Abstract
Objective
This study aimed to investigate the effect of the patented herbal formula Fanqi Tea (Guava leaf, Panax notoginseng flower, Momordica grosvenori, and Jasmine flower) and swimming exercise intervention on hepatic glucolipid metabolism and inflammatory response in db/db mice, to explore whether a combination intervention could bring in a synergistic effect on the treatment of type 2 diabetes mellitus (T2DM) and non-alcoholic fatty liver disease (NAFLD).
Method
Network pharmacology and molecular docking were used to predict the mechanism of action of Fanqi Tea on T2DM and NAFLD. 36 db/db mice were divided randomly into six groups: the model control (Control) group, the metformin (MET) group, the Fanqi Tea low-dose (FT-L) group, the Fanqi Tea high-dose (FT-H) group, the swimming exercise (Exercise) group, and the Fanqi Tea high-dose combined with exercise (FT-H + Exercise) group. Six wt/wt mice were set as the (Normal) group.
Results
The screened main active ingredients and the key targets PIK3R1, AKT2, and GSK3β may have good binding activity. The hepatic Pik3r1 and Akt2 mRNA expression, as well as p-PI3K and p-AKT level tended to increase in the intervention groups, and the expression level of Gsk3β mRNA and p-GSK3β was significantly inhibited. Fanqi Tea and swimming exercise interventions differentially decreased CD86 fluorescence intensity and increased CD206 fluorescence intensity. The expression of pro-inflammatory cytokines, including interleukin 6 (IL-6) and tumor necrosis factor-alpha (TNF-α), was significantly decreased. While the anti-inflammatory factor IL-10 was significantly increased.
Conclusions
Fanqi Tea and swimming exercise may regulate hepatic glucolipid metabolism by acting on the PI3K/AKT/GSK3β pathway. These interventions can decrease hepatic inflammatory responses, which may be attributed to the modulation of macrophage polarization. However, the effect of Fanqi Tea combined with exercise in improving glucolipid metabolism and alleviating inflammation was not significantly better than exercise alone or Fanqi Tea alone.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disease characterized by elevated blood glucose levels due to insulin resistance (IR) or relative deficiency in secretion. 1 Non-alcoholic fatty liver disease (NAFLD), now recognized as metabolic dysfunction-associated fatty liver disease (MAFLD) or metabolic dysfunction-associated steatotic liver disease (MASLD), is a metabolic-associated liver disease in which hepatic lipid accumulation occurs in the absence of significant alcohol intake or other definitive hepatic injury factors. 2 Currently, T2DM has a global prevalence of approximately 500 million, while NAFLD affects up to 25% of adults. Both conditions are increasing in prevalence, thereby constituting a pervasive global public health concern.3,4 NAFLD is a risk factor for T2DM, and there is a bidirectional causal relationship between hepatic lipid accumulation and T2DM. The prevalence of NAFLD in patients with T2DM exceeds 70%, and more than one-fifth of adults with NAFLD are also affected by T2DM.5,6 T2DM has been demonstrated to accelerate the course of NAFLD towards non-alcoholic steatohepatitis (NASH) and to heighten the risk of cirrhosis and hepatocellular carcinoma, whereas NAFLD has been shown to lead to a significant increase in the incidence of macrovascular and microvascular lesions in patients with T2DM. 7
T2DM and NAFLD are both metabolic and inflammatory diseases that share a common pathological basis, which includes obesity, IR, and dyslipidemia.8,9 The liver functions as both a target organ for insulin action and an organ involved in lipid accumulation and lesions in the disease states of T2DM and NAFLD. It plays an important role in maintaining glucose and lipid metabolic homeostasis. 10 The metabolic processes of hepatic glucolipid metabolism are mainly regulated by the insulin signaling pathway, and phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) are key signaling factors in the downstream of insulin receptor substrate (IRS). Activated phosphorylated AKT (phospho-AKT, p-AKT) has been shown to regulate several key factors, including glycogen synthase kinase-3 beta (GSK3β), which is associated with glycogen synthesis, as well as other gluconeogenesis-related factors.11,12 Disorders of glucose and lipid metabolism lead to excessive ectopic accumulation of lipids, increasing the risk of obesity, T2DM, hepatic dysfunction, and cardiovascular disease. These pathologies affect insulin signaling and increase systemic inflammation. 13 In systemic chronic inflammatory disease states such as obesity and T2DM, an imbalance in the ratio of M1/M2 type macrophages has been observed. When the polarization phenotype of macrophages is converted from M2 to M1, the number of M1 pro-inflammatory macrophages is increased in comparison to the number of M2 anti-inflammatory macrophages that are downregulated. The resultant increase in chronic inflammation and metabolic dysfunction is closely associated with the generation of IR, which contributes to the impairment of both insulin sensitivity and insulin secretion, as well as the development and progression of T2DM. The restoration of the M1/M2 macrophage imbalance has the potential to attenuate the inflammatory response and promote damaged tissue repair. 14
Current treatment options for the comorbidities of T2DM and NAFLD are limited. Exercise has been demonstrated to enhance body metabolism. However, it may not result in substantial biopsy-proven enhancements in liver histopathology, including inflammation and fibrosis. 15 Statins, silymarin, and vitamin E appear to possess promise in the treatment of NAFLD. Nevertheless, these substances have not yet received formal approval, and they can not regulate blood glucose levels. The selection of hypoglycemic drugs that reduce body weight may be an important strategy to improve the prognosis of both diseases. 16 Natural herbal products may have the potential to serve a complementary role in the early prevention and treatment of metabolic and inflammatory diseases. One example is Fanqi Tea, a Chinese national invention patent consisting of Guava leaf, Panax notoginseng flower, Momordica grosvenori, and Jasmine flower (Patent No. CN101606978B). This patent has the potential to be applied in the prevention and treatment of T2DM and its related complications such as NAFLD. 17 The objective of this study is to investigate the effect of Fanqi Tea and swimming exercise intervention on hepatic glucose and lipid metabolism, as well as inflammatory response in db/db mice, to explore whether a combination intervention could bring in a synergistic effect on the treatment of these comorbid diseases.
Methods
Aqueous Extract Preparation and Quality Control of Fanqi Tea
Guava leaf (Cat. No. a10000602), Panax notoginseng flower (Cat. No. a10000601), Momordica grosvenori (Cat. No. a100010203), and Jasmine flower (Cat. No. a10000603) were purchased from Beijing Shuangqiao Yanjing Pharmaceutical Co., Ltd (Brand: Shiweicao). The method of preparing the aqueous extract of Fanqi Tea was to weigh Guava leaf : Panax notoginseng flower : Momordica grosvenori : Jasmine flower = 8 : 2 : 1 : 1. Then these herbs were crushed into powder form, which was subsequently added to 10 times distilled water (mass/volume) and soaked for 2 h. The mixture was heated to 100°C for reflux extraction for 2 h twice, and then filtered. The filtered extract was then concentrated at 60°C and dried under reduced pressure to get the desired aqueous extract. The ultra-high-performance liquid chromatography-mass spectrometry/mass spectrometry (UHPLC-MS/MS) was utilized for the quality control of Fanqi Tea, with the technical parameters detailed in Table S1.
Network Pharmacology Prediction and Molecular Docking
The TCMSP database (https://tcmsp-e.com/) and HERB database (http://herb.ac.cn/) were used to collect active ingredients in Fanqi Tea (Guava leaf, Panax notoginseng flower, Momordica grosvenori, and Jasmine flower).18,19 The oral bioavailability (OB) and drug likeness (DL) parameters were set to ≥20% and ≥0.1, respectively, in the TCMSP database. The SwissADME platform (http://www.swissadme.ch/) was utilized to conduct further screening of active ingredients, requiring a minimum of two “yes” in the drug-likeness parameters and “high” GI absorption. To supplement the ingredients not included in the database or not passing the screening parameter, but reported in the relevant research literature, a search was conducted on PubMed and China National Knowledge Infrastructure (CNKI). The target prediction of the active ingredients was performed by the SwissTargetPrediction platform (http://www.swisstargetprediction.ch/) (Probilitity > 0.1) and the SuperPred platform (https://prediction.charite.de/index.php) if the number of atoms is greater than 200.
“Type 2 diabetes mellitus” and “non-alcoholic fatty liver disease” were used as the search terms to collect related disease targets from three databases: GeneCards (https://www.genecards.org) (limited to the top half of the Score median value), OMIM (https://omim.org/), and TTD (http://db.idrblab.net/ttd). The GEO database (https://www.ncbi.nlm.nih.gov/gds/) was queried to identify liver tissue gene chips for normal populations, comparing patients with T2DM and patients with NAFLD. The GEO2R online analysis tool was applied to analyze the concatenated set of differentially expressed genes with P value <0.5 and |logFoldChange| > 0.5 as screening conditions. 20 The SRplot platform was used to draw the Venn diagram of predicted ingredient action targets and disease targets. 21
The intersection targets were imported into the String database (https://string-db.org/) to derive the protein-protein interaction (PPI) relationship (interaction score: 0.900). Drug-ingredient-target network visualization and key target screening were performed using Cytoscape 3.10.2 software with the cytoHubba 0.1 plug-in by degree, MCC, and MNC topological analysis methods. 22 The selection of the top 10 active ingredients was based on the ingredients in the drug-ingredient-target network, as determined by the intersection of their betweenness centrality, closeness centrality, and degree ranking. R 4.4.1 software was applied for Gene Ontology (GO) annotation and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis. Key target protein structures were downloaded from the RSCB PDB database (https://www.rcsb.org/), and molecular docking of active ingredients and key targets was performed by AutoDock Vina 1.1.2 software and PyMOL 2.5 software. 23
Animals and Experimental Protocol
Thirty-six 5-week-old male db/db mice (BKS-Leprem2Cd479/Gpt) and six 5-week-old male wt/wt mice were purchased from Beijing YaoKang Biotechnology Co., Ltd, Beijing, China [Animal license number: SCXK (Jing) 2023-0008], and the mice were fed standard maintenance feed under SPF conditions at Liangxiang Campus of Beijing University of Chinese Medicine [License number: SYXK (Jing) 2023-0011]. The reporting of this study conforms to ARRIVE 2.0 guidelines. 24
On the third day during the 10-day acclimation period, disease modeling was considered to be established in db/db mice with tail-end fasting blood glucose (FBG) ≥ 11.1 mmol/L. db/db mice were divided into six groups according to FBG and fasting body weight by the random number method, which were the model (Control) group, the metformin (MET) group, the Fanqi Tea low-dose (FT-L) group, the Fanqi Tea high-dose (FT-H) group, the swimming training (Exercise) group, and the Fanqi Tea high-dose combined with exercise (FT-H + Exercise) group. Six wt/wt mice were set as the (Normal) group.
The mice included in the exercise intervention were trained for adaptive swimming after grouping during the adaptive feeding period. The first swimming duration was 10 min, the daily increase was increased by 5 min to 30 min for 5 d, and the daily swimming was 30 min starting in the second week, 5 d per week for a total of 4 weeks.25,26 According to the traditional herbal usage and the recommended ratio of invention patents, the adult dosage of Fanqi Tea was 24 g/kg/d (Guava leaf: 16 g, Panax notoginseng flower: 4 g, Momordica grosvenori: 2 g, Jasmine flower: 2 g), and the FT-H group and FT-L group were equivalent to 1 and 0.5 times the normal adult dose, respectively. The dose was determined based on the body surface area normalization method between adult and mouse. The daily oral drug dose was 1.56 g/kg/d in the FT-L group, 3.12 g/kg/d in the FT-H group and the FT-H + Exercise group, 65 mg/kg/d in the MET group, and the mice in the Normal, Control, and Exercise groups were given the same volume of distilled water (0.01 mL/g) for 4 w. Swimming should not be started until 30 min after administration, The mice were not weight-bearing during the swimming. The temperature of the water was controlled to be 35 ± 1°C. The time that the mice were suspended against the wall and rested should not exceed 30 s. Other non-exercise groups were fasted for the same length of time during the swimming exercise period.
During the experiment, the general conditions, such as FBG, body weight, and food intake of the mice, were observed and recorded, and the oral glucose tolerance test (OGTT) was performed at the end of the intervention. After the mice were euthanized via isoflurane anesthesia (R530, Shenzhen RWD Life Science Co., Ltd, China), samples were collected, and serum was obtained after an additional 48 h to avoid the immediate effects of exercise and then used for subsequent testing.
Serum Biochemistry Analysis
The levels of blood lipids total cholesterol (TC), triglycerides (TG), low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C), as well as liver and kidney function indexes alanine aminotransferase (ALT), aspartate aminotransferase (AST), Urea, creatinine (Cr), uric acid (UA) in serum, were measured using an automatic biochemical analyzer (BS-420, Shenzhen Mindray Biomedical Electronics Co., Ltd, China).
Liver Biochemistry Analysis
Liver TC (Cat. No. A111-1-1), liver TG (Cat. No. A110-1-1), and liver total protein (Cat. No. A045-4-2) were detected according to the manufacturer's instructions of Nanjing Jiancheng Institute of Bioengineering, China. The optical density (OD) value was read by the GloMax Multi + Detection System (E9032, Promega, USA), and the corresponding content was calculated.
Liver Histopathological Analysis
Hematoxylin-eosin (H&E) staining, periodic acid-schiff (PAS) staining, and oil red O staining were performed on paraffin sections and frozen sections of formaldehyde-fixed liver tissues of mice. Pathologic sections were observed using a light microscope (BX53, Olympus, Japan).
Reverse Transcription-quantitative PCR (RT-qPCR)
Total RNA of liver tissue was extracted (Cat. No. AG21017) and reverse transcribed into cDNA (Cat. No. AG11706) according to the instructions of Accurate Biotechnology (Hunan) Co., Ltd, Changsha, China. The cDNA was amplified using the SYBR Green Pro Taq HS premixed qPCR kits (Cat. No. AG11701) from the same company. RT-qPCR was used to detect the expression of the PI3K/AKT/GSK3β signaling pathway and macrophage polarization-related mRNA by CFX Duet Real-Time PCR System (12016265, Bio-Rad, USA). The primer sequences table was shown in Table S2. The relative expressions of the mRNA were determined by the 2−ΔΔCt method using β-actin as the internal control.
Immunohistochemistry
Immunohistochemistry was used to detect the relative protein expression levels of p-PI3K p85 (Tyr458)/p55 (Tyr199) (1:50, Cat. No. 17366, Cell Signaling Technology, USA), p-AKT Ser473 (1: 100, Cat. No. 66444-1-Ig, Proteintech Group, USA), and p-GSK3β Ser9 (1: 50, Cat. No. 9323, Proteintech Group, USA) in liver tissue. The secondary antibody was ultrapolymer goat anti-rabbit IgG-HRP (Cat. No. PR30011, Proteintech Group, USA). After staining with the DAB Kit (Cat. No. PV-6000D, Beijing Zhongshan Golden Bridge Biotechnology Co. Ltd, China), liver immunohistochemical sections were observed by light microscope (400×). The relative protein expression levels were measured by the average OD value and calculated using ImageJ.
Immunofluorescence
Immunofluorescence was used to analyze the polarization of liver macrophages. CD86 (1: 1000, Cat. No. YM8023, Immunoway, USA) was used to label M1 macrophages, and M2 macrophages were labeled with CD206 (1: 4000, Cat. No. 24595, Cell Signaling Technology, USA). Average fluorescence intensity was calculated at 400 × fluorescence microscope magnification (DM3000, Leica, Germany).
Enzyme-Linked Immunosorbent Assay (ELISA)
Serum fasting insulin (FINS) was detected by Mouse Ultrasensitive Insulin ELISA Kit (Cat. No. 80-INSMSU-E01, ALPCO Diagnostics, USA). Mouse Interleukin 6 (IL-6) ELISA Kit (Cat. No. E-EL-M0044) and Mouse Interleukin 10 (IL-10) ELISA Kit (Cat. No. E-EL-M0046) were used to detect the expression of IL-6 and IL-10 in liver tissue (Wuhan Elabscience Biotechnology Co., Ltd, China). Homeostatic model assessment of insulin resistance (HOMA-IR) was calculated using the following formula: HOMA-IR = FINS (μIU/mL) × FBG (mmol/L)/22.5.
Statistical Analysis
GraphPad Prism 9.5 software was used to analyze the experimental data. Two-way or one-way analysis of variance (ANOVA) and Tukey's multiple comparisons test were used for comparison among multiple groups, whereas Welch's ANOVA and Dunnett's T3 test were used when the data had uneven variance. In instances where the data did not conform to the normal distribution, the non-parametric test was employed. P < 0.05 was considered statistically significant.
Results
UHPLC-MS/MS Result of Fanqi Tea
The ingredients in Fanqi Tea include alkaloids such as tetrahydropentoxyline, pterolactam, and huperzine. Flavonoids like quercetin-3-O-pentoside and avicularin. Phenolic acids, including 3,4-Dihydroxybenzoic acid, 3,5-Dihydroxybenzoic acid, gentisic acid, and 2,3-Dihydroxybenzoic acid, as well as fatty acids and derivatives, such as (R)-2-aminoheptoheptic acid and succinic acid, have been identified. The results of qualitative matching of substances and total ion chromatogram were shown in Table S3 and Fig. S1.
Network Pharmacology Prediction of Fanqi Tea
Guava leaf and Jasmine flower were not included in the TCMSP database. 14 active ingredients of Panax notoginseng flower and 32 active ingredients of Momordica grosvenori were obtained after being limited to OB ≥ 20% and DL ≥ 0.10. The HERB database contained the following ingredients: 13 from Guava leaf, 69 from Panax notoginseng flower, 194 from Momordica grosvenori, and 8 from Jasmine flower. 8 ingredients: gallic acid, quercetin, corosolic acid, maslinic acid, asiatic acid, oleanolic acid, guajadial, and psiguadial A of Guava leaf were supplemented according to relevant research literature,27–34 as well as mogroside V, an ingredient of Momordica grosvenori.35,36 These ingredients are further screened by the SwissADME platform. The active ingredients of the network pharmacology screening of Fanqi Tea were presented in Table S4.
A total of 827 targets of Fanqi Tea were obtained following prediction on the SwissTargetPrediction platform and SuperPred platform (mogroside V). A comprehensive search of the database was conducted for common targets of T2DM and NAFLD. 870 targets were retrieved from the GeneCards database, 34 from the OMIM database, and 3 from the TTD database. After removing duplicates, a total of 901 targets were obtained. The GEO database obtained liver tissue sequencing gene chips of the healthy control population, comparing T2DM (GSE23343) and NAFLD population (GSE89632), in which GSE23343 included 7 normal subjects and 10 T2DM patients, and GSE89632 included 24 normal subjects and 39 NAFLD patients (20 patients with simple steatosis and 19 cases of NASH). 1039 genes were up-regulated, and 1891 genes were down-regulated in the liver tissues of T2DM patients relative to the normal population in GSE23343. 2378 genes were up-regulated, and 1975 genes were down-regulated relative to the normal population in NAFLD patients in GSE89632 (Fig. 1A). 539 differentially expressed genes in the two datasets were obtained by taking the intersection of the two datasets. Eventually, a total of 190 possible targets of action of Fanqi Tea for the treatment of T2DM-NAFLD were obtained (Fig. 1B, Table S5).

Network pharmacology prediction of Fanqi Tea on T2DM and NAFLD. (A) Volcano plots of GSE23343 and GSE89632. (B) Venn diagram of possible targets. (C) Drug-ingredient-target network. (D) Key targets screen. (E) GO analysis. (F) KEGG enrichment analysis.
Mogroside V, siraitic acid B, bryodulcosigenin, oleic acid, oleanolic acid, maslinic acid, mogrol, flazin, kaempferol, and β-sitosterol may be the main active ingredients (Fig. 1C). Phosphatidylinositol 3-kinase regulatory subunit alpha (PIK3R1), AKT1, and AKT2 may be key targets (Fig. 1D). GO analysis results cover regulation of small molecule metabolic process, membrane raft, and nuclear receptor activity (Fig. 1E). KEGG enrichment identified 164 signaling pathways. The pathways primarily associated with T2DM and NAFLD included the AGE-RAGE signaling pathway in diabetic complications, the insulin resistance signaling pathway, and non-alcoholic fatty liver disease. Especially, the insulin resistance signaling pathway analysis revealed an enrichment of PIK3R1, AKT2, and GSK3β, which are closely related to glucose and lipid metabolism (Fig. 1F). KEGG enrichment analysis of the insulin resistance pathway was detailed in Fig. S2.
The molecular docking results demonstrated that the binding energies of the key active ingredients in Fanqi Tea with PIK3R1, AKT2, and GSK3β were all negative, suggesting that the ingredients may exhibit superior binding activities with the targets (Fig. 2).

Molecular docking results. (A) PIK3R1 and Bryodulcosigenin. (B) AKT2 and Flazin. (C) GSK3β and β-sitosterol. (D) Binding energy results.
Effect of Fanqi Tea and Swimming Exercise Alone or Their Combination on Glucolipid Metabolism, IR, and Liver and Kidney Function Indexes
During the intervention period, db/db mice exhibited a higher food intake compared to wt/wt mice in the Normal group. The FBG levels of the MET group, FT-L group, and FT-H group demonstrated a significant decrease, suggesting that Fanqi Tea exerts a comparable effect in reducing FBG as metformin. However, the FBG levels in the Exercise group and the FT-H + Exercise group tended to decrease, though this decrease did not reach statistical significance. The FT-L group, FT-H group, Exercise group, and FT-H + Exercise group all performed a significant decrease in serum TC and LDL-C compared with the Control group. Additionally, there was a tendency for TG to decrease while HDL-C increased; however, none of these differences were statistically significant. The FT-L group and FT-H group exhibited a substantial decrease in FINS level and HOMA-IR, while the FINS level was elevated in the Exercise group and the FT-H + Exercise group. These findings suggest that Fanqi Tea could alleviate IR, and exercise intervention may stimulate insulin secretion (Fig. 3).

Effects of Fanqi Tea and swimming exercise alone or their combination on FBG, blood lipids, FINS, and HOMA-IR. (A) Experimental protocol. (B) Changes in FBG. (C) Serum TC. (D) Serum TG. (E) Serum LDL-C. (F) Serum HDL-C. (G) FINS. (H) HOMA-IR (n = 6). Compared with the Control group, *P < 0.05, **P < 0.01, ***P < 0.001. Compared with the Normal group, #P < 0.05, ##P < 0.01, ###P < 0.001.
The OGTT result showed that the FT-L group, FT-H group, Exercise group, and FT-H + Exercise group demonstrated a reduction in blood glucose levels, though this reduction did not reach statistical significance. This phenomenon may be attributed to the occurrence of certain acoustic stimuli during the measurement process, which resulted in stress-induced elevated blood glucose levels. It is noteworthy that the actual blood glucose values of the db/db mice, particularly those in the Control group, might have exceeded the range of the glucose meter. Consequently, the underestimation of blood glucose values in the Control group during the calculations may have resulted in the production of negative results (Fig. S3).
Serum liver function indicators ALT and AST in the Control group mice were significantly elevated compared to the Normal group, indicating possible liver injury. Only the serum Urea index of the FT-L group, FT-H group, exercise group, and FT-H + exercise group revealed a significant reduction compared to the Control group. No substantial changes were observed in other liver and kidney function indexes, including ALT, AST, Cr, UA, and Urea/Cr. These findings suggest that Fanqi Tea and the exercise intervention are well-tolerated and safe (Fig. S4).
Effect of Fanqi Tea and Swimming Exercise Alone or Their Combination on Reducing Liver Steatosis
The FT-L group, Exercise group, and FT-H + Exercise group exhibited a significant delay in the rate of weight gain compared to the Control group, suggesting that Fanqi Tea and exercise interventions have a potential impact on weight loss. The liver weight and liver index of the Control group were found to be significantly greater than those of the Normal group. In contrast, the FT-L group and FT-H group demonstrated a significant decrease in liver weight compared with the Control group, while the FT-H + Exercise group exhibited a significant reduction in liver index. In comparison with the Normal group, the liver TC and liver TG levels in the Control group exhibited a significant increase. A tendency to reduce the liver TC was observed in the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group. However, the statistical significance of this reduction remained to be determined. Except for the MET group, the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group significantly reduced liver TG (Fig. 4).

Effects of Fanqi Tea and swimming exercise alone or their combination on body weight, liver weight, liver index, and liver lipids. (A) Body weight. (B) Liver weight. (C) Liver index (%). (D) Liver TC. (E) Liver TG (n = 6). Compared with the Control group, *P < 0.05**P < 0.01, ***P < 0.001. Compared with the Normal group, #P < 0.05, ##P < 0.01, ###P < 0.001.
Hepatic H&E staining revealed that the hepatocytes in the Control group exhibited enlarged dimensions and disordered arrangement. A substantial number of fat vacuoles and areas of inflammatory cell infiltration were observed within the field of view under microscopic examination. In the MET group, FT-L group, FT-H group, Exercise group, and FT-H + Exercise group, a decrease in the number of fat vacuoles was observed, indicating an improvement in hepatic steatosis. Hepatic PAS staining indicated that the diminished hepatic glycogen levels observed in the Normal group could be attributed to the lower food intake, which resulted in a limited supply of raw materials available for glycogen synthesis. The augmented purple-red areas and darker coloration detected in the MET group, FT-L group, FT-H group, Exercise group, and FT-H + Exercise group in comparison to the Control group suggested that hepatic glycogen synthesis might be elevated and that hepatic glycogen synthesis has undergone enhancement. In the Control group, a significant number of red lipid droplets were observed in the oil red O staining. Conversely, the MET group, the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group exhibited a notable decrease in red lipid droplets, indicating a potential reduction in hepatic lipid accumulation (Fig. 5).

H&E staining, PAS staining, and oil red O staining of the liver in mice.
Fanqi Tea and Swimming Exercise Alone or Their Combination Activated the PI3K/AKT/GSK3β Pathway
RT-qPCR results demonstrated that the Control group exhibited diminished expression of Pik3r1 and Akt2 mRNA, accompanied by augmented relative expression of Gsk3β mRNA compared with the other groups. A tendency towards heightened hepatic Pik3r1 and Akt2 mRNA expression was observed in the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group. However, only the FT-H group demonstrated a statistically significant increase in Pik3r1 mRNA expression. A statistically significant decrease in Gsk3β mRNA expression was observed in the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group compared to the Control group.
Immunohistochemical analysis results showed that the p-PI3K and p-AKT expression levels in the mice liver of the Control group were significantly lower than those of the Normal group. In contrast, the phosphorylation level of PI3K and AKT were significantly activated in the FT-L group, the FT-H group, and the FT-H + Exercise group, except the p-PI3K expression level in the Exercise group exhibited no statistical difference compared with the Control group. The expression level of p-GSK3β was elevated in the Control group relative to the Normal group. Conversely, the expression level of p-GSK3β was significantly repressed in the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group (Fig. 6).

Fanqi Tea and swimming exercise alone or their combination activated the PI3K/AKT/GSK3β pathway. (A) Relative expression level of PI3KR1 mRNA. (B) AKT2 mRNA level. (C) GSK3β mRNA level. (D) Liver immunohistochemistry of p-PI3K, p-AKT, and p-GSK3β protein. (E) Relative expression level of p-PI3K protein. (F) p-AKT protein level. (G) p-GSK3β protein level (n = 3). Compared with the Control group, *P < 0.05, **P < 0.01, ***P < 0.001. Compared with the Normal group, #P < 0.05, ##P < 0.01, ###P < 0.001.
Fanqi Tea and Swimming Exercise Alone or Their Combination Regulated Liver M1/M2 Macrophage Polarization
CD86 predominantly labeled M1 macrophages, whereas CD206 mainly labeled M2 macrophages. M1 and M2 macrophages secreted different inflammatory cytokines. M1 macrophages predominantly secreted pro-inflammatory factors such as tumor necrosis factor-alpha (TNF-α) and IL-6. In contrast, M2 macrophages produced more anti-inflammatory factors such as IL-10 and transforming growth factor beta 1 (Tgf-β1). 37 The immunofluorescence results were labeled as CD86 in red, and the average fluorescence intensity of CD86 in the liver was increased in the Control group compared with the Normal group, while the average fluorescence intensity of CD206 was decreased. The FT-H group, the Exercise group, and the FT-H + Exercise group exhibited a significant decrease in the fluorescence intensity of CD86 compared with the Control group. Moreover, the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group all demonstrated an increase in CD206 fluorescence intensity. These findings suggest that Fanqi Tea and swimming exercise interventions may regulate hepatic M1/M2 macrophage polarization.
RT-qPCR result indicated that, in comparison with the Control group, the FT-L group, the FT-H group, and the FT-H + Exercise group exhibited a significant decrease in IL-6 and TNF-α mRNA expression. A downward trend in IL-6 and TNF-α mRNA expression was observed in the Exercise group, though this trend did not reach statistical significance. Whereas the Exercise group exhibited a significant increase in IL-10 mRNA expression compared to the Control group. Additionally, TGF-β1 mRNA expression exhibited a tendency to increase in the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group, although statistical differences were not observed.
ELISA results revealed that the Control group mice exhibited a significant increase in hepatic IL-6 levels and a significant decrease in hepatic IL-10 levels compared to the Normal group. In contrast, the FT-L group, the FT-H group, the Exercise group, and the FT-H + Exercise group demonstrated an increase in hepatic IL-10 expression, while also exhibiting different degrees of reduction in IL-6 levels (Fig. 7).
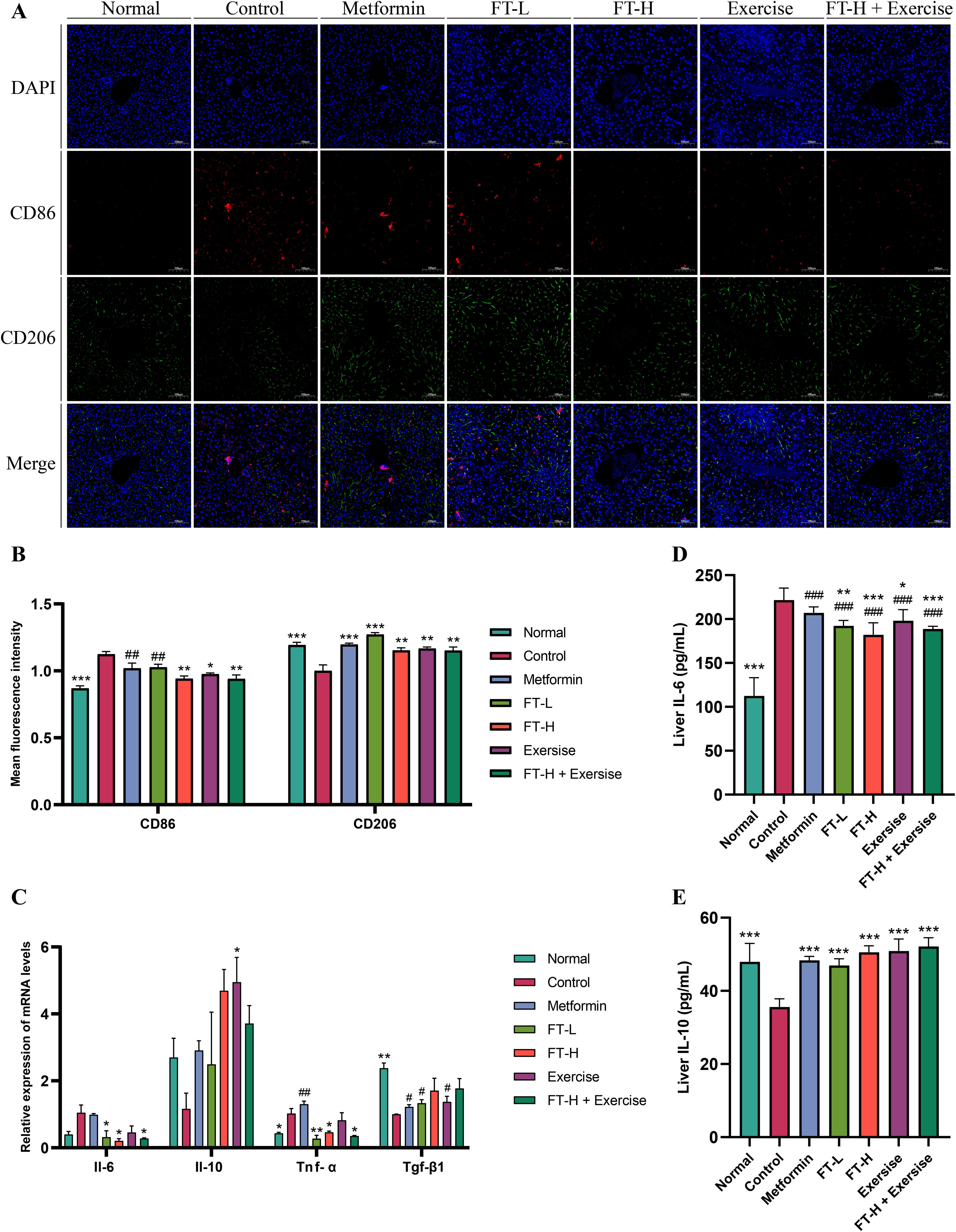
Fanqi Tea and swimming exercise alone or their combination regulated liver M1/M2 macrophage polarization. (A) Immunofluorescence of CD86 and CD206. (B) Mean fluorescence intensity of CD86 and CD206. (C) Relative expression levels of Il-6, Il-10, Tnf-α, and Tgf-β1 mRNA (n = 3). (D) Liver IL-6 level (n = 6). (E) Liver IL-10 level (n = 6). Compared with the Control group, *P < 0.05, **P < 0.01, ***P < 0.001. Compared with the Normal group, #P < 0.05, ##P < 0.01, ###P < 0.001.
Discussion
In the course of the experiment, a notable increase in food intake was observed in db/db mice, concomitant with the development of obesity, hyperglycemia (FBG ≥ 11.1 mmol/L), dyslipidaemia, and hyperinsulinemia phenotypes. The serum levels of ALT, AST, as well as liver TC and TG, were found to be significantly elevated. Concurrently, pathological changes in the liver were detected. Liver H&E staining showed obvious steatosis and inflammatory cell infiltration, especially in the Control group. PAS staining and oil red O staining suggested a decrease in glycogen synthesis and an increase in lipid accumulation. In general, the T2DM and NAFLD disease model was stable and credible.As a basic lifestyle intervention, moderate exercise has been demonstrated to possess significant health benefits in the prevention of many metabolic diseases. Nevertheless, its application is limited by specific requirements for location, frequency, and intensity, as well as poor patient compliance. As conditions progress, exercise alone may become insufficient or unachievable at the needed intensity. Fanqi Tea, a patented tea substitute, has demonstrated weight-loss, hypoglycemic, and hypolipidemic effects without substantially increasing hepatic or renal burden, showing promise in metabolic disorder management. This study preliminarily investigates the mechanism of Fanqi Tea and its potential synergistic effects with exercise.
Network pharmacological predictions indicated that Fanqi Tea may act on the PI3K/AKT/GSK3β pathway of the insulin resistance signaling pathway, thereby regulating hepatic glucose and lipid metabolism. In this experiment, the mRNA of pathway-related signaling molecules PIK3R1, AKT2, and GSK3β, as well as the protein expression of p-PI3K, p-AKT, and p-GSK3β, were verified. PIK3R1, also known as p85α, is one of the three regulatory subunits of PI3K and forms a heterodimer with the catalytic subunit. As a downstream signaling molecule of the insulin receptor, class I PI3K plays an important role in the regulation of insulin signaling and maintenance of metabolic homeostasis, and activates and recruits AKT to the cell membrane to participate in various metabolic functions.38,39 AKT2 is one of the three isoforms of AKT, which is predominantly expressed in insulin-sensitive tissues, such as the liver. The activation of AKT results in the phosphorylation and subsequent inhibition of GSK3β. This, in turn, leads to the removal of the inhibitory state of gluconeogenic synthase, the promotion of glucose uptake and glycogen synthesis, and the improvement of insulin resistance.40,41 Although downstream signaling molecules glycogen synthase and liver glycogen level were not quantitatively analyzed, PAS staining and oil red O staining of the liver suggested that hepatic glycogen synthesis was increased and lipid accumulation was reduced in the Fanqi Tea and exercise groups. Liver TG level was significantly reduced, while liver TC also exhibited a tendency to decrease.
T2DM and NAFLD are diseases of dual etiology, metabolic and inflammatory. The important role of inflammation in increasing the risk of IR is increasingly recognized and can be considered an etiological starting point for the decline of metabolic function in the body. A reduction in the inflammatory response has been shown to exert a metabolic protective effect. Metabolic inflammation is characterized by the presence of tissue-resident macrophages, which have the potential to influence the progression and severity of the disease. Macrophages, vital components of the immune system, are predominantly Kupffer cells derived from the liver, with a smaller percentage of bone marrow-derived macrophages. These macrophages fulfill a critical function in maintaining metabolic balance and repairing liver tissue. Macrophage polarization can be categorized into two distinct classifications: classically activated M1 macrophages, which are believed to elicit pro-inflammatory responses, and alternatively activated M2 macrophages, which secrete anti-inflammatory factors. The role of macrophages in the pathogenesis of these diseases has attracted increasing attention from researchers. Monocytes are recruited to peripheral tissues, such as the liver, pancreas, and adipose tissue. In these tissues, monocytes become resident macrophages. These macrophages promote local inflammation and the development of IR.42,43 The conversion of macrophage polarization phenotype from M2 to M1 in systemic chronic inflammatory disease states leads to increased chronic inflammation and metabolic dysfunction, which is closely associated with the development of patients with obesity and T2DM. The presence of IR, persistent glycolipotoxicity, and systemic inflammatory responses contributes to the development of ectopic fat deposition. This process primarily affects the liver, progressing from steatosis, inflammation, fibrosis, and cirrhosis. Consequently, the restoration of M1/M2 macrophage polarization has been demonstrated to attenuate the inflammatory response and promote damaged tissue repair, thereby playing an important role in systemic chronic inflammatory diseases such as T2DM and NAFLD.44,45 Some researchers have found that reduced hepatic endothelial-type nitric oxide synthase activation in Otsuka Long-Evans Tokushima Fatty (OLETF) rats was significantly associated with reduced insulin secretion, elevated blood glucose, and diminished AKT activation. Moreover, active running-wheel exercise was found to activate endothelial-type nitric oxide synthase and AKT protein phosphorylation, thereby alleviating this pathological change. 46 Tang demonstrated that swimming training could inhibit the activation of macrophage migration inhibitory factor, AKT, and its downstream signaling molecule GSK3β. In addition, it has been shown to promote autophagy, thereby ameliorating hepatic steatosis in high-fat-fed C57BL/6 mice. 47
Our findings contradicted several previously published studies reporting synergistic effects of lifestyle and herbal interventions.48–50 Despite the similarities between the effects of Fanqi Tea and swimming exercise in reducing body weight, blood glucose, blood lipids, and hepatic lipid accumulation, there may be an antagonistic effect of Fanqi Tea and swimming exercise on the regulation of insulin secretion. Swimming exercise was found to promote insulin secretion but did not significantly improve IR in this experiment, which is contrary to the results of some studies with similar experimental designs.51,52 However, there was also evidence from other studies that swimming exercise promoted insulin secretion.53,54 This discrepancy may be attributable to variations in the weekly age and severity of IR in mice, differences in the intensity and number of weeks of exercise intervention, and inadequate recovery period from the end of the intervention to the index measurements. Water temperature may also be one of the influencing factors. A study indicated that swimming training in cold water (5°C) instead of warm water (35°C) combined with herbal intake improved insulin sensitivity in diabetic rats. 55 A thoroughgoing comparison of phenotypic data and regulatory effects on glycolipid metabolism factors and inflammatory factors revealed that Fanqi Tea and swimming exercise did not appear to exert synergistic interactions. The efficacy of Fanqi Tea combined with exercise to improve IR and alleviate inflammation was not significantly better than that of exercise alone or Fanqi Tea alone. Exercise and herbal intervention may improve glucolipid metabolism through different mechanisms while sharing common results. The Pik3r1 and Akt2 mRNA expression, as well as the semi-quantitative results of p-PI3K protein expression level of the Exercise group was not significantly increased. The specific mechanism of swimming exercise may not be completely consistent with the PI3K/AKT/GSK3β pathway of herbal intervention Fanqi Tea predicted by network pharmacology. Further exploration is necessary to elucidate the specific mechanism of action. The possible mechanism of Fanqi Tea and swimming exercise on hepatic glucolipid metabolism and inflammatory response was depicted in Fig. 8.

Graphical abstract. Note: Pik3r1, Phosphatidylinositol 3-kinase regulatory subunit alpha; Akt2, Protein kinase B beta; Gsk3β, Glycogen synthase kinase-3 beta; IL-6, Interleukin-6; IL-10, Interleukin-10; TNF-α, Tumor necrosis factor-alpha.ALT, Alanine aminotransferase; AST, Aspartate aminotransferase; Cr, Creatinine; ELISA, Enzyme-linked immunosorbent assay; FBG, Fasting blood glucose; FINS, Fasting insulin; H&E, Hematoxylin-eosin; HOMA-IR, Homeostatic model assessment of insulin resistance; OGTT, Oral glucose tolerance test; PAS, Periodic acid-schiff; PI3K, Phosphatidylinositol 3-kinase; RT-qPCR, Reverse transcription-quantitative PCR; TG, Triglyceride; Tgf-β1, Transforming growth factor beta 1; UA, Uric acid; UHPLC-MS/MS, ultra-high-performance liquid chromatography-mass spectrometry/mass spectrometry.
Limitation
First, our study only used male db/db mice with leptin receptor mutation as the disease model. The comparative application of multiple models and different genders, such as leptin-mutant ob/ob mice and high-fat diet models, may better simulate the development and pathological changes of human diseases to fully understand the relevant mechanisms. Second, immunohistochemistry and immunofluorescence were used for key protein expression and macrophage polarization, which were qualitative and semi-quantitative. Future studies using other experimental techniques, such as Western blotting and flow cytometry, may provide a more precise and quantitative evaluation in future studies. Moreover, the study had a small sample size and did not classify the exercise intervention by dosage. The dose-response relationships or differences between intervention groups could not be well verified, making the results still require cautious interpretation.
Conclusion
In summary, Fanqi Tea and swimming exercise have a certain effect in regulating disorders of glucolipid metabolism. Fenqi Tea can enhance insulin sensitivity, while swimming exercise may stimulate insulin secretion. Moreover, both interventions have no significant impact on liver and kidney function. Fanqi Tea and exercise may increase glycogen synthesis and reduce liver lipid accumulation by acting on the PI3K/AKT/GSK3β pathway. These interventions have also been observed to decrease hepatic inflammatory responses, which may be attributed to the modulation of macrophage polarization. However, the effect of Fanqi Tea combined with exercise in improving glucolipid metabolism and alleviating inflammation was not significantly better than exercise alone or Fanqi Tea alone.
Supplemental Material
sj-doc-1-npx-10.1177_1934578X261420563 - Supplemental material for Effects of Fanqi Tea and Swimming Exercise Intervention on Hepatic Glucolipid Metabolism and Inflammatory Response in db/db Mice
Supplemental material, sj-doc-1-npx-10.1177_1934578X261420563 for Effects of Fanqi Tea and Swimming Exercise Intervention on Hepatic Glucolipid Metabolism and Inflammatory Response in db/db Mice by Ruifeng Yang, Jiangfan Guo, Ning Wang, Zhiwei Qi, Congyi Liu, Qingsong Li, Lili Wu, Lingling Qin and Tonghua Liu in Natural Product Communications
Footnotes
Abbreviations
Ethical Approval
Ethical approval to report this case was obtained from the Animal Experimental Ethics Committee of Beijing University of Chinese Medicine (No. BUCM-2023111306-4294).
Author's Contributions
RY: Conceptualization, formal analysis, methodology, writing – original draft, writing – review and editing. JG: Writing – review and editing. NW: Writing – review and editing. ZQ: Visualization. CL: Data curation. QL: Data curation. LW: Supervision, validation. LQ: Funding acquisition, supervision, validation. TL: Funding acquisition, supervision. All authors reviewed and approved the final paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Traditional Chinese Medicine Intervention of Metabolic Diseases Research Project, (grant number 90020165120003), Creation and Talent Introduction Base of Prevention and Treatment of Diabetes and Its Complications with Traditional Chinese Medicine (grant number B20055), Key Laboratory of Health Cultivation of the Ministry of Education Preservation (grant number 2024-JYB-KYPT-002), (22) Beijing City “Double First-Class” to Support High-Level Key Disciplines of TCM Funding of 23 years – TCM Health Cultivation (grant number 90010963420018).
Declaration of Conflicting Interests
Tonghua Liu has patent #CN101606978B licensed to Beijing Shuangqiao Yanjing Pharmaceutical Co., Ltd. The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data are available upon reasonable request from the first author or the corresponding authors.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Animal Experimental Ethics Committee of Beijing University of Chinese Medicine (No. BUCM-2023111306-4294) approved protocols.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
