Abstract
Background/Objectives
Bacterial infections caused by resistant strains such as Pseudomonas aeruginosa, Staphylococcus aureus, Klebsiella pneumoniae, Enterococcus spp. and Escherichia coli represent a growing threat to human and animal health. In the face of rising resistance, essential oils derived from medicinal plants offer a promising natural alternative. The present study aims to characterize the phytochemical composition and evaluate the antioxidant and antimicrobial activities of the essential oil extracted from the aerial part of Ononis natrix.
Methods
The phytochemical composition, antioxidant activity and antimicrobial efficacy of the essential oil were assessed using gas chromatography-mass spectrometry (GC-MS), the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging and Ferric-Reducing Antioxidant Power (FRAP) assays, as well as the disk diffusion method, supplemented by determining of the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC), respectively.
Results
Our findings revealed that Ononis natrix essential oil contains a high level of oxygenated monoterpenes (45.6%) and oxygenated sesquiterpenes (35.35%). The lowest IC50 value obtained, of the order of 339 μg/mL, was observed with the reducing power test, thus reflecting a reducing capacity of this oil towards the ferric ion Fe3+. In addition, the essential oil showed marked antimicrobial effects against the Candida strains and multi-resistant bacteria tested.
Conclusion
The findings of the current study demonstrated strong antioxidant and antimicrobial properties of O. natrix, which could potentially provide novel treatments, thus, addressing antibiotic resistance issues in the clinical setting.
This is a visual representation of the abstract.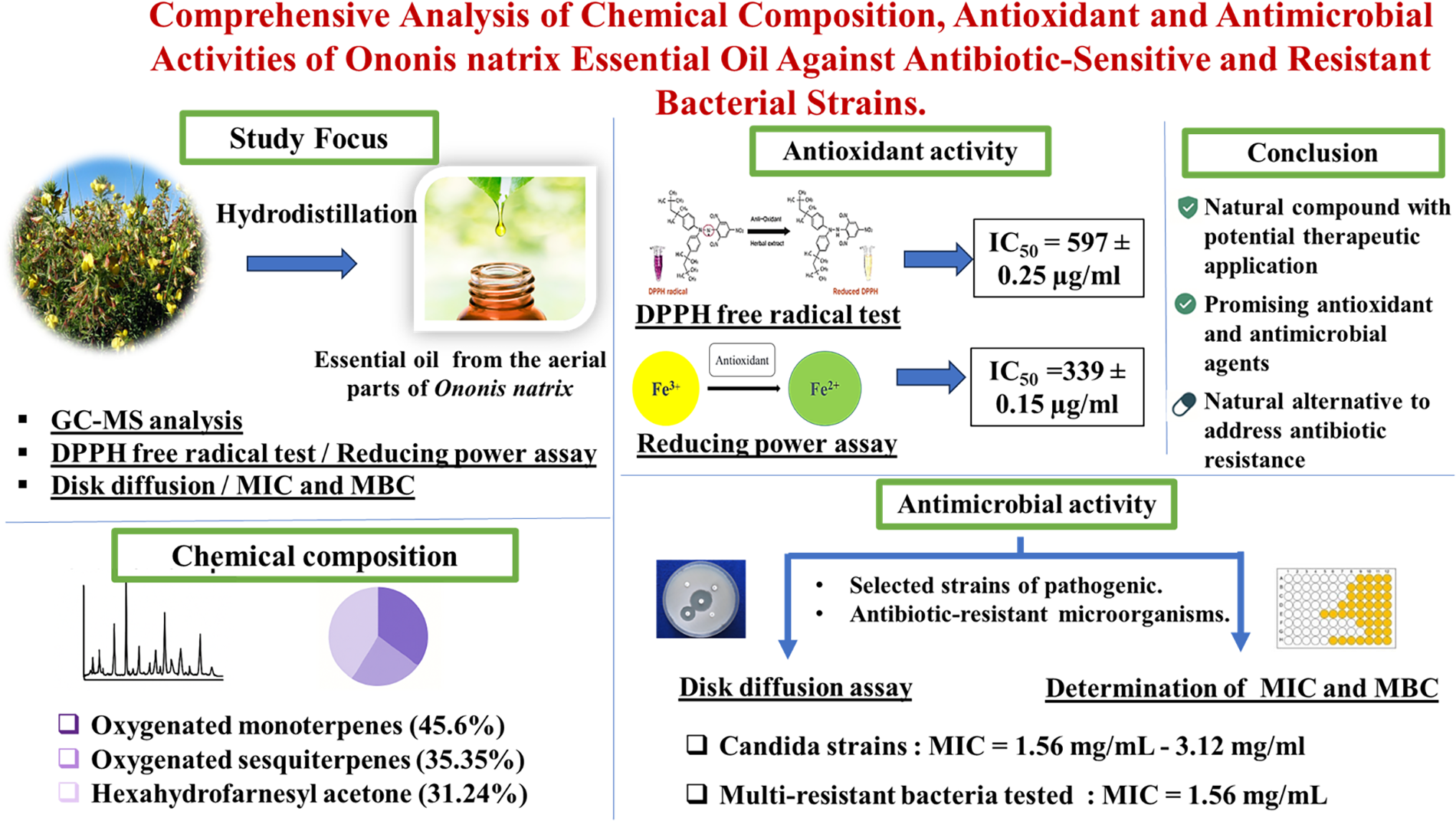
Keywords
Introduction
Antibiotic resistance is one of the most critical health challenges worldwide, causing around 25 000 deaths every year. 1 This phenomenon is due in particular to the excessive and inappropriate use of antibiotics, as well as the poor renewal of the molecules available for several decades. 2 Infections caused by multi-resistant bacteria, such as Salmonella spp., Enterococcus spp., Pseudomonas aeruginosa, Staphylococcus aureus and Escherichia coli, have become a particular cause for concern in both humans and animals. 3 Moreover, the possibility of resistance transfer through the food chain, in particular via products of animal origin, constitutes a major public health risk. 4
In this context, essential oils (EOs) derived from medicinal and aromatic plants represent promising natural alternatives. Rich in secondary metabolites such as monoterpenes and sesquiterpenes, they possess recognized antimicrobial and antioxidant properties. 5 Several plant species have already been studied in this context, but the majority of work focuses on well-known plants such as Origanum vulgare, Thymus vulgaris or Rosmarinus officinalis. To date, very little data is available on the detailed chemical composition and biological activities of the essential oil of Ononis natrix, a species widely used in traditional medicine in Morocco.
Ononis natrix, a species of the Fabaceae family (Leguminosae), is widespread in several regions of Morocco, notably Bou-Azzer, the Oued Tensift basin, the Rif, the Anti-Atlas (Taznakht), and the Mamora forest. Known by the Berber vernacular names âfzzaz, âfesdad or fezzaz. Traditionally, its roots and flowers are used in infusions to treat urinary and liver disorders. 6 Recent studies have highlighted the richness of its essential oil in volatile bioactive compounds.7–10 However, to our knowledge, no in-depth research has yet evaluated its antioxidant and antimicrobial activities against sensitive and resistant pathogenic strains.
In light of these findings, the present study has been produced to fill these gaps. It aims to:
✓ Characterize the phytochemical composition of the essential oil extracted from the aerial part of Ononis natrix using GC-MS; ✓ Evaluate its antioxidant activity using in vitro methods (DPPH and FRAP); ✓ Examine its antimicrobial efficacy against a wide range of pathogenic strains, including multi-resistant strains.
The aim of this research is to scientifically valorize a little-researched medicinal species and explore its potential as a natural source of therapeutic agents.
Material and Methods
Plant Material
In spring 2022, aerial parts (stems, leaves and flowers) of Ononis natrix were collected in their natural habitat in February 2019 in the region of Ouarzazate (Southeastern Morocco). 11 Identification was made by Professor Ouhammou Ahmed, and a reference specimen was deposited at the “MARK” Regional Herbarium of the Semlalia Faculty of Sciences, Cadi Ayyad University, Marrakech, Morocco, under reference specimen code MARK-13792. 11 The material was air-dried at room temperature and stored until use.
Extraction of Essential Oil
EO from air-dried Ononis natrix aerial parts was extracted by hydrodistillation using a Clevenger apparatus. Briefly, 150 g of dried plant material were placed in the Clevenger apparatus and hydrodistilled with 2000 mL of distilled water for four hours at boiling temperature. This process was performed in triplicate. The essential oil was collected from the Clevenger column and stored at 4 °C until chemical composition analysis and biological tests. The essential oil yield was determined using the subsequent formula: - RHE: Essential oil yield (mL/100 g dry matter). - VHE: Volume of essential oil recovered (mL). - MS: Quantity of dry plant matter used for extraction (g).
Analysis of the Essential Oil's Chemical Composition Using Gas Chromatography-Mass Spectrometry
Separation of the constituents of the essential oil extracted from Ononis natrix aerial parts was performed using GC/MS. This analysis was carried out at the Center for Analysis and Characterization, Faculty of Sciences Semlalia, Cadi Ayyad University. The essential oil was analyzed using a gas chromatograph with a TG-5MS column (30 m × 0.25 mm × 0.25 μm). A temperature of 230 °C was used for the injector and 250 °C for the detector, and an electron impact ionization energy of 70 eV was used. The column temperature was ramped at 3 °C/min from 60 °C to 230 °C, where it was held for 10 min. A 1 μL aliquot of the diluted sample (1:100 in acetone) was injected into the GC/MS system. Helium served as the carrier gas at a flow rate of 1 mL/min. 12
The chemical components of the essential oil were identified by comparing their mass spectra with those in the NIST library and by calculating their retention indices (RI), which were determined using a homologous series of n-alkanes (C7–C40) under the same experimental conditions of the essential oils. The components were quantified using the external standard method, based on calibration curves generated from routine GC analyses of authentic standard compounds.
Antioxidant Activity
DPPH Free Radical Scavenging Test
The 2,2′-diphenyl-1-picrylhydrazyl (DPPH) test was used to assess free radical scavenging activity according to the protocol described by. 13
Concentration series of the essential oil (EO) were previously prepared in methanol. Then, 50 μL of each concentration was added to 2 mL of a methanolic solution of DPPH at a concentration of 60 μM. For twenty minutes, the mixture was allowed to sit at room temperature in the dark. Parallel preparation of a negative control involved combining 50 μL of methanol with 2 mL of the DPPH• methanolic solution. Quercetin was used as a reference antioxidant. After incubation, absorbances were measured at 517 nm and converted to percentage DPPH• radical inhibition using the following formula:
The IC50, indicating the concentration of EO or reference antioxidant needed to decrease DPPH radical activity by 50%, was determined from the graph showing the reduction of DPPH (%) as a function of concentration. IC50 values were expressed as the mean of three replicates.
Reducing Activity on Potassium Ferricyanide
In this study, the reducing power of the essential oil (EO) was determined according to the protocol described by. 14 Briefly, 1 mL of different concentrations of EO or quercetin (standard) prepared in methanol, was mixed with 2.5 mL of PBS (0.2 M; pH 6.6) and 2.5 mL of 1% potassium ferricyanide solution (K3Fe(CN)6). Tubes were incubated at 50 °C for 20 min. After incubation and cooling, 2.5 mL of 10% trichloroacetic acid was added to stop the reaction. Tubes were centrifuged at 3000 g for 10 min. Subsequently, 2.5 mL of the supernatant from each concentration was mixed with 2.5 mL distilled water and 0.5 mL 0.1% FeCl3. Absorbance was measured at 700 nm against a blank prepared in parallel, containing methanol instead of samples. The concentration of EO with an absorbance of 0.5 is considered the IC50.
Antimicrobial Assays
Microbiological tests were carried out at the Laboratory of microbiology and virology, Department of medical biology, Faculty of medicine and pharmacy, Cadi Ayyad University Marrakech, Morocco.
Strains Tested
EO extracted from Ononis natrix was evaluated for its antimicrobial activity against 9 human pathogenic microorganisms. This collection included five susceptible bacteria (Escherichia coli ATCC 25922, Klebsiella pneumoniae ATCC 700603, Pseudomonas aeruginosa ATCC 27653, Enterococcus faecium ATCC 4602, Staphylococcus aureus ATCC 25923), Two multi-resistant bacteria (methicillin-resistant Staphylococcus aureus NCTCC 12493, vancomycin-resistant Enterococcus spp) and two yeasts (Candida albicans BU 7098 and Candida tropicalis BU 1430).
Agar Diffusion Method
The antimicrobial activity of Ononis natrix essential oil was assessed using the agar disc diffusion method, following the guidelines set by the Clinical and Laboratory Standards Institute (CLSI) M07-A10. 15
A microbial suspension inoculated with one or two colonies isolated from an 18-24 h culture was adjusted to approximately 106 CFU/mL for bacteria and 1-2 × 103 cells/mL for yeast. The suspension was then spread evenly onto Mueller-Hinton (MH) agar plates for bacteria or Sabouraud agar plates for yeast. The plates were dried in a laminar flow hood for 15 min, then 6 mm diameter discs were soaked with 10 μL of pure essential oil (10 μL/disc). The disks were deposited on the surface of the agar previously seeded with the test microorganisms.
In each Petri dish, discs soaked with essential oil were placed alongside discs impregnated with positive controls (ciprofloxacin 5 μg/disc, gentamicin 15 μg/disc for bacteria and fluconazole 40 μg/disc for yeast). The dishes were placed at 4 °C for 4 h to allow diffusion of any bioactive molecules that might be produced, while stopping the growth of the test microorganisms. Finally, the plates were incubated at 37 °C for 18 to 24 h for bacteria and at 28 °C for 48 h for yeasts. After incubation, the absence of microbial growth is reflected by a zone of inhibition around the disks, whose diameter was measured in mm with a graduated ruler applied in contact with the agar surface. Each test was repeated three times in two replicates. 15
Microdilution Method
Quantitative evaluation EO antimicrobial activity was performed through the determination of Minimum Bactericidal Concentration (MBC) and Minimum Inhibitory Concentration (MIC) or Fungicidal Concentration (MFC) on liquid medium, by the standard liquid microdilution method using 96-well plates.
Determination of Minimum Inhibitory Concentration (MIC)
A series of ½ dilutions of essential oils were prepared at different concentrations in dimethyl sulfoxide (2% DMSO). In each microplate, the essential oil dilutions were distributed at 100 μL/well. Subsequent, 100 μL of suitable freshly prepared new liquid culture medium (MH broth for bacteria and Sabouraud broth for yeast) previously seeded with 50 μL of the microbial suspension were added to the wells. The mixture was then thoroughly mixed and the microplates incubated at 37 °C (for 18-24 h for bacteria) and at 28 °C (for 48 h for yeast). Positive controls (ciprofloxacin, gentamycin and fluconazole) were also tested. The Minimum Inhibitory Concentration (MIC) was defined as the lowest concentration of the essential oil that inhibits the visible growth of the strain tested. 16
Determination of Minimum Fungicidal Concentration (MFC) and Minimum Bactericidal Concentration (MBC)
Wells showing no visible growth after incubation were inoculated onto Petri dishes containing nutrient agar for bacteria or Sabouraud agar for yeasts. Readings were taken after incubation under the same conditions as above. The minimum bactericidal concentration (MBC) or fungicidal concentration (MFC) was defined as the lowest concentration of the sample tested for which there was a total absence of colonies after incubation. 16
Data Availability
The data underlying this article are available in the article and in its online supplementary material.
Statistical Analysis
All antioxidant activity assays and antimicrobial activity tests were performed in triplicate (n = 3). In contrast, the GC-MS analysis of the essential oil was each conducted once. The data were statistically analysed using SPSS Statistics software (version 24.0 for Windows, SPSS Inc.). Differences between means were assessed using the least significant difference test at a significance level of P < .05. The results are expressed as the mean value ± standard deviation.
Results and Discussions
Chemical Composition
The yield of the extracted EO was noted as 0.3%. The results of the identification of the constituents of O. natrix essential oil are presented in Table 1. A total of 18 compounds were identified by GC-MS (Figure 1), representing 99.3% of all constituents present in the oil. This EO is mainly composed of oxygenated monoterpenes (45.6%), oxygenated sesquiterpenes (35.35%), hydrocarbon monoterpenes (13.27%) and hydrocarbon sesquiterpenes (5.08%). The main compound present is hexahydrofarnesyl acetone (31.24%). In addition, other compounds are also present in significant quantities, including (+)-2-Bornanone (14.82%), endo-Borneol (6.91%) and carvone (6.15%).

Chromatographic profile of Ononis natrix essential oil.
Chemical Composition of the Essential oil Isolated from Ononis natrix Aerial Parts.

RT: Retention times on DB-5 column; RI exp: Retention index relative to C8-C25 n-alkanes on DB-5 column; RI lit: Retention indices reported in the literature taken from NIST.
The EO yields obtained in our study are higher than those reported in other research. For instance, a study of Ononis natrix EO in Jordan reported a yield of 0.04%. 8 However, the yield obtained in our study remain lower than those of O. natrix harvested in Guelmim, Morocco. 7
Our study corroborates the results obtained by, 7 who reported oxygenated monoterpenes as the majority compounds in the EO of Ononis natrix collected in Guelmim, with farnesyl acetone as the main compound (27.3%). However, the chemical profile of O. natrix EO in our study differs from that found for the same species collected in Jordan by. 9 In this latter study, the composition was dominated by hydrocarbon sesquiterpenes, with α-copaene and germacrene D as the main constituents. Nevertheless, common constituents are observed between the two samples, notably α-pinene, α-terpinene and γ-cadinene, although their respective contents vary considerably. Furthermore, our results show significant differences from those reported by. 8 This study on O. natrix in Jordan identified a preponderance of oxygenated sesquiterpenes (66.29%) and sesquiterpene hydrocarbons (20.10%), with (2E,6E)-farnesol (18.83%) as the major constituent.
It is worth mentioning that some factors might affect the essential oil's chemical composition within the same species which include plant material, environment, soil, harvesting period, and genetic factors as well as preservation techniques, and extraction methods. 17 Consequently, these variability factors play a decisive role in shaping the chemical profile of essential oils, and, by extension, their therapeutic properties.
Antioxidant Activity
The antioxidant activity of Ononis natrix essential oil (EO) was evaluated using DPPH radical scavenging and FRAP (ferric reducing antioxidant power) assays. The IC50 values, summarized in Table 2, indicate that the EO showed stronger antioxidant potential in the FRAP assay (IC50 = 339 ± 0.15 μg/mL) than in the DPPH assay (IC50 = 597 ± 0.25 μg/mL). In both assays, the EO exhibited significantly lower antioxidant activity compared to the reference compound quercetin (P < .001), as confirmed by one-way ANOVA followed by Tukey's post hoc test. Different letters (a for quercetin, b for EO) indicate statistically distinct groups. While quercetin remains a more potent antioxidant, this comparison does not exclude the notable antioxidant capacity of O. natrix essential oil.
Antioxidant Activities of Ononis natrix EO Expressed as IC50 Values (μg/mL).
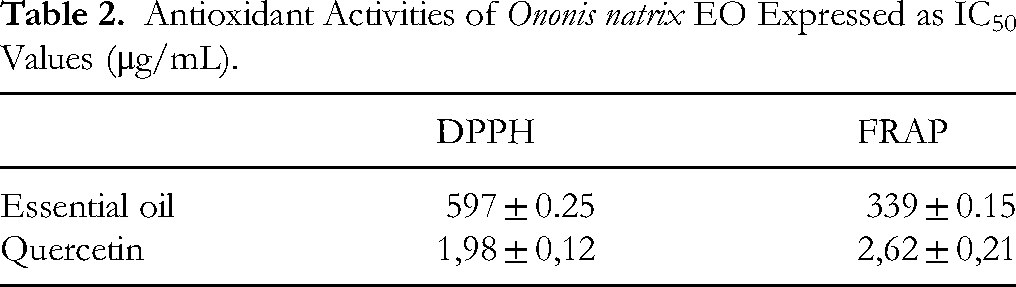
Essential oils are complex mixtures of volatile compounds, each contributing, alone or in synergy, to the biological activities observed. It has now been established that the antioxidant properties of essential oils do not depend solely on the majority compounds, but can also be influenced by the presence of minor compounds with complementary or synergistic effects.
In Ononis natrix essential oil, the antioxidant activity observed could be attributed to the notable presence of hexahydrofarnesyl acetone and (+)-2-Bornanone, two compounds for which previous studies have reported marked antioxidant effects.18,19 In addition, minority constituents such as thymol and carvacrol, well known for their antioxidant power, could also enhance this activity through synergistic mechanisms.20,21
Our research confirmed DPPH free radical scavenging activity, in agreement with the results previously reported by Al-Qudah et al 8 However, it should be emphasised that our study is, to our knowledge, the first to investigate the ability of O. natrix essential oil to chelate ferric ions, which broadens our understanding of its antioxidant potential.
In comparative terms, other species in the Ononis genus have also shown antioxidant capacities, albeit to varying degrees. For example, the essential oil of Ononis angustissima subsp. filifolia showed moderate activity against DPPH radicals (IC50 = 102.30 µg/mL) and strong inhibition in the β-carotene test (91.35%). 22 For its part, Ononis aurasiaca, a species endemic to Algeria, showed significant activity in the β-carotene assay (IC50 = 87.5 µg/mL), but less activity towards DPPH (IC50 = 231.87 µg/mL) and a low capacity to reduce ferric ions (IC50 = 751.3 µg/mL). 23
These comparisons highlight the variability in antioxidant potential between species of the Ononis genus, probably linked to differences in chemical composition, influenced by genetic, ecological and geographical factors. The results obtained with O. natrix therefore show a better overall antioxidant profile, particularly in terms of chelation of ferric ions, and merit further study, particularly in vivo, in order to better define their potential biological and therapeutic implications.
Antimicrobial Activity
Qualitative Assessment
The results presented in Table 3 show the mean values for the zones of inhibition of the essential oil and the antimicrobials tested on various strains of Candida and bacteria, both antibiotic-sensitive and antibiotic-resistant.
Inhibition Zone Values of Ononis natrix EO and Antimicrobials Tested on Candida Strains, Antibiotic-Sensitive and Antibiotic-Resistant bacteria.

Values are expressed as mean ± standard deviation; NT: not tested; NI: no inhibition
These results show that the essential oil studied has a growth inhibiting effect on all the microorganisms tested. Notably, a particularly marked inhibition was observed on Candida strains, with inhibition diameters ranging from 25 ± 1.2 mm to 85 ± 2.45 mm. As for bacteria, zones of inhibition ranging from 11 ± 0.17 mm to 20 ± 0.7 mm were recorded for antibiotic-resistant strains, while for sensitive strains, these values were between 11 ± 0.35 mm and 25 ± 0.88 mm. It is worth noting that the Gram-negative E. coli strain proved to be the least sensitive to the essential oil studied, while the S. anteuphorbium strain showed the greatest zone of inhibition, reaching 25 ± 0.88 mm.
It is also remarkable to note that the diameters of inhibition observed with the essential oil tested are sometimes comparable to, or even greater than, those of standard antibiotics (ciprofloxacin, gentamicin and fluconazole). This finding is particularly notable in the case of S. aureus bacteria (Gram-positive), K. pneumoniae (Gram-negative) and Candida strains (C. tropicalis and C. albicans).
Quantitative Evaluation
The antibacterial activity of Ononis natrix EO was evaluated by measuring the minimum inhibitory concentration (MIC), minimum bactericidal concentration (MBC), and minimum fungicidal concentration (MFC) using the liquid microdilution technique. The findings, shown in Table 4, indicate that the EO antimicrobial effectiveness varied depending on the strain tested.
Values of Minimum Inhibitory Concentrations (MIC) and Minimum Microbicidal Concentrations (MMC) of Essential oil, Antibacterial Agents and Reference Antifungal Agent Tested in mg/mL.

Abbreviations: MBC, minimum bactericidal concentration; MFC, minimum fungicidal concentration.
MIC values showed that antibiotic-resistant bacteria were inhibited at a relatively low concentration of 3.12 mg/mL. In contrast, sensitive strains required higher concentrations, ranging from 1.56 to 6.24 mg/mL, to observe growth inhibition. The essential oil also demonstrated antifungal properties, with MIC values ranging from 1.56 mg/mL to 3.12 mg/mL. Notably, the Gram-negative P. aeruginosa strain proved to be the most resistant to the essential oil tested, with an MIC of 6.24 mg/mL.
For some strains, the MIC value corresponded to the MBC or MFC, indicating a microbicidal action of the essential oil, notably against E. coli, K. pneumoniae, S. aureus and C. tropicalis. Comparatively, the reference antibacterial and antifungal agents showed lower MIC values than the essential oil (Table 4).
Ononis natrix essential oil (EO) has demonstrated significant antimicrobial activity against a range of pathogenic strains, including Gram-positive and Gram-negative bacteria and Candida strains. Our study is the first, to our knowledge, to evaluate the effect of this essential oil against multi-resistant strains, including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus spp. (VRE) and Candida strains.
When compared to the previous study carried out on O. natrix from Morocco by, 7 which reported MICs of 7.81 mg/mL against E. coli ATCC 25922 and 15.60 mg/mL against S. aureus ATCC 25923, our results reveal a more marked antimicrobial activity, with MICs/MBCs of 1.56 mg/mL for these two strains. For P. aeruginosa ATCC 27853, the same study reported high resistance (MIC = 60.96 mg/mL), which is in line with our results: this strain was also the most resistant in our study, with an MIC of 6.24 mg/mL. This confirms the data in the literature highlighting the high intrinsic resistance of this bacterium to essential oils.24,25
Other species of the Ononis genus have also been studied recently. The essential oil of Ononis angustissima subsp. filifolia has shown efficacy against Gram-positive strains such as S. aureus (ATCC 25923 and 43300), but limited efficacy against Gram-negative strains. 22 Likewise, Ononis aurasiaca, a species endemic to Algeria, showed marked activity against S. aureus (ATCC 25923 and 43300), but little effect on E. coli or P. aeruginosa. 23 These comparisons suggest that, although antimicrobial activity seems more marked against Gram-positive bacteria in the Ononis genus, O. natrix essential oil stands out for its more balanced efficacy against both types of bacteria, as well as for its antifungal activity.
The variations in efficacy observed between sensitive and resistant strains could be linked to the structure of the bacterial wall (more complex in Gram-negatives) and the ability of certain bacteria to express defence mechanisms against lipophilic agents, such as efflux pumps or degrading enzymes. 26
The antimicrobial activity of O. natrix EO can be attributed to its wealth of bioactive compounds such as oxygenated monoterpenes (thymol, carvacrol, (+)-2-bornanone), oxygenated sesquiterpenes (hexahydrofarnesyl acetone, 1,10-di-epi-cubenol) and sesquiterpene hydrocarbons (caryophyllene, γ-cadinene). These molecules are known to alter the integrity of cell membranes, disrupt permeability, inhibit bacterial enzyme systems, and affect the biosynthesis of essential compounds such as ergosterol in fungi.27–30 Compounds such as camphene and D-limonene, also detected in our sample, have shown significant antimicrobial and antifungal properties in other studies. 31
Despite these promising results, our study has several limitations. It is based exclusively on in vitro tests, and the absence of in vivo tests limits the transposability of the results to clinical contexts. In addition, the exact mechanism of the essential oil's antimicrobial action remains to be elucidated, particularly in terms of the specific interactions between the compounds and the microbial targets. No tests have been carried out to assess a possible synergy between the essential oil and traditional antibiotics, although this is a promising avenue of research in view of the increase in microbial resistance. Future studies could therefore explore the combination of O. natrix EO with conventional antibiotics in order to determine potential synergistic effects and reduce effective concentrations.
Conclusion
Our study highlighted the antioxidant and antimicrobial properties of Ononis natrix essential oil. Antioxidant activity, illustrated by its ability to scavenge free radicals and chelate ferric ions, suggests a potential for protection against oxidative stress. Furthermore, the antimicrobial activity observed, notably against resistant pathogenic strains such as methicillin-resistant Staphylococcus aureus and vancomycin-resistant Enterococcus spp. (VRE), testifies to potential therapeutic interest. The presence of bioactive compounds such as oxygenated monoterpenes and sesquiterpenes reinforces these observations.
However, it should be emphasized that these results are preliminary and based exclusively on in vitro tests. In vivo studies are essential to confirm these biological effects, particularly with regard to safety, bioavailability and therapeutic efficacy. Consequently, further research, including tests on animal models and clinical studies, is required before proposing medical or industrial applications for this essential oil.
Footnotes
Abbreviations
Acknowledgements
The authors would like to thank Mrs. Arifi Rabia and Mrs. El Khiat Kbira for collecting the plant material. They also express their gratitude to the Analysis and Characterization Centre of Cadi Ayyad University for conducting the essential oil analyses.
Ethical Approval
This study does not require ethical approval since it does not involve human participants, animal subjects, or any other elements requiring ethical approval. All procedures and analyses carried out complies with applicable guidelines and requirements for research integrity.
Author Contributions
Conceptualization, A.E.K., H.G.; methodology, A.E.K., S.O. and I.D.; software, Y.B. and A.A.; validation, A.E.K. and H.G.; formal analysis, S.O., I.D. and M.A.T.; investigation, S.O. and I.D.; data curation, S.O., I.D.; writing—original draft preparation, A.E.K., S.O.; and M.A.T.; writing—review and editing, A.E.K., S.O. and B.E.M .; supervision, A.E.K. and H.G. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.


