Abstract
Background
This study focuses on the extraction and application of a natural dye from the Hulela Zard plant (Terminalia chebula L.), which is rich in tannins. The aim is to develop an eco-friendly dyeing process for cotton fabric using this plant-based dye and optimize the dyeing parameters for efficiency.
Method
The microwave-assisted extraction (MAE) method was employed to extract the colorant from Hulela Zard. Four independent dyeing parameters—extraction time, pH, temperature, and salt concentration—were optimized using new design of response surface methodology. Furthermore, the effects of sustainable chemical mordants (0.5-2.5% concentrations of aluminum, iron, and tartaric acid) and bio-mordants (pomegranate and madder at 0.5-2.5% concentrations) were evaluated for shade development on the cotton fabric.
Results
Optimal dyeing conditions were determined to maximize the dye yield by using innovative response surface design. The dyed fabric was then subjected to ISO-standard tests for colorfastness to light, washing, and rubbing. The fabric showed a good light fastness rating of 4-5 and wash fastness rating of 4-5. The optimization process was validated by computing the coefficient of determination (R² = 0.9549), ensuring reliable outcomes.
Conclusion
The findings demonstrate that the MAE method, combined with sustainable mordants, can effectively produce desirable shades on cotton fabric while maintaining good colorfastness properties. This eco-friendly process offers a viable alternative for natural dyeing with potential industrial applications.
Keywords
Introduction
The world is beautiful due to the variety of colors gifted by nature. However, some technological advancements, particularly in industrial processes, have had unintended negative impacts on the environment and natural beauty. 1 Various sectors have developed to improve human lifestyle, with industrialization playing a key role. 2 While agriculture and food production are fundamental to human survival, the textile sector also plays a significant role in meeting essential needs and enhancing quality of life through clothing, handicrafts, and fashion. 3 Previously since the development of mankind, natural sources of colors were used to enhance the beauty of body parts, house painting, traditional, cultural gathering but the rapid advancement of industrialization, the sudden discover of Mauvein has revolutionized the world of dyes and pigments. 4 Now thousands of dyes are synthesized daily and employed in almost all walks of life. However, the global safety and protection agencies who are keenly observing their role found that many of these dyes after application or during their synthesis shed toxic effluents. 5 There effluents when come in contact with water, produce water borne diseases, barren agriculture land by disturbing soil pH and disturb the water ecosystem. 6 These wastes when released into the atmosphere also raise global heat, disturb water quality parameter and cause many hazardous and toxic aftermaths for the people. A great awareness about reducing the widespread use of these toxic colorants in all fields is nowadays the talk of the globe. The global protection agencies have now spread awareness about green products, their benefits and after reuse aspects. Among them green products, plant base natural dyes have been revived in all fields particularly in textiles. 7
Natural dyes are functional bio-colorants extracted from plants, minerals, and animals using various isolation methods, many of which aim to minimize chemical interventions. Plant parts such as roots, flower, barks etc are one of the biggest sources of natural dyes. 8 Many natural dyes, particularly those derived from plants, possess beneficial bioactive compounds that contribute to various medicinal activities. Unlike some synthetic dyes, these natural colorants often exhibit low toxicity while offering potential health benefits. For example, curcumin from turmeric has demonstrated anti-inflammatory and antioxidant properties, while anthocyanins found in many blue and purple plant-based dyes have shown potential in preventing cardiovascular disease and have anticancer, neuroprotective, and antidiabetic properties. 9 These colorants, when isolated do not process any harmful effluents. Most of the plants have also the ability to contain such bio-colorants that insect repellant and attractive eye soothing shades. 10 However, their isolation process using conventional methods have some limitations. These conventional methods such as heating, soaking, stirring, etc take a lot of time, energy, solvent and cost. 11 Now researchers are using such methods which are taking less solvent, labor, energy and cost. Among these modern methods microwave treatments are eco-friendly, uniform and rapid in action. 12 These rays have two-fold functions ie improvement in extraction via excellent mass transfer kinetics and surface modification of fiber to enhance its substantivity. 13
Natural dyes also face poor fastness ratings which have been overcome by using some metallic mordants. However, these metallic mordants such as salts of copper, cobalt, tin, chromium etc are carcinogenic and need replacement with some eco-friendly anchors. 14 Now bio-mordants have been included to make color fast shades which employed on fiber before and after dyeing. Now after utilization of microwave treatment and bio mordants there is need of standardization of extraction and optimization of effective dyeing variables at low levels. 15 Nowadays, response surface methodology (RSM) and other statistical optimization techniques have been extensively employed for the optimization of various chemical and biological processes. These methods can be used to search for optimal values of process parameters and enhance the overall response.16,17 In our study, we apply similar optimization approaches to natural dyeing processes. While Deepanraj et al 17 applied these techniques to biogas production, similar principles can be used to optimize natural dyeing processes, as both involve complex biological and chemical interactions.
In this study, Hulela Zard (Terminalia chebula) belongs to the family Combretaceae has been used as a source of natural colorant for the dyeing of cotton fabric. This plant has been reported good antioxidant, anti-bacterial, anti-carcinogenic, anti-microbial and anti-inflammatory.18,19 While the cited study focuses on Terminalia ferdinandiana, both T. chebula and T. ferdinandiana are closely related species within the Terminalia genus, known for their similar bioactive properties. Various Terminalia species, including T. chebula and T. ferdinandiana, contains high levels of bioactive compounds such as polyphenols and vitamin C, contributing to their medicinal properties.20,21 The colorant tannin from Hulela Zard has been used to dye the wool, yarn, cotton, silk and leather. 22 Cotton is the most important and widespread natural textile fiber in the world. It has cellulosic part where its terminal -OH group binds with dye. 23 Natural herbs such as Hulela Zard have been widely employed in the treatment of chronic fever, skin and eye conditions, allergies, asthma, rheumatism, body pain, diabetes, as well as skin infections and allergic reactions. 24
Here, the coloring behavior of Hulela Zard based colorant for cotton was studied by considering new subset design under RSM. Additionally, microwave treated as pollution free heating trial has been included to get effective yield of colorant onto fabric. The other purpose was to develop colorfast shades using ecofriendly chemicals and bio-mordants.
Material and Methods
Material Required
The ripe yellow fruit of Hulela Zard (Terminalia chebula L.) has been chosen as a source of plant dye. A laboratory grinder was then used to grind the dried fruit into a fine powder, which was subsequently sieved to produce particles that were 250-300 μm in size. Standard AOAC procedures were used to determine the powder's moisture content (7.8 ± 0.5%), ash content (3.3 ± 0.08%), and volatile matter (75.2 ± 0.6%). The produced powder was then kept at room temperature in an airtight container. Additionally, tannin source of bio-mordants and alizarin source of bio-mordants have been cleaned, and prepared for further mordanting. Chemical mordants such as Aluminum, Iron, and Tartaric acid have been utilized for the process of extraction, dyeing, and mordanting.
Extraction and Irradiation Process
The extraction process involved mixing 4 g of finely ground crude powder with 100 mL of distilled water and boiling it for 45 min. The mixture was then filtered using muslin fabric, and the resulting extract was used for bio-coloration. The extraction process was conducted in three different mediums: aqueous (distilled water), acidic (pH adjusted with hydrochloric acid), and alkaline (pH adjusted with sodium hydroxide). 25 The pH values for the acidic and alkaline extractions were 4 and 10, respectively. Next, the three extracts were treated with microwave (MW) exposure for up to 5 min at high power using a domestic oven, following the method of Bashir et al 18 and Elez Garofulić et al. 12 With 2 min intervals, the extracts were treated at high power for 2 to 10 min. To stop evaporation and allow pressure release, the 100 mL extract samples were put in glass beakers that were safe to use in the microwave and covered with perforated plastic wrap. An infrared thermometer was used to measure the extracts temperature just after each irradiation interval; the temperature was 70°, depending on the length of exposure. To improve dye absorption, samples of cotton fabric (5 g each) were also exposed to microwave irradiation under comparable circumstances before dyeing. The MW-cured (RE) and uncured (NRE) extracts were used to dye both MW-cured (RC) and uncured (NRC) cotton samples at 70 ˚C for 60 min, with an extract to fabric ratio of 25:1. While efficient extraction was achieved using microwave-assisted extraction (MAE), the primary focus of this study was on optimizing the dyeing parameters for cotton fabric to achieve optimal coloration properties using the extracted natural dye. The schematic representation of the dye extraction from Hulela Zard plant and their application on cotton fabric is shown in Figure 1. Every experiment was carried out three times under the same circumstances to ensure the stability and repeatability of the extraction and dyeing procedure. 26

Schematic Representation of the dye Extraction from Hulela Zard Plant and Their Application on Cotton Fabric.
Design of Experiment, Modeling and Process Optimization
RSM is a group of statistical techniques that have been employed to select dyeing variable using multiple connections27,28 by analyzing results using equation (1) given below
Design Structure with Coded Levels Used to Optimize the Dyeing Parameters.

In order to optimize the dyeing parameters and to examine the effects of factor variability on the K/S value of Hulela Zard; a three-level subset design (S4 + 2S1) comprising of factorial runs and axial runs was considered (Table 1). Cuboidal-region based design was employed and the axial design points were obtained by setting axial distance at
Table 1 presents the coded levels for the experimental design. Coded levels are used to standardize factors with different units of measurement, allowing for direct comparison. In this study, four independent variables (dyeing parameters) were investigated: pH, salt concentration, temperature, and time. The un-coded levels and units for these parameters are presented in Table 2.
Statistical Analysis
The statistical methods that were employed includes; regression analysis, response surface methodology (RSM) comprising analysis of variance (ANOVA) and coefficient of determination.
Shade Development Through Mordanting
The crude extracts of Punica granatum (pomegranate) and Rubia cordifolia (madder) were prepared by mixing finely mashed powders (0.5-2.5 g each) with 100 ml of boiling water. After boiling, each mordant was filtered and the resulting liquid was used to bio-mordant the cotton before and after dyeing at 70 ˚C for 45 min, using a ratio of 25 parts liquid extract to 1 part fabric. For chemical mordanting, 0.5-2.5 g of aluminum salt, iron salt, and tartaric acid were heated with 100 mL of water. Then, 25 mL of each chemical mordant was used to pre and post mordant of cellulosic fabric (18 each) at 70 ˚C for 45 min.
Dyed Fabric Assessment
The color analysis of the dyed fabrics obtained at each stage involved evaluating the color depth K/S, which was determined using the Kubelka-Munk equation (2)
32
and measured with a colorimeter spectrometer (SF600, Data color USA). The K/S was measured at a wavelength of 400 nm, which corresponded to the maximum absorbance (λ max) for the Hulela Zard dye on cotton fabric. Total variables for shade appearance (L*) Ladder/greener (a*) and yellowish/brownish (b*) have been taken for selected fabrics.
33
Three separate measurements were taken of each sample, and the averages of those measurements were reported. The dyed fabric was initially chosen and then subjected to ISO standard testing procedures for light (ISO 105 B02), washing (ISO 105 C03), and rubbing (ISO 105 X12). In the light testing, a Xenon lamp was used, while neutral soap was employed for washing, and rubbing was done with 10 strokes. The resulting changes in color were measured in terms of color change (CC) and color stain (CS).
34
The water used in the research was deionized and distilled, and all of the chemicals employed were analytical grade.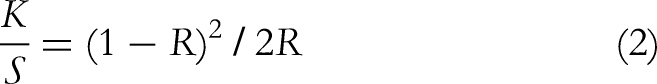
Here, K/S = color strength, K = absorption coefficients, S = scattering coefficients, and R = reflectance%.
Results
Optimization of the Dyeing Parameters
Extraction of biomaterials from plants using classic methods always has been found in trouble in terms of economy, labor energy and chemicals. Now in order to save solvents, money, time and cost, modern approaches have been adopted. Microwave treatment is a promising source for the isolation of colorants because these rays transfer energy into solvent molecules and raise their kinetics which in turn collapses with the plant cell wall. 35 During this solid-liquid interaction, the plant cell bonding is ruptured and colorant molecules evolved within a short period of time. 36 MW treatment time matters, because less time treatment does not allow plant binding to rapture whereas overheating may break molecules into byproducts which may get the maximum chance to sorb onto fabric. 37 Also, the surface tannin of fabric by MW rays plays its role because the peeled surface sorb dyes up to a maximum extent. The results after assessment of dyed fabric have been presented in Table 3. Our previous studies reveal that MW ray tunes the fabric surface for raising its sorption behavior. Also, previous studies reveal that MW rays cannot chemically change the nature of the functional site of fabric which is a positive feature of MW rays. 38 The results given in Figure 2(a-2c) show that using aqueous extract radiation up to 6 min has given good strength (K/S = 2.7756), whereas using basic extracted after treatment up to 6 min has also given better results (K/S = 3.60301). Using a basic medium, the treatment up to 6 min has given excellent results (K/S = 5.80143). Hence for getting excellent results stable medium is basic and radiation time for fabric tannin and extract is 6 min.

Influence of Microwave Radiation for Isolation of Colorant from Hulela Zard in Aqueous (a) Acidic (b) Basic (c) for Isolation of Colorant and its Dyeing of Cotton.
Assessment of Dyed Fabric.

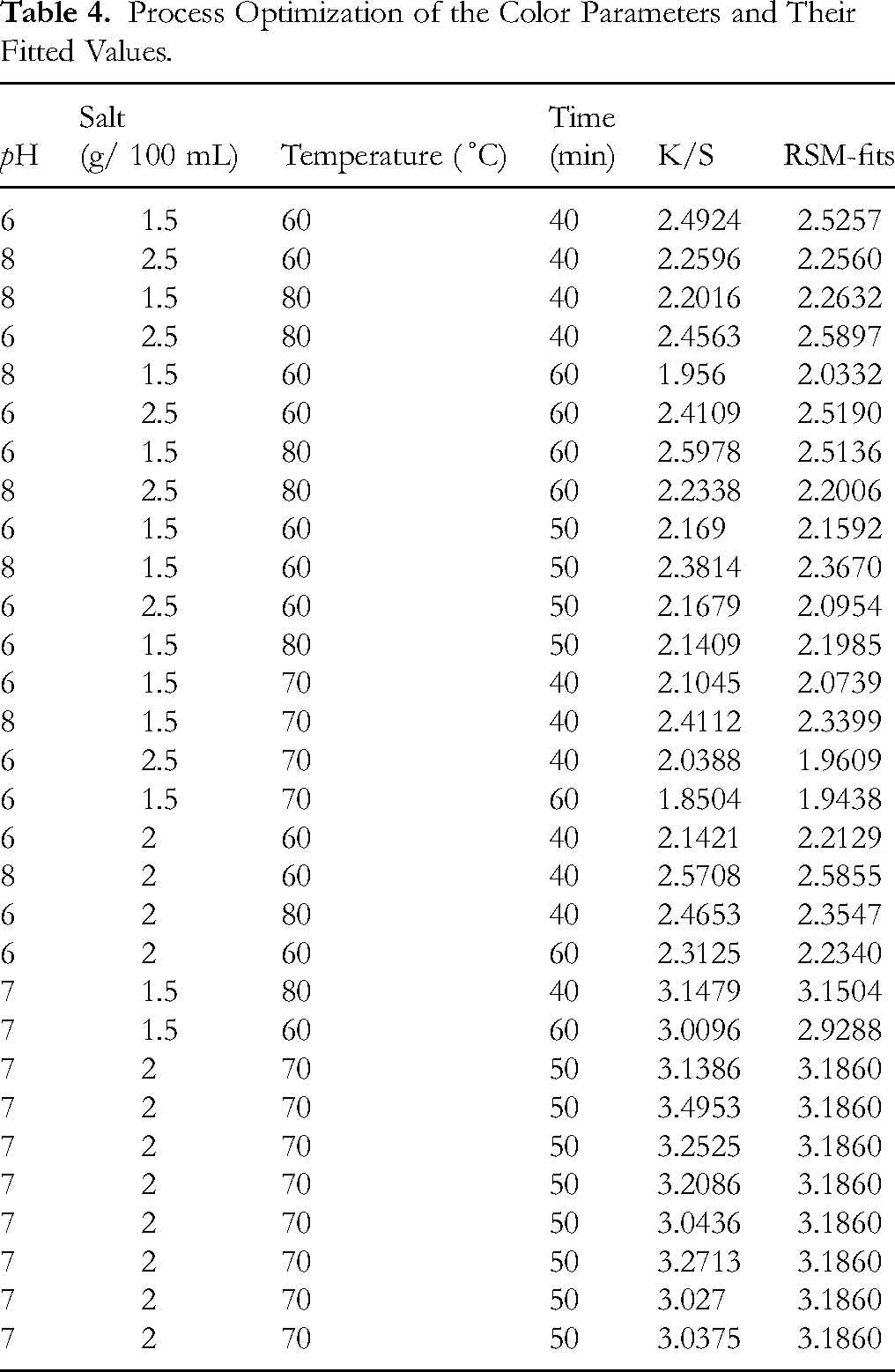
The results in Table 2 are based upon the values of Table 4; it shows that pH is linearly playing a significant role in the response. At the same time all two-way interaction terms of the dyeing variables were observed to be highly significant and contribute significantly to the response (p < 0.01). Currently used second order polynomial model was found effective since the insignificant value of lack of fit (0.803) was found. The model was found to be highly significant as F-value was high (22.70) with smaller probability value (0.000). The coefficient of determination (R2 = 0.9549) which is high, indicates that 95.49% of the total variance was explained by the given model. The experiments conducted were observed to be precise and reliable on the basis of low value (0.1379) of standard deviation (SD). Hence the dyeing process parameters, namely dyeing time, extract pH, temperature, and salt concentration, at their particular levels, have shown a key role in achieving the desired color results.
Performance Evaluation of the Experimental Data by Using Analysis of Variance.

Here, DF: degrees of freedom, Adj. SS: adjusted sum of squares, Adj. MS: Adjusted mean square, p-value: probability value or significance value, and “*” indicates significant terms of the model.
Process Optimization of the Color Parameters and Their Fitted Values.
Graphical Performance Evaluation of Observed Responses (K/S) and Fitted Responses
In Figure 3 presents closeness of experimental K/S and its RSM-fits. Practical K/S values were used on the Y-axis and experimentation order on the X-axis. Purple rectangular and blue circular dots are for observed K/S and fitted values of the model respectively. From Figure 2, it can be noticed that observed and fitted values under the given model are closer and indicates that the given data is fitted well by RSM.
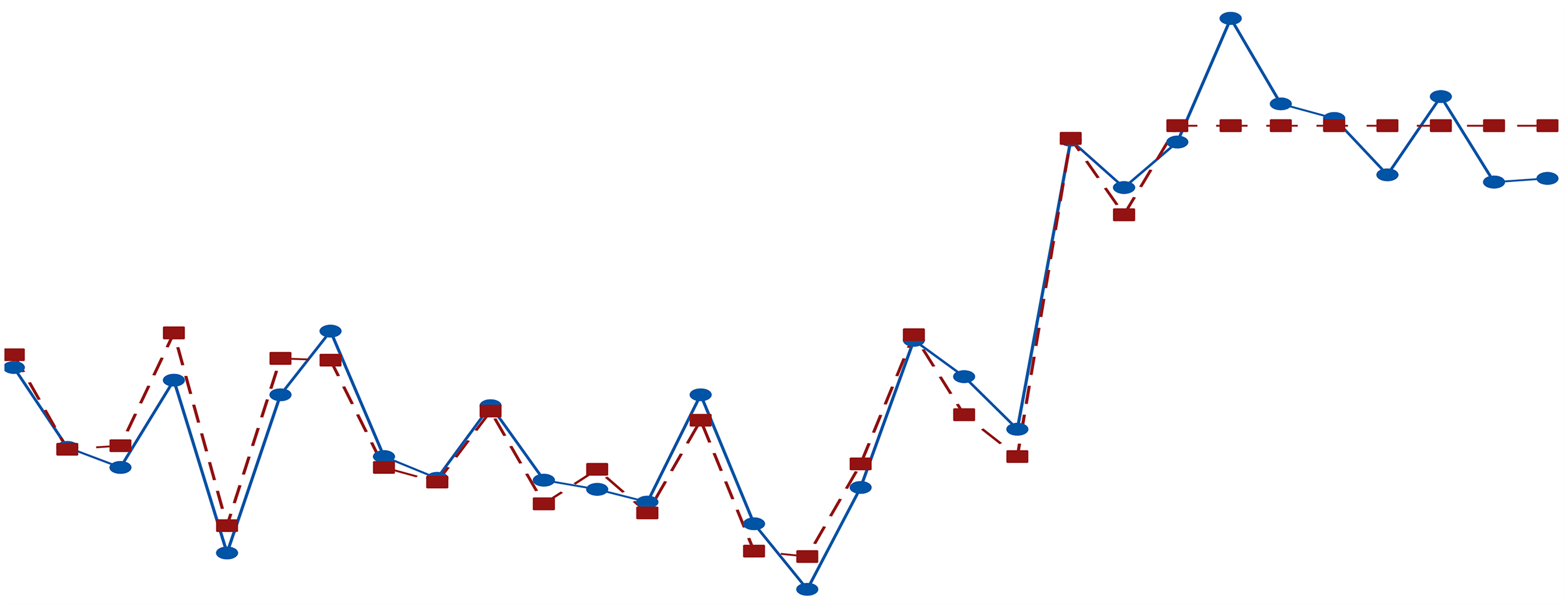
Model Adequacy of Observed Responses (K/S) and Fitted Model Values.
Mordanting is an important part of fixing the colorant firmly onto the fabric, it can be bio or chemical & depending upon its nature, the tint is developed. The new colorfast tint is due to the formation of natural dye complex onto fabric or by developing additional H-bonding through -OH of bio-mordant. 39 In this study, this chemical and these bio-mordants have been used before and after dyeing. It is shown that before dyeing, 2.5% madder, 1% pomegranate, 0.5% Aluminum, 2.5% Fe and 1.5 T.A.A. has given good yield and after dyeing 0.5% madder, 1% pomegranate, 0.5% Aluminum, 1.5% Fe and 2% T.A.A. has given good results (Figure 4a and b).
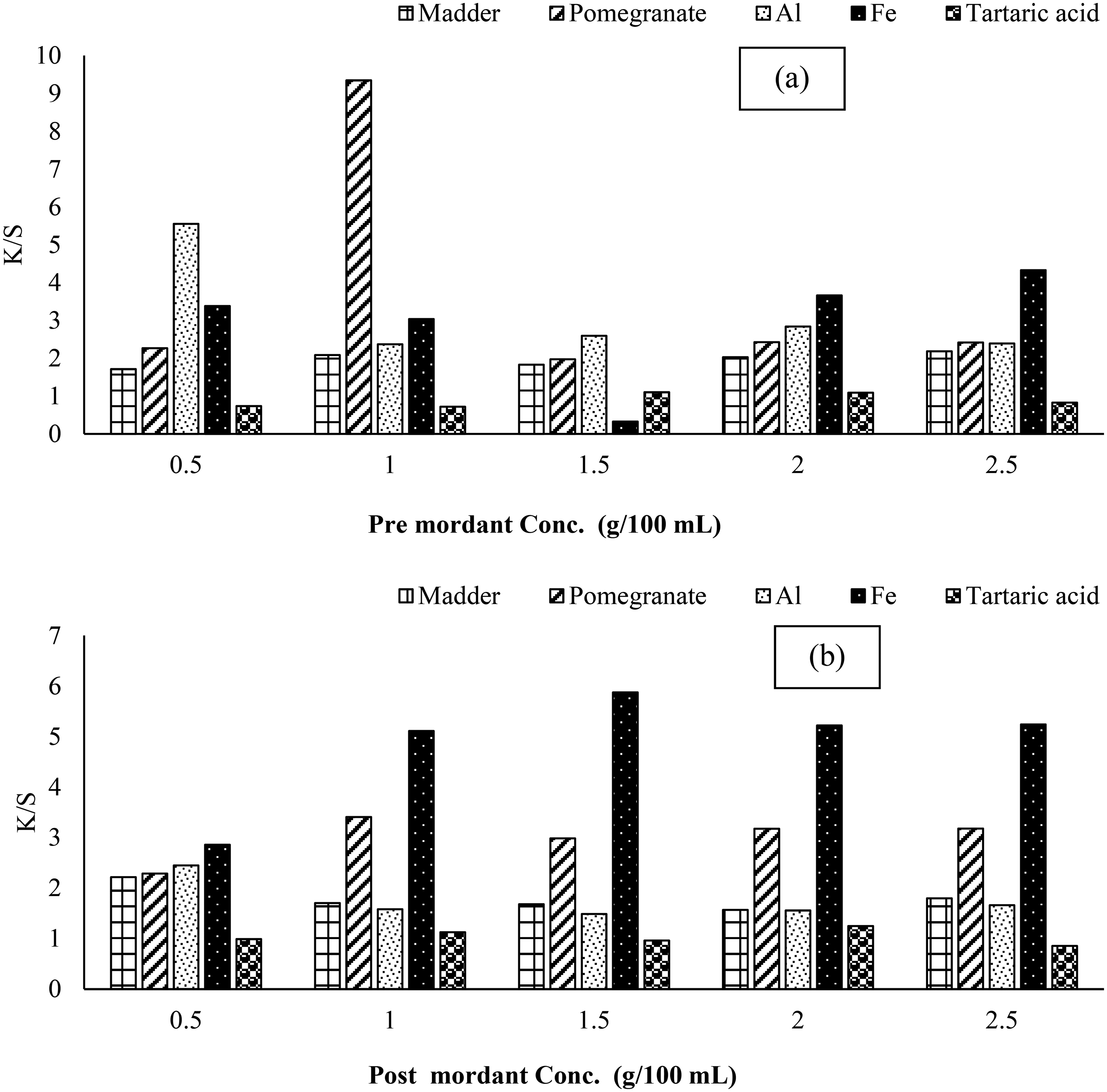
Effect of Pre (a) and Post (b) Chemical Mordants on Color Strength of Irradiated Cotton Fabric (RCF) with Irradiated Extract RE.
The dyeing mechanism of Hulela Zard dye extractions on cotton fabric involves multiple interactions (Figure 5). In the presence of chemical mordants, the metal ions form coordination complexes between the hydroxyl (-OH) groups of cellulose and the phenolic groups of tannins present in the dye. For bio-mordants, additional hydrogen bonding occurs through the -OH groups of bio-mordants, creating a stable bridge between the fabric and dye molecules. The tannin molecules from Hulela Zard form firm and stable complexes with both types of mordants, leading to the development of colorfast shades on cotton through these multiple bonding mechanisms. 40
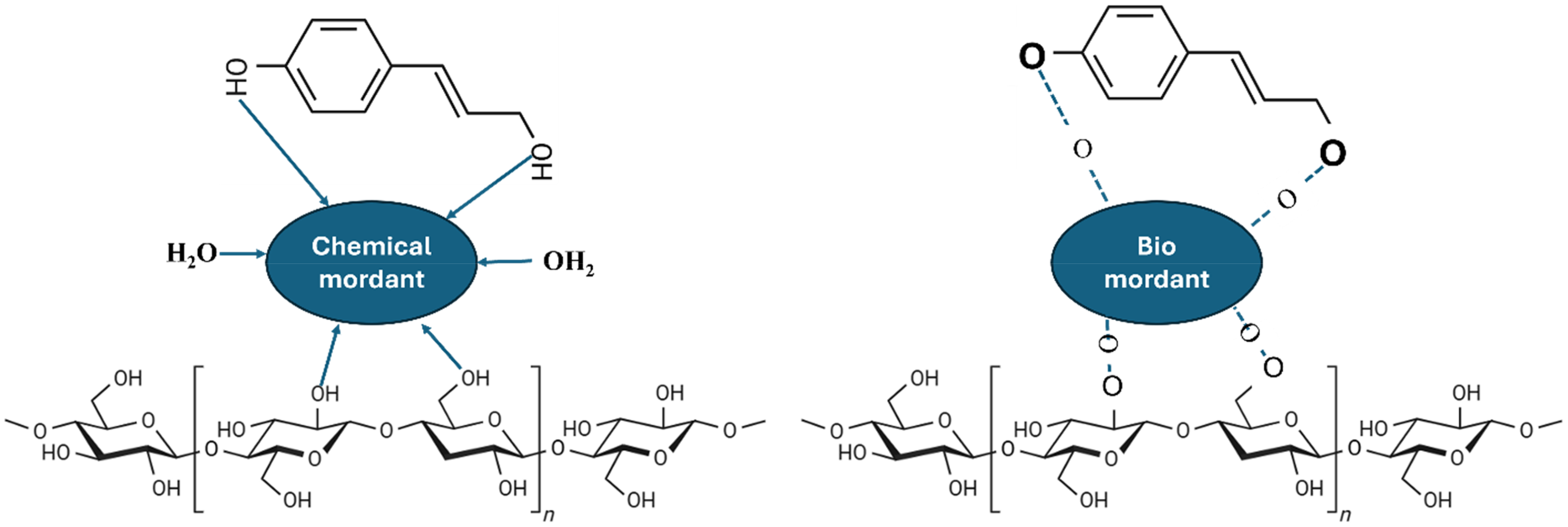
Hulela Zard Dye and Cotton Fabric Chemical Interactions in the Presence of Chemical and Bio Mordant.
Discussion
The colorfast properties of natural dyes are displayed upon proper mordanting done as per the weight of fabric (owf). The firm and stable metal dye complex, and nature of fabric and dye play their role in tint formation and resistance against foreign agencies such as heat, light, soaping crocking, etc. 41 The results given in Table 5 show that using selected mordanted condition, the moiety shades developed are brighter redder and yellowish in tone. The fastness properties found poor before mordanting whereas after mordanting found good to excellent. Dry and wet fabrics were also found good. Hence the selected number of mordants has given the colorfast and sustainable use of mordants in fewer amounts has made the process greener and eco-friendly.
Fastness Grading of Cotton Fabric Dyed with Hulela Zard Before and After Mordanting at Selected Conditions.

Here, LF = light fastness, WF = washing fastness, DRF = dry rubbing fastness, WRF = wet rubbing fastness.
The fastness properties of the cotton fabric dyed with Hulela Zard extract were evaluated according to the ISO standards, demonstrating promising results. The fabric showed a good light fastness rating of 4-5 and wash fastness rating of 4-5. Rubbing fastness ratings were 4-5 for dry and 3-4 for wet, with slightly lower wet rubbing fastness still within acceptable ranges. 42 These results suggest the fabric's suitability for clothing, home textiles, light-use upholstery, and accessories. While caution is advised for wet abrasion-prone uses, the dyed fabric meets industry standards, showcasing Hulela Zard as a sustainable natural dye with fastness properties comparable to synthetic dyes.
Based on the comparative analysis shown in Table 6, this study using Hulela Zard dye with microwave-assisted dyeing shows superior performance compared to previous research. The color strength (K/S) value of 5.80 achieved in this work is significantly higher than other natural dyes like Ficus amplissima (K/S 3.60) and Acacia catechu (K/S 2.97). 43 Additionally, our dyeing process demonstrates excellent colorfastness properties (LF-5, WF-5, DRF-5, WRF-5) compared to other studies which generally showed lower fastness ratings in the range of 3-4. Even compared to synthetic reactive dye J4 (K/S 5.02), our natural dye exhibits better color strength and fastness properties while being more environmentally sustainable.
Comparative Analysis of the Performance of Dyed Cotton Fabric Using Different Natural Dyes and Dyeing Methods.

Here, LF = light fastness, WF = washing fastness, DRF = dry rubbing fastness, WRF = wet rubbing fastness.
Overall, the study demonstrates several ecologically friendly advancements in coloring and extraction techniques. This study showed that when compared to conventional extraction methods, the MAE approach offers significant improvements. Compared to traditional methods, MAE reduces extraction time, energy consumption, and solvent usage. For instance, the extraction method took only 6 min, whereas conventional methods usually take several hours. 48 This time savings leads to increased efficiency and energy conservation.
The study was also able to utilize less water and chemicals to improve dyeing process by the use of RSM. By reducing dependency on metallic mordants, the use of bio-mordants like pomegranate and madder highly enhances the process's environmental sustainability. In addition, compared to conventional high-temperature dyeing techniques, the use of low-temperature dyeing (70 °C) allows for energy conservation. 13
Therefore, the study clearly combines MAE, RSM optimization, and bio-mordants in a comprehensive approach for the extraction and application of Hulela Zard dye, even though previous research has looked at these specific components separately. This combination represents an incremental but significant step towards more sustainable textile dyeing techniques.
Limitations of the Study
While the study successfully demonstrated the eco-friendly extraction and application of natural dye from Terminalia chebula L. using microwave-assisted extraction (MAE), certain limitations should be acknowledged. First, the research focused on a single type of natural dye and limited combinations of mordants. Expanding the range of bio-mordants or evaluating different plant species may provide broader insights into dyeing processes. Second, the study primarily assessed dyeing performance on cotton fabric, which may limit the generalizability of the findings to other textile materials like wool or synthetic fibers. Further research on different substrates would be necessary to confirm the effectiveness of this method across various textiles. Additionally, while the response surface methodology provided useful insights for optimizing dye extraction parameters, the scalability of the MAE process to an industrial level remains untested. Potential challenges in large-scale production, such as equipment costs, energy consumption, and consistency in dye yield, need further exploration. Finally, the study did not assess the long-term environmental impact or biodegradability of the mordants, and dyes used, which is essential for a comprehensive evaluation of the sustainability of this process. To ensure its viability for industrial applications, further studies should examine the scalability of this procedure, including tests with larger fabric samples (10-50 g). In addition, to provide a more thorough understanding of the characteristics and performance of the Hulela Zard dye, future research should include chemical characterization of the crude extract, identification of particular dye components present, and assessment of color stability over extended periods of time.
Conclusion
In the current scenario, microwave treatment day by day plays an important role in natural dyeing of fabrics. The present study demonstrated the K/S optimization for Hulela Zard plant, the important color parameters such as pH, salt, temperature and time of dyeing concentration were used on cotton fabric. Current work provides a road map to reduce the environmental impact and achieve sustainability in textile industry by utilizing eco-friendly methods in natural dyeing. By implementing these techniques, we can advance to cleaner and greener textile sector, living pleasantly nature. For the process optimization RSM was used as an advanced technique. The subset design (S4 + 2S1) was implemented in this study to obtain experimental data for chemical processes, offering a novel approach in this context. It explored experimental space in different ways so that it may provide some additional information to facilitate the researchers. This design exhibits favorable statistical properties, including sufficient experimental trials (replications) to estimate pure error during experimentation. It also satisfies multiple optimality criteria such as D-, A-, and G-optimality. These properties enhance the design's efficiency and provide researchers with a reliable framework for assessing experimental outcomes. This new subset design may be an addition to the designs toolkit already been used to facilitate the researchers. For future studies, such statistical tools are useful in finding suitable parameters for dyeing of cotton, wool, silk, polyester and blend fabrics with natural plant colorants. Also, bio-mordants should be used to make processes green and ecofriendly. Additionally microwave rays have made environment more clean and effective. These rays should be used for isolation of plant dyes with effective yield.
Footnotes
Acknowledgement
This work is part of PhD studies. The authors are grateful to the Department of Statistics and Department of Applied Chemistry, Government College University Faisalabad for providing the facilities for smooth running of experiments and assessment of the data.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by Department of Statistics and Department of Applied Chemistry, Government College University Faisalabad, Pakistan.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies on human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.

