Abstract
Introduction
The increasing resistance of microorganisms to antibiotics poses a significant threat to public health, prompting researchers to seek new natural products that can offer novel and effective antimicrobial strategies to their diverse chemical structures and interactions. 1 Plants and their products, including essential oils (EOs), are frequently used in traditional medicine and play a crucial role in plant protection. Essential oils are rich in a wide array of secondary metabolites that can inhibit or slow bacterial growth. 2 These oils and their constituents affect various cellular pathways, particularly targeting cytoplasm and the cell membrane, and in some cases, they can significantly alter gene expression and cell morphology. 3
Oxidative stress in another crucial issue, as il leads to numerous pathologies with harmful effects. The use of synthetic antioxidants, such as butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), and tertiary butylhydroquinone (TBHQ), poses considerable health risk and toxicity. 4
Oxidative stress is implicated in several chronic diseases, including cardiovascular diseases, neurodegenerative diseases, chronic kidney disease, diabetes and cancer. 5 Thus, there is an urgent need to identify new sources of antimicrobial and antioxidant compounds.
Plants represent one of the most promising avenues for discovering medicinal properties and therapeutic virtues. 6 The pharmacological properties of plant species are typically attributed to their bioactive compounds. 7 Medicinal plants are a primary source of a variety of bioactive compounds, 8 and many are used in microbial treatment9,10 and as antioxidants.11,12
Anacyclus is a genus belonging to the Asteraceae family known for its significant antimicrobial activity. Previous studies have reported the antimicrobial effects of A. pyrethrum,13,14 A. clavatus, 15 A. valentinus 16 and A. cyrtolepidioïdes. 17 Additionally, the genus has been largely studied for its antioxidant activity18,19,20 Despite the interest in Anacyclus species, the bioactivities of Anacyclus maroccanus Ball, an endemic plant of Moroccan flora, and Anacyclus radiatus Loisel have been scarcely investigated. Furthermore, The infusion of A. radiatus flower is traditionally used to alleviate stomach aches. 21 Recently, we demonstrated the antioxidant and hypoglycemic properties of methanolic and ethyl acetate extracts obtained from the flowered plants. 22 However, the antimicrobial properties of their essential oils have never been studied before.
The current study was carried out to assess the antioxidant activity of A. maroccanus and A. radiatus essential oils for the first time using two different tests: diphenyl-1-picrylhydrazyl (DPPH) tests and ferric reducing antioxidant power (FRAP). Moreover, the antimicrobial activity against 16 pathogen strains, including multidrug-resistant (MDR) strains, was conducted using the agar diffusion method, the Minimum Inhibitory Concentration (MIC), and the Minimum Bactericidal Concentration (MBC) methods. The components of the essential oils were determined by gas chromatography coupled with mass spectrometry (GC-MS) analysis to identify the component responsible for the activity.
Results and Discussion
Phytochemical Composition
The yield of the extracted EOs was noted as 0.13 and 0.16% for A. maroccanus and A. radiatus, respectively. The results of the GC-MS analysis of the volatile EOs constituents are summarized in Table 1 and Table 2. Mass spectrometry analysis of A. maroccanus EO revealed the presence of thirty compounds representing 93.73% of the total EO composition, with α-farnesene (9.83%), β-thujone (8.84%), neointermedeol (7.47%) Pentadecanal (6.27%) as the majority compounds (Table 1 and Figure 1). The analysis also revealed that the dominant class was oxygenated sesquiterpenes (25.67%), followed by oxygenated monoterpenes (24.18%), sesquiterpene hydrocarbons (19.201%), and oxygenated diterpenes (5.15%).
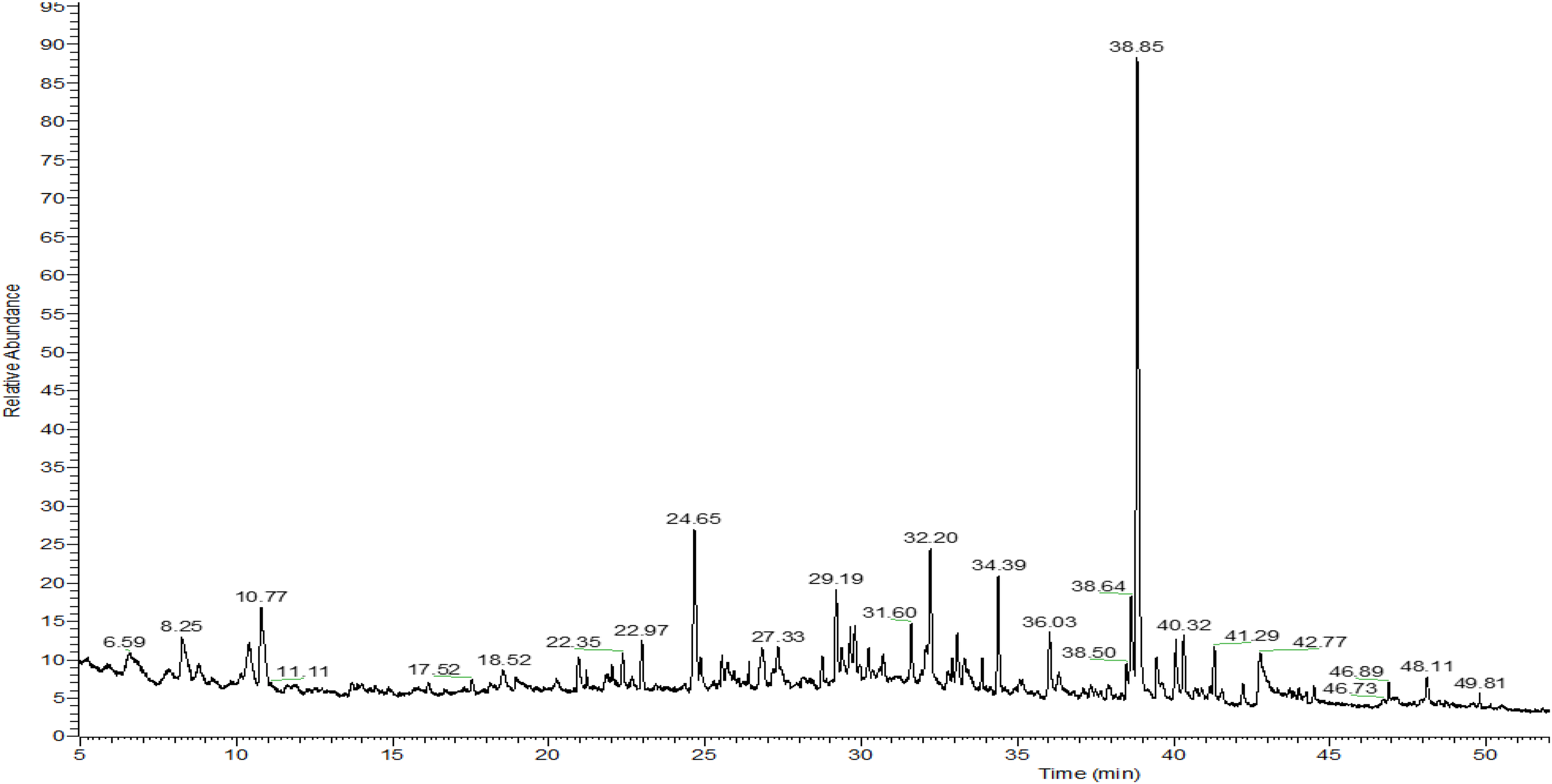
Chromatographic profiles of the A. maroccanus essential oil.
Chemical Composition of of A. maroccanus EO.

RT: Retention times on DB-5 column; RI exp: Retention index relative to C8-C25 n-alkanes on DB-5 column; RI lit: Retention indices reported in the literature taken from NIST. By Dr ROBERT P. ADAMS Professor Biology Department Baylor University ed. 4.1, 30 Jan. 2017.
Chemical Composition of A. radiatus EO.

RT: Retention times on DB-5 column; RI exp: Retention index relative to C8-C25 n-alkanes on DB-5 column; RI lit: Retention indices reported in the literature taken from NIST. By Dr ROBERT P. ADAMS Professor Biology Department Baylor University ed. 4.1, 30 Jan. 2017.
On the other hand, twenty-eight compounds were found in A. radiatus EO, representing 99.2% of the total composition. The main compounds identified were chrysanthenyl acetate (10.18%), salvial-4(14)-en-1-one (7.24%), caryophyllene oxide (6.77%), -copaene (6.56%), 4-methylamyl angelate (6.55%), and (-)-spathulenol (6.23%) (Table 2 and Figure 2). The major class in this EO was oxygenated sesquiterpenes (43.7%), followed by sesquiterpene hydrocarbons (26.85%), oxygenated monoterpenes (11.61%), monoterpene hydrocarbons (3.35%) and oxygenated diterpenes (1.97%).

Chromatographic profiles of the A. radiatus essential oil.
The yields of plant EOs in our study are higher than those reported in other studies. For instance, a study on Algerian A. clavatus EO reported a yield of 0.041%. 18 However, the yields obtained in our study are lower than those of A. pyrethrum harvested in Timehdit Morocco. 14 The chemical compositions of A. maroccanus and A. radiatus EOs exhibit significant differences. Notably, both EOs contain (-)-spathulenol, caryophyllene oxide and salvial-4(14)-en-1-one, albeit in varying percentages. The observed differences between species within the same genus can be attributed to several factors, including environmental conditions, geographical location, climate, soil characteristics, and exposure to soil. These factors can influence the activation or inactivation of some enzymatic groups, leading to a particular biosynthetic pathway. 23
Extensive investigations have been conducted on the essential oils of various Anacyclus species. However, the chemical characterisation of the EOs derived from A. maroccanus and A. radiatus has not been previously documented. Consequently, our results cannot be directly compared to existing data. Nonetheless, the chemical composition of our EOs is consisting with findings from numerous studies involving species within the same genus. For instance, spathulenol, germacra-trien-1-α-ol, and caryphyllene oxide were identified as the principal volatile components of A. pyrethrum EO harvested from Timehdit, Morocco. 14 According to Selles and his collaborators. oxygenated sesquiterpenes constitute the majority of A. pyrethrum EO. 24 Additionally, another study reported α-thujone (11.9%) as a major component of the essential oil extracted from A. clavatus inflorescences collected in Corso, Algeria. 18 These comparisons suggest a degree of chemical similarity among Anacyclus species while highlighting the unique profiles of A. maroccanus and A. radiatus EOs.
Antioxidant Activity
The reducing power and DPPH essays of A. maroccanus and A. radiatus EOs are represented in Table 3. Lower IC50 values indicate stronger antioxidant activity. In the DPPH test, the IC50 of A. maroccanus and A. radiatus EOs were 167.36 ± 6.2 µg.mL
Antioxidant Activities of A. maroccanus and A. radiatus EOs Expressed as IC50 Values ± SE (n = 3).
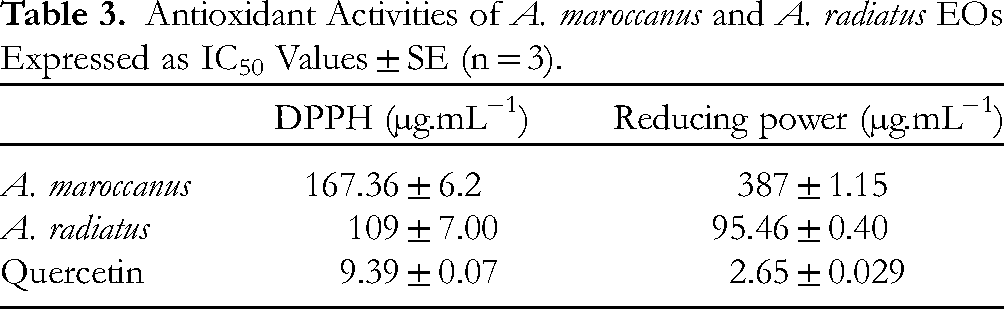
In this study, the antioxidant activity of EOs from the aerial parts of A. maroccanus and A. radiatus was evaluated using two complementary assays: DPPH free radical scavenging and ferricyanide of potassium reducing power, comparated with reference antioxidants quercetin. To the best of our knowledge, these results represent the first published data on the antioxidant activity of A. maroccanus and A. radiatus EOs. The antioxidant properties of A. pyrethrum EO from the Timahdite region of Morocco have been reported with an IC50 of 30.05 mg.mL-1 for the DPPH assay, indicating low radical scavenging activity when compared to the species in our study. 14
The high antioxidant properties of EOs can be attributed to a complex mixture of volatile compounds; which are related to various mechanisms, such as redox properties. EO compounds can act as reducing agents, hydrogen donors, and singlet oxygen quenchers. 14 The antioxidant activity of EOs may attributed to major or minor compounds, or to synergic and antagonistic interactions among these compounds that enhance their effect against free radicals.25,26 Numerous studies have established that the antioxidant activity EOs is due to oxygenated terpenes27,28,29,30 as well as the phenolic content. 31 The strong antioxidant effectiveness of A. maroccanus and A. radiatus EOs can be primarily attributed to the high concentrations of oxygenated terpenes (56.8% and 57.28%, respectively) which are known for their radical scavenging proprieties. Species rich in these Oxygenated monoterpene compounds, such as; thujone, β-thujone, 32 carvacrol, 29 β-ionon, 33 dihydroactinolide, 34 chrysanthenyl acetate 35 and thymol 36 have demonstrated appreciable antioxidant activity. Furthermore, oxygenated sesquiterpenes are also known to have significant antioxidant potential, including (-)-spathulenol, 37 salvial-4(14)-en-1-one, 21 tau-cadinol, 38 cedrenol, 48 neointermedeol, 39 caryophyllene oxide, 40 β-elemol, 41 isoaromadendrene epoxide, 42 β-eudesmol, 43 ledene oxide-(II). 44
Antimicrobial Activity
The antimicrobial activity of A. maroccanus and A. radiatus essential oils was evaluated against 16 human pathogenic microorganisms, including nine multidrug-resistant bacteria (MDR), five sensitive bacteria and two yeasts.
The assessment of antibacterial properties was conducted using the minimal inhibitory concentrations (MICs), minimal bactericidal and fungicidal concentrations (MBCs and MFCs) assays (Table 4). The results demonstrated that A. maroccanus EO inhibited all tested bacteria at MICs ranging from 1.56 to 25 mg/mL, while A. radiatus EO showed MICs ranging from 0.19 to 12.5 mg/mL. Additionally, antifungal activity was observed with MICs ranging from 0.78 to 12.5 mg/mL for A. maroccanus and from 1.56 to 3.12 mg/mL for A. radiatus. The most potent activity was exhibited by A. radiatus essential oil against methicillin-resistant S. aureus (0.19 mg.mL
Minimum Inhibitory Concentrations (MIC) and Minimum Bactericidal Concentrations (MBC) Values (mg/ml) of A. maroccanus and A. radiatus Essential Oils Compared Gentamicin and Fluconazole Against Pathogenic Strains.
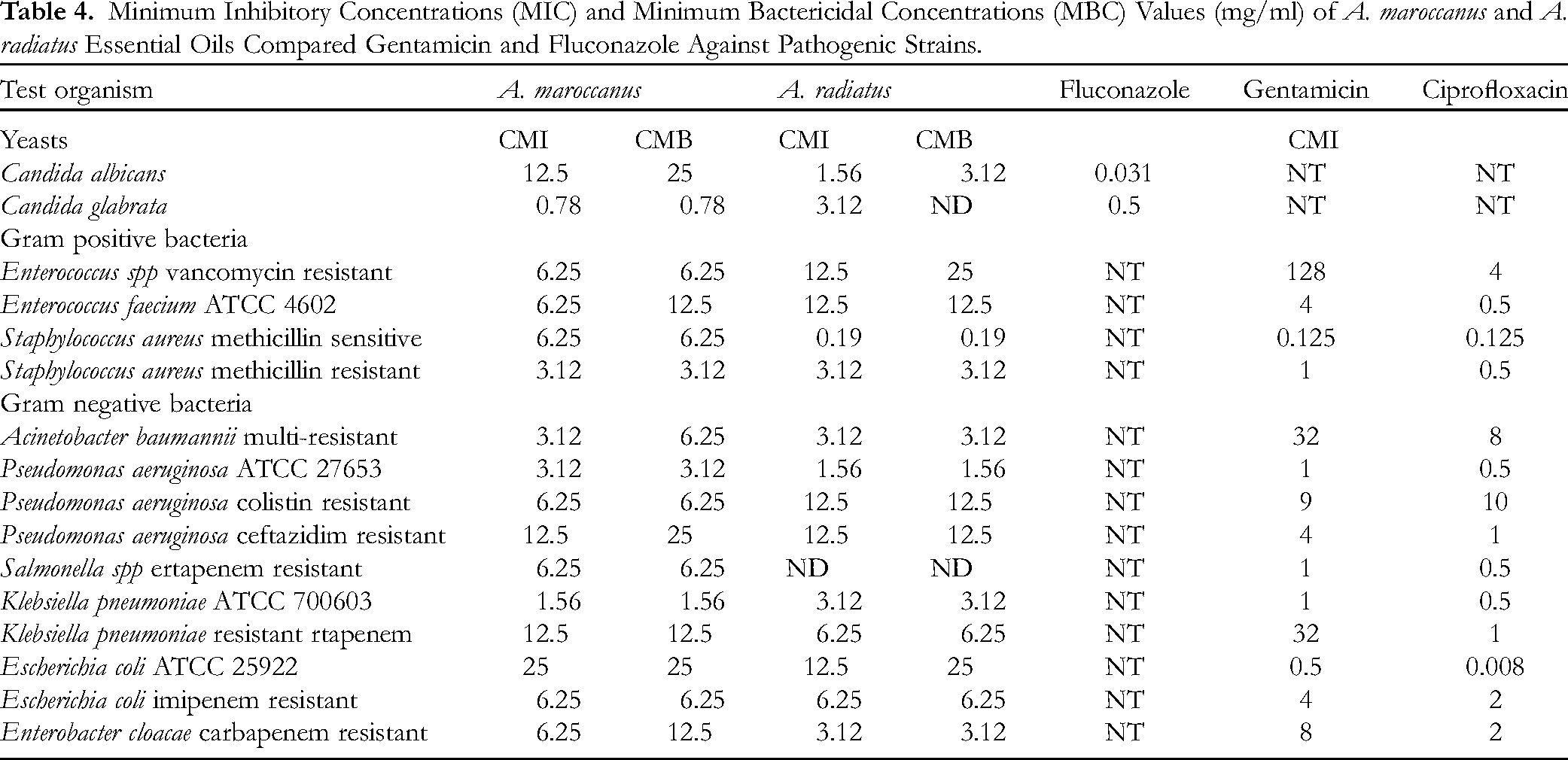
The essential oils exhibited significant antibacterial effects against both Gram-negative and Gram-positive strains to varying degrees. Notably, this study represents the inaugural investigation into the antibacterial proprieties of A. maroccanus and A. radiatus EOs against sensitive and Multidrug Resistance (MDR) pathogens. Consequently, our findings can be comprehensively contextualized and contrasted solely within the Anacyclus genus.
Limited research has delved into antimicrobial potentials of Anacyclus genus. For instance, a study on Tunisian A. clavatus highlighted its efficacy against S. aureus ATCC 27853 whit minimum inhibitory concetration (MIC) of 1.8 mg.mL
The antimicrobial potency observed in the EOs of A. maroccanus and A. radiatus can likely be attributed to their predominant constituents. Notably, A. maroccanus EO is characterized by notable level of α-farnesene (9.83%), β-thujone (8.84%), neointermedeol (7.47%) and (-)-spathulenol (5.06%), wile A. radiatus EO exhibits significant quantities of chrysanthenyl acetate (10.18%), salvial-4(14)-en-1-one (7.24%), caryophyllene oxide (6.77%), α-copaene (6.56%), 4-methylamyl angelate (6.55%) and (-)-spathulenol (6.23%). The intricate composition of these essential oils likely underlies their notable antimicrobial activity.
Various factor can contribute to the observed activities of the EOs, including synergistic interactions among major and minor compounds, the functional groups present in the active constitute, and the chemical nature of terpenes particularly oxygenated hydrocarbons. 45
Notably, both A. maroccanus and A. radiatus EOs are rich in oxygenated monoterpenes sesquiterpenes and diterpenes constituting approximatively 55% and 57.48% of their compositions, respectively. These compounds renowned for their profound antibacterial potential, further corroborating the observed antimicrobial effects. 46
Materials and Methods
Plant Material
In the spring of 2021, aerial parts of A. maroccanus and A. radiatus were harvested from Demnate in the High Atlas Mountains (Alt. 31°39’22’’ N and Lon. 7°25’28’’ W) and Essaouira on the Atlantic coast (Alt. 31°29’13’’ N and Lon. 9°45’44.8’’ W), respectively. The aerial parts of the plants, especially during the flowering stage, were used during this study. The identification was done by Professor Ouhammou Ahmed, and a voucher specimen was deposited at the Regional Herbarium “MARK” of the Faculty of Sciences Semlalia, University of Cadi Ayyad, Marrakech, Morocco, under the voucher specimen codes MARK-13830 and MARK-13829 for A. maroccanus and A. radiatus, respectively. The material was air-dried at room temperature and stored until use.
Essential oil Extraction
EOs from the air-dried aerial parts of each plant (3 × 150 g) were extracted by hydrodistillation using a Clevenger apparatus. Briefly, 150 g of dried plant material were placed in the Clevenger apparatus and hydrodistilled with 2000 ml of distilled water. Each extraction was carried out for five hours at boiling temperature. The essential oil was collected in the column of the Clevenger apparatus. The essential oils were stored in dark, dry glass bottles at 4 °C until use.
Phytochemical Composition
The qualitative and quantitative analysis of EOs was conducted using a gas chromatograph with a mass spectrometer detector, as previously described in the work of Ouhaddou et al 23 A Trace GC-MS system from Thermo ScientificTM (Trace 1300 GC, USA) was used. The detector and a nonpolar column TG-5MS (30 m × 0.25 mm. × 0.25 μm) were used in electron-impact ionization mode. The MS transfer line temperatures were set at 250 °C and 230 °C, respectively. The temperatures of the detector and the injector were set at 250 °C and 230 °C, respectively. The electron-impact ionization energy was70 eV. The run time was 67.59 min and the masse range wase between 40 m/z and 400 m/z. 1 μL of each sample was injected in splitless mode. The carrier gas used was Helium, with a flow rate of 1 Ml.min−1. The samples were pre-diluted in pure dichloromethane solvent at a 1:100 ratio. The oven temperature was programmed to increase at a rate of 3 °C per minute from an initial temperature of 60 °C to a final temperature of 230 °C, held for 10 min. 23 A calibration curve was used to identify the volatile compounds of A. maroccanus and A. radiatus EOs. The chemical components of EOs were identified by comparing their retention indices (RI) and mass fragmentation patterns with those on the stored NIST library (National Institute of Standards and Technology).
Finally, the chemical components were quantified by external standard method using calibration curves generated by running GC analysis of representative compounds.
Antioxidant Activity
The EOs extracted from A. maroccanus and A. radiatus were evaluated for their antioxidant activity using DPPH and ferric reducing antioxidant power (FRAP) assays,47,48 with quercetin used a positive control. In the DPPH assay, a 60 µM DPPH solution in methanol was prepared, and 2 mL of this solution was mixed with 50 µL of sample, previously dissolved in methanol at varying concentrations. The resulting mixture underwent agitation and was then incubated in darkness for 20 min. The absorbance of the samples was measured at 517 nm using a UV spectrophotometer. The DPPH % scavenging activity was calculated using the following equation:
The ferric-reducing power was determined using the ferric reducing antioxidant power method. Different concentrations of EOs were prepared in methanol. 1 mL of each concentration was mixed with 2.5 mL of the phosphate buffer solution (0.2 M, pH 6.6) and 2.5 mL of a 1% solution of potassium ferricyanide [K3Fe(CN)6], then the mixture was incubated at 50 °C for 20 min. After incubation, 2.5 mL of a trichloroacetic acid solution (10%) were added and 2.5 mL of the mixture was mixed with 2.5 mL of distilled water and 0.5 mL of iron chloride (1%). The absorbance of the final mixture was measured at 700 nm. The reducing power was expressed as an absorbance. The concentration of EOs with an absorbance of 0.5 is known as IC50.
The results of the DPPH and ferric reducing antioxidant power (FRAP) scavenging activities were expressed as IC50 values, representing the concentration of the antioxidant necessary to scavenge 50% of the DPPH or FRAP present in the test solution. The experiments were conducted independently at least three times. The IC50 values for both DPPH and FRAP assays were calculated through linear regression analysis using Microsoft Excel 2010 (Microsoft Corporation, Redmond, WA, USA) as the analytical tool.
Antimicrobial Assays
The EOs extracted from A. maroccanus and A. radiatus were evaluated for their antimicrobial activity against 16 human pathogenic microorganisms. This collection comprised nine multidrug-resistant bacteria (methicillin-resistant Staphylococcus aureus NCTCC 12493, colistin-resistant Pseudomonas aeruginosa P623, impenem-resistant Escherichia coli H1764, ceftazidim-resistant Pseudomonas aeruginosa BU2966, ertapenem-resistant Klebsiella pneumoniae H2121, vancomycin-resistant Enterococcus spp, multi-resistant Acinetobacter baumannii P1415, carbapenem-resistant Enterobacter cloacae P611, ertapenem-resistant Salmonella spp H18), five sensitive bacteria (Klebsiella pneumoniae ATCC 700603, Enterococcus faecium ATCC 4602, Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27653, methicillin-sensitive Staphylococcus aureus P1085), and two yeasts (Candida albicans BU 7098 and Candida glabrata BU7170.) The bacteria and fungi were cultured on Mueller Hinton gelose (MH) and Sabouraud Agar, respectively.
The MIC and MBC assays of A. maroccanus and A. radiatus essential oils were performed using the microdilution technique following CLSI guidelines.49,50
For this purpose, the EO was dissolved in 1% dimethylsulfoxide (DMSO) and subsequently diluted to the highest concentration. Serial doubling dilutions of the EO were then prepared in a 96-well microtiter plate. Overnight broth cultures of each microbial strain were prepared, and the final concentration in each well was adjusted to 106 CFU/mL for bacteria and 2 × 103 CFU/mL for yeasts. Subsequently, the bacteria and yeasts were incubated for 24 h at 37 °C and 28 °C, respectively.
Positive controls, including Gentamicin and Ciprofloxacin for bacteria, and Fluconazole for fungi, were prepared under the same experimental conditions. Additionally, a negative control using the vehicle (DMSO) was included. The MIC was defined as the lowest concentration showing no visible growth of the microorganism strains after 24 h of incubation.
Wells showing no visible growth after incubation were inoculated onto Petri dishes containing nutrient agar for bacteria or Sabouraud agar for yeasts. Readings were taken after incubation under the same conditions as above. The minimum bactericidal concentration (MBC) or fungicidal concentration (MFC) was defined as the lowest concentration of the sample tested for which there was a total absence of colonies after incubation.
Statistical Analysis
The data were statistically analyzed using SPSS Statistics software (version 24.0 for Windows, SPSS Inc.). Differences between means were assessed using the least significant difference test at a significance level of P < .05. The results are presented as the mean value ± standard deviation.
Conclusion
This article presents novel research focusing light on endemic species native to Morocco. Our findings highlight the significant antioxidant and antibacterial proprieties of essential oils extracted from the aerial parts of A. maroccanus and A. radiatus, demonstrating their efficacy against spectrum of microorganisms, including multidrug-resisting strains. Moreover, the chemical analysis reveals that these EO are abundant in bioactive compounds, particularly oxygenated monoterpenes and sesquiterpenes. Future investigations should be directed towards validating these results and assessing the safety and efficacy of A. radiatus and A. maroccanus EOs for potential therapeutic applications.
Footnotes
Acknowledgements
The authors express their gratitude to the Analysis and Characterization Centre of Cadi Ayyad University for conducting the essential oils analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
