Abstract
Aim
The aim of this study was to evaluate the in vitro antidiabetic activity of Clerodendrum splendens and to identify its bioactive constituents.
Methods
The plant was extracted with ethanol using the cold maceration method and the crude extract (CE) was partitioned in a separating funnel into n-hexane (HF), ethyl acetate and aqueous ethanol fractions. Phytochemical screening of CE was done following standard methods while HF which gave the best activity was further sub-fractioned using Vacuum Liquid Chromatography into final 5 sub-fractions as guided by analytical thin layer chromatography. The in-vitro alpha-amylase assay was done using standard procedure and concentrations of 1.0, 0.5, 0.25 and 0.125 mg/mL were prepared for all the groups including the standard drug, acarbose in triplicates. The process was repeated for the sub-fractions using 0.5, 0.25 and 0.125 mg/mL and the sub-fraction that gave the best activity, VHF-2 was analyzed using chromatography-mass spectrometry (GC-MS).
Results
The phytochemical analysis revealed the presence of glycosides, tannins, alkaloids steroids and terpenoids and flavonoids in CE. The results of the alpha-amylase assay revealed that HF produced the highest percentage inhibition of 63.97% and half-maximal inhibitory concentration (IC50) of 0.49 mg/mL when compared with other fractions. The IC50 of VHF-2 and the acarbose were 4.19 and 0.485 mg/mL respectively and GC-MS characterization revealed the presence of 14 major compounds.
Conclusion
The ethanol leaves extract of C. splendens holds potential antidiabetic agents which can be used in the management of diabetes mellitus.
Introduction
There is a growing interest in the increasing prevalence of non-communicable diseases (NCD) in Nigeria. These diseases are non-infectious health conditions that cannot be spread from person to person. NCDs are long-lasting diseases that often proceed slowly and are caused by a confluence of behavioral, physiological, environmental, and hereditary variables.1,2 One such disease of increasing prevalence in Nigeria is diabetes mellitus (DM); a complicated metabolic condition when the pancreas fails to make enough insulin (type 1 diabetes mellitus), or a condition where a person's system fails to react to insulin as necessary (type 2 diabetes mellitus). 3
DM is among the most serious issues in public health which is regarded as the twenty-first century's hidden plague. 4 It is a long-term condition that progressively affects several organs within the body. This disease condition develops gradually, with symptoms showing up several years after its first manifestation with serious and permanent complications developing simultaneously. 5 Although DM is manageable, but the uncontrolled condition of the disease raises the risk of other various complications, particularly cardiovascular-related complications such as hypertension and hyperlipidemia. The reduction in physical activity and obesity contributes significantly to the etiology of cardiovascular diseases and according to estimates, persons who have both hypertension and diabetes have double risk of developing cardiovascular complications. Hyperlipidemia, which is primarily linked to early coronary heart disease, is common in diabetic people and it also causes an increase in insulin resistance. Furthermore, obesity is a significant risk factor for heart disease and is linked to insulin resistance. More significantly, being sedentary increases the risk of insulin resistance and cardiovascular disease and it has been proven that regular exercise and weight loss lower blood pressure, prevent type 2 diabetes, and lower the risk of cardiovascular complications. 6
DM is characterized by abnormal hyperglycemia, abnormalities of lipoproteins and metabolic rate, change in intermediate metabolism of the main nutrients in diet and malfunction in the enzymes that scavenge reactive oxygen species. 7 Abnormal increase in thirst, frequent urination, weight loss and excess ketones in urine are some of the early symptoms of diabetes which are caused by an inadequate amount of insulin produced by the pancreatic islet, or by a hereditary and/or acquired impairment in insulin production. 8 The kidney, heart, and eyes are among the vital organs of the body that are harmed by chronic hyperglycemia. 9
According to the literature, approximately 50 000 plant species are said to have medicinal properties, 10 and many of the approved pharmaceuticals used today have their roots in the herbal traditions of many civilizations. Galegine (isoamylene guanidine), a toxic bio-isolate from Galega officinalis has been reportedly used to treat DM in the seventeenth Century. 11 Modern methods of analysis have made it possible to isolate the active principles of medicinal plants and, these active principles have been obtained synthetically in the laboratory to produce medicines. 12 The knowledge of the chemical structure of galegine was used to develop a synthetic biguanide antidiabetic drug, metformin (dimethyl biguanide) that is less toxic, effective and safe. 11 Thus, herbal therapy was scientifically validated to provide a base for the synthesis of contemporary medications like quinine, morphine, aspirin and digitoxin.13,14
The knowledge of plant-based medications has progressively grown and been transmitted, laying the groundwork for several traditional medical systems around the world. 15 Local traditional medicine, including herbalism, is the sole form of healthcare available to people in most developing countries, particularly in rural regions. In contrast, alternative medicine, which includes the use of nutritional supplements, is mostly sold in the industrialized world under the guise of traditional medicine. Until today, the direct use of medicines from medicinal plants is apparently displaced in modern medicine. 16 Modern medicine needs pharmaceutical industries, that use active principles from plants and/or their derivatives as raw materials in their production. 17
C. splendens is a native of tropical western Africa and is popularly known as a flaming glory bower plant belonging to the genus Clerodendrum and family Verbenaceae. 18 It is an evergreen, twining vine with woody stems that can reach lengths of 10 to 12 feet. It has shiny dark green, elliptic to ovate leaves and can grow up to 7 inches in length.18,19 It has salverform, vibrant red flowers that bloom in dense terminal clusters up to 4-5 inches long, and with an abruptly spreading corolla. 19 C. splendens is a woody climbing shrub that has a stem with a quadrangular section that is often seen for its striking inflorescences that seldom adapt to warm temperate conditions. 19 The leaves of C. splendens are used to treat shingles, spleen in children, asthma, rheumatism, ulcers and malaria. 20 The plant is used in Ghana to treat vaginal thrush, bruises, wounds and various skin infections, 21 and the antimicrobial and wound healing properties of C. splendens have been established.22,23
This study is aimed at investigating the in vitro antidiabetic activity of C. splendens and identifying some bioactive constituents that contributed to the plant's potential antidiabetic activity.
Methodology
Collection of Plant Material
The fresh Clerodendrum splendens leaves sample was collected in August 2023 from Nsukka in Enugu North and was verified by Mr A. O. Ozioko, a taxonomist from the International Centre for Ethnomedicine and Drug Development. (InterCEDD), A voucher specimen with number esut/cog/202008 was deposited at the herbarium of the Department of Pharmacognosy, Enugu State University of Science and Technology, Agbani, Enugu, Nigeria.
Preparation of Plant Extract
The plant sample was air-dried under a shade at room temperature for ten days and after which a suitable grinder was used to grind it into a coarse powdered form, which was then stored in an airtight container in preparation for extraction. 350 g of the powdered plant sample was macerated in 1500 mL of ethanol for 48 h and then filtered with the aid of a muslin cloth. The marc was further rinsed with ethanol to remove all the extractable constituents after which the Whatman (No1) filter paper was used to filter the extract. The filtrate was concentrated at 40 °C using a rotary evaporator and the concentrated extract was further dried in a water bath set at 40 °C. 21 A weighing balance was used to obtain the weight of the dried extract, from which the percentage yield was calculated. 75 g of the ethanol extract was partitioned in a separating funnel into n-hexane (HF), ethyl acetate (EF) and aqueous ethanol (AEF) fractions. The HF which gave the best activity was further sub-fractioned using Vacuum Liquid Chromatography. Mobile phases of 100% n-hexane, n-hexane: ethyl acetate (7:3), n-hexane: ethyl acetate (1:1), n-hexane: ethyl acetate (3:7), 100% ethyl acetate, ethyl acetate: ethanol (7:3), ethyl acetate: ethanol (1:1) and 100% ethanol were used to elute the system. Silica gel (200-400 mesh) was used as the stationary phase and analytical thin layer chromatography (TLC) was used to merge sub-fractions with similar constituents resulting in the final five sub-fractions.
Phytochemical Screening
The analyses were carried out based on outlined standard procedures.21,24,25
Test for alkaloids: 20 mL of 3% sulfuric acid in 50% ethanol was added to 2 g of the extract and heated in a boiling water bath for 10 min, cooled and filtered. 2 mL of the filtrate was tested with a few drops of Mayer's reagent (Potassium mercuric iodide solution), Dragendorff's reagent (Bismuth potassium iodide solution), Wagner's regent (iodo- potassium iodide solution) and Picric acid solution (1%). Alkaloids give a milky precipitate with Mayer's reagent; a reddish-brown precipitate with Wagner's reagent; a yellowish precipitate with picric acid and a brick-red precipitate with Dragendorff's reagent.
Test for saponins: 5 mL of the filtrate was diluted with 15 mL of distilled water and shaken vigorously. The formation of stable froth shows the presence of saponins.
Test for flavonoids: 4 mL of the filtrate was shaken with 1 mL of dilute ammonia solution. The layers were allowed to separate. A yellow color in the ammoniacal layer shows the presence of flavonoids.
Test for tannins: A few drops of ferric chloride were added to the filtrate. Formation of a greenish black precipitate shows the presence of tannins.
Test for glycosides: 10 mL of 1% sulfuric acid was added to 0.5 g of the powder in a test tube and boiled for 15 min in a water bath, then cooled and neutralized with 20% potassium hydroxide solution. 10 mL of a mixture of equal volume of Fehling's solution I and II was added and boiled for 5 min. A brick-red precipitate shows the presence of glycosides.
Test for carbohydrates: 0.5 g of the plant extract was boiled with 2 mL of distilled water and filtered. A few drops of α- naphthol solution in ethanol (Molisch reagent) was added to the filtrate and concentrated sulfuric acid was then gently poured down the side of the test tube to form a lower layer. A purple interfacial ring shows the presence of carbohydrates.
Test for reducing sugars: 5 mL of a mixture of equal volume of Fehling's solution I and II were added to 5 mL of the extract and then heated in a water bath for 5 min. A brick red precipitate shows the presence of reducing sugar.
Test for proteins: Two drops of million's reagent were added to a little part of the filtrate in a test tube. A white precipitate shows the presence of proteins.
Test for steroids: 1 mL of concentrated sulfuric acid was carefully added to 0.5 mL of chloroform extract in a test tube to form a lower layer. A reddish-brown interface shows the presence of steroids.
Test for terpenoids: 0.5 mL of the chloroform extract was evaporated to dryness in a water bath and heated with 3 mL of concentrated sulfuric acid for 10 min in a water bath. A gray color shows the presence of terpenoids.
Alpha-Amylase Inhibitory Test
Alpha-amylase inhibitory activity test refers to the minimally modified standard protocol by Wulandari et al 26 Concentrations of 1, 0.5, 0.25 and 0.125 mg/mL were prepared for crude extract (CE), fractions, and the standard drug, acarbose in triplicates. 26 While 0.5, 0.25 and 0.125 mg/mL were prepared for the sub-fractions and the acarbose. Test tubes were kept as the negative control and blank. 200 uL of the samples and the acarbose were introduced into clean test tubes except the ones labeled as control after which 50 uL of the enzyme solution (alpha-amylase) was pipetted into the test tubes holding the samples, the acarbose, and the control. The volumes of the various solutions in the test tubes were adjusted to 1 mL using a phosphate buffer solution at pH 6.9. The test tubes were incubated for 15 min at 37 °C. They were then allowed to cool and 200 uL of starch substrate solution was added to the solution, mixed gently, and incubated for 10 min at 37 °C. 800 uL of dinitro salicylic acid reagent (DNSA) was added to the various test tubes, mixed gently and then heated in boiling water for 15 min. After allowing the test tubes to cool at room temperature, the absorbances were then measured at 540 nm using a UV-visible Spectrophotometer. 27
Quantification by Gas Chromatography-Mass Spectrophotometry (GC-MS)
The sample which gave the best activity was further evaluated using Gas Chromatography-Mass Spectrophotometry (GC-MS). High energy electrons (70 eV) were used in an electron ionization system for spectroscopic detection by GC-MS. The carrier gas, which had a flow rate of one milliliter per minute, was pure helium gas (99.995%). With a holding time of 10 min and an increase rate of 3 °C per minute, the starting temperature was set between 50 and 150 °C. At last, the temperature was raised to 300 °C at the rate of 10 °C per minute. Splitless injection of one microliter of 1% of the extract diluted with the right solvents was performed and based on the peak area created in the chromatogram, the relative number of chemical components in the extract was displayed in percentages. 28
Identification of Chemical Constituents
The identification of compounds in the most active sub-fraction was based on GC retention time in an HP-5MS column and the comparison of the spectra with standard computer software data (Replib and Mainlab data of GC–MS systems).
Statistical Analysis
Statistical analysis of the results was done by one-way analysis of variance (ANOVA) using GraphPad software followed by Tukey comparison for test of significance (P < .05). The half maximal inhibitory concentration (IC50) which is the concentration of the drug that inhibited 50% of the alpha-amylase enzyme was extrapolated and computed from a graphical plot of percentage inhibitions versus concentrations using Microsoft Excel software, 2010 version.
Results
Results of Phytochemical Screening
The CE showed a diverse array of phytochemicals such as glycosides, alkaloids, saponins, tannins, proteins, carbohydrates, steroids, terpenoids and flavonoids as shown in Table 1.
Phytochemical Screening of the Ethanol Crude Extract of C Splendens.

Key: + = Present.
Results of Alpha-Amylase Enzyme Inhibitory Assay
The results of percentage inhibition of alpha-amylase enzyme by CE, HF, EF and AEF, and the acarbose groups are represented in Table 2 and Figures 1 to 4. Table 3 represent the results of IC50 of the treatment groups.
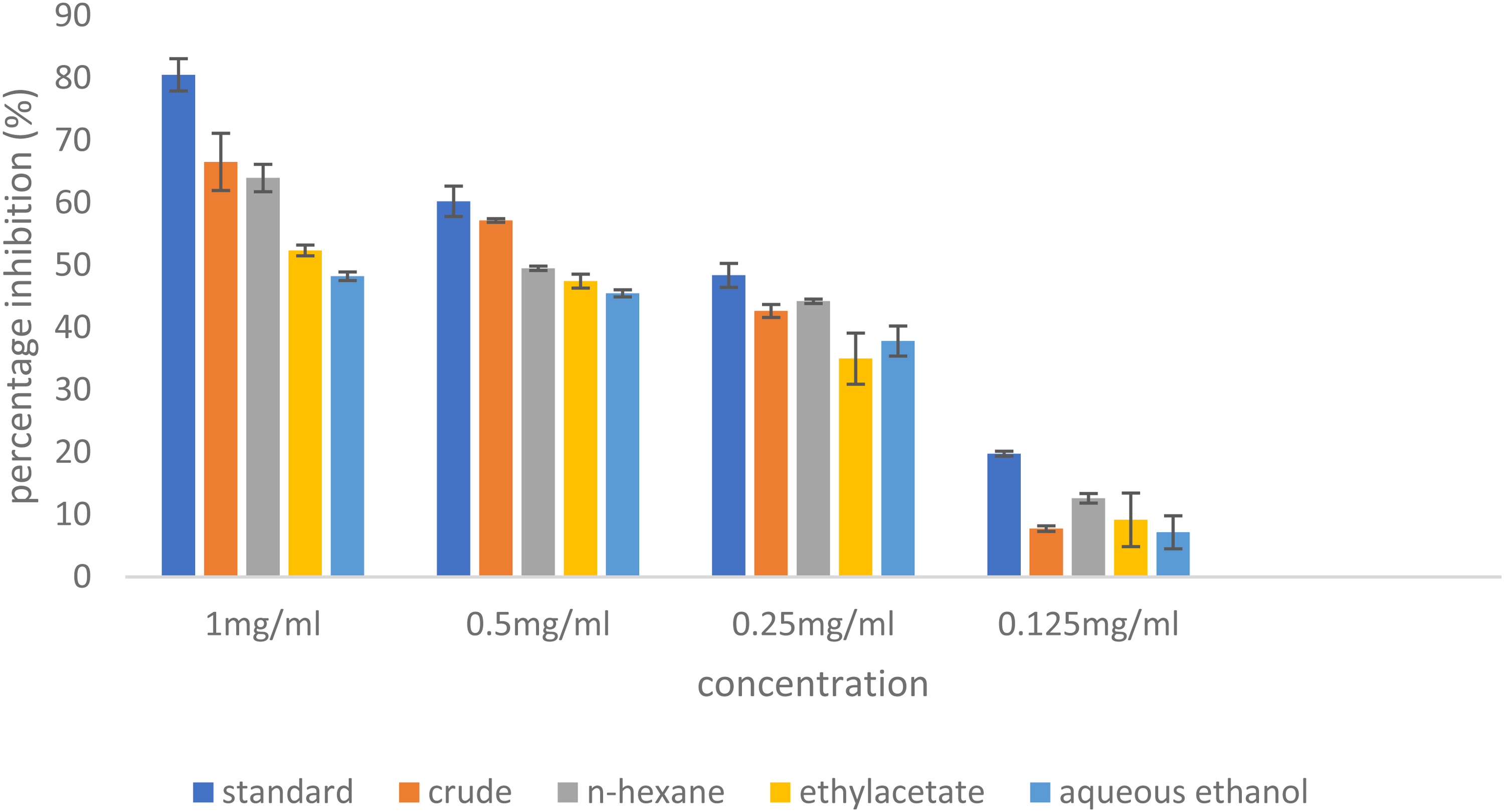
Inhibitory Effect of Standard Drug, Crude Extract, and Fractions of C. splendens Leaf Extract on Alpha-Amylase Enzyme Against.
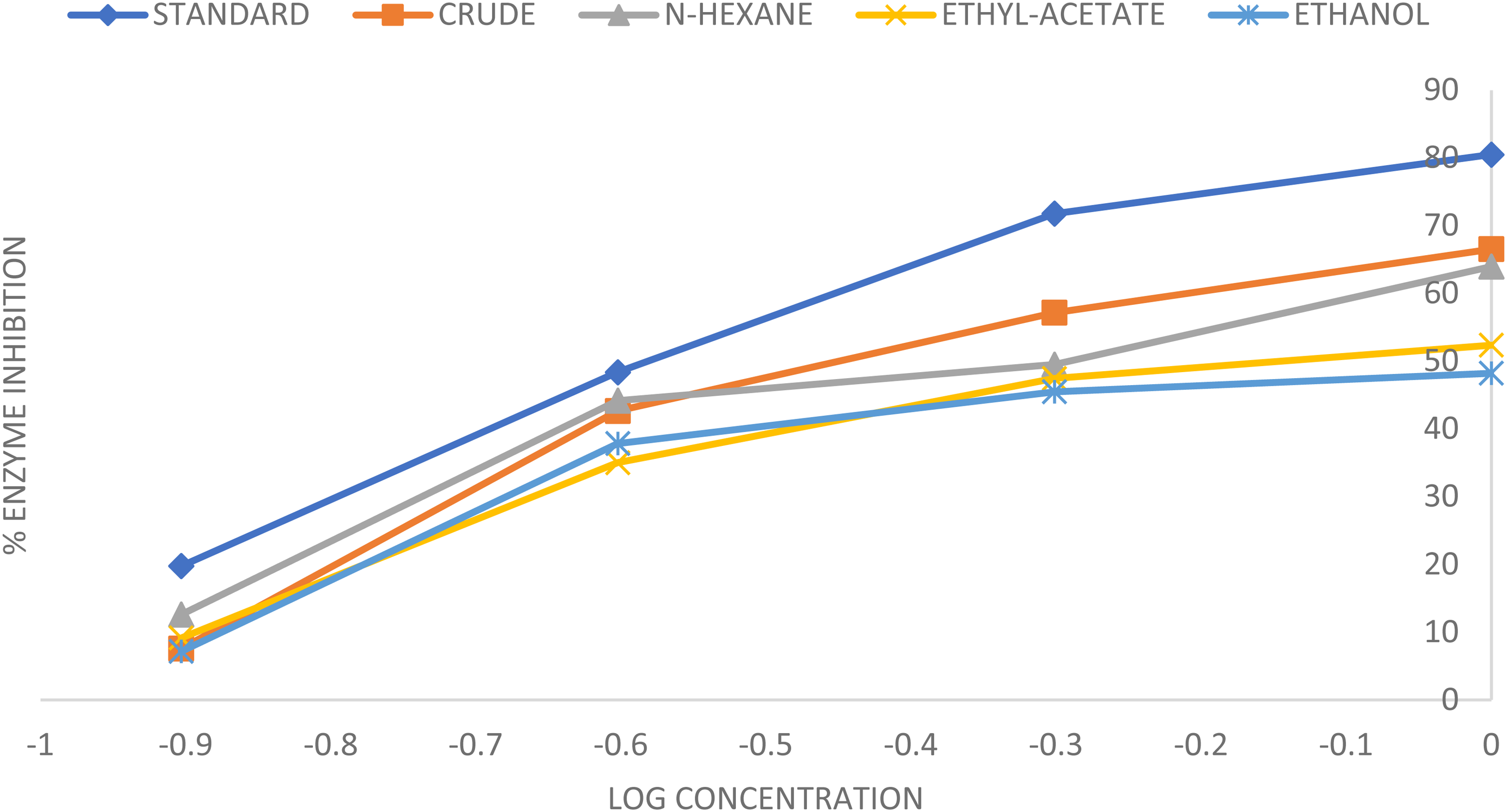
A Chart Showing the Change in Mean % Inhibition of the Alpha-Amylase Enzyme Activity with Change in Log Concentration of the Various Treatment Groups.

Graphical Representation of Percentage Inhibition of Standard Drug at Various Concentrations.

Graphical Representation of Percentage Inhibition Against Concentrations Used to Determine the IC50 of HF.
The Percentage Inhibition of Alpha-Amylase Enzyme.
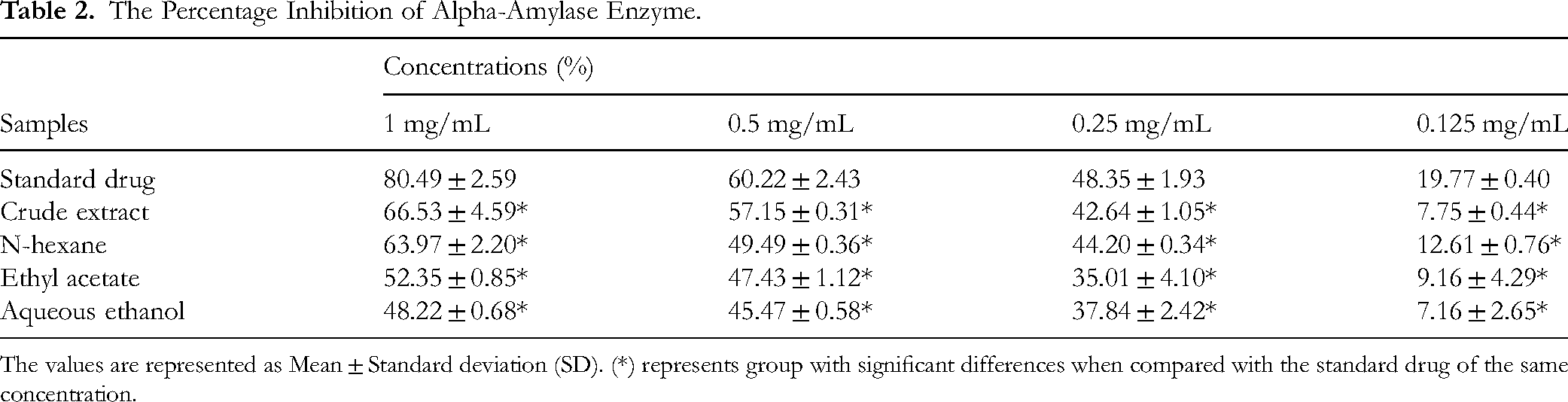
The values are represented as Mean ± Standard deviation (SD). (*) represents group with significant differences when compared with the standard drug of the same concentration.
The Results of Half Maximal Inhibitory Concentration (IC50).

Tables 4 and 5, and Figures 5 and 6 represent the results of percentage inhibition of alpha-amylase enzyme by the HF sub-fractions and the IC50 of HF sub-fractions and the acarbose. The data were analyzed using a one-way ANOVA at P < .05.

Alpha-Amylase Enzyme Inhibitory Activities of the Standard Drug and HF Sub-Fractions.
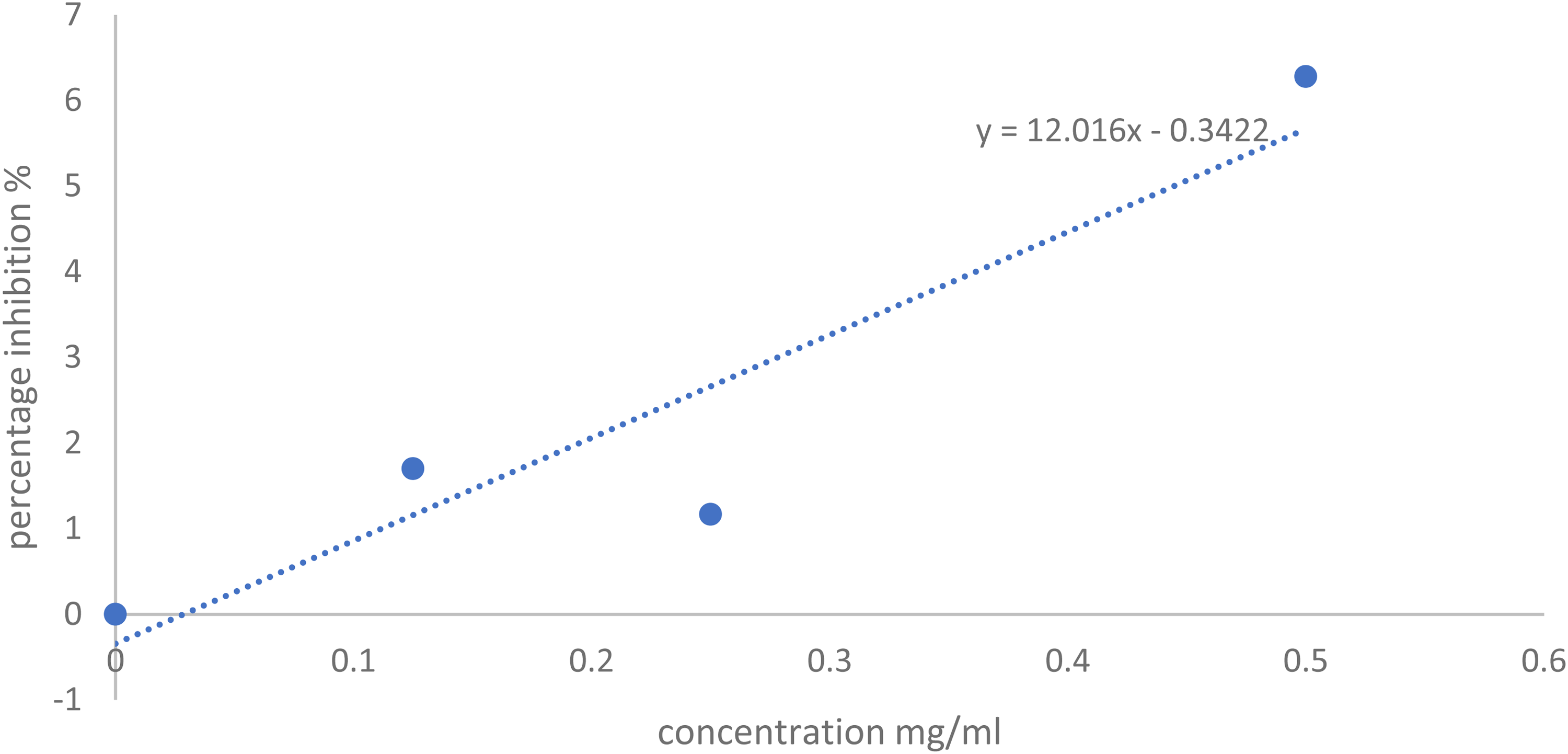
Graph of Alpha-Amylase Percentage Inhibition Power Against Concentration for VHF-2 (SUB-1).
The Results of Percentage Inhibition of HF Sub-Fractions.

The values are represented as Mean ± SD. (*) stands for groups with significant differences when compared with the standard drug of the same concentration.
Half-Maximal Inhibitory Concentration (IC50) of HF Sub-Fractions.

Results of GC-MS Analysis
The GC-MS analysis of the most active sub-fraction, SUB-1 revealed the presence of 14 compounds as represented in Table 6 and Figure 7 which, by matching their spectra and their retention time, were identified with those in the in-built library of NIST 14 as shown in Table 6 and Figure 7.

GC-MS Chromatogram Showing the Phytoconstituents in SUB-1.
Compounds Identified by GCMS from SUB-1.

Discussion
Phytochemical Screening
The investigation into the phytoconstituents of C. splendens leaves aimed at elucidating its potential antidiabetic properties. The CE showed a diverse array of phytochemicals such as glycosides, alkaloids, saponins, tannins, proteins, carbohydrates, steroids, terpenoids and flavonoids as shown in Table 1. There are alkaloids that are recognized as hypoglycemic agents. 29 For example, clinical investigations and experimental models of DM have shown effectiveness with berberine, a quaternary ammonium salt belonging to the protoberberine group of isoquinoline alkaloids. 30 Notably, HF retained some key constituents, emphasizing its potential significance in the plant's antidiabetic activity. The presence of steroids, alkaloids, and terpenoids in this extract aligns with their known bioactive properties, suggesting their contributions to the observed antidiabetic effects. It is an established fact that some pentacyclic triterpenes have an improving potential for insulin sensitivity, dyslipidemia, and lipoprotein lipase expression.31–34
Alpha-Amylase Enzyme Inhibitory Assay
The results of percentage inhibition of alpha-amylase enzyme showed significant antidiabetic actions of CE, HF, and the acarbose groups when analyzed in a two-way ANOVA at P < .05. All the treatment groups showed increased levels of alpha-amylase enzyme inhibition with increasing concentrations as represented in Table 2. The acarbose gave the highest percentage inhibition of 80.49% at a concentration of 1 mg/kg and 19.77% at 0.125 mg/kg as shown in Figure 3, while among the various fractions, HF exhibited the highest level of inhibition of 63.97% at 1 mg/kg and 12.61% at 0.125 mg/kg concentrations shown in Table 2 and represented in Figure 4. These inhibitory activities were represented in graphical representation as shown in Figures 1 and 2 for more emphasis and clarity. A key digestive system enzyme, pancreatic α-amylase hydrolyzes starch into a variety of oligosaccharide units, which are then broken down into glucose by α-glucosidase. After digestion of carbohydrate, the resultant glucose is absorbed into the circulatory system, raising the amount of postprandial hyperglycemia (PPHG). 35 Therefore, plant extracts with the ability to inhibit the alpha-amylase enzyme may be useful in reducing the complications that are linked to PPHG in type 2 diabetes. 35
The IC50 was determined for all the treatment groups and is presented in Table 3. The acarbose gave the best IC50 value of 0.49 while CE and HF gave IC50 values of 0.604 and 0.64, respectively. The EF, and AEF groups gave the least IC50 activities of 0.79 and 0.85, respectively. It is an established fact that a lower IC50 value signifies better potency. Hence the acarbose, CE and HF groups gave significant alpha-amylase inhibitory potentials at various concentrations.
The observed variations in inhibitory effects among HF sub-fractions highlighted the complexity of the fraction. The HF sub-fraction, VHF-2 (SUB 1) at 0.5 mg/kg showed 6.28% inhibition, which decreased to 1.17% at 0.25 mg/kg and 1.70% at 0.125 mg/kg as represented in Table 4. The VHF-2 gave significant antidiabetic activity when compared with other sub-fractions. Although the reported activity was lesser than the standard drug and the significant difference between VHF-2 and the standard drug at the same concentrations, denoted by the asterisk (*), emphasizes that VHF-2 percentage inhibitory effects are notably weaker than those of the acarbose. It is pertinent to note that acarbose is a single pure compound, while VHF-2 have a mixture of compounds, hence the concentration of the antidiabetic bioactive compound in VHF-2 in each treatment concentration may be relatively comparable to acarbose concentrations. The other n-hexane sub-fractions (VHF 1, 3, 4, and 5) displayed minimal inhibitory effects, with percentages often close to or at zero percentage.
Table 5 and Figure 5 show the IC50 of the standard drug and individual sub-fractions. The standard drug and VHF-2 gave IC50 values of 0.49 and 4.19, respectively. There were no visible activities seen in the other sub-fractions. These findings suggest that the HF sub-fractions (VHF 1, 3, 4, and 5) may not possess insignificant or no anti-diabetic activity under the conditions tested and the statistical analysis, indicating a significant difference from the standard drug, acarbose at corresponding concentrations, underscores the importance of considering the potency and specificity of the sub-fractions.
GC-MS Analysis
The major class of compounds identified in the extract are more of fatty acid esters: limonene, 2-propanol,1-(1-methyl-2-(2-propenyl)ethoxy)-, 4,8,12-tetraoxaeicosan-1-ol, butylated hydroxytoluene, undec-10-ynoic acid dodecyl ester, hexadecanoic acid methyl ester, butanoic acid, 4-nitro,9,12-octadecadienol, heptadecanoic acid, 16-methyl methyl ester, L–ribulose, tetrakis(trimethylsilyl)ether pentafluorobenzyl oxime(isomer 2), tri(propylene glycol)propyl ether and oleic acid. The mass spectrum of each compound showing their respective fragment ion (m/z) are presented in Table 6. The GC-MS chromatogram showing the phytoconstituents of SUB-1 is presented in Figure 7 while the chemical structures of some of the identified compounds are presented in Figure 8. Limonene is a monoterpene that has been reported to exhibit antidiabetic, antioxidant, anticancer, anti-inflammatory, cardioprotective, and gastroprotective properties. 36 Limonene inhibits protein glycation by stabilizing protein structure and preventing alpha-beta transitions, 37 and the in vivo antidiabetic study by Bacanlı et al 38 reported the antidiabetic activity of d-limonene in streptozotocin-induced diabetic rats. The hexadecanoic acid methyl ester is a palmitic acid group compound that binds a methyl group and has been reported to have antidiabetic and antioxidant activities and help to decrease cholesterol levels.39,40 The hexadecanoic acid methyl ester and oleic acid have been reported to be responsible for the antidiabetic activity of Carica papaya seed extract. 41 Nevertheless, oleic acid has been reported to inhibit inflammatory cytokine TNF-α and increases insulin secretion in type II diabetes. 42 Su et al also reported that oleic acid and linoleic acid successfully inhibited α-glucosidase enzyme activity. 43 One of the limitations of this study is the inability to ascertain the mechanism of action of the individual bioactive antidiabetic constituents.

Some Chemical Structures Obtained from the Dereplication Result of Sub-Fraction (VHF-2).
Conclusion
In conclusion, this study unveils the antidiabetic potential of C. splendens leaves, particularly within its n-hexane fraction. The investigation found a diverse array of phytochemicals in the ethanol extract, with HF keeping crucial constituents such as terpenoids. The GC-MS analysis provided a detailed profile of bioactive compounds in the HF sub-fraction, SUB-1 that was found to contain compounds such as oleic acid, limonene and hexadecanoic acid methyl ester that have been reported in some publications as antidiabetic agents. The observed complexity among different compounds within the sub-fraction emphasized the need for further exploration to fully harness the therapeutic potential of C. splendens in managing and treating other diseases.
Footnotes
Acknowledgements
We sincerely acknowledged the Almighty God for his guidance.
Ethical Considerations
Experimental animals were not used in this study. Hence, ethical approval is not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Limitations of the Study
Lack of funding deprived us from isolating bioactive compound(s) from the C. splendens leaf.
Statement of Human and Animal Rights
Human or animal subjects were not used in this research work.
Statement of Informed Consent
Statement of informed consent is not applicable in this study.
