Abstract
The aim of this work was to study the chemical composition and antioxidant activity of essential oils of Thymus ciliatus (Desf.) Benth from middle Algeria obtained from the aerial parts by hydrodistillation using a Clevenger-type apparatus. The oil was analyzed by gas chromatography (GC) and gas chromatography coupled with mass spectrometry (GC-MS). The antioxidant capacity values of T ciliatus extracts were determined using 2 different and complementary assays: free radical scavenging (2,2-diphenyl-1-picrylhydrazyl [DPPH]) and ferric reducing antioxidant power (FRAP) assays. Six samples of T ciliatus collected in 2019 were used in the study. GC and GC-MS were used to determine the chemical composition of the essential oils. Twenty-five compounds were identified with a percentage of 89.3%–97.7%. The essential oils of T ciliatus were characterized by the presence of myrcene (9.6%-26.9%), p-cymene (7.6%-17.0%), and borneol (13.4%-30.2%) as principal components.
Introduction
Essential oils from plants have found their place in aromatherapy, pharmacy, perfumery, cosmetics, and food preservation. Their use is linked to their broad spectra of recognized biological activities.1–4 Thyme essential oils are widely used as antiseptic agents in several pharmaceutical fields and as flavoring agents for many types of food products. 5 Thymus ciliatus (Desf.) Benth of the family Lamiaceae is a small shrub but can form well spread tufts on the ground. The floral leaves are different from the stem leaves, in general strongly dilated at their lower portion. The plants are found in scrub, swamps, on calcareous and siliceous substrates, and on rocky and well-drained soils; the plants are locally called Djeirtel.6,7 The objective of this study was to track the change in the percentages of the main volatile components extracted from the aerial parts of T ciliates due to the season of harvest and the corresponding interchangeability in the antioxidant activities due to the chemical content variation.
Materials and Methods
Plant Materials
Six samples of the aerial parts of wild T ciliatus (wild plants) were collected in the spring of 2019 in the Djelfa region of Algeria with the following geographical coordinates: Latitude: 34° 44’47,229 “N, Longitude: 3° 18 19, 841 inches east, altitude: 1191 m. The third (20th April) and fourth (1st May) samples were collected at the time of flowering. All collected samples were kept in paper bags from the moment of cutting. Samples were dried in a dark laboratory herbarium at air temperature. A voucher sample (T ciliatus – Dj/19-n) was deposited in the research laboratory herbarium and conserved for use in the future.
Chemicals and Reagents
Absolute ethanol (99.8%), absolute methanol (≥99.7%), sodium carbonate, Folin–Ciocalteau reagent, gallic acid, quercetin, ferrous chloride, ascorbic acid, 2,2-diphenyl-1-picrylhydrazyl (DPPH), α-tocopherol, and 24,6-tri [2-pyridyl]-s-triazine (TPTZ) were procured from Sigma-Aldrich. All the chemicals used were of an analytical grade.
Methods
Preparation of Extracts
Air-dried aerial parts of the plants (100 g) were subjected to hydrodistillation using a Clevenger-type apparatus until there was no significant increase in the volume of oil collected (4 h). The essential oil obtained was separated from the water, treated with anhydrous sodium sulfate to remove all water traces, and stored at 4°C in a sealed vial until required.
Essential Oil Analysis
GC
Gas chromatography (GC) analyses were carried out using a Chrompack type CP 9002, equipped with a FID (set at 280°C) and a fused silica capillary column of type DB-5 (30 m × 0.32 mm, film thickness: 0.25 μm). The oven temperature was programmed from 50°C to 250°C at a rate of 2°C/min and then the temperature was kept at 250°C for 10 min. It was supplied with an H2/air, gas mixture and a split injector set at 250°C. The carrier gas was nitrogen with a flow rate of 1 mL.min−1. The injection mode was split (with leakage rate of 1/50 and a leak ratio of 66 mL.min−1).
GC/MS
The isolated volatile compounds were analyzed by gas chromatography–mass spectrometry (GC/MS) using a Shimadzu GC/MS-QP2010 ultra fused DB-5 capillary column (the same program as that used in the GC/FID analysis). The injection port temperature was 250°C and the detector was at 280°C (split ratio: 1/100). The carrier gas was helium (99.995% purity) with a flow rate of 1.2 mL.min−1. The MS conditions were ionization voltage 70 eV, ion source temperature 150°C; electron ionization mass spectra were acquired over the mass range 50 to 550 m/z.
Identification of Components
Linear retention indices (LRIs) were calculated for the separate compounds relative to C9 to C25 n-alkanes. Constituents were identified by comparison of their calculated LRI with those in the literature, 8 and by comparison of their MS with those recorded in the NIST library “NIST14” and “WILEY 229.” Standard pure compounds were also used for identification.
Measurement of in Vitro Antioxidant Capacity
DPPH Assay
The free radical scavenging activity was measured in terms of radical scavenging ability (hydrogen donating) using the stable radical DPPH. This was determined according to the technique employed earlier by Molyneux
9
with some modifications. Three hundred μL of different test compounds (or extracts) at different concentrations was added to 700 μL of 100 μM ethanol solution of DPPH. The mixture was kept in the dark for 30 min. Absorbance values of these solutions were recorded on an ultraviolet and visible (Kyoto JAPAN, Model UV-1800) spectrophotometer at 517 nm. Inhibition of DPPH-free radicals in percent (I %) was calculated as follows:
Asample is the absorbance value of the tested extracts.
Ascorbic acid and α-tocopherol were used as positive controls. All tests were carried out in triplicate.
FRAP Assay
The iron reduction procedure (ferric reducing antioxidant power [FRAP] assay) used was as reported by Benzie and Strain
10
with some modifications. The FRAP working reagent was prepared by mixing the 3 following solutions with respective ratios of 10:1:1 (v:v:v): acetate buffer solution 300 mM (pH = 3.6), (3.1 g of sodium acetate trihydrate was added to 16 mL glacial acetic acid and made up to 1 L with distilled water), plus 10 mM TPTZ in 40 mM HCl solution and 20 mM Fe2Cl3.6H2O. Fifty μL of several different prepared dilutions of extracts in absolute ethanol was mixed with 1 mL of FRAP reagent. After 7 min, the absorbance value of the reaction solution was read at 593 nm on a UV-Vis spectrophotometer (Kyoto JAPAN, Model UV-1800). The inhibition percentage
Asample is the absorbance value of the tested extracts.
All the tests were performed in triplicate.
Statistical Analyses
Statistical analysis was carried out using IBM SPSS Statistics 22. All the tests were carried out in triplicate.
Results and Discussion
Essential oil Yield
The essential oil of T ciliatus was a transparent yellow color with a strong odor. The yield of oil, obtained by hydrodistillation of the aerial parts, ranged between 2.2% and 2.4% (Table 1). The yield was similar for all samples with a slight difference between flowering time (2.4%), before flowering (2.3%), and after flowering (2.3%). There are many studies on the essential oil of T ciliatus from several regions, including the Tlemcen region of western Algeria, for which the yield ranged between 3.0% and 5.1%, while it reached 2.1% for the Batna region in Algeria, 1.2% in the Azrou region in the Middle Atlas in Morocco, and 0.46% for the M'sila region in Algeria. In the Annaba region of Algeria, the plant was collected at different stages of growth, with the yield ranging from 0.54% to 1.2%, with a maximum obtained during the post-flowering phase. This difference is due to the time of harvest, which has a direct effect on the quantity and composition of the oil obtained.11–14 In fact, the yield of essential oil increases more in the flowering stage,15,16 and this is what was achieved in our study.
Chemical Composition of Thymus ciliatus Essential oil Collected in 2019.

Abbreviations: LRI : Linear retention index; Tr: Retention time; tr: trace (< .01%).
Chemical Composition of the Essential oil
The characterization of the essential oils of 6 samples by GC-FID and GC/MS analyses resulted in the identification of 25 volatile constituents, accounting for 89.3% to 97.7% of the total essential oil composition (Table 1). In previous studies, 29 compounds were identified with a ratio of 96.6% to 98.2%, 17 18 components representing 99.6%, 18 15 components representing 97.41%, 19 24 components representing 99.87%, 20 75 components representing 95.57%, 21 and 50, 54, and 55 components, representing 98.6%, 99.5%, and 99.3% of the total essential oil in the vegetative, flowering, and post-flowering stages of growth, respectively. 22 The difference in the detection of the No. of compounds and the total percentage of the selected compounds can be attributed to the detection technique, picking area, and time, as climatic conditions have a major impact on the chemical composition of essential oils in terms of quality and quantity. 23
The predominant classes of compound were oxygenated monoterpenes (31.5-57.1%) and monoterpene hydrocarbons (30.1-61.4%), while sesquiterpenes were found in low percentages (0.9-2.8%). The oxygenated monoterpenes are represented mainly by borneol, α-terpineol, and linalool, while the monoterpene hydrocarbons are represented mainly by p-cymene, γ-terpinene, β−pinene, myrcene, and β-ocimene. Monoterpene hydrocarbons recorded their highest value (61.4%) during the flowering stage, while oxygenated monoterpenes and sesquiterpenes showed their highest values before the flowering period (50.7%, 2.6%, respectively) (Table 1).
The 3 dominant compounds were myrcene (9.6%-26.9%), p-cymene (7.6%-17.0%), and borneol (13.4%-30.2%), which differed significantly from previous studies in different regions. For example, in the Tlemcen region of western Algeria, the predominant compounds were carvacrol (72.4%-80.3%) and p-cymene (4.2%-7.2%), while in the Batna region of Algeria, the predominant compounds were thymol (79.1%) and p-cymene (5.6%). In the Azrou region in the Middle Atlas in Morocco, the major compounds were thymol (44.2%), β-E-ocimène (25.8%), and α-terpinène (12.3%). In the region of Tawra (northeast Algeria), the main components were thymol (67.78%), p-cymene (12.25%), pseudo-limonene (5.10%), and γ-terpinène (4.42%), while in the Annaba region of Algeria, thymol (33.39%) and carvacrol (31.37%) were predominant.
For myrcene, an increase was observed in May compared to April, while the percentage of p-cymene decreased in April, but its percentage was almost fixed in May. For borneol, the largest percentage was recorded in April and decreased in May. As shown in Figure 1, the highest percentage of myrcene was recorded in the sixth sample, with a percentage of 26.9%. The highest percentage of p-cymene (17.0%) was in the first picking (April 1st), which also had the highest percentage of borneol (30.2%).

Change in the percentages of different major components of the essential oil of Thymus ciliatus (Desf.) in line with the samples of collection.
Our results show major differences from previous studies. In our oils, thymol did not exceed 0.4%, and we did not record the presence of carvacrol, but these two compounds were found in relatively high proportions in previous studies; these differences may be due to the region of plant growth, climate, nature of the land, and time of harvesting.
Ten secondary compounds present in significant amounts were α-pinene(0.7%-3.0%), sabinene(0.5%-10.1%), β−pinene(2.6%-6.1%), β-ocimene(1.7%-4.4%), γ-terpinène(1.3%-6.8%), linalool(1.9%-17.3%), α-terpineol(3.0%-7.7%), β-bourbonene(1.8%- 7.3%), geranyl acetate(2.0%-8.0%), and β-caryophyllene (1.0%-2.8%); average values were recorded as: 1.7%, 3.0%, 4.3%, 3.0%, 2.8%, 3.8%, 4.9%, 3.9%, 4.2%, and 2.2%, respectively (Table 1). Accordingly, the variance of each of the major and minor components has a close relationship with environmental and climatic factors represented by temporal and spatial variance, and, therefore, does not allow for definitive results. 24
The second sample gave the highest percentage of α-pinene (3.0%), while we recorded the lowest percentage in the fifth sample (0.7%). Sabinene had a significant percentage in the second (5.0%) and fourth (10.1%) samples, while it was almost nonexistent in the rest of the samples. The percentage of β-pinene increased in May (3.8%) compared to April (2.2%). The highest percentage of β-ocimene was estimated at 4.4% in the fifth sample. Sample 1 recorded the highest percentage of γ-terpinene (6.8%) compared to the rest of the samples, and the second and fourth samples recorded large percentages of linalool (7.3%, 6.1%, respectively) compared to the other four samples. The third sample agreed with a higher percentage of α-terpineol (7.7%) and β-bourbonene (7.3%). The fifth sample contained the highest percentage of geranyl acetate (3.0%), while the percentages of β-caryophyllene in the samples are close, but almost nonexistent in the fourth sample (Figure 2).
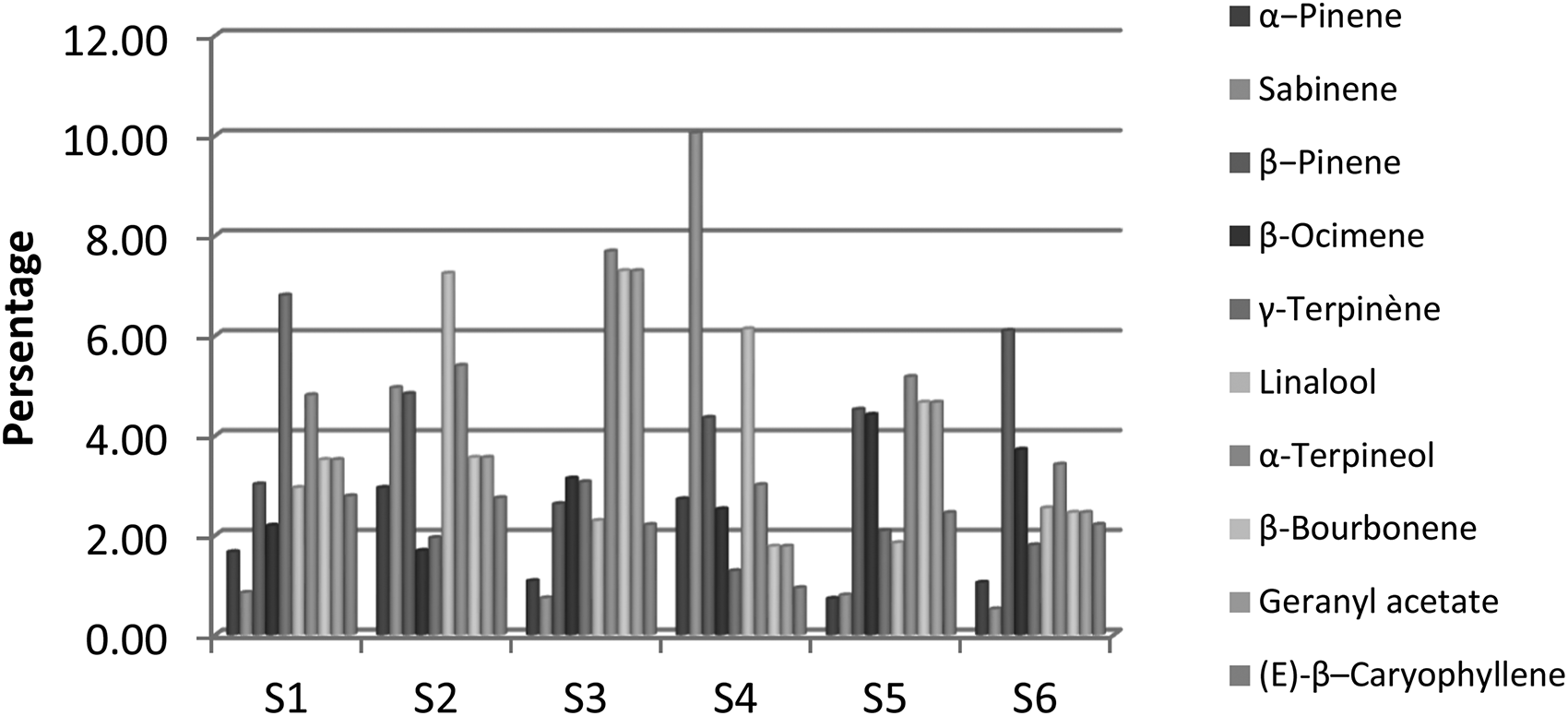
Change in the percentages of different minor components of the essential oil of Thymus ciliatus (Desf.) in line with the samples of collection.
Accordingly, the importance of the essential oil of T ciliatus lies in the dominant compounds (borneol > myrcene >p-cymene). Borneol has been widely used in traditional Chinese medicine as an adjuvant drug facilitating the transport of many drugs to specific sites and coordinating the effects of those drugs.23,25 Myrcene is an intermediate used in the perfumery industry, and p-cymene exhibits a variety of biological activities that include antioxidant, antidepressant, anti-inflammatory, anticancer, and antimicrobial activities. It is used to prevent coughing and is also used in the production of fungicides and insecticides. 26
Antioxidant Activity
Table 2 shows the antioxidant activity of the EOs during plant growth. For the DPPH assay, the IC50 values varied from 3.00 to 4.31 mg/mL, while for the FRAP assay, the range was 3.87 to 4.55 mg/mL; the best value was recorded on the 20th April for the third sample for both assays; this is the time of the beginning of flowering. As shown in Table 2, DPPH and FRAP assays showed the importance of T. ciliatus essential oil in the antioxidant activity during the flowering stage (IC50 [DPPH] = 3.12mg/mL and IC50 [FRAP] = 3.88mg/mL). However, the antioxidant activity of the samples was lower than that of vitamins C and E. However, the essential oil obtained in a previous study showed the best antioxidant activity in the DPPH assay after flowering (438 ± 9.07 μg/mL) compared with the vegetative (580 ± 1.15 μg/mL) and flowering stages (590 ± 8.62 μg/mL); the same applied for the FRAP assay.
Antioxidant Activity of Thymus Ciliates Essential oil Collected in 2019.
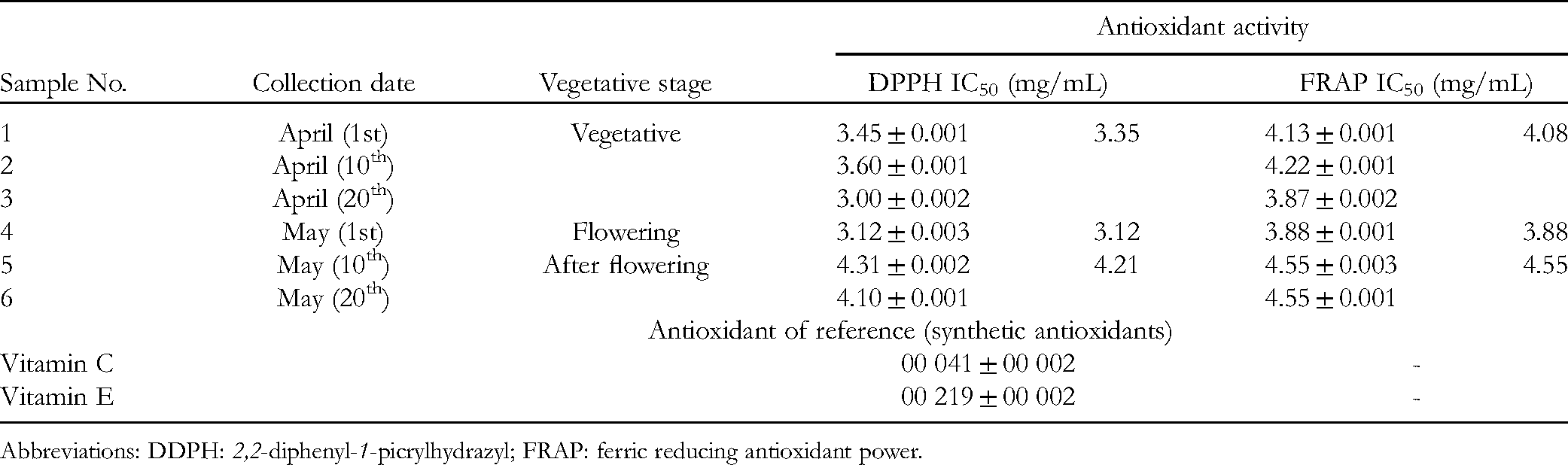
Abbreviations: DDPH: 2,2-diphenyl-1-picrylhydrazyl; FRAP: ferric reducing antioxidant power.
The antioxidant activity can be attributed to its three main components (eg myrcene, p-cymene, and borneol), as shown by previous studies, in which myrcene had significant antioxidant activity, with an IC50 of 4.5 µg/mL . 27 p-Cymene has an antioxidant potential and may constitute a new strategy for the development of treatments for various diseases in which oxidative stress plays an inherent part in their physiology. 28 Borneol showed only moderate antioxidant activity compared to synthetic antioxidants in the antioxidant activity assays. 29
Statistical Analyses
We obtained the results as shown in Table 3 using ANOVA statistical analysis for the following groups: oil yield, percentage of the No. of detected substances, and percentage of the No. of monoterpene hydrocarbons, oxygenated monoterpenes, and sesquiterpenes. Since the calculated F value is greater than the tabulated F value (192.1 > 2.8) at the .01 significance level, and the P value is almost 0 (2.2 * 10−18), we can say that the means of the 5 groups are unequal and there are significant differences between them, and, in this case, we resorted to dimensional comparison tests, such as the Tukey’s test. We calculated the true significant difference (HSD) by Tukey’s method and found: 8.0. Accordingly, it can be said that there are statistically significant differences between the approved binaries for differences greater than the value of the real difference (HSD) as follows:
- There is a big difference between the specific groups and the rest of the 4 groups. - There are significant differences between monoterpene hydrocarbons and sesquiterpenes. - There is a significant difference between the oxygenated monoterpenes and sesquiterpenes.
Results of one-way ANOVA Test for the Ratio Variable.
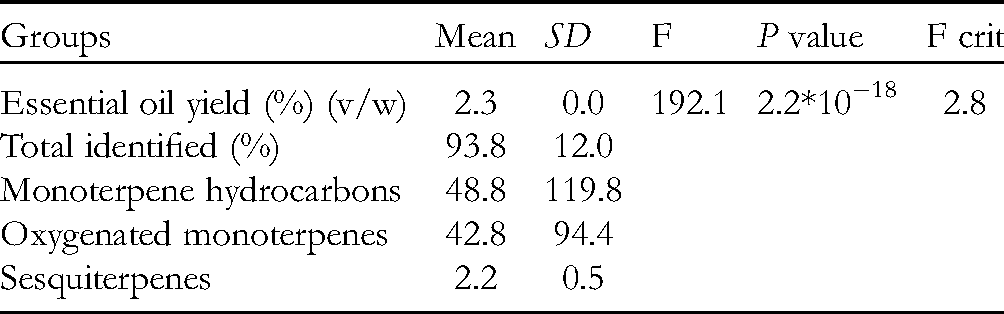
Table 4 shows the results of a one-way ANOVA test between antioxidant activity and major compounds. In the same way as the previous one, we found the following:
- There are significant differences between the activity of borneol and antioxidants (DPPH, FRAP). - There is a significant difference between the effectiveness of p-cymene and antioxidants (DPPH, FRAP). - There is a significant difference between the activity of myrcene and antioxidants (DPPH, FRAP).
Results of the one-way ANOVA Test Between Antioxidant Activity and Major Compounds Discovered.

Abbreviations: DDPH:2,2-diphenyl-1-picrylhydrazyl; FRAP: ferric reducing antioxidant power.
By relying on the Pearson correlation matrix, we obtained the results as shown in Table 5, where the relationship between the components of the essential oil and the effectiveness of the antioxidant activity can be summarized as follows:
- There is a strong distinct relationship between DPPH and the following compounds: β-pinene (0.7), myrcene (0.6), β-ocimene (0.6), eugenol (0.8), and geranyl acetate (0.6). - There is a strong distinct relationship between FRAP and the following compounds: β-pinene (0.7), myrcene (0.6), β-ocimene (0.6), eugenol (0.8), and geranyl acetate (0.5).
Results of the Strong Correlation Between Antioxidant Activity and Chemical Compounds According to Pearson Correlation Matrix.

Abbreviations: DDPH:2,2-diphenyl-1-picrylhydrazyl; FRAP: ferric reducing antioxidant power.
Conclusion
This study was concerned with the essential oil of several samples of T ciliatus in line with its growth to give a comprehensive study and try to know the best times to harvest it according to the efficiency of essential oil extraction, amount of major compounds, and best antioxidant activity. The essential oils consist of monoterpene hydrocarbons, oxygenated monoterpenes, and sesquiterpenes. The obtained essential oils are rich in monoterpene compounds (hydrocarbons and oxygenated), which are represented by 3 major compounds: myrcene, p-cymene, and borneol. These compounds were the major ones in all samples analyzed. Evaluating the extracts in terms of their ability to scavenge free radicals using DPPH and FRAP assays showed that T ciliatus is of great importance as a source of natural antioxidants.
Footnotes
Acknowledgments
The author would like to thank the researchers in the Physico-Chemistry of Materials and Environment Laboratory, Ziane Achour University of Djelfa, Algeria.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.


