TG, and low-density lipoprotein cholesterol (LDL-C) concentrations without affecting high-density lipoprotein cholesterol (HDL-C) levels, thereby increasing lipid metabolism. 17 Berberine hydrochloride alleviates metabolic disorders by exerting anti-inflammatory and antioxidative stress effects. 18 Gamma-oryzanol, a bioactive compound derived from O. sativa, acts primarily functions as an antioxidant, hypoglycemic agent, and cholesterol-lowering substance. 19 It decreases insulin resistance, which is elevated in the skeletal muscle of obese animals, by increasing the expression of GLUT-4 and improving metabolic dysfunction.20,21
Dihydromyricetin is the major bioactive component isolated from the tender stems and leaves of the traditional Chinese medicinal plant A. grossedentata. It has various biological effects, including antialcohol toxicity, anti-inflammatory, antibacterial, antioxidant, anti-tumor properties, and lipid and BG regulatory effects.22,23 Clinical and experimental studies have demonstrated its preventive and therapeutic potential in treating T2DM and its complications.24,25 Dihydromyricetin can improve glucose and lipid metabolism, 26 alleviate oxidative stress, 27 and reduce inflammatory reactions. 28 Dihydromyricetin exerts antidiabetic effects by activating the AMPK signaling pathway. Its mechanism of action is associated with the AMPK/mTOR/PGC-1α and MEK/ERK signaling pathways, highlighting that it may serve as a therapeutic agent for metabolic disorders. 29
Network pharmacology integrates systems biology and computational biology, it is a powerful method for elucidating the intricate interactions among drugs, targets, and diseases. 30 In this study, through network pharmacology approaches and empirical validation, we achieved a deeper understanding of the therapeutic effects of the Hengtang capsule in mitigating T2DM and the underlying molecular mechanisms of action. Given the pharmacological properties of its components, the ability of the Hengtang capsule to alleviate diabetes by reducing oxidative stress and regulating lipid metabolism needs further investigation. We also investigated the ameliorative effects of the Hengtang capsule on hyperglycemia, hyperlipidemia, and oxidative stress indices in db/db mice and the effects of the Hengtang capsule on the function and pathological changes in the liver and pancreas to determine whether the Hengtang capsule can improve BG, hyperlipidemia, and oxidative stress in db/db mice.
Materials and Methods
Main Reagents and Instruments
The Hengtang capsule containing 2000 mg contents was manufactured and provided by Beijing Meibo High-tech Co., Ltd. The composition of each the Hengtang capsule is comprised of berberine hydrochloride (500 mg), gamma-oryzanol (50 mg), gastrointestinal support cream (1450 mg). The gastrointestinal support cream consists of dihydromyricetin, sesame oil, and beeswax. Metformin was purchased from Dalian Meilun Biotechnology Corporation. Kits for TC, TG, LDL-C, HDL-C, AST, and ALT were purchased from Shanghai Enzyme-linked Biotechnology Corporation, whereas kits for MDA, SOD, CAT, and GSH, were purchased from Shanghai Biyuntian Biotechnology Corporation.
Network Pharmacology Analysis
A network-based pharmacological strategy was used to investigate the molecular pathways through which the Hengtang capsule influences the treatment and regulation of T2DM. First, the potential targets of berberine hydrochloride, gamma-oryzanol, and dihydromyricetin were identified through the Traditional Chinese Medicine Systems Pharmacology (TCMSP), PubChem, Pharm Mapper, Comparative Toxicogenomics Database (CTD), and Swiss Target Prediction platforms. To determine T2DM-related targets, the Therapeutic Target Database (TTD), GeneCards, and Online Mendelian Inheritance in Man (OMIM) databases were queried using the keyword “type 2 diabetes mellitus”. All databases were restricted to the species “Homo sapiens”.
The common targets shared between the Hengtang capsule and T2DM patients were subsequently identified by constructing a Venn diagram. A protein-protein interaction (PPI) network for common targets was developed using the Search Tool for the Retrieval of Interacting Genes/Proteins (STRING) database. The results and network data were imported into Cytoscape (Version 3.9.1) for graphical representation and analysis. Finally, the Database for Annotation, Visualization, and Integrated Discovery (DAVID) was used to perform enrichment analyses for Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG). The GO biological function analysis results were categorized into molecular function (MF), cellular component (CC), and biological process (BP) categories. The top 10 GO terms and the top 20 KEGG pathways significantly enriched at a false discovery rate threshold of < 0.05 were selected for further investigation.
Animals and Models
In total, 72 male db/db mice (40-42 g, 8 weeks old, specific pathogen-free grade) were obtained from Beijing Weitonglihua, permit No. SCXK (Beijing) 2022–0016. The animals were kept in a controlled environment at 22 ± 2 °C and a relative humidity of 40%–60%. The animals were kept in a 12-h/12-h light/dark cycle and provided free access to food and water. During the feeding period, the animals were provided a standard diet consisting of 12.4% kcal from fats, 24.2% kcal from proteins, and 63.4% kcal from carbohydrates (Synergetic Pharmaceutical Bioengineering Co., Ltd, Jiangsu, China; Lot No. XTI01FZ-013). The animal study protocol was approved by the Ethics Review Board of Binzhou Medical University. The animal research was conducted strictly following the ARRIVE 2.0 guidelines. 31
Before the experiment, 72 db/db mice were adaptively fed with a free diet and water intake for 7 days. The mice were then randomly divided into 6 groups (n = 12 per group): normal control (Nor-Con group), negative control (Neg-Con group), low-dose Hengtang capsule (Low-HTC group), medium-dose Hengtang capsule (Med-HTC group), high-dose Hengtang capsule (High-HTC group) and positive control groups (Pos-Con group). The recommended human dosage of the Hengtang capsule is 6 g/person/day, which contains 1500 mg of berberine hydrochloride and 150 mg of gamma-oryzanol. The corresponding mouse doses were determined using the body surface area-based dose conversion method between humans and standard laboratory animals. 32 Thus, the Hengtang capsule treatment group was divided into Low-HTC, Med-HTC, and High-HTC groups according to 5, 10, and 20 times the recommended human dose. Specifically, the Low-HTC group received 3.9 g/kg Hengtang capsule by gavage, the Med-HTC group received 7.8 g/kg Hengtang capsule by gavage, the High-HTC group received 15.6 g/kg Hengtang capsule by gavage, and the Pos-Con group received 300 mg/kg metformin by gavage. 33 For the Neg-Con group, the gavage volume of sesame oil beeswax was calculated based on the amount of sesame oil beeswax (excluding the weight of berberine hydrochloride and gamma-oryzanol) corresponding to the gavage dose in the High-HTC group. The mass fractions of sesame oil and beeswax in the Hengtang capsule ranged from 97% and 3%, respectively. This amount constituted 72.5% of the total mass fraction, resulting in a dosage of 11.31 g/kg.
Determination of FBG in Experimental Animals
The FBG levels in the mice were determined after 4 weeks of intragastric administration. The mice were subjected to 12 h of fasting before FBG levels were determined, and hematological samples were collected from the distal region of the tail. The first drop of blood was wiped using a cotton ball, and the second drop was applied to the test strip's suction area. Then, the glucose meter was used to analyze the sample and display the FBG values, which were recorded for further analysis. 34
Assessment of the OGTT in Animal Models
Before the OGTT experiment, the experimental animals were starved for 12 h. Baseline BG levels were measured using a glucose meter and then recorded. The test mice were then administered 40% glucose solution by gavage (2 g of glucose/kg body weight). BG levels were subsequently measured at the 30th, 60th, 90th, and 120th min and recorded as BG0, BG30, BG60, BG90, and BG120, respectively. Then, the AUC was then calculated using the following formula: AUC (mg/dL/h) = (BG0 + BG30) × 15/60 + (BG30 + BG60) × 15/60 + (BG60 + BG90) × 15/60 + (BG90 + BG120) × 15/60. 35
Sample Collection
After the experiment ended, the mice were fasted overnight for 12 h, and anesthetized by inhaling isoflurane the following day. Blood samples were collected via retro-orbital bleeding, transferred to centrifuge tubes, and processed immediately to minimize coagulation. The collected blood was allowed to clot at room temperature for 30 min before it was centrifuged at 1200 rpm for 15 min at 4 °C. The serum samples were aliquoted to about 20 μL per tube and stored at −80 °C for long-term preservation. Additionally, liver and pancreatic tissues were fixed in 4% paraformaldehyde and preserved for further analysis. 36
Effects of the Hengtang Capsule on Serum Biochemical Indexes of Experimental Mice
Frozen serum samples were retrieved from storage at −80 °C, thawed on ice, and subsequently centrifuged at 2000 rpm for 30 min at 4 °C. The contents of TC, TG, LDL-C, HDL-C, AST, and ALT in the serum were measured following the manufacturer's instructions.
Effect of the Hengtang Capsule on the Oxidative Stress Levels of Experimental Mice
Liver tissue homogenates were prepared, and the levels of MDA, SOD, CAT, and GSH were detected following the manufacturer's instructions.
Histology and Immunofluorescence Staining
The mice were euthanized via cervical dislocation, following which the pancreas and liver tissues were excised, rinsed with isotonic saline, blotted dry, weighed, and subsequently fixed in 4% paraformaldehyde. The tissue samples were subjected to paraffin embedding and sectioning following standard protocols. Liver tissue sections were subjected to HE staining, 37 whereas pancreatic sections were subjected to immunofluorescence staining. 38
Statistical Analysis
The GraphPad Prism software (Version 8.0.1, GraphPad Software, USA) was used for comprehensive data analysis. The data were presented as the mean ± standard error of the mean. The differences among multiple groups were determined by conducting the one-way analysis of variance, followed by the least statistically significant post hoc test. All differences among and between groups were considered to be statistically significant at p < 0.05 (p < 0.01 indicated high statistical significance).
Results
Identification and Investigation of Shared Therapeutic Targets of the Hengtang Capsule in Improving T2DM Management
We identified 745 and 5701 potential targets for the Hengtang capsule and T2DM, respectively. Among them, 529 common targets were identified as potential therapeutic targets of the Hengtang capsule for treating T2DM (Figure 1A). The PPI network for these common targets were generated using the STRING database platform, which consists of 525 nodes and 5446 edges. The interaction information of the PPI network was imported into Cytoscape for visualization (Figure 1B). In terms of degree, the top 10 genes were identified as hub genes for the use of the Hengtang capsule in treating T2DM (Figure 1C). The results of the GO enrichment analysis revealed 2112 enriched GO terms, including 1575 BP, 188 CC, and 349 MF terms. The enrichment of the top 10 GO terms suggested that multiple cellular functions may be involved in the broad effects of the Hengtang capsule in managing T2DM. In BP, the key terms were predominantly associated with positive regulation of gene expression and negative regulation of apoptosis (Figure 1D). The potential targets were strongly associated with different CC, including the cytoplasm and the nucleus, etc (Figure 1E). The MF were associated primarily with protein binding, identical protein binding, ATP binding, etc (Figure 1F). The results of the KEGG pathway enrichment analysis revealed 189 significant entries, suggesting that the Hengtang capsule might be involved in the PI3K-Akt signaling pathway, lipid and atherosclerosis, the TNF signaling pathway, insulin resistance, the FoxO signaling pathway, etc (Figure 1G).

Results of the Component and Disease Target Analysis, Along with the GO and KEGG Enrichment Results are Presented. (A) the Venn Diagram Illustrates the Commonalities Between Active Ingredients and Disease Targets. (B) the PPI Network Links the Hengtang Capsule to Common Targets Associated with T2DM. (C) the 10 Most Prominent Targets of the Hengtang Capsule in the Management of T2DM. (D) the top 10 Enriched Terms Identified in the BP Category. (E) the 10 Most Significantly Enriched Terms Associated with CC. (F) the 10 Most Notably Enriched Terms Related to MF. (G) the top 20 Signaling Pathways Were Identified Through the KEGG Enrichment Analysis.
Effects of the Hengtang Capsule on AUC and FBG Levels in Db/db Mice
The AUCs of the Neg-Con, Low-HTC, Med-HTC, and High-HTC groups were significantly greater than those of the Nor-Con group (p < 0.001). The Pos-Con group also exhibited a significant increase in the AUC compared to the Nor-Con group (p < 0.01). However, the AUCs of the Med-HTC, High-HTC, and Pos-Con groups were significantly lower than those of the Neg-Con group (p < 0.001) (Figure 2A). Similarly, significant differences in FBG levels were found across the experimental groups. The FBG levels were significantly greater in the Neg-Con, Low-HTC, and High-HTC groups than in the Nor-Con group (p < 0.01). Additionally, the FBG levels were significantly higher in the Med-HTC group (p < 0.05). However, the FBG levels were significantly lower in the Med-HTC and Pos-Con groups than in the Neg-Con group (p < 0.01) (Figure 2B).

The Effect of the Hengtang Capsule on the Glucose AUC and FBG Levels in db/db Mice is Shown. (A) The Effect of the Hengtang Capsule on Glucose AUC Levels. (B) The Effect of the Hengtang Capsule on FBG levels. The Data are Presented as the Mean ± Standard Error of the Mean; *p < 0.05, **p < 0.01 and ***p < 0.001, Compared to the Nor-Con Group; ##p < 0.01 and ###p < 0.001, Compared to the Neg-Con Group. Nor-Con Group (n = 12), Neg-Con Group (n = 12), Low-HTC Group (n = 12), Med-HTC Group (n = 12), High-HTC Group (n = 12), and Pos-Con Group (n = 12).
Effects of the Hengtang Capsule on Serum Levels of TC, TG, LDL-C, and HDL-C in Db/db Mice
The TC concentration in the Neg-Con group were significantly greater than that in the Nor-Con group (p < 0.001). Moreover, TC levels were significantly higher in the Low-HTC and High-HTC groups (p < 0.01). The increase in TC levels were significantly attenuated in the Low-HTC group compared to that in the Neg-Con group (p < 0.05). The increase in TC levels were significantly attenuated in the Med-HTC, High-HTC, and Pos-Con groups compared to that in the Neg-Con group (p < 0.01) (Figure 3A). The TG concentration was significantly greater in the Neg-Con and Low-HTC groups than in the Nor-Con group (p < 0.01). However, the TG levels were significantly lower in the Med-HTC, High-HTC, and Pos-Con groups than in the Neg-Con group (p < .01) (Figure 3B). The HDL-C concentration was significantly lower in the Neg-Con group than in the Nor-Con group (p < 0.05), and a more pronounced reduction was observed in the Low-HTC group (p < 0.01) (Figure 3C). The LDL-C levels were significantly higher in the Neg-Con group than in the Nor-Con group (p < 0.05). In contrast, LDL-C levels were lower in the Low-HTC and High-HTC groups than in the Neg-Con group (p < 0.05), and more significant reductions were observed in the Med-HTC and Pos-Con groups (p < 0.01) (Figure 3D).
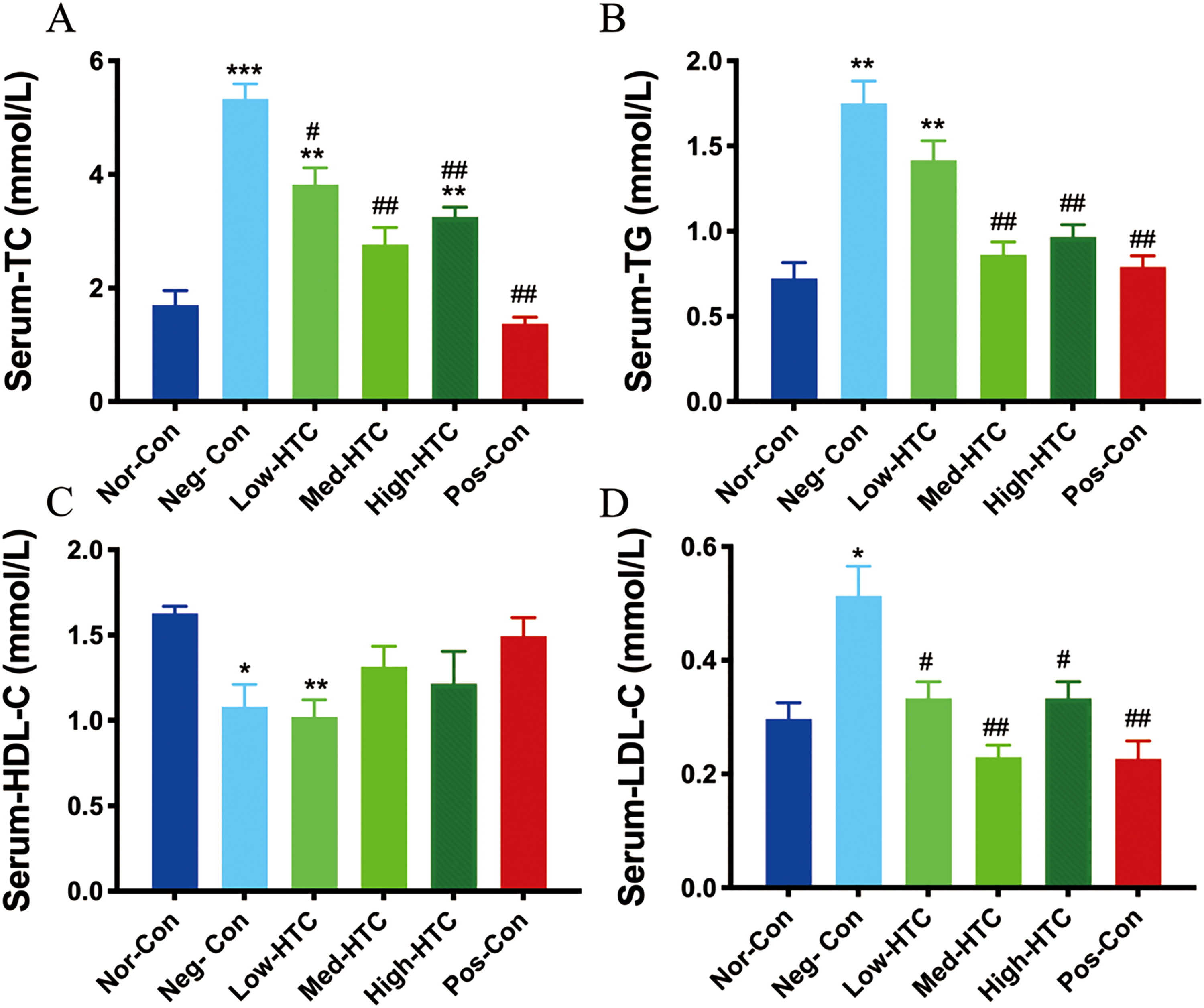
The Effect of the Hengtang Capsule on Lipid Profiles in db/db Mice. (A) The Effect of the Hengtang Capsule Administration on TC Levels. (B) The Effect of the Hengtang Capsule Therapy on TG Levels. (C) The Effect of the Hengtang Capsule Intervention on HDL-C Levels. (D) The Effect of the Hengtang Capsule Treatment on LDL-C Levels. The Data are Presented as the Mean ± Standard Error of the Mean; *p < 0.05, **p < 0.01, and ***p < 0.001, Compared to the Nor-Con Group; #p < 0.05 and ##p < 0.01, Compared to the Neg-Con Group. Nor-Con Group (n = 12), Neg-Con Group (n = 12), Low-HTC Group (n = 12), Med-HTC Group (n = 12), High-HTC Group (n = 12), and Pos-Con Group (n = 12).
Effects of the Hengtang Capsule on Serum AST and ALT Levels of Db/db Mice
The effects of the Hengtang capsule on liver function-related biochemical markers were assessed in db/db mice. AST concentrations were significantly elevated in the Neg-Con group compared to those in the Nor-Con group (p < 0.01). AST levels were also significantly higher in the Low-HTC, Med-HTC, and High-HTC groups (p < 0.05). However, AST levels were significantly lower in the Med-HTC group than in the Neg-Con group (p < 0.05). AST levels were also significantly lower in the Pos-Con group (p < 0.01) (Figure 4A). Similarly, significant differences in ALT concentrations were found across the experimental groups. ALT concentrations were significantly greater in the Neg-Con group than in the Nor-Con group (p < 0.05). Additionally, ALT levels were significantly higher in the Low-HTC, Med-HTC, and High-HTC groups (p < 0.01). However, the ALT concentrations were significantly lower in the Med-HTC and Pos-Con groups than in the Neg-Con group (p < 0.05) (Figure 4B).
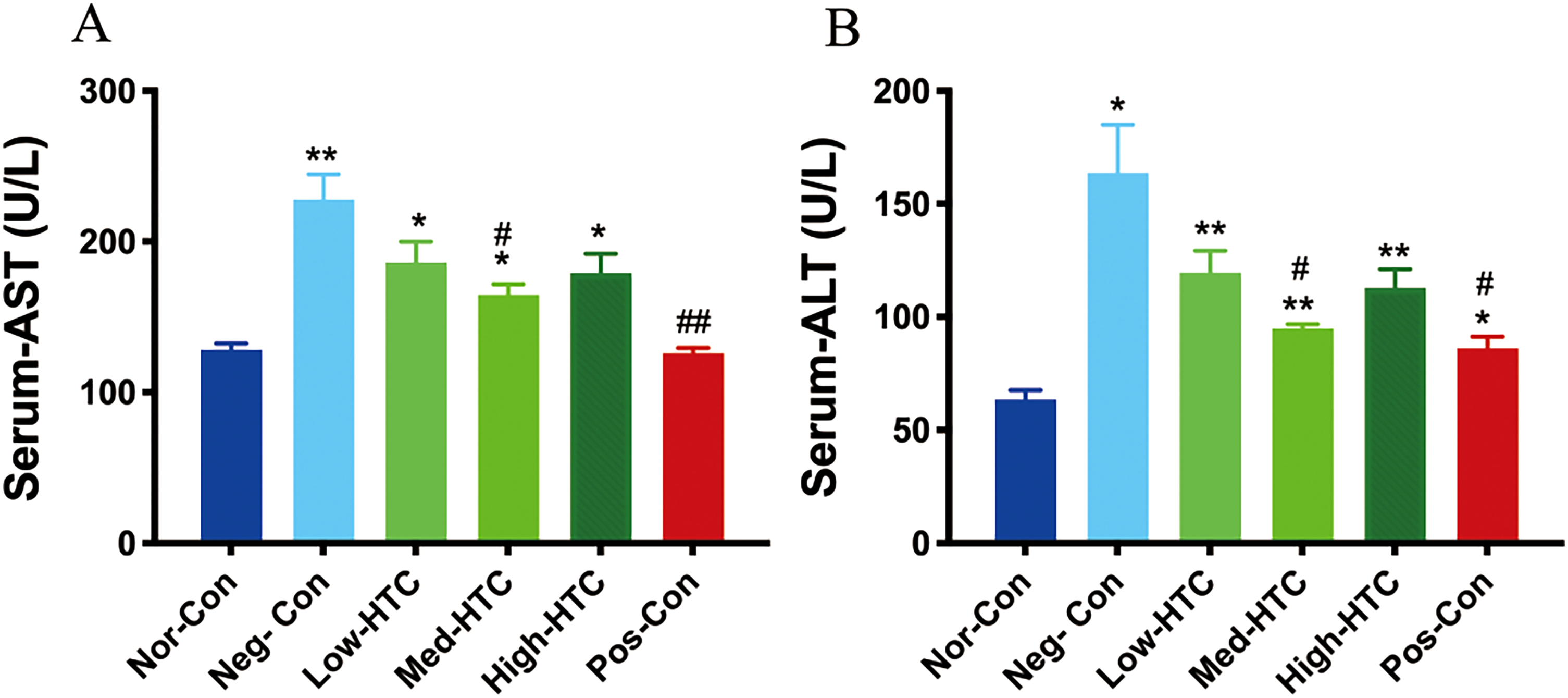
Effects of the Hengtang Capsule on Biochemical Indices Related to Liver Function in db/db Mice. (A) Effect of the Hengtang Capsule Administration on AST Levels. (B) Effect of the Hengtang Capsule Administration on ALT levels. The Data are Presented as the Mean ± Standard Error of the Mean; *p < 0.05 and **p < 0.01, Compared to the Nor-Con Group; #p < 0.05 and ##p < 0.01, Compared to the Neg-Con Group. Nor-Con Group (n = 12), Neg-Con Group (n = 12), Low-HTC Group (n = 12), Med-HTC Group (n = 12), High-HTC Group (n = 12), and Pos-Con Group (n = 12).
Effects of the Hengtang Capsule on Liver Oxidative Stress in Db/db Mice
The liver MDA levels in the Neg-Con group were significantly greater than that in the Nor-Con group (p < 0.01). Similarly, the liver MDA concentrations in the Low-HTC and High-HTC groups were significantly higher (p < 0.01), and those in the Med-HTC group were significantly greater (p < 0.05). However, compared to those in the Neg-Con group, the MDA levels in the livers of the Low-HTC group were significantly lower (p < 0.05), whereas those in the Low-HTC, High-HTC, and Pos-Con groups were significantly lower (p < 0.01) (Figure 5A). As shown in Figure 5B, compared to that in the Nor-Con group, the liver SOD levels in the Neg-Con group were significantly lower (p < 0.05), and the liver SOD levels in the low-dose group were significantly lower (p < 0.01). Compared to that in the Neg-Con group, the MDA levels in the liver of the medium-dose group were significantly greater (p < 0.05), and the MDA levels in the liver of the Pos-Con group were significantly greater (p < 0.01).
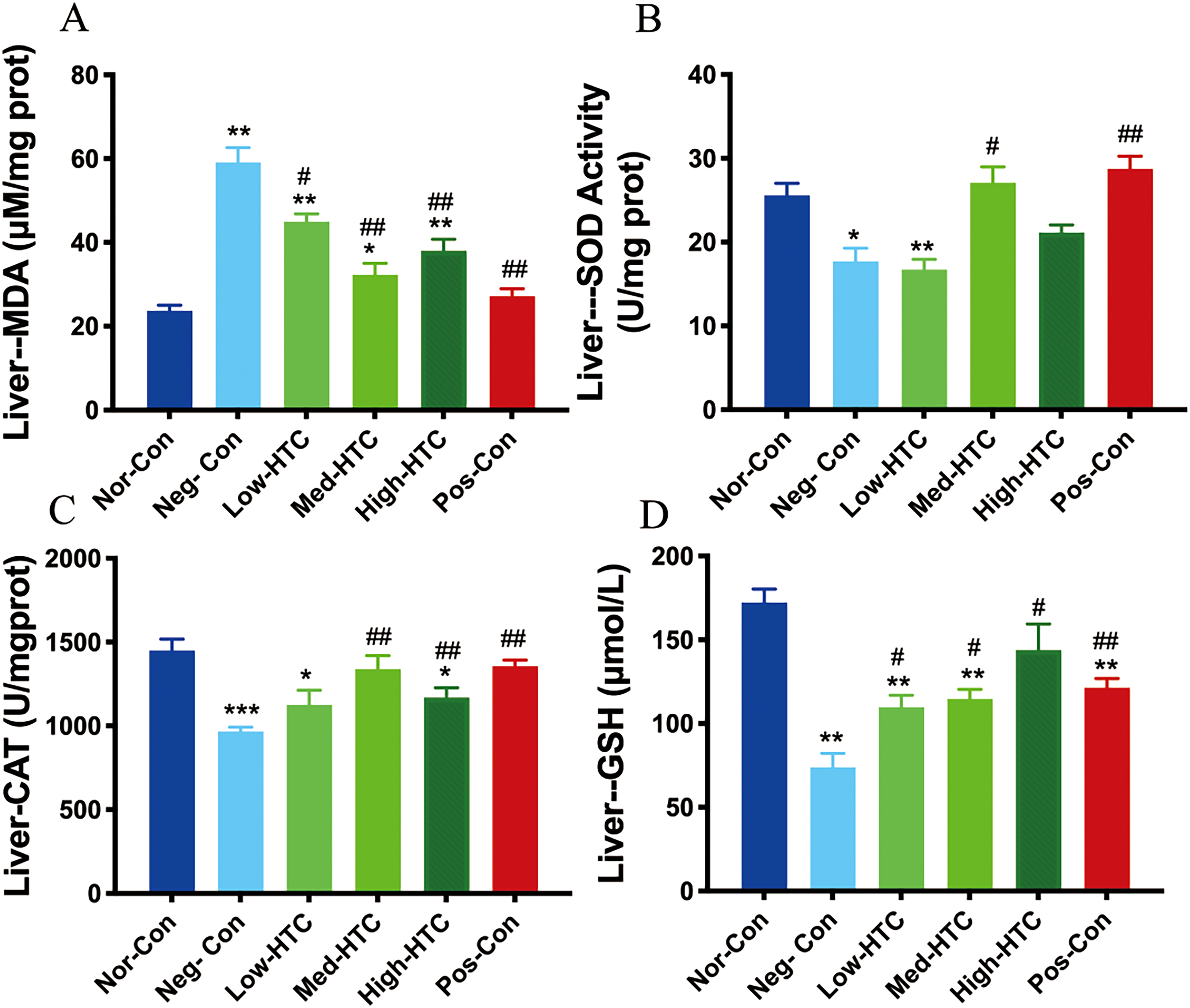
Effects of the Hengtang Capsule on Hepatic Oxidative Stress-related Indices in db/db Model Mice. (A) Effect of the Hengtang Capsule Administration on Liver MDA Levels. (B) Effect of the Hengtang Capsule Administration on Liver SOD Activity. (C) Effect of the Hengtang Capsule Administration on Liver CAT Activity. (D) Effect of the Hengtang Capsule Administration on Liver GSH Levels. The Data are Presented as the Mean ± Standard Error of the Mean; *p < 0.05, **p < 0.01, and ***p < 0.001, Compared to the Nor-Con Group; #p < 0.05 and ##p < 0.01, Compared to the Neg-Con Group. Nor-Con Group (n = 12), Neg-Con Group (n = 12), Low-HTC Group (n = 12), Med-HTC Group (n = 12), High-HTC Group (n = 12), and Pos-Con Group (n = 12).
The liver CAT levels were significantly lower in the Neg-Con group than in the Nor-Con group (p < 0.001). Similarly, CAT levels were significantly lower in the Low-HTC and High-HTC groups (p < 0.05). However, the Med-HTC, High-HTC, and Pos-Con groups presented greater liver CAT levels compared to the Neg-Con group (p < 0.01). The liver CAT levels were also significantly higher in the Pos-Con group (p < 0.01) (Figure 5C). The liver GSH levels were significantly lower in the Neg-Con, Low-HTC, Med-HTC, and Pos-Con groups than in the Nor-Con group (p < 0.01). However, the liver GSH levels were greater in the Low-HTC, Med-HTC, and High-HTC groups than in the Neg-Con group (p < 0.01). The liver GSH levels were significantly higher in the Pos-Con group (p < 0.01) (Figure 5D).
Effects of the Hengtang Capsule on the Histopathological Alterations in the Liver of Db/db Mice
We performed HE staining to examine the hepatic histopathological alterations associated with the Hengtang capsule (Figure 6). In the Neg-Con group, large and densely distributed lipid droplets were found in the liver tissue, resulting in hepatic cellular morphological alterations, including loosely arranged cytoplasm, indistinct cell boundaries, and inflammatory cell infiltration. In contrast, the liver tissue from the Hengtang capsule intervention groups presented a significant reduction in the accumulation of lipid droplets and inflammatory cell infiltration. Additionally, hepatocytes were more neatly arranged, and the liver cell morphology appeared closer to normal. These findings suggested that the Hengtang capsule intervention alleviates hepatic pathological changes in db/db mice.

Effect of the Hengtang Capsule on Liver Histopathological Changes in the db/db Mice Model. Note: Black Arrows Indicate Pathological Changes in Liver Cells; Scale Bars: 40×: 570 µm; 100×: 230 µm; 200×: 110 µm.
Effects of the Hengtang Capsule on Insulin-Immunoreactive Positive Cells in Pancreatic Tissue of Db/db Mice
Immunofluorescence staining was performed to visualize insulin-immunoreactive cells in the pancreatic islet tissue. After the Hengtang capsule treatment at a specific dose, the islet structure in diabetic model mice appeared well-defined, and the area occupied by insulin-positive cells increased significantly (Figure 7).

Effect of the Hengtang Capsule on the Islet Tissue of the db/db Mouse Model. Scale bars: 130 µm.
Discussion
T2DM is a heterogeneous chronic metabolic disease caused by insulin resistance or deficiency and mainly manifests as hyperglycemia or hyperlipidemia. 39 Despite the development of medicines such as metformin, regorazine, and pioglitazone, which provide considerable benefits to T2DM patients, these drugs are associated with adverse reactions due to their “one drug targeting one target” mechanism. 40 Therefore, complementary and alternative drugs need to be developed for effective intervention in patients with T2DM.
The Hengtang capsule contains several key bioactive ingredients, such as berberine hydrochloride, gamma-oryzanol, and dihydromyricetin. Studies have demonstrated that these compounds play critical roles in preventing hyperglycemia and dyslipidemia through multiple mechanisms, such as inhibiting lipid accumulation, regulating insulin metabolism, and suppressing inflammation and oxidative stress. Berberine hydrochloride targets the PPARγ/FGF21/GLUT2 signaling pathway to improve insulin sensitivity in cells and metabolic disorders in T2DM mice. 41 Gamma-oryzanol is another core compound that directly alleviates endoplasmic reticulum oxidative stress, downregulates the expression of endoplasmic reticulum stress-related genes, and inhibits apoptosis signaling pathways in pancreatic islets. These effects contribute to a decrease in β-cell apoptosis and improvement in glucose metabolism in db/db mice induced by a high-fat diet and streptozotocin. 42 Gamma-oryzanol also increases the secretion of adiponectin in mouse adipocytes by inhibiting the activation of PPAR-γ by NF-κB 1 and promotes the differentiation of adipocytes by increasing the expression of proteins that generate fat marker genes, such as PPAR-γ and C/EBP alpha, in vitro. Additionally, glucose transporter type 4 stores glucose from the cell to the cell surface, enhancing glucose uptake in insulin-resistant cells. 43 Dihydromyricetin has hypoglycemic effects on T2DM by improving glucose and lipid metabolism, reducing the inflammatory response, and decreasing oxidative stress. The signal transduction pathway is the basis for regulating AMPK or mTOR/autophagy and related downstream cascade reactions, including the PGC-1 α/SIRT3, MEK/ERK, and PI3K/Akt signaling pathways. 29 Therefore, in this study, we investigated the therapeutic effects of the Hengtang capsule on T2DM in mice and performed network pharmacology analysis to identify the molecular targets and underlying mechanisms of action.
Network pharmacology analysis revealed that the top 20 targets with the highest degree values included various important targets for developing anti-T2DM drugs, such as AKT1, TP53, CTNNB1, STAT3, EGFR, IL6, BCL2, TNF, and INS, highlighting the multitarget and multicomponent nature of the Hengtang capsule in treating T2DM.44,45 AKT1 is one of the three subtypes of AKT and is an important signaling molecule involved in various cellular processes, including cell growth, survival, proliferation, and differentiation. 46 It regulates glucose uptake in the body by mediating the insulin-induced transport of SLC2A4/GLUT4 glucose transporters to the cell surface and plays an important role in maintaining the stability of BG. 47 INS, which is secreted by β-cells, is important for regulating BG levels. The impaired secretion and function of insulin leads to chronic hyperglycemia, a hallmark of diabetes. 48 TP53, a key tumor suppressor gene, plays an important role in T2DM. It is associated with damage to pancreatic beta cells, glucose homeostasis disorders, and peripheral tissue insulin resistance, contributing to the progression of T2DM. 49
Our study also revealed that the common targets of T2DM and the Hengtang capsule are involved in many diabetes-related signaling pathways, such as the PI3K/Akt pathway, lipid and atherosclerosis, the TNF pathway, insulin resistance, and the FoxO pathway. Insulin resistance can impair glucose uptake and storage in skeletal muscle cells, decrease hepatic glycogen synthesis, and increase hepatic gluconeogenesis, ultimately leading to high BG levels. 50 The PI3K/Akt pathway is the main pathway of insulin signal transduction and plays an important role in regulating lipid metabolism and glucose homeostasis. Regulating the activity of proteins related to the PI3K/Akt signaling pathway in liver tissue can decrease the occurrence of abnormal glucose and lipid metabolism and the oxidative stress response in T2DM. 51 The FoxO signaling pathway mainly regulates oxidative stress and controls cell apoptosis and glucose metabolism by converting extracellular signals into cell-specific biological responses. 52 Other T2DM-related pathways, such as the AGE-RAGE pathway and lipid and atherosclerosis pathways, are also involved. These pathways are regulated by bioactive compounds such as berberine hydrochloride, gamma-oryzanol, and dihydromyricetin. For example, berberine hydrochloride has beneficial effects on polycystic ovary syndrome through the PI3K/Akt pathway, including the improvement of insulin resistance and cell viability and the inhibition of apoptosis. 53 Dihydromyricetin activates the FoxO signaling pathway, improving tolerance and lipid levels, slowing intestinal dysfunction, and prolonging the lifespan of fruit flies. 54 Gamma-oryzanol decreases renal inflammation and oxidative stress by regulating the AGE/RAGE axis in animals fed a high-sugar and high-fat diet. 55 To summarize, these findings confirmed that the Hengtang capsule exerts therapeutic effects on T2DM through the abovementioned multiple targets and signaling pathways.
Studies have shown that the concentrations of BG, TG, and TC can serve as indicators of metabolic processes related to carbohydrates and lipids in animals and humans. 56 Diabetes is closely associated with the levels of blood lipids, and reducing these levels can improve diabetes. 57 Glucose tolerance is an indicator of the clinical diagnosis of diabetes. 58 The results of this study showed that the Hengtang capsule effectively reduces FBG levels and the AUC in db/db mice, indicating its regulatory effects on BG in a T2DM model. Under diabetic conditions, the liver loses its ability to regulate BG, which increases glucose levels and the disruption of both glucose and lipid metabolism. Dysregulation of glycolipid metabolism is a key factor in the onset and progression of T2DM and its associated complications. 59 AST and ALT are frequently measured as biomarkers of liver dysfunction. 60 The findings of this study revealed significant increases in the serum levels of TC, TG, LDL-C, ALT, and AST in db/db mice, whereas HDL-C levels were significantly lower. Treatment with a specific dose of the Hengtang capsule significantly decreased the serum levels of TC, TG, LDL-C, ALT, and AST, whereas the HDL-C levels increased, suggesting that the Hengtang capsule modulates lipid metabolism and liver function in T2DM mice.
Diabetes impairs glucose utilization and increases the production of ROS, these changes disturb antioxidant defense capacity, resulting in an imbalance between ROS and antioxidants. This imbalance ultimately leads to cellular oxidative stress. MDA is a byproduct generated from lipid peroxidation intermediates and serves as a biomarker of oxidative stress. SOD is an important antioxidant enzyme that can convert ROS to hydrogen peroxide in the body to eliminate ROS and provide a defense mechanism to inhibit free radical-induced oxidative damage. 61 The results of this study revealed that SOD, CAT, and GSH levels were significantly lower in db/db mice, whereas MDA levels were higher. Treatment with a specific dose of the Hengtang capsule significantly increased the levels of SOD, CAT, and GSH in db/db mice and decreased the levels of MDA, indicating that the Hengtang capsule can inhibit oxidative stress in db/db mice. Therefore, the potential of the Hengtang capsule against diabetes-induced liver oxidative damage may be due to its antioxidant effect, which further helps improve hepatic insulin action. Moreover, histopathological analysis revealed that the Hengtang capsule effectively alleviated liver and pancreatic damage in db/db mice, demonstrating its protective effects and suggesting that the Hengtang capsule protects organ injury associated with metabolic dysfunction in db/db mice.
Abnormal lipid metabolism and oxidative stress are interrelated, and lipid metabolism disorders can increase oxidative stress, which in turn aggravates lipid metabolism disorders. 62 Therefore, based on the results of network pharmacology, the Hengtang capsule may alleviate oxidative stress in db/db mice, protect the liver from damage, and regulate blood lipid and glucose levels by regulating targets (such as AKT1, TNF, TP53, INS, MAPK3, and CASP3) and signaling pathways (such as PI3K/Akt, FoxO, insulin resistance, and MAPK). These findings suggested that the Hengtang capsule can comprehensively improve T2DM through multi-target and multi-pathway approaches. Although network pharmacology provides a theoretically robust prediction of the targets and pathways of the Hengtang capsule in treating T2DM, this study has several limitations. First, we did not conduct in vivo molecular biology experiments to investigate the precise regulatory effects of the Hengtang capsule on potential targets and pathways. Second, we did not include in vitro experiments to establish a T2DM cell model. Therefore, future studies should include in vitro and in vivo experiments based on the predictions of network pharmacology. Changes in the mRNA and protein levels of key target proteins in relevant pathways, such as PI3K/Akt and FoxO, warrant further investigation. Additionally, genetic knockout or pharmacological inhibition of these pathways should be performed to determine whether blocking these mechanisms affects the therapeutic efficacy of the Hengtang capsule.
Conclusions
The Hengtang capsule can regulate BG and lipid levels, liver function, and liver oxidative stress in db/db mice and improve glucose and lipid metabolism and liver and pancreatic injury. These findings suggested that the Hengtang capsule, at a specific dose, can further regulate the onset and progression of T2DM by modulating the antioxidant response of the liver to glucose and lipid metabolism. A systematic study was conducted using network pharmacology to elucidate the pharmacological mechanism underlying the multi-component, multi-target, and multi-signaling pathway intervention of the Hengtang capsule in T2DM. These results indicated that the Hengtang capsule may exert therapeutic effects by acting on key targets, such as AKT1, TNF, JUN, INS, MAPK3, and CASP3, to modulate critical signaling pathways, including the PI3K/Akt, FoxO, insulin resistance, and MAPK pathways. These actions decrease oxidative stress and thereby alleviate disturbances in glucose and lipid metabolism, playing an important role in treating T2DM. However, we did not conduct relevant experiments for verification and only investigated mechanisms along with previous studies to provide a reference for subsequent research.
Footnotes
Ethical Approval
The animal study protocol was approved by the Committee on the Ethics of Animal Experiments of the Binzhou Medical University (No. 2023-121), following the guidelines of animal use and care.
Author Contributions
Zhiqi Li: Operation of the experiment, Statistics, Analysis, Interpretation of relevant data, Writing of the initial manuscript. Xiaoran Liu: Operation of the experiment. Analysis of experimental data, Plot figures. Zhiqi Li and Xiaoran Liu contributed equally to this work and thus should be considered as co-first authors. Wenguo Jiang: Analysis of experimental data, Plot figures. Wenzhuo Ming: Supervision, Writing-reviewing, and editing. Zhenhong Xia: Supervision, Writing-reviewing and editing, Funding of experiments, Overall project mana. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the [Major Basic Research Project of Shandong Province Natural Science Foundation] under Grant [ZR2019ZD27], [Key R&D Program of Shandong Province, China] under Grant [2023CXPT012], and [Natural Science Foundation of Shandong Province] under Grant [ZR2021QH305], and [Startup Foundation for Introducing Talent of Binzhou Medical University] under Grant [BY2020KYQD24].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Statement of Human and Animal Rights
All experimental procedures involving animals followed the Binzhou Medical University Institutional Animal Care Guidelines and approved by the Binzhou Medical University Laboratory Animal Management Committee.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
