Abstract
Hepatic fibrosis (HF), a critical intermediate stage in the progression of chronic liver diseases to cirrhosis and hepatocellular carcinoma (HCC), represents a growing global health burden due to its increasing morbidity and mortality. To date, no targeted anti-fibrotic therapies specifically for hepatic fibrosis have been clinically approved. Hepatic stellate cells (HSCs), the primary source of extracellular matrix (ECM) production, play a pivotal role in the pathogenesis of hepatic fibrosis and have thus become a central focus in both mechanistic and therapeutic research. Relevant research indicates that traditional Chinese medicine (TCM) demonstrates promising anti-fibrotic efficacy by suppressing HSC activation and proliferation, inducing apoptosis, and reverting them to a quiescent state, thereby effectively reversing hepatic fibrosis. This review systematically synthesizes current knowledge on the pathogenesis of hepatic fibrosis and the mechanistic actions of various TCM in modulating this process, aiming to provide a theoretical foundation for developing novel TCM-based anti-fibrotic therapeutics and to advance clinical translation through innovative formulation strategies.
Introduction
Chronic liver disease is a major burden on global health, resulting in approximately 2 million deaths annually. Potential causes of chronic liver disease include viral-related chronic liver disease, alcoholic steatohepatitis (ASH), and non-alcoholic steatohepatitis (NASH), as well as autoimmune and genetic disorders. In the liver, fibrosis levels are associated with liver function and represent a major risk factor for hepatocellular carcinoma (HCC) development. 1 The prevalence of liver fibrosis ranges from 2% to 19%, remaining one of the primary causes of morbidity and mortality worldwide. 2 It leads to severe complications such as ascites, portal hypertension, hepatic encephalopathy, and liver failure, and increases the risk of hepatocellular carcinoma, imposing a heavy burden on individuals, society, and the healthcare system. 3 Currently, Western medical treatments for liver fibrosis include antiviral drugs, corticosteroids, hepatoprotective medications, and liver support therapies, but their efficacy is low, they exhibit strong drug resistance, and they have significant side effects. 4
In liver fibrosis, the liver cells secrete a variety of growth factors and inflammatory cytokines (or chemokines) that stimulate hepatic stellate cells (HSCs) so that they are converted into myofibroblasts (MFBs). 5 HSCs secrete a large amount of extracellular matrix (ECM) and are diffusely deposited in the liver, and fibrous scar tissue is formed. 6 According to relevant literature reports, early effective drug intervention in HF can delay or even reverse the occurrence of fibrosis, thereby further reducing the incidence of liver cirrhosis and even liver cancer. 7 Western medicine treatment involves inhibiting the activation and proliferation of HSCs and promoting the decomposition of ECM, but because of its single target, there are many side effects (especially liver and kidney function damage), and currently, there are no other effective treatment methods.
In recent years, relevant studies have indicated that, compared with Western drugs, there are many advantages of using traditional Chinese medicine (TCM) preparations for inhibiting and reversing HF, such as the advantages of multiple components, multiple pathways, and multiple targets. Thus, diagnosis and treatment based on TCM syndrome differentiation has been adopted, and a large number of TCM anti-liver fibrosis preparations and empirical prescriptions in hospitals have achieved satisfactory clinical treatment effects.8,9 Traditional Chinese Medicine (TCM), as a unique medical approach in China, offers superior safety and efficacy and is widely used in the treatment of liver fibrosis. 10 In this article, recent research on TCM preparations for HF reversal were reviewed to provide a reference for the development and application of new TCM preparations in the future.
Understanding of TCM on Liver Fibrosis
In TCM, the liver is the viscera of wind wood, and the liver is characteristically Yin in physique and Yang in function. The liver prefers to grow freely and dislikes depression, therefore, it pertains to wood. When the liver promotes the heart, this is akin to wood generating fire, such as the liver storing blood to supplement the heart. When it is affected by many internal and external causes, such as external pathogenic factors, epidemic toxins, phlegm and blood stasis, deficiency of vital energy, and internal injury of the seven emotions, this affects the liver Qi and blood, and Yin and Yang, and results in body fluid dysfunction. After a long time, there will be the accumulation of symptoms and pains, which will further lead to the formation of liver fibrosis and even cirrhosis.11,12
Although there is no clear record of the name of HF in ancient medical books, it is usually described as jaundice, hypochondriac pain, accumulation, bulge, and hepatic accumulation in terms of its clinical manifestations and related symptoms. For example, in the “Theory of the Origin of Diseases,” it was said that “The whole body is yellow in the face of the claw armor and urine, and wants to lie down, the face is yellow, the tooth scale is yellow, the claw nail is yellow, and that is the jaundice.” Suwen ·Dirty Qi Law recorded that “Liver disease, under the two threats of pain to lead to less abdomen, making people angry.” In “The Origin of Diseases and Syndromes of Accumulation Diseases,” it is stated that “The organs are evil, they fail to accumulate at the beginning, they do not go to the end, but they accumulate,” which coincides with the developmental order of: chronic hepatitis-liver fibrosis-cirrhosis-liver cancer. The “Suwen Abdominal Treatise” noted: “If there is a sick heart and stomach, if you eat it, you cannot eat twilight, which is called bulging.” According to the “Testament of Zhou Shenzhai”: “The lumps, the liver accumulation, the liver through the damp and hot Qi, gathered into also.”
The above-mentioned ancient books and documents summarize the main etiology and pathogenesis of liver fibrosis as being related to dampness, heat, blood stasis, and toxins. It was previously noted that the changes involved in chronic HF are a dynamic process. 13 If the hot and humid poison and blood stasis continue to progress, their evolution characteristics proceed from light to heavy. The Qi is the basic pathogenesis characteristic, according to the theory and the treatment principle of support (ie, detoxification, tonify, eradicate deficiency, build blood). In addition, an empirical formula for treating HF, Qiwei chemical fiber decoction, was established and achieved a satisfactory clinical effect. Chen et al noted that normal liver drainage function is an important condition for the coordination of the ascending and descending functions of the spleen and stomach. 14 If the drainage function is maladjusted, the transportation and metabolism of the spleen and stomach will be affected so that water and liquid cannot accumulate in sputum, and Qi and blood cannot remain in the liver. Therefore, the main pathogenesis of HF is blood stasis and phlegm-dampness blockage, and phlegm-stasis intercoagulation.
In 2019, Xu et al also mentioned in the newly released “Guidelines for the diagnosis and treatment of liver fibrosis with integrated traditional Chinese and Western medicine” that the basic pathogenesis of HF is “virtual damage and accumulation”. 15 The shape and quality of the liver are damaged, resulting in the loss of Yin and essence, and therefore, it cannot be used for Qi, resulting in poor blood circulation and long-term coagulation. 16 He also mentioned in the guide that “deficiency loss” mainly manifests as deficiency of spleen Qi and deficiency of Yin and jing of the liver and kidney. Qi deficiency reflects the impairment and decrease in body function, and deficiency of Yin and jing of the liver and kidney denotes the damage to the liver shape, which is a deeper pathogenesis of deficiency loss. Deficiency loss is mainly manifested by the decrease in the number of liver parenchymal cells with subsequent decrease in liver function, as well as damage to the hepatic sinusoid wall, while blood stasis is mainly manifested by the excessive deposition of liver ECM and the occurrence of a series of physiological and pathological processes such as hepatic sinusoid capillarity. 17
The Relationship Between Hepatic Stellate Cells and Their Main Cells and Hepatic Fibrosis
Normal liver tissue is composed of non-parenchymal cells and parenchymal cells (ie, hepatocytes). In the former, HSCs, Kupffer cells, and inflammatory cells play an important role in the process of HF. In particular, the continuous activation of HSCs plays a central role in the occurrence and development of HF. 18 Under normal circumstances, HSCs are in a quiescent state. When the liver is affected by internal and external factors such as inflammatory factors or mechanical stimulation, the phenotype of the activated HSCs changes from quiescent to activate. The activated HSCs increase the internal pressure of the hepatic sinusoid through the contraction of the cells, and the pressure of the portal vein increases. This results in the formation of HF and the reconstruction of the internal structure of the liver through its own proliferation and secretion of a large amount of ECM.
The activation of HSCs mainly includes two stages (ie, the initiation stage and the persistent stage). The initiation stage refers to the change in early gene expression and the change in cell phenotype under the stimulation of a series of cytokines. The specific process consists of the loss of liver parenchymal cells, which results in the nearby Kupffer cells, hepatocytes, hepatic sinusoidal endothelial cells, and platelets secreting a variety of different cytokines such as hepatocyte growth factor, factor-1, transforming growth factor-β (TGF-β), tumor necrosis factor, insulin growth factor, and platelet-derived growth factor (PDGF) under paracrine action. They can be used to activate HSCs, and the activated HSCs gradually transform into a myofibroblast (MFB) phenotype, which leads to cell proliferation, increased ECM synthesis, and secretion of endothelin-1 through its own secretory mechanism. 19
According to Seki et al HSCs secrete a variety of cytokines during activation, among which PDGF and TGF-β are the two main cytokines that transform HSCs into MFBs. TGF-β, PDGF, and other cytokines promote the entire activation process. If external stimuli are removed, cessation of the entire HF process will not occur, as this stage mainly relies on paracrine stimuli for completion. 20 The persistent stage refers to maintaining the activation state of HSCs so that fibrogenesis can occur under the action of various cytokines in the initiation stage. During the entire duration, a series of characteristic changes occurs that includes cell proliferation, chemotactic aggregation, cell contraction, promotion of fiber formation, matrix degradation, retinol disappearance, leukocyte chemotaxis, and cytokine release. However, the ultimate result of these changes is a direct or indirect increase in ECM deposition, with HSC activation at this stage governed by paracrine and autocrine regulation. 21 The former is a cytokine that strongly promotes the proliferation of HSCs, while the latter is a cytokine that most strongly activates HSCs. If PDGF and TGF-β can be regulated to further delay and reverse the process of HF, then the activation of HSCs can be inhibited.20,22
There are five members of the PDGF family: PDGF-AA, PDGF-BB, PDGF-AB, PDGF-CC, and PDGF-DD. PDGF is composed of two highly homologous PDGF-A chains and PDGF-B chains, and connects the dimer structure of the A chain and B chain through disulfide bonds. PDGF is stored as α-particles in blood platelets (PLTs) in the physiological state. To function, it needs to bind to PDGF-α receptor and PDGF-β receptoron the cell membrane. The α-subunit has a strong affinity for both PDGF-A and PDGF-B chains, while the β-subunit only has a strong affinity for PDGF-B chains. The former can combine with PDGF-AA, PDGF-BB, and PDGF-AB, while the latter can only combine with PDGF-BB and PDGF-AB. Ying et al found that the progression of HF can be inhibited by inhibiting the expression of PDGF-BB and its downstream signal molecules, and therefore, it can be used as an effective target for the treatment of HF. 23 Related studies showed that during liver fibrosis, when priority is given to PDGF-β receptors, then PDGF-BB activates intracellular tyrosine residues before and after phosphorylation (or unique target protein tyrosine residue phosphorylation), and signal transduction of the intracellular cascade amplification process can control the activity of cells. Thus, the activation, proliferation, and migration of HSCs will occur.23-26 Wang et al found that Qianggan capsule inhibited the activation and proliferation of HSCs and achieved anti-HF effects by inhibiting the expression of PDGF-BB mRNA in rat liver tissues and downregulating the signaling pathway of TGF-β1/Smads. 27 Wang et al found that the Da Huang Tuo Chong pill combined with astragalus (Astragalus membranaceus [Fisch.] Bunge) and safflower seed (Carthamus tinctorius L.) significantly inhibited the expression of PDGF-BB protein and PDGF-R protein in liver tissue, decreased the proliferation and activation of HSCs, and reduced collagen deposition, so as to delay and reverse the process of HF. 28
The TGF-β superfamily comprises over 30 cytokines, including TGF-β, activin, inhibin, bone morphogenetic protein, and Müllerian inhibitory substance. These cytokines play crucial roles in tissue repair, embryo development, wound healing, cell growth and differentiation, immune function, and other processes. 29 The human TGF-β1 gene is located on chromosome 19Q3 and contains 7 exons. The terminal sequence contains five regulatory regions: one enhancer-like active region, two promoter regions, and two negative regulatory regions. It can stimulate mesenchymal cells while inhibiting cells from the neuroectoderm or epithelium, and its concentration is highest in liver tissues.30,31 In the resting state, TGF-β1 is mainly secreted by hepatocytes, HSC, MFB, PLT, library cells, hepatic sinus endothelial cells, and other cells. Secreted TGF-β1 in the cell is mainly synthesized for two types of peptide structures, that is the probody propeptide of the mature domain and relevant latent peptides. The former is secreted into the ECM post-synthesis and stored in a readily accessible TGF-β pool, eliminating the need for re-synthesis. 32
When the liver is affected by inflammatory factors, mechanical stimulation, or other internal/external factors, the mature domain probody propeptide stored in the TGF-β pool is cut and activated, and with the assistance of Smad protein-mediated intracellular signal transduction, it binds to the specific type I and type II receptors on the membrane of the activated HSCs and other cells. Guo et al found that Chai Jie Yiganxian, a Chinese herbal compound preparation, inhibited the proliferation and activation of HSC-T6 cells and decreased the expression of its marker, the α-SMA protein, by regulating the TGF-β/Smad signal transduction pathway.33,34 TCM compounds can inhibit the TGF-β/Smad signal transduction pathway and decrease the activity of HSCs and the synthesis of ECM, and delay and reverse the process of HF.35,36 Tian et al found that Chaihu Shugan SAN inhibited the activation of HSCs and decreased the expression of TGF-β1 and α-SMA proteins, which subsequently decreased the synthesis of the ECM through the TGF-β1/p38MAPK signaling pathway. 37
The inhibition of TIMP-1 protein expression led to a decrease in MMP-9 protein expression, promoting ECM degradation and reversing HF progression. Cai et al found that Yinchen Hao decoction downregulated the TGF-β1, Smad3, P-ERK1/2, and α-SMA protein expression in a mouse model of HF by regulating the TGF-β1/Smad/ERK signal transduction pathway, which then decreased the production of ECM and reversed the occurrence and development of HF. 38
Clinical Application of TCM in the Treatment of Liver Fibrosis
TCM is widely used in China and is increasingly recognized as a complementary and alternative form of modern medicine, encompassing herbal remedies, acupuncture, and other physical therapies such as massage. 39 Holistic concepts and dialectical treatment are theoretical guidelines in TCM, reflecting systemic physiology approaches and personalized medicine in Western medicine, which demonstrate their advantages in the treatment of liver fibrosis. 40 The theoretical guidance of TCM for the treatment of hepatic fibrosis includes resolving phlegm and removing blood stasis, nourishing the liver and kidneys, soothing the liver and strengthening the spleen, and promoting diuresis and clearing heat.
There are numerous drugs available for treating HF, as its causes often involve blood stasis, dampness and heat, Qi deficiency, and heat toxicity. Treatment primarily focuses on promoting blood circulation, removing stasis, clearing heat and dampness, and invigorating Qi and the spleen. Single drugs such as Salvia miltiorrhiza, A. membranaceus, Angelica sinensis, and turtle shell appear frequently in TCM compound preparations.41,42 These ingredients play an important role by inhibiting the proliferation of hepatic fibrous connective tissue, and decreasing the deposition of ECM and the activation of HSCs. Therefore, they offer a curative effect by protecting liver function and inhibiting the further progression of HF.
Clinical Applications of Common TCMs in the Treatment of Liver Fibrosis
Salvia Miltiorrhiza
The root of Chinese salvia (Salvia miltiorrhiza) is used as medicine (Figure 1). It has a bitter taste and a slightly cold nature, and acts upon the heart and liver meridians, which was first recorded in the Shennong Bencao Jing. It has the functions of promoting blood circulation and removing stasis, counteracting against inflammation, calming and removing irritation, reducing swelling, and relieving pain, and is one of the important components of the compound preparation for treating liver fibrosis. 43 Studies show that a water extract from S. miltiorrhiza roots enhances liver microcirculation, removes endotoxins, and protects liver cells by neutralizing HF. It has been widely used in the treatment of chronic hepatitis, liver fibrosis, and other diseases with satisfactory efficacy.

The Chinese Traditional Medicine Salvia miltiorrhiza.
Liu et al studied danshensu, which is the water-soluble active component of S. miltiorrhiza, and determined that it regulated the degradation of the ECM by promoting the apoptosis of HSCs. 44 It also decreased the expression of angiogenic regulatory factors to inhibit the activation of HSCs. Danshensu also effectively improves liver function indexes (aspartate transaminase (AST) and alanine transaminase (ALT)) and liver fibrosis indexes (HA, collagen IV, LN, and PIIINP) in a CCl4-induced hepatic fibrosis rat model. Hematoxylin and eosin (HE) staining and VG staining revealed the significant recovery of liver tissue morphology, indicating that danshensu can effectively decrease the damage to liver function and prevent liver fibrosis. Wang et al discovered that danshensu increases the levels of superoxide dismutase (SOD) and glutathione peroxidase while decreasing malondialdehyde levels in liver tissue. This action helps inhibit oxidative stress by modulating the nuclear factor erythrocyte 2-related factor 2/heme oxygenase-1 signaling pathway. 45 In addition, danshensu inhibited the expression of TGF-β1 and cycde-2 by regulating the signal transduction pathway of the transcription factor nuclear factor κB (NF-κB), thus delaying the process of liver fibrosis.
Astragalus Membranaceus
Astragalus (Astragalus membranaceus) is a compound leaf with a sweet taste and mild nature, and affects the liver, kidney, spleen, and lung meridians, which was first recorded in the Shennong Materia Medica (Figure 2). The pharmacological effects of this herb include protecting the liver, promoting diuresis and detoxification, clearing heat, resisting oxidative stress, and enhancing immunity. 46 Astragaloside, the main active component of A. membranaceus, protects the nervous, immune, and cardiovascular systems in an oxidative stress model. Oxidative stress is a key factor that activating HSCs and contributing to HF. Consequently, A. membranaceus plays a crucial role in combating liver fibrosis.47,48 Li et al found that baicalin notably reduced CCl4-induced liver fibrosis in rats, leading to less liver damage, improved liver function, and normalized biochemical indicators to varying extents. 49 The SOD levels in liver tissue significantly increased, indicating that baicalin's anti-fibrotic effect is due to its antioxidant properties. Lei et al found that A. membranaceus inhibited the expression of upstream activator MKK3 and downstream transcription factor AFT-2, and phosphorylated P38MAPK in a liver fibrosis rat model. 50 It also prevented liver fibrosis by reducing the expression of factors that contribute to its development.

The Chinese Traditional Medicine Astragalus membranaceus.
Angelica Sinensis
Angelica (Angelica sinensis) is a perennial herb in the umbelliferae family (Figure 3). It tastes sweet and warm, and acts on the heart, liver, and spleen meridians. It was first recorded in the Shennong Materia Medica, and its therapeutic effects include invigorating blood circulation, regulating menstruation, relieving pain, protecting the liver, resisting inflammation, counteracting tumors, and enhancing immunity. The main chemical components of A. sinensis include volatile oils, amino acids, organic acids, flavonoids, and polysaccharides, among which polysaccharides can activate the autophagy system mediated by the MEK/ERK pathway in the human body, so as to further inhibit hepatotoxicity induced by large doses of diphosphocholine B and protect the liver. 51

The Chinese Traditional Medicine Angelica sinensis.
Turtle Shell
Turtle shell is the dried shell of trionychidae animals (Figure 4). It has a salty taste and a slightly cold nature, affecting the liver and kidney meridians. It serves to nourish Yin and Yang, disperse soft and firm elements, reduce fever, and eliminate steam. Modern pharmacological studies have confirmed that powdered turtle shell inhibits the body's inflammatory response, thereby blocking the expression of growth factors. This action helps to prevent the active proliferation of HSCs, the antioxidant reaction, and the excessive growth of the ECM, thus playing an anti-HF role. 52 Sun et al found that proteins and peptides with a molecular weight under 6000 in turtle shell can induce G1 phase arrest in HSC-T6 cells, thereby inhibiting their proliferation and reducing ECM synthesis to prevent liver fibrosis. 53 In addition, turtle shell decoction administered to rats with liver fibrosis resulted in anti-lipid peroxidation, regulation of cytokine levels to inhibit the activation of HSC proliferation, and degradation of the ECM to reverse or further slow the occurrence of HF development.

The Chinese Traditional Medicine Turtle Shell.
Clinical use of Common Chinese Herbal Compound Preparations for Treating Liver Fibrosis
Clinical use of Drugs to Enhance Blood Circulation and Eliminate Stasis in the Treatment of Liver Fibrosis
Clinical studies have shown that using Traditional Chinese Medicine to improve blood circulation and eliminate blood stasis can lower serum liver fibrosis markers (HA, collagen IV, LN, and PIIINP) and liver function indicators (AST and ALT) by increasing the diameter of the main portal vein and enhancing blood flow velocity (Table 1). This decreases liver damage, and will subsequently delay or reverse the occurrence and development of HF. Drugs promoting blood circulation and removal of blood stasis will inhibit the active proliferation of HSCs by regulating the expression of cytokines and their receptors, regulating the cell signal transduction pathway, and decreasing the inflammatory response and anti-oxidative stress response, which reflects the multi-layered, multi-pathway, and multi-target treatment characteristics of TCMs in the treatment of HF.
Common Clinical Anti HF Compound Preparations Based on Activating Blood Circulation and Removing Blood Stasis.

Regulation of Cell Signal Transduction Pathways
The activation and proliferation of HSCs is one of the important links in the genesis and development of liver fibers, and the activated HSCs can play a role in the regulation of upstream and downstream cytokines through multiple molecular signaling pathways. TCM compound preparations exert multi-level, multi-pathway, and multi-target therapeutic effects. Therefore, in the treatment of HF, multiple signaling pathways can be altered to upregulate or downregulate inhibitory cytokines, which will induce the apoptosis of activated HSCs and inhibit additional activation of HSCs, so as to achieve an anti-HF effect.62-64
According to current relevant studies, the regulation of HF molecular signal transduction pathways can be roughly divided into two categories: (1) Inhibiting the molecular signaling pathways of liver fibrosis: the Wnt/β-catenin signaling pathway (β-catenin), Toll-like receptor 4/nuclear factor κB signaling pathway (TLR4/NF-κB), and p53 signaling pathway; (2) Promoting the molecular signal transduction pathways for liver fibrosis: the TGF-β/Smad signal transduction pathway, mitogen-activated protein kinase (MAPK) signal transduction pathway, peroxisome proliferator activated receptor (PPAR) signal transduction pathway, JAK kinase/signal transduction pathway, transcription activator (JAK/STAT) signal transduction pathway, and the phosphatidylinositol 3-kinase (PI3K) signal transduction pathway.65,66
Chen et al confirmed that ligustrazine promoted the release of cytochrome c by inhibiting the expression of anti-apoptotic protein Bcl-2 and apoptotic protein Bax, and activated the cleavage of substrates by apoptotic protease caspases to induce the apoptosis of activated HSCs. 67 Ligustrazine regulates the p53 signaling pathway, increasing the expression of P21, P27, and other proteins, which causes HSCs to stall in the G0/G1 phase of the cell cycle, blocking cell proliferation and inhibiting liver fibrosis development.
Agents That Decrease Inflammation
miR-122 and interleukin-10 (IL-10) play significant roles in the inflammatory response during chronic liver injury. miR-122, which accounts for approximately 70% of liver miRNA, mainly inhibits viral replication and antigen expression of viral hepatitis B. IL-10 releases proteases and free radicals to regulate the generation of inflammatory mediators, which results in a cascade effect. 68 Kong et al found that curcumin significantly decreased the amounts of glutamic-oxalacetic transaminase and glutamic-pyruvic transaminase in the serum of hepatic fibrosis mice with bile duct ligation. 69 Curcumin also inhibited the deposition of ECM and infiltration of inflammatory cells, and alleviated liver injury caused by inflammatory reaction.
Gan et al proposed that ursolic acid may alleviate liver inflammation and collagen matrix deposition, potentially by activating the NOX2/ROS/NLRP3 inflammasome pathway, which converts the inactive caspase-1 precursor into active caspase-1. 70 The latter, in turn, transforms inactive IL-1β precursors into active IL-1β and IL-18, which are extracellularly released and cause downstream inflammatory responses. 71
Antioxidant Stress Response
Oxidative stress refers to the imbalance between ROS and active nitrogen produced by the body in the process of aerobic metabolism and the elimination of antioxidant defense. In essence, the ROS generated by the body in a pathological state surpasses the capacity of its antioxidant defense system, leading to the buildup of ROS in the body. Oxidation of some macromolecular substances such as DNA, protein, and lipids can also occur. 72 The change in the oxidative stress state in the liver is closely related to the occurrence and development of liver fibrosis. When ROS increases, the anabolic function of normal liver cells, such as liver cells, HSCs, and Kupffer cells, will be affected. 73 When Fuzheng Huayu capsule was administered, Ou et al observed a significant anti-fibrotic effect in rats with CCl4-induced liver fibrosis, Qi deficiency, and blood stasis. By inhibiting the expression of autophagy LC3II protein, the expression of p62 protein was increased, which thereby activated an increase in Nrf2/Keap1 anti-oxidative stress Nqo1 protein expression. 74
Clinical Application of Heat-Clearing (Detoxification) and Dampening Drugs in the Treatment of Liver Fibrosis
In TCM, when dampness and heat invade the body, it can accumulate in the liver, and over the long-term, will result in liver and Qi stagnation, which manifests with symptoms of liver blood stasis such as pale complexion and purple tongue (Table 2). Therefore, it is likely that damp-heat toxin plays a leading role in the pathogenesis of liver fibrosis, and blood stasis runs through the entire process of the occurrence and development of the disease. Therefore, the main treatment goals should be to detoxify poison, regulate Qi, promote blood circulation, and disperse soft and firm junctions.
Common Clinical Anti HF Compound Preparations Based on Clearing Heat (Detoxification) and Diuresis.

Drugs that clear heat and reduce dampness are crucial for protecting liver cells and lowering inflammation. The possible mechanisms are as follows: (1) Enhance the body's immunity, reduce or block the damage caused by the immune system's response to hepatitis B virus infection. (2) Inhibit the replication of hepatitis virus, reduce the damage caused by inflammatory reaction to liver cells, and block the active proliferation of HSCs. (3) Heat-clearing (detoxification) and dampening drugs can promote the degradation of ECM and inhibit the synthesis of ECM to a certain extent, and also improve the microcirculation function of the liver so that the fibrosis can be dissolved and absorbed.79,80
Wan et al found that Huagan Jiadu decoction significantly decreased the serum liver fibrosis indexes in rats, alleviated liver damage, and further slowed the occurrence and development of liver fibrosis. 81 It was speculated that the possible mechanism involved inhibition of the TLR4 signal transduction pathway and downregulation of TLR4, MyD88, and NF-κB mRNA and protein levels. Inhibition of nuclear displacement of NF-κB resulted in inhibition of the downstream TGF-β1 signal transduction pathway and decreased serum hyaluronic acid and laminin levels.
Clinical Application of Invigorating Qi and Strengthening the Spleen in the Treatment of Liver Fibrosis
In the clinical treatment of liver fibrosis, liver stagnation, spleen deficiency, and damp-heat internal knots are common regarded as the main pathogenesis and syndrome types, and the treatment of this disease is based on syndrome differentiation, such as soothing the liver and strengthening the spleen, clearing heat, and invigorating dampness (Table 3). 90 Xia et al found through experimental study that administering a prescription to regulate liver function, strengthen the spleen, and promote blood circulation prescription to rats with dimethylnitrosamine-induced liver fibrosis decreased the severity of rat liver fiber, inhibited the activation of the local renin-angiotensin-aldosterone system, and thus regulated the signaling pathway of TGF-β1/Smad. 91 Additionally, HIF-1α protein and VEGF mRNA expression were downregulated, which inhibited liver fibrosis.
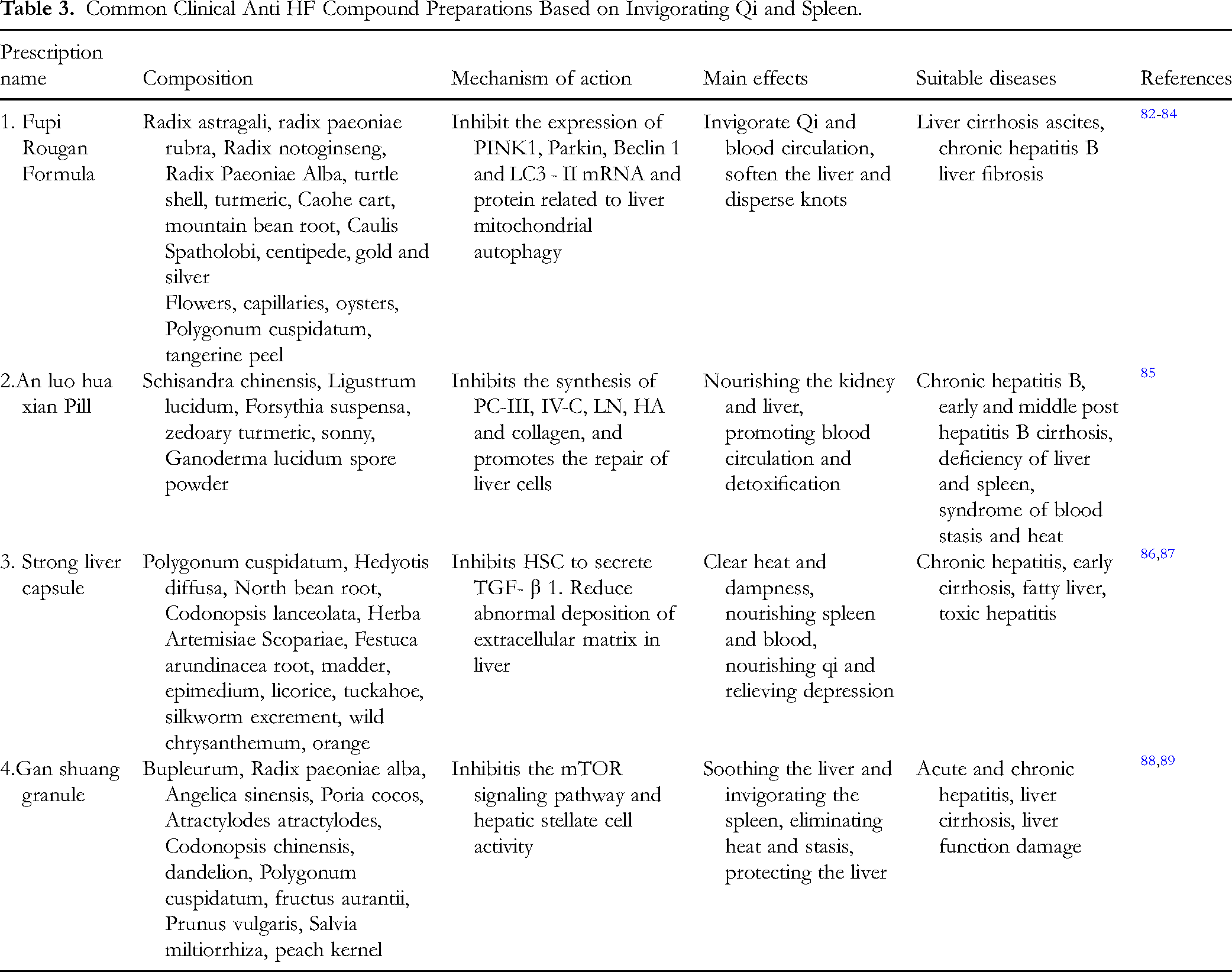
Common Clinical Anti HF Compound Preparations Based on Invigorating Qi and Spleen.
Summary and Prospects
Hepatic fibrosis, a well-established clinical manifestation of chronic liver disease, typically requires approximately 10 years to progress to advanced stages. It serves as a critical transitional phase in the progression of various chronic liver diseases toward cirrhosis and eventual hepatic failure. However, there are currently no guideline-endorsed pharmacological interventions specifically targeting hepatic fibrosis in clinical practice. During the early stages, therapeutic strategies remain predominantly focused on anti-inflammatory and hepatoprotective measures to address the underlying etiology. TCM is a holistic medical system encompassing disease prevention, diagnosis, and therapeutic intervention, guided by the theoretical framework of TCM that emphasizes dynamic balance and pattern differentiation. In clinical practice, TCM is not administered as isolated herbs but rather prescribed as compound formulations based on compatibility principles (Jun-Chen-Zuo-Shi) to achieve synergistic therapeutic effects. To date, numerous studies have demonstrated that bioactive components in TCM possess the potential to selectively target HSCs for the treatment of liver fibrosis.92-94 This therapeutic effect involves multiple pathways, with a particular focus on the transforming growth factor-β (TGF-β)/Smad signaling pathway. However, most current studies on the mechanisms of TCM in treating liver fibrosis remain in the preliminary stage, primarily focusing on in vitro cell lines and animal models that moderately simulate liver fibrosis. Therefore, in modern medicine, it is crucial to clearly and accurately understand the active components, key targets, and related pathways of TCM. In summary, TCM can regulate a series of biological processes related to the activation, proliferation, apoptosis, autophagy, and ferroptosis of HSCs through targeted interventions.
Currently, the principal therapeutic approaches for hepatic fibrosis management involve elimination of etiological factors inducing chronic hepatic injury, eradication or inactivation of myofibroblasts, suppression of inflammatory responses, and degradation of extracellular matrix (ECM). 95 Nevertheless, substantial clinical breakthroughs in reversing fibrotic progression remain elusive. Because of the significant role of hepatic macrophages in the progression and regression of liver fibrosis, currently growing number of researches focused on macrophage-based therapies to improve liver fibrosis, demonstrating considerable therapeutic potential. 96 To date, the main therapeutic strategies for reversing fibrosis based on macrophages include antifibrotic treatments targeting macrophage immune metabolism, 97 anti-liver fibrosis treatments targeting macrophage-related signaling pathways, 98 and the use of autologous macrophages for treating liver fibrosis. 99
TCM is a treasure of Chinese traditional medicine, possessing unique advantages and profound connotations. 100 Single compounds, single-herb extracts, and traditional Chinese medicine formulations are increasingly attracting researchers’ attention due to their low adverse reactions, cost-effectiveness, and broad-targeting effects on multiple pathways, playing a significant role in the prevention and treatment of liver fibrosis. Research on the molecular mechanisms of antifibrotic effects of single compounds, single herb extracts, and traditional Chinese medicine formulations targeting oxidative stress and intracellular signaling pathways has been extensively conducted based on the development of modern scientific technologies such as pharmacology and genomics, achieving favorable outcomes and demonstrating broad application prospects. 101 Furthermore, due to insufficient research on the structure-activity relationship of the active ingredients. Most polysaccharides, single-herb extracts, and traditional Chinese medicine formulations are mixtures, making their pharmacological mechanisms difficult to fully elucidate. This difficulty may be due to the accumulation of active components in single-herb extracts or because the mixtures form systemic pharmacological mechanisms. 55 This limitation restricts the application of these traditional Chinese medicines in clinical practice. Based on previous research, it is necessary to analyze the individual differences in pathogenesis and disease progression, and further explore how traditional Chinese medicine can inhibit oxidative stress to treat liver fibrosis.
The occurrence and development of liver fibrosis is a complex pathophysiological process involving multiple genes and multiple pathways. There are numerous advantages to using TCM for the treatment of liver fibrosis, such as its multiple components, multiple pathways, and multiple targets. It assumes a synergistic role in inhibiting the active proliferation of HSCs and promoting their apoptosis, inhibiting the synthesis and degradation of ECM, inhibiting hepatic sinusoidal capillarization, and alleviating the inflammatory response and anti-oxidative stress of the liver. Therefore, early intervention can effectively block or even reverse the occurrence and development of liver fibrosis. This is of great significance for the clinical treatment of liver cirrhosis and hepatocellular carcinoma.
Most TCM preparations are based on activating blood circulation and removing stasis, draining turbidity and dredging collaterals, and on this basis, supplementing the Qi and spleen, so as to achieve the effect of strengthening the viscera. It should be noted that there are many different dosage forms of TCM that are usually used in combination with symptom-relieving and antiviral drugs to prevent and cure liver fibrosis. At present, TCM research on liver fibrosis has gradually developed to the cellular and molecular levels. The current body of research has indicated that the actions that can prevent fibrosis are mainly related to inhibiting the active proliferation of HSCs, protecting liver cells, and anti-inflammatory and anti-oxidant effects. 102
Combination therapy for liver fibrosis has the potential for greater effectiveness, eg, etiological therapy, immune therapy, protecting regenerated liver cells, activation of HSC suppression, and adjustment of the ECM. A variety of different treatment methods may increase effectiveness and decrease unpleasant symptoms for patients such as abdominal pain, diarrhea, rib pain, jaundice, and nausea and vomiting. These methods have the ability to inhibit the progression of liver fibrosis at different levels through different mechanisms.
Previous studies have found that Chinese medicine acts through some mechanism to suppress the development of liver fiber, but the research on this mechanism of action is limited to only a few signaling pathways. Additional detailed studies are required to elucidate the specific mechanisms of action in complete signaling pathways. Therefore, systems biology research methods have been adopted for liver fibrosis that are multi-angle and examine multiple signaling pathways that regulate networks. Thus, applying TCM ingredients to HSC activation with different targets is a reasonable formula that might result in some significant findings for liver fibrosis therapy. 103
TCM typically consists of various herbs/ingredients with known or unknown active components that can target different pathways for specific medical indications and can be adjusted based on individual symptoms. TCM typically consists of various herbs/ingredients with known or unknown active components that can target different pathways for specific medical indications and can be adjusted based on individual symptoms. Used clinically as formulations, herbal medicines, and monomeric compounds. TCM demonstrates unique therapeutic advantages, whether used independently or in conjunction with standard biomedicine. Although TCM has demonstrated promising clinical outcomes, it faces significant challenges in standardization, mechanism research, and clinical validation. Future research should aim to solidify the scientific foundation of TCM, expand its application in disease management, and strive for seamless integration between traditional and contemporary medical practices. Given the current state of research on Chinese medicine for the treatment of liver fibrosis, experimentation is currently conducted to study the curative effect during the primary stage. Specific mechanisms of action and effects of related target proteins remain unclear, and therefore, genomics, proteomics, and metabonomics are required for in-depth study of TCM treatment for liver fibrosis. These will provide a broader theoretical basis for later development of safe and effective drugs to counteract liver fibrosis based on a system of regulating targets for inhibition of liver fiber.
Footnotes
Acknowledgements
Author Contributions
Ming Yang: Conceptualization and Writing—Original Draft Preparation; Qi Ge: Conceptualization and Writing—Original Draft Preparation, Supervision, Funding Acquisition; Jingxue Zhang: Manuscript Proofreading, Writing-Review & Editing; Jiayi Yao: Manuscript Proofreading; Keping Chen: Supervision, Funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China [grant numbers 31872425 and 31861143051], Natural Science Research Project of Anhui Educational Committee [grant number 2024AH051415], and Scientific Research Starting Foundation of Chuzhou University [grant number 2024qd43].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
