Abstract
Keywords
Introduction
Ultraviolet (UV) light is the primary contributor to skin wrinkles, erythema, aging, and skin tumors. Sunlight comprises three UV wavelengths: UVA (320-400 nm), UVB (275-320 nm), and UVC (230-275 nm). Atmospheric ozone absorbs 100% of UVC, over 90% of UVB, and nearly none of UVA. Consequently, UVA and UVB are the primary contributors to UV-induced skin damage.1,2 Acute UV exposure typically results in skin erythema, sunburn, tanning, and other manifestations that impact external appearance. Prolonged UV radiation induces skin laxity and pigmentation, contributing to photoaging. Concurrently, the accumulation of epigenetic alterations can transform normal cells into precancerous lesions, ultimately culminating in skin cancers, including basal cell carcinoma (BCC), cutaneous squamous cell carcinoma (cSCC), and melanoma.3,4 In recent years, the incidence of skin diseases caused by UV radiation has increased, especially in high-altitude areas. Research indicates that the intensity of UV radiation in high-altitude regions is 1.47 to 2.50 times greater than in lowland areas, with ultraviolet reflectance on sunny days and during snow accumulation being even more pronounced. Furthermore, the incidence of skin diseases is projected to rise by 25% to 32% for every 2% increase in UV radiation5,6; this indicates that those in plateau regions are more susceptible to UV-induced skin damage, necessitating a significantly greater need for protection against UV harm compared to those in plain locations. Consequently, it is crucial to investigate the pathophysiology of UV damage, identify effective and safe treatment options, and create new drugs for the theoretical study of UV damage and the clinical treatment of plateau skin damage diseases.
Sanhuang Erxiang Powder (SHEX) from the Qing Dynasty, Wu-Tang “Wen Bing Tiao Bian,” consisting of Rheum officinale Baill. (rhubarb), Coptis chinensis Franch. (Chinese goldthread), Phellodendron amurense Rupr. (amur cork tree), Boswellia carterii Birdw. (frankincense), and Commiphora myrrh (Nees) Engl. (myrrh), has the effect of clearing heat and detoxification. It is clinically used in the treatment of herpes zoster, contact dermatitis, and other skin diseases with oozing and vesiculation. Contemporary pharmacological research has demonstrated that the herbs in SHEX have anti-UV-damaging properties. Silveira et al 7 investigated the photoprotective effect of rhubarb extract on UV-induced human epidermal melanocytes and found that rhubarb extract inhibited the activities of interleukin-1α (IL-1α), tumor necrosis factor α (TNF-α), α-MSH, and tyrosine kinases in UV-irradiated melanocytes, thus exerting a photoprotective effect. Berberine is the most representative and most abundant alkaloid compound in Coptis chinensis and Phellodendron amurense, demonstrated to diminish UV-induced matrix metalloproteinase (MMP)-1 expression and enhance type I procollagen fiber expression, thereby exhibiting an anti-aging effect.8,9 In addition to non-ionizing radiation, Coptis chinensis has a protective effect against skin damage caused by radioactive ionizing radiation. 10 In addition, sunscreens prepared from myrrh extracts can prevent UV-induced skin damage. 11 Boswellic acid, a pentacyclic triterpenoid derived from Boswellia serrata, and its topical formulations can mitigate skin photoaging. 12 The findings indicate that SHEX possesses research potential concerning UV damage. However, whether SHEX can be used to prevent UV-induced skin damage has not yet been demonstrated in a study.
In recent years, the application of network pharmacology based on systems biology and biological network equilibrium to explain diseases has been widely used in the study of traditional Chinese medicine (TCM), and as the relationship between the chemical composition and efficacy of natural medicines has been gradually revealed by more and more scholars, the use of network pharmacology to explore the potential links between compounded Chinese medicines and related diseases has become a commonly used tool. 13 Therefore, the present study aims to predict the molecular network mechanism of SHEX for UV damage using a network pharmacology approach and validate the therapeutic effect of SHEX on UV-induced skin damage and potential mechanisms of action using UV-induced human fibroblasts and SD rat skin damage model experiments. To provide theoretical support for the further development and clinical application of SHEX as an anti-ultraviolet damage drug.
Materials and Methods
Network Pharmacology
Collection of SHEX Active Ingredients
The Traditional Chinese Medicine Systematic Pharmacology Database and Analysis Platform (TCMSP) (https://old.tcmsp.com/tcmsp.php), Swisstargetprediction (http://swisstargetprediction.ch/), and Pharmmapper (https://www.lilab-ecust.cn/pharmmapper/) databases search all active compounds of five herbs in SHEX. The above constituents could be connected to its human target gene biomolecules directly or indirectly by the UniProt database (http://www.uniprot.org/).
Collection of UV-Induced Skin Damage Diseases Target Genes
The keywords “sunburn,” “photoaging,” “light aging,” and “light allergy” were respectively entered into the following five databases: GeneCards (https://www.genecards.org/), DisGeNET (https://www.disgenet.org/), PharmGkb (https://www.pharmgkb.org/), TTD (https://db.idrblab.net/ttd/), and CTD (https://ctdbase.org/) for searching for all target genes. The search was performed to obtain potential targets of UV damage, combine the search results, and remove duplicates to obtain the relevant UV damage targets.
Protein-Protein Interaction (PPI) Network and key Target Acquisition
The Venny 2.1 platform (https://bioinfogp.cnb.csic.es/tools/venny/) was utilized to illustrate the intersecting targets of SHEX and UV damage. The common targets of each therapeutic disease were uploaded to the STRING online platform (http://string-db.org), with the species restricted to “Homo sapiens,” and interactions between target proteins were retrieved at a confidence level of 0.400 to generate the PPI network diagram. The CytosSape 3.10.1 software was utilized to create a “TCM-ingredient-target” network for SHEX. The built-in “Analyse Network” of CytoScape 3.10.1 assessed the network topology metrics of SHEX in the context of UV damage treatment, including degree, betweenness, and closeness, to identify the principal target proteins and the key active constituents through which SHEX exerts its pharmacological effects.
Gene Ontology (GO) and KEGG Pathway Enrichment Analysis
To further explore the pathways of disease occurrence, the core targets were submitted to the DAVID database (https://david.ncifcrf.gov/) for GO function enrichment and KEGG pathway enrichment analyses, in which the GO analyses were divided into three parts, annotating the roles of drug therapy disease-interacting target proteins in gene function in terms of biological process (BP), cellular component (CC), and molecular function (MF); the KEGG analyses were mainly for pathways, which aim to elucidate the main signaling pathways of drug therapy diseases. Selected the top 10 GO terms and the top 20 KEGG pathways with the smallest P value and imported them into the bioinformatics mapping website (http://www.bioinformatics.com.cn/) to draw bar graphs and bubble graphs.
Experimental Verification of the Anti-UV Damage Effect of SHEX and Possible Mechanism
Medicinal Herbs and Reagents
Rhubarb (batch number: 220501), Chinese goldthread (batch number: 220501), amur cork tree (batch number: 220501), frankincense (batch number: 220503), and myrrh (batch number: 220412) were purchased from Anhui Aikang (Anhui, China). Ethanol and methanol (chromatographic purity) were purchased from Thermo Fisher Scientific (Carlsbad, USA). Dimethyl sulfoxide (DMSO) was acquired from Sigma-Aldrich (St. Louis, USA). Minimum essential medium (MEM), fetal bovine serum (FBS), phosphate-buffered saline (PBS), and penicillin/streptomycin (P/S) were obtained from Gibco BRL (Gaithersburg, MD, USA). Cell counting kit 8 (CCK-8) was purchased from Elabscience Biotechnology (Hubei, China). Catalase (CAT; 22070435N), superoxide dismutase (SOD; 22070435N), glutathione peroxidase (GSH-Px; 22070435N), and hyaluronic acid (HA; 22070435N) assay kits were acquired from Shanghai Kexing (Shanghai, China). The Trizol kit (15596-026) was obtained from Invitrogen (Carlsbad, USA). HiScript® II Q Select RT SuperMix for qPCR (R233) and SYBR Green Master Mix (Q111-02) were purchased from Vazyme Biotechnology (Nanjing, China). Taq Plus DNA Polymerase and DL2000 DNA Marker (MD114-02) were acquired from TIANGEN BIOTECH (Beijing, China). Radiation immunoprecipitation assay (RIPA) lysate (P0013B) and phenylmethylsulphonyl fluoride (PMSF) protease inhibitor (P105539) were obtained from Beijing Solarbio (Beijing, China). The BCA protein concentration measurement kit (P0010) was purchased from Beyotime Biotechnology (Shanghai, China). The antibodies used are as follows: rabbit GAPDH polyclonal antibody (AF7021) was produced by Affinity Biosciences (Jiangsu, China). Rabbit p38 MAPK polyclonal antibody (14064-1-AP) and rabbit MMP-1 polyclonal antibody (10371-2-AP) were purchased from Proteintech Group (Hubei, China).
Preparation of SHEX Extracts
Five herbs, rhubarb, Chinese goldthread, amur cork tree, frankincense, and myrrh, were weighed in the mass ratio of 2: 2: 2: 1: 1. Frankincense and myrrh were processed by steam distillation to obtain volatile oils and residues. Concentrate the above extract and add 95% ethanol to concentrate the solution to precipitate and filter; the filtrate is concentrated and dried to a constant weight to obtain the aqueous extract. The rhubarb, Chinese goldthread, and amur cork tree add a volume concentration of 60% ethanol solution for ethanol heating and reflux extraction, filtration, and filtrate concentration of recycling ethanol, and then dried to constant weight to obtain the alcoholic extract, mixed with the aqueous extract. The extract of SHEX was dissolved in DMSO for cellular administration. For easy administration to animals, the prepared extracts were made into a cream and applied to the skin surfaces of rats. Cream preparation: weigh the appropriate proportion of vaseline, stearic acid, liquid paraffin, glyceryl monostearate, span-60, and volatile oil mixed well at 80°C as the oil phase; weigh the appropriate proportion of tween-80, glycerol, ethylparaben, paste extract, and water mixed well at 80°C as the aqueous phase; add the aqueous phase to the oil phase; and emulsify quickly to obtain the required cream. The extraction of SHEX and the preparation of creams are carried out with reference to the patented method. 14
Cell Culture, pre-Treatment with SHEX, and UVB Radiation
The BJ human dermal fibroblast cell line was purchased from iCell Bioscience Inc. (Shanghai, China) and cultured in MEM medium containing 10% FBS and 1% P/S. Cells were cultured in a humidified atmosphere of 95% air and 5% CO2 at 37°C. The SHEX extract was dissolved in DMSO and diluted to different concentrations with the medium. The cells were divided into five groups: control group (Control), UVB irradiation group (Model), SHEX low dose group (2.5 μg/mL; L), SHEX medium dose group (5 μg/mL; M), and SHEX high dose group (10 μg/mL; H). Fibroblasts were subjected to UVB irradiation to induce UV damage, as previously described.15,16 For the L, M, and H groups, cells were first treated with a medium for 24 h and then with a drug-containing medium for 24 h. Then, the drug solution was discarded, PBS was added, and the cells were placed under irradiation at a distance of 30 cm from the UVB lamps (TL20 W/01, PHILIPS, Netherlands). The UVB lamps had emission spectra between 311 and 313 nm (peak at 311 nm), and the irradiation intensity was set at 0.425 mW/cm2. The irradiation duration was 15 min, resulting in a total irradiation dose of 382.5 mJ/cm2. The UV output was calibrated before each experiment using an LS125UV UV radiometer (Shenzhen Linshang Technology Co., Ltd, Shenzhen, China), and the radiation dose was verified at several time points during the exposure period. The control group was neither exposed to UVB nor treated with drugs, and the model group was irradiated only without drug administration. After irradiation, PBS was replaced with fresh medium, and the cells were incubated for 24 h in an incubator and then processed for subsequent analysis.
Cell Viability Assay
The CCK-8 kit was used to determine the effect of different concentrations of SHEX and different doses of UVB on cell viability. After cell treatment, the supernatant of the cell plate was absorbed and incubated with CCK-8 working solution at 37°C. The optical density (OD) values were measured by reading the enzyme marker at 450 nm.
Animal Modeling and Grouping
Male Sprague-Dawley (SD) rats with a weight of 180-220 g were obtained from the Laboratory Animal Centre of Xi’an Jiaotong University Medical College (certificate No. 2018-001). The rats were housed in a specific pathogen-free (SPF) animal facility with unrestricted access to food and water. The facility maintained a 12-h light/dark cycle and a constant temperature of 24°C. Rats were randomly divided into seven groups: control group (Control), UV model group (Model), vitamin E group (10 mg/cm2; Pos), SHEX-free cream group (Base), SHEX cream low-dose group (1.64 mg/cm2; L), SHEX cream medium-dose group (3.28 mg/cm2; M), and SHEX cream high-dose group (6.56 mg/cm²; H). A rat model of UV-induced skin damage was established following established literature protocols.17,18 During the experimental period, rats were periodically anesthetized using a respiratory anesthesia machine. After the anesthesia, the long hairs on the backs of the rats were shaved with a small animal shaver, with the shaved area measuring 4 cm × 5 cm. The skin on the backs of the rats was exposed to UVA lamps and UVB lamps. The UVA lamps (TLK40 W/10R, PHILIPS, Netherlands) have emissions in the range of 300-460 nm (peak at 365 nm), and UVB lamps (TL20 W/01, PHILIPS, Netherlands) have emission spectrums between 311 and 313 nm (peak at 311 nm). The lamps were placed 20 cm away from the skin, and rats were placed in special rat cages. The lamps were allowed to warm up for 15 min before each irradiation, and the positions of the rat boxes were rotated every 5 min during the irradiation process to ensure that the rats in each cage received the same light intensity. The UV output was calibrated before each experiment using an LS125UV UV radiometer, and the radiation dose was verified at several time points during the exposure period. The irradiation intensity was 0.3 mW/cm2 for UVB and 1.3 mW/cm2 for UVA, and the starting intensity of UV was based on the minimum erythemal dose (MED) (180 mJ/cm2 for UVB and 780 mJ/cm2 for UVA). Irradiation was performed every other day, and the irradiation intensity was increased by 0.5 MED every 4 sessions until reaching 2.5 MED. Then, a constant dose of 2.5 MED was administered for 16 additional irradiations. The overall radiation dose was 10.44 J/cm2 for UVB and 45.22 J/cm2 for UVA. The control group was not treated after shaving; the model group was irradiated with UV rays only without any drug treatment; and the base group, the pos group, the L group, the M group, and the H group were given the corresponding drugs on the back 30 min before each irradiation. The skin on the back of the animals was observed daily for blisters or vesicles during the modeling process, and irradiation was stopped for 2-3 days if the above symptoms appeared and then continued after the symptoms had disappeared until modeling was successful. The reporting of this study conforms to ARRIVE 2.0 guidelines. 19
General Observation
The body mass of rats was recorded weekly, and macroscopic changes in rat skin were photographed and documented.
Tissue Sampling and Sample Processing
After the last irradiation, the rats were anesthetized by isoflurane inhalation, and after anesthesia, the rats were fixed on the operating table, and whole blood was collected by the abdominal aortic method for routine blood examinations. Then, the damaged skin on the back was quickly removed, and the subcutaneous fat was removed and rinsed in cold saline. The tissue was wiped dry with filter paper and then divided into three small parts: one part was fixed with neutral formaldehyde for hematoxylin-eosin (HE) staining, one part was fixed with 3% glutaraldehyde for transmission electron microscope (TEM), and one part was stored in the refrigerator at −80°C for enzyme-linked immunosorbent assay (ELISA), real-time polymerase chain reaction analysis (RT-PCR), and western blot analysis.
Routine Blood Examinations
Whole blood was taken, and routine blood parameters such as white blood cells (WBC), hemoglobin (HGB), red blood cells (RBC), and platelets (PLT) were measured using an XN-10 automatic hematology analyzer (Sysmex Corporation, Japan).
HE Staining
The neutral formaldehyde-fixed samples were taken, washed, and dehydrated with anhydrous ethanol from low to high concentration, treated with xylene for 2 h for transparency, and then transferred to paraffin solution, placed in a warm box overnight, stained with hematoxylin-eosin solution, and dehydrated. The sections were sealed with neutral resin, and the pathological changes were observed under a Pannoramic 250 digital sectional scanner (3DHISTECH, Hungary).
TEM
Samples prefixed with 3% glutaraldehyde were refixed with 1% osmium tetroxide, dehydrated in series with acetone, infiltrated in Epox 812 for a longer time, and embedded. The semithin sections were stained with methylene blue, and the ultrathin sections were cut with a diamond knife and stained with uranyl acetate and lead citrate. Sections were examined with a JEM-1400FLASH transmission electron microscope (JEOL, Japan).
ELISA Assay
The cells were removed from the culture medium, washed once with PBS, and then lysed by adding lysis solution and centrifuged to take the supernatant. Skin tissues kept at −80°C were homogenized by adding a cold saline ice bath and centrifuged to take the supernatant. And then the enzyme antioxidant activities were detected by SOD, GSH-Px, CAT, and HA ELISA kits, and the operation was carried out according to the instructions of the kits.
RT-PCR Analysis
A total of 100-200 mg of skin tissue was homogenized, and the total RNA was extracted using a total RNA extraction kit. The RNA solution was evaluated using a NanoDrop 2000c spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA), and cDNA was synthesized using a HiScript II kit. The relative mRNA expression of the target gene was analyzed using a Bio-Rad CFX-ligated real-time PCR system (Bio-Rad Laboratories, Hercules, CA, USA). The PCR products were pre-denatured at 95°C for 10 min, followed by 40 cycles of amplification at 95°C for 15 s and 60°C for 60 s. Calculations were performed using the 2−ΔΔCt method. Amplification primers for the target gene and internal reference GAPDH gene are listed in Table 1.
Design and Sequence of the Primers.

Western Blot Analysis
A small amount of clipped skin tissue block was placed in an EP tube and homogenized in an automated homogenizer by adding RIPA lysis solution (containing PMSF). After homogenization, it was placed on ice for 30 min for complete lysis. After lysis, the supernatant was centrifuged at 12 000 rpm for 5 min at 4°C, and the protein concentration was determined using the BCA kit. The protein samples and BSA standards were diluted with PBS, the diluted protein samples and standards were added to the microtiter plate, and the BCA working solution was added, mixed well, and incubated at 37°C for 30 min under light protection, and the absorbance value at 568 nm was measured by the enzyme counter, and the concentration of the samples was calculated based on the concentration of the standard proteins and the corresponding OD value. The sample concentration was diluted to a uniform value, and the supernatant of the extracted protein was mixed with 5× bromophenol blue loading buffer at 95°C for 10 min until translucent. The protein samples were electrophoresed on a 10% SDS polyacrylamide gel and then transferred to a polyvinylidene difluoride (PVDF) membrane. The PVDF membrane was immersed in TBST containing 5% skimmed milk powder and sealed on a room-temperature shaker for 2 h. After discarding TBST, incubated with primary antibodies GAPDH (1:1000), p38 MAPK (1:1000), and MMP-1 (1:1000) diluted with skimmed milk powder, respectively, and then incubated with a shaking machine at 4°C overnight. The PVDF membrane was washed with TBST five times, and the secondary anti-rabbit antibody HRP was diluted with TBST. The PVDF membrane was immersed in the secondary antibody incubation solution and incubated in a shaking bed at room temperature for 2 h. The PVDF membrane was washed with TBST five times. ECL reagent in the enhancement solution and stabilized peroxidase solution according to the ratio of 1:1 mixing, drop and fully cover the surface of the PVDF membrane, and place it into the ChemiScope 6100 Chemiluminescence Imager (CLINX Qinxiang Co., Ltd, Shanghai, China) for imaging. The optical density values of the target bands of each sample were analyzed using Image J software.
Statistical Analysis
Data are expressed as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used to compare the groups, and two-by-two comparisons were made using the least significant difference method (LSD). A test level of P < .05 was considered statistically significant. All data were processed using SPSS 22.0 statistical software.
Results
Network Pharmacology-Based Analysis
Screening of Active Compounds and Targets in Herbal Medicine
A total of 120 compounds (16 from rhubarb, 14 from Chinese goldthread, 37 from amur cork tree, 8 from frankincense, and 45 from myrrh) from SHEX were screened in the TCMSP, Swisstargetprediction, and Pharmmapper databases. Of these, phellamurin_qt, beta-sitosterol, stigmasterol, quercetin, palmidin A, berberine, coptisin, obacunone, dihydroniloticin, worenine, magnograndiolide, and palmatin are common constituents of drugs, which suggests at least two or more drugs contain this constituent. The search also yielded 3002 potential targets, which were combined and narrowed down to a total of 707 targets.
Acquisition of Potential Targets for SHEX Intervention in UV Damage and Construction of Component-Target Network Diagrams
GeneCards, DisGeNET, PharmGKB, TTD, and CTD databases were used to search for UV damage-related targets, and 7244 targets were obtained by merging and de-emphasizing, and the targets were plotted on a Venn diagram, resulting in 651 overlapping targets in SHEX and disease (Figure 1A). The 651 overlapping targets were imported into CytoScape 3.10.1 software to construct a “TCM-component-target” network diagram (Figure 1B), in which the purple, pink, light green, dark green, and yellow squares represent the active ingredients of rhubarb, Chinese goldthread, amur cork tree, frankincense, and myrrh, respectively. Circles represent common constituents, blue represents common targets of drugs and diseases, and connecting lines represent interactions between constituents and targets. The more components correspond to the target, the higher the importance of the target. CytoScape network analysis was performed to obtain the core components of SHEX for the treatment of UV damage, and the top 10 core components were screened according to degree, as shown in Table 2.

Construction Results of Herb-Component-Target Analysis Diagram. (A) Venn Diagram of the Intersection of SHEX and UV Damage Disease. (B) SHEX Network Diagram of “TCM-Component-Target” for the Treatment of UV Damage Disease. (DH: Rhubarb, HL: Chinese Goldthread, HB: Amur Cork Tree, RX: Frankincense, MY: Myrrh).
Summary of SHEX Main Bioactive Components.

PPI Network Construction, GO Enrichment Functional Analysis, and KEGG Pathway Enrichment Analysis
The drug and disease common targets were imported into the STRING online platform for analysis, and the highest confidence level of 0.400 was taken to obtain the interactions between the target proteins. The PPI network graph was constructed using the CytoScape software platform (Figure 2A). The number of nodes in the SHEX therapeutic targets gene interactions network was counted about the number of connections to other nodes, with a total of 646 nodes and 17 042 edges. The core target proteins were screened according to the degree value, and the results showed that the targets of AKT1, TNF, IL6, TP53, ALB, SRC, IL1B, STAT3, EGFR, and CTNNB1 were highly ranked, indicating that these targets play an important role in SHEX intervention in UV damage. GO enrichment analysis yielded 3516 BP entries, mainly including drug response, peptidyl-tyrosine phosphorylation, etc; 201 CC entries, mainly including membrane raft, membrane microdomain, etc; and 381 MF entries, mainly including protein tyrosine kinase activity, protein serine/threonine kinase activity, etc An enrichment analysis by KEGG revealed 196 pathways, mainly involving the neuroactive ligand-receptor interaction pathway, the lipid and atherosclerosis pathway, and the MAPK signaling pathway. The top 10 entries of the GO enrichment analysis were plotted as color bar graphs (Figure 2B), and the top 20 entries of the KEGG pathways were plotted as bubble graphs (Figure 2C).

PPI, GO, and KEGG Diagram. (A) PPI Network Diagram of “SHEX-UV Damage” Intersection Target. (B) GO Functional Enrichment Analysis. (C) KEGG Pathway Enrichment Analysis Results.
Effects of SHEX Anti-UV Damage and Possible Mechanisms
SHEX Protects BJ Cells from UV-Induced Oxidative Damage
The cell viability assay showed that the cell viability decreased after UVB irradiation, and when the irradiation time was 15 min, the viability was 50%, so we determined that the irradiation time was 15 min (Figure 3A). It was also found that the concentration of SHEX at 2.5-10 μg/mL showed no significant cytotoxicity at 24 h (Figure 3B). Therefore, we fixed the concentrations of SHEX at 2.5, 5.0, and 10 μg/mL for further cell-based experiments. Microscopic observations revealed that SHEX reduced the number of UV-induced suspended and rounded wrinkled cells and improved cell morphology (Figure 3C). Determination of antioxidant enzymes such as GSH-Px, SOD, CAT, and HA in BJ cells revealed that the levels of antioxidant enzymes were reduced in BJ cells from the UV model group compared to the control group, suggesting that UV irradiation causes oxidative damage to the cells. After administration of SHEX extracts, compared to the model group, CAT content was significantly higher in groups L, M, and H (P < .05), GSH-Px content was significantly higher in groups M and H (P < .05), and HA content was significantly higher in group H (P < .05) (Figure 3D). These results suggest that SHEX has antioxidant capacity and protects BJ cells from photodamage induced by UVB radiation.
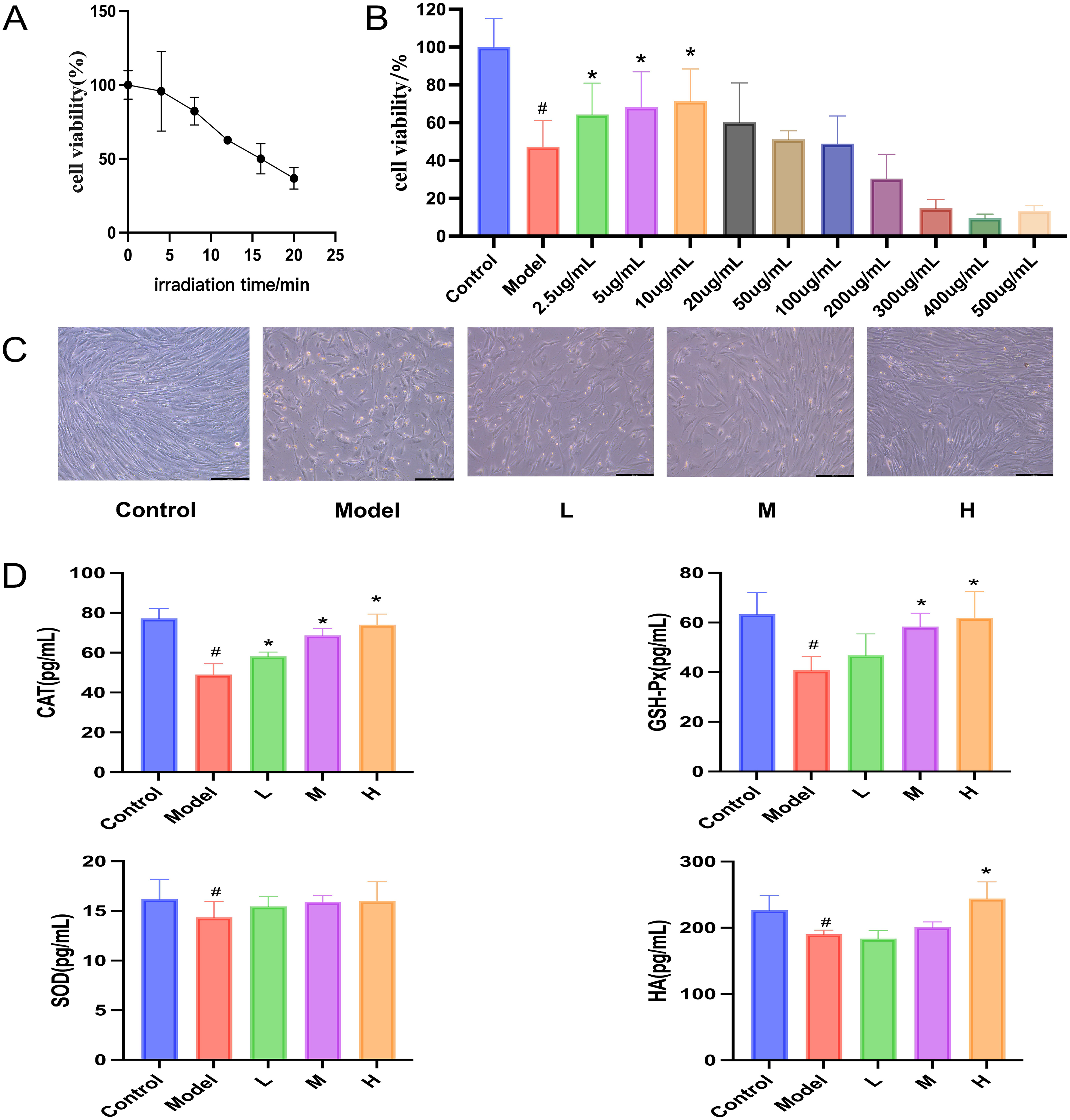
SHEX Protects BJ Cells from UV-Induced Damage. (A) Effect of Different Doses of UVB on Cell Viability (n = 6). (B) Effect of Different Concentrations of SHEX on Cell Viability (n = 6). (C) Effect of SHEX on Cell State After UVB Irradiation. (D) Effect of SHEX on the Content of Antioxidant Enzymes in Cells After UVB Irradiation (n = 6). #P < .05 Compared to the Control Group; *P < .05 Compared to the Model Group.
SHEX Protects Rats from UV-Induced Skin Damage
In the present study, after UV irradiation of SD rats, the weight gain of rats in the model group was relatively slow compared to the other groups, and the weight was increased by the use of SHEX cream (Figure 4A). The results of appearance observation showed that at the end of the eighth week of modeling, the back skin of rats in the control group was smooth and full, and the skin condition was normal; the back skin of the rats in the model group was reddish and rough, wrinkled, with skin peeling and cracking, and purple-red ulceration and scabbing, which was scaly, indicating that the UV damage model of the rat was successfully established. The skin of the rats in the L group was rough, with mild redness and a small amount of peeling, which was improved compared with that of the model group; the skin of the rats in the POS group, the M group, and the H group did not show any redness or peeling, and the skin condition was close to that of the control group. The skin condition of the base group was close to that of the model group. It is tentatively suggested that SHEX can improve UV-induced skin damage, and the cream matrix has no effect (Figure 4B).
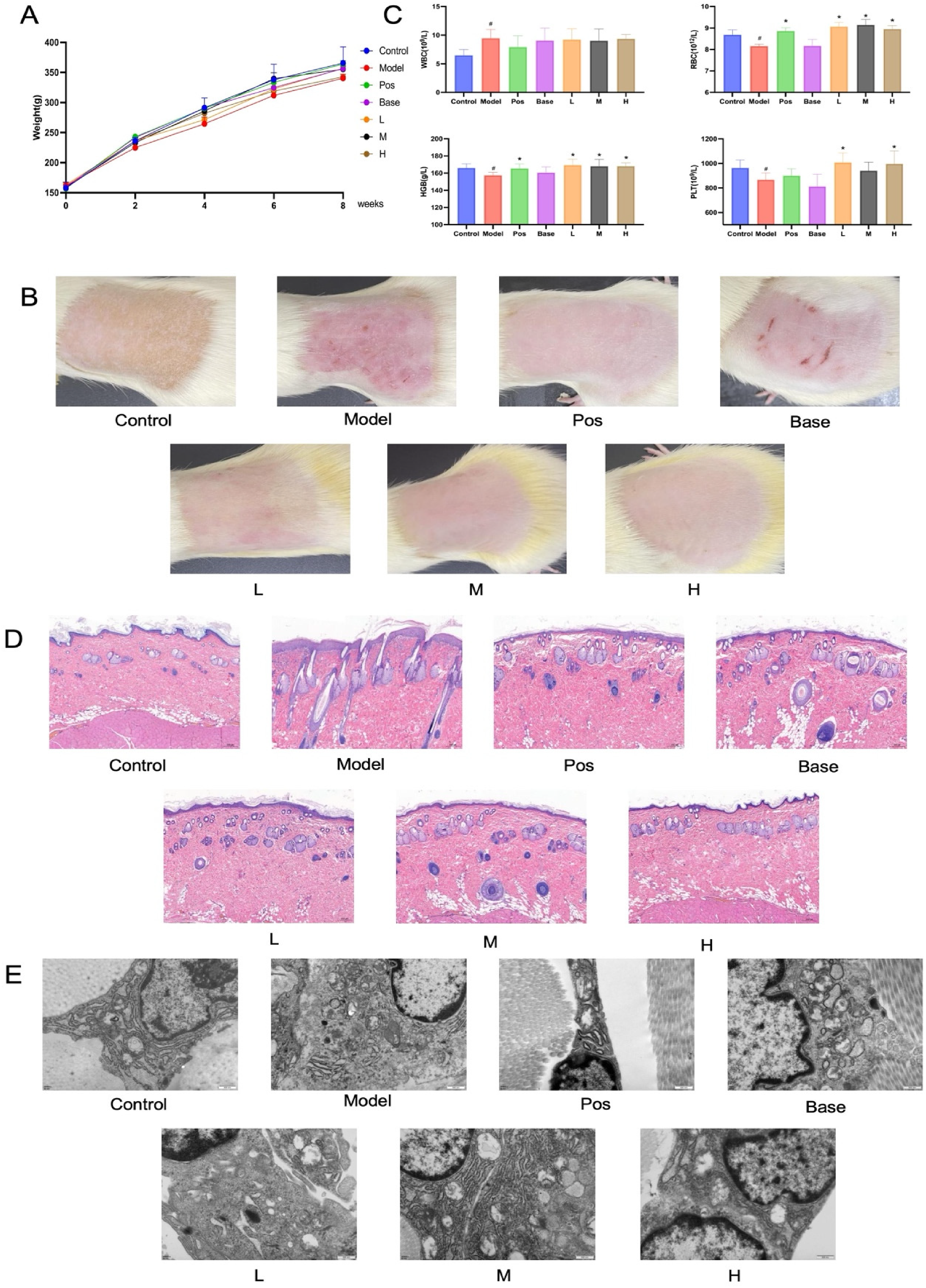
SHEX Protects Rats from UV-Induced Skin Damage. (A) Effect of SHEX on Body Weight of UV Damage Model Rats (n = 6). (B) Effect of SHEX on Blood Routine in UV Damage Model Rats (n = 6). (C) Effect of SHEX on the Skin Appearance Performance of UV Damage Model Rats. (D) Effect of SHEX on Histopathology of UV Damage Model Rats (HE, × 50). (E) Effect of SHEX on Skin Fibroblasts and Collagen Fibers in a UV-Damaged Rat Model under Transmission Electron Microscopy (TEM, × 30 000). #P < .05 Compared to the Control Group; *P < .05 Compared to the Model Group.
Previous studies have shown that the body protects itself by regulating physiological and biochemical indices to mitigate or protect itself from UV-induced damage. In the present study, we found that WBC was significantly higher (P < .05) and RBC, PLT, and HGB were significantly lower (P < .05) in the model groups compared with the control group, and RBC, PLT, and HGB were significantly higher (P < .05) in the drug group compared with the model group, whereas there was no significant difference in the base group (Figure 4C). These results suggest that SHEX attenuates UV-induced changes in physiological indices in SD rats.
The results of HE staining illustrated that SHEX attenuated UV-induced rat skin keratinization insufficiency, epidermal layer thickening, collagen fiber necrosis in the dermis, loss of collagen fiber structure, and inflammatory cell infiltration in the dermis of the skin (Figure 4D). TEM results illustrated that SHEX attenuated fibroblast necrosis, chromatin lysis in the nucleus, disorganization of cytoplasmic structures, loss of ribosomes, and expansion of rough endoplasmic reticulum (Figure 4E). These results suggest that SHEX attenuated UV-induced histological damage in the skin of SD rats.
SHEX Inhibits UV-Induced Oxidative Damage in rat Skin
The antioxidant index measurements revealed a significant reduction in CAT, SOD, GSH-Px, and HA levels in the model group compared to the control group after UV irradiation (P < .05). Conversely, the drug group exhibited a significant increase in CAT, SOD, GSH-Px, and HA levels compared to the model group (P < .05), while no significant difference was observed in the base group (Figure 5). These findings indicate that SHEX has the potential to enhance antioxidant levels and decrease UV-induced lipid peroxidation.
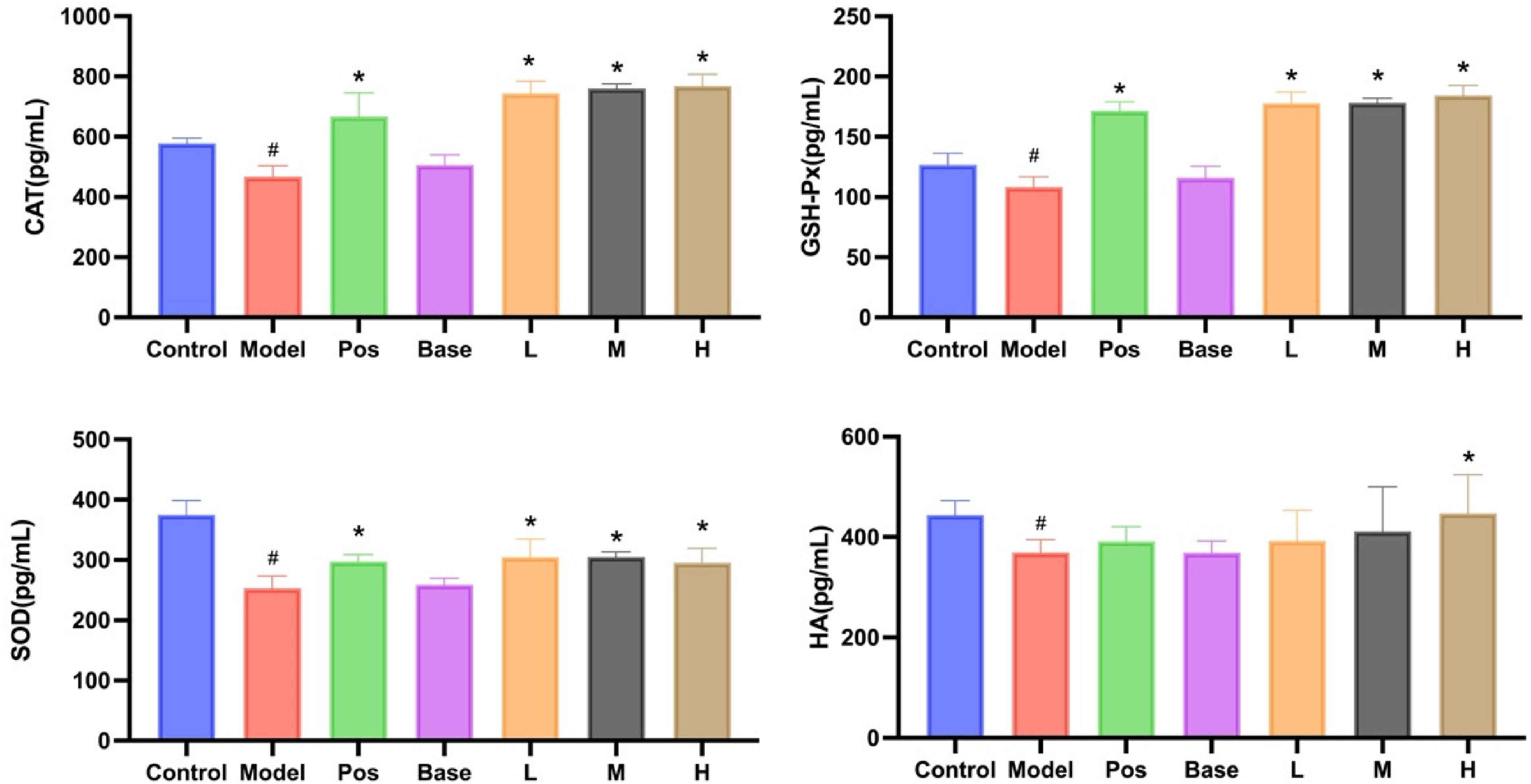
Effect of SHEX on Antioxidant Enzymes in the Skin of UV Damage Model Rats (n = 6). #P < .05 Compared to the Control Group; *P < .05 Compared to the Model Group.
Potential Mechanisms of SHEX Resistance to UV-Induced Skin Damage
According to the results of the KEGG analysis and literature references, UV irradiation activates the MAPK signaling pathway. To investigate the effect of SHEX on the MAPK pathway, protein and mRNA expression of two key proteins in the MAPK pathway, p38 MAPK and MMP-1, were determined by Western blot and RT-PCR. The experimental findings demonstrated the presence of p38 MAPK and MMP-1 mRNA expression in normal rat skin tissues. In comparison to the control group, the model group showed a significant increase in p38 MAPK and MMP-1 mRNA levels (P < .05). In contrast, the drug group exhibited a significant decrease in p38 MAPK and MMP-1 mRNA levels compared to the model group (P < .05), and this reduction was dependent on the dosage, whereas there was no significant difference in the base group (Figure 6A). The Western blot results demonstrated a significant increase in the protein content of p38 MAPK and MMP-1 in the model group compared to the control group (P < .05). Additionally, the drug group exhibited a significant decrease in p38 MAPK and MMP-1 protein content compared to the model group (P < .05), and this decrease was dependent on the dosage, whereas there was no significant difference in the base group (Figure 6B). These results suggest that SHEX may play a role in the protection against UV-induced skin damage by decreasing the expression of p38 MAPK and MMP-1.

Effect of SHEX on the Expression of p38 MAPK and MMP-1 in the Skin of UV-Damaged Model Rats. (A) Effect of SHEX on p38 MAPK and MMP-1 mRNA Content in the Skin of UV-Damaged Model Rats (n = 3). (B) Effect of SHEX on the Expression of p38 MAPK and MMP-1 Proteins in the Skin of UV-Damaged Model Rats (n = 3). #P < .05 Compared to the Control Group; *P < .05 Compared to the Model Group.
Discussion
With the growth of tourism in the plateau and an aging population, the prevention and treatment of skin damage diseases caused by ultraviolet radiation are becoming more important. Although laser technology has made significant progress in the treatment of UV damage, its application and equipment are limited and unsuitable for widespread use. 20 Drug therapy is the most convenient and effective way to treat UV damage. However, traditional Western medicine treatments can cause adverse reactions, negatively impacting the patient's condition. 21 Traditional Chinese medicines and compounds have promising applications in preventing and treating UV damage. 22 As a result, it is critical to develop herbal topical preparations with preventive or therapeutic effects on UV damage. We found that SHEX, a traditional Chinese medicine preparation, is appropriate for the treatment of UV damage based on both traditional Chinese medicine theory and modern pharmacological research, so we investigated the anti-UV damage effects of SHEX. Using network pharmacology combined with experimental validation, this study found that SHEX significantly attenuated UV-induced deleterious effects in in vitro and in vivo models and that this effect was associated with inhibition of the MAPK/p38 MAPK/MMP-1 signaling pathway.
In this study, we first screened the active ingredients of SHEX by network pharmacology, obtained 120 active ingredients and 651 therapeutic targets related to UV damage in SHEX, and then predicted that the network pharmacology was used to construct the “TCM-active ingredient-target-disease” network map, which predicted quercetin, obacunone, and dihydroniloticin as the core components of SHEX for the treatment of UV damage. Quercetin is a plant flavonoid, and more and more studies have found that quercetin can delay skin aging, reduce melanin production, and prevent skin oxidation, so the development of quercetin nanopreparations has become a new direction in skin antioxidant research in recent years.23,24 Obacunone, a triterpene, has been identified as a potent Nrf2 agonist, and some studies have found that obacunone attenuated the photodamage response in keratinocytes and full-thickness skin models exposed to solar radiation.25,26 These studies suggest that quercetin and obacunone may be the pharmacodynamic material basis of SHEX for the treatment of UV damage. The KEGG signaling pathway analysis showed that the main targets were the neuroactive ligand-receptor interaction pathway, the lipid and atherosclerosis pathway, and the MAPK signaling pathway. Research has demonstrated that exposure to radiation has neurotoxic effects, resulting in aberrant behavior and damage to neurons in embryos. Hou et al proposed that the mechanism of interaction between neuroactive ligands and receptors might play a role in radiation-induced neurotoxicity. 27 However, further investigation is required to determine whether non-ionizing radiation from UV light has a similar impact. Furthermore, lipid and atherosclerotic pathways are also controlled by UV because human skin is continuously exposed to UV radiation, and the accumulation of subcutaneous fat in peripheral regions may provide some protection against UV radiation. The reason for this is that subcutaneous adipose tissue in skin that has been damaged by continuous sun exposure has a lower amount of fat compared to naturally aged skin. 28 This indicates that UV radiation has a suppressive effect on the synthesis of lipids in the subcutaneous layer. In addition to the above 2 pathways, another noteworthy pathway is the MAPK signaling pathway. MAPK is an extensive group of protein kinases consisting of three primary subfamilies: extracellular signal-regulated kinases, c-Jun amino-terminal kinases (JNK), and p38 MAPK. It has been shown that a number of the above molecules can be activated by UV radiation. Prasanth et al discovered that gene-specific mutants of the p38 MAPK pathway had reduced survival upon exposure to UVA, whereas there was no effect on the lifespan of mutations in the JNK pathway, 29 and upon UV irradiation of HaCaT cells as well as A431 cells (a human epidermoid carcinoma cell line), a phosphorylated form of ERK 1/2. 30 Furthermore, the study found that UV radiation causes the production of ROS, which activates cell surface growth factor receptors and apoptosis signal-regulated kinases. This activates MAPK, which further increases the levels of NF- κB, TNF-α, and MMP. The result is the degradation of the extracellular matrix (ECM) and accelerates skin aging. 31 These results suggest an important role for the MAPK pathway in initiating the host response to UV exposure.
This study employed in vivo and in vitro experiments to thoroughly assess the protective effect of SHEX against UV damage. However, due to the absence of a standardized approach for modeling and validating animal models of UV damage, we first developed a new in vivo and in vitro model of UV damage. Firstly, the model selection for the in vitro research is based on fibroblasts, which are the primary constituent cells of the skin's dermal layer and the target cells for UV radiation. 8 Consequently, we selected BJ human fibroblasts as the subject for the in vitro study. In the in vivo investigations, we selected the widely utilized SD rat as a model organism due to its skin structure's resemblance to that of human skin. 32 Furthermore, when it comes to choosing the type of radiation, past research has shown that both UVA and UVB exposure can damage skin. Although the UVA to UVB ratio in the dermis is approximately 20:1, an equivalent dose of UVB radiation causes approximately 800 to 1000 times more skin damage than UVA. This disparity is due to the fact that UVB photons have energy that can be directly absorbed by nucleic acids and proteins, causing DNA damage. In addition, UVB can increase the expression of oncogenic genes, thereby increasing the risk of skin cancer, and UVA mediates energy transfer through photosensitizers in cells, forming free radicals and indirectly inducing cellular damage through oxidative stress and other pathways. 33 Taking these characteristics into account, BJ human fibroblasts were exposed to UVB radiation in this study to establish an in vitro model of UV damage. To accurately replicate UV damage in a natural setting, we irradiated SD rats with a combination of high-dose UVA and low-dose UVB to examine the in vivo protective effects of SHEX against UV damage. These modelings provide a reference for subsequent related research.
In vitro experiments demonstrated that SHEX enhanced the viability and condition of BJ human fibroblasts post-UV irradiation. Furthermore, topical application of a cream containing SHEX significantly improved the appearance and pathological condition of UV-damaged skin tissues in SD rats, implying that SHEX has potential therapeutic value for UV skin injury disorders. Prior research has shown that the five herbs in SHEX have distinct pharmacological effects. Quercetin, obacunone, rhododendron, rhubarbic acid, and various alkaloids screened by us through network pharmacology have good antioxidant effects, so we hypothesized that the anti-UV damage effects of SHEX might be related to its antioxidant properties. Previous studies have shown that overexposure to UV radiation leads to the production of large amounts of reactive oxygen species (ROS), such as superoxide (O2−), hydrogen peroxide (H2O2), and hydroxyl radicals (OH•), whereas SOD, GSH-Px, and CAT can scavenge excessive ROS, reduce lipid peroxidation, and improve the antioxidant capacity of the body.34,35 In addition, HA is an acidic mucopolysaccharide present in the intercellular matrix of skin tissues, which not only has a certain water-retaining effect but also effectively removes reactive oxygen species generated by UV radiation.36,37 Therefore, the activity levels of these enzymes can be used as markers of oxidative stress in UV-induced skin damage. Notably, the application of SHEX led to a substantial elevation in the levels of SOD, GSH-Px, CAT, and HA in fibroblasts and skin tissues, indicating that SHEX enhances the skin's antioxidant capacity by promoting the expression of these enzymes, providing protection against UV-induced skin damage. Furthermore, it is important to highlight that vitamin E (VE), the selected therapeutic agent in the in vivo studies, is a clinically utilized medication for the treatment of UV-induced skin damage, which exerts its therapeutic effect by enhancing the activity of the body's defense systems against oxidative stress. 38 The findings indicated that the antioxidant efficacy of SHEX was analogous to that of VE. In conclusion, these results suggest that SHEX attenuates UV-induced oxidative damage both in vivo and in vitro, and that SHEX may have the same potential for clinical application as VE.
Using network pharmacology, we discovered that UV irradiation activates the MAPK signaling pathway. Previous research has shown that UV irradiation can strongly activate p38 MAPK in the MAPK signaling pathway and that activated p38 MAPK undergoes phosphorylation and promotes the activation of various transcription factors, regulating UV-induced cell cycle arrest, DNA repair, inflammatory responses, and apoptosis. 39 It has been shown that pharmacological inhibition of p38 MAPK can be used to prevent UV-induced damage to the skin. Kim et al found that blocking p38 MAPK prevented hyperpigmentation induced by UVB exposure, 40 and Papoutsaki et al found a significant reduction in UV-induced tumor number and growth in p38 dominant-negative (p38DN) hairless mice. 41 The aforementioned data suggest that p38 MAPK is an effective target for pharmacological intervention in the context of skin damage induction. Activation of the p38 MAPK pathway further regulates the transcription of MMPs, which are important genes for the degradation of ECM proteins (including collagen, fibronectin, elastin, and proteoglycans) and inhibition of neocollagenesis. Among them, MMP-1 is the major gene that destroys type I collagen, and the results of network pharmacology suggest that MMP-1 is a key target for SHEX to combat UV-induced skin damage. Therefore, we investigated the molecular mechanism of SHEX against UV damage by evaluating the expression levels of p38 MAPK and MMP-1. The results confirmed that SHEX significantly downregulated the expression of p38 MAPK and MMP-1 in the MAPK signaling pathway, suggesting that SHEX can prevent UV-induced skin damage by inhibiting the MAPK/p38 MAPK/MMP-1 signaling pathway. It has been suggested that UV-induced oxidative damage also stimulates the synthesis of MMPs, 42 whereas in our study we found that SHEX increased the expression of antioxidant enzymes in the organism, so the SHEX-mediated reduction of MMP-1 may also be related to the antioxidant effect of SHEX, but further experimental verification is needed.
In conclusion, we speculate that the anti-ultraviolet damage effect of SHEX may be related to the following two mechanisms: one is to directly up-regulate the body's antioxidant defense system to directly scavenge or inhibit the production of reactive oxygen species from keeping the body in dynamic equilibrium to prevent the damage, and the other is to inhibit the expression of p38 MAPK in the skin tissue and reduce the production of MMP-1 to reduce the degradation of the ECM (Figure 7). This study not only provides a theoretical foundation and technical support for the clinical application of SHEX, but it also offers potential clues for the development of novel anti-UV damage formulations. However, it should be noted that the UV-induced signaling pathway is extremely complex, and thus SHEX's mechanism of action for UV damage requires further investigation.
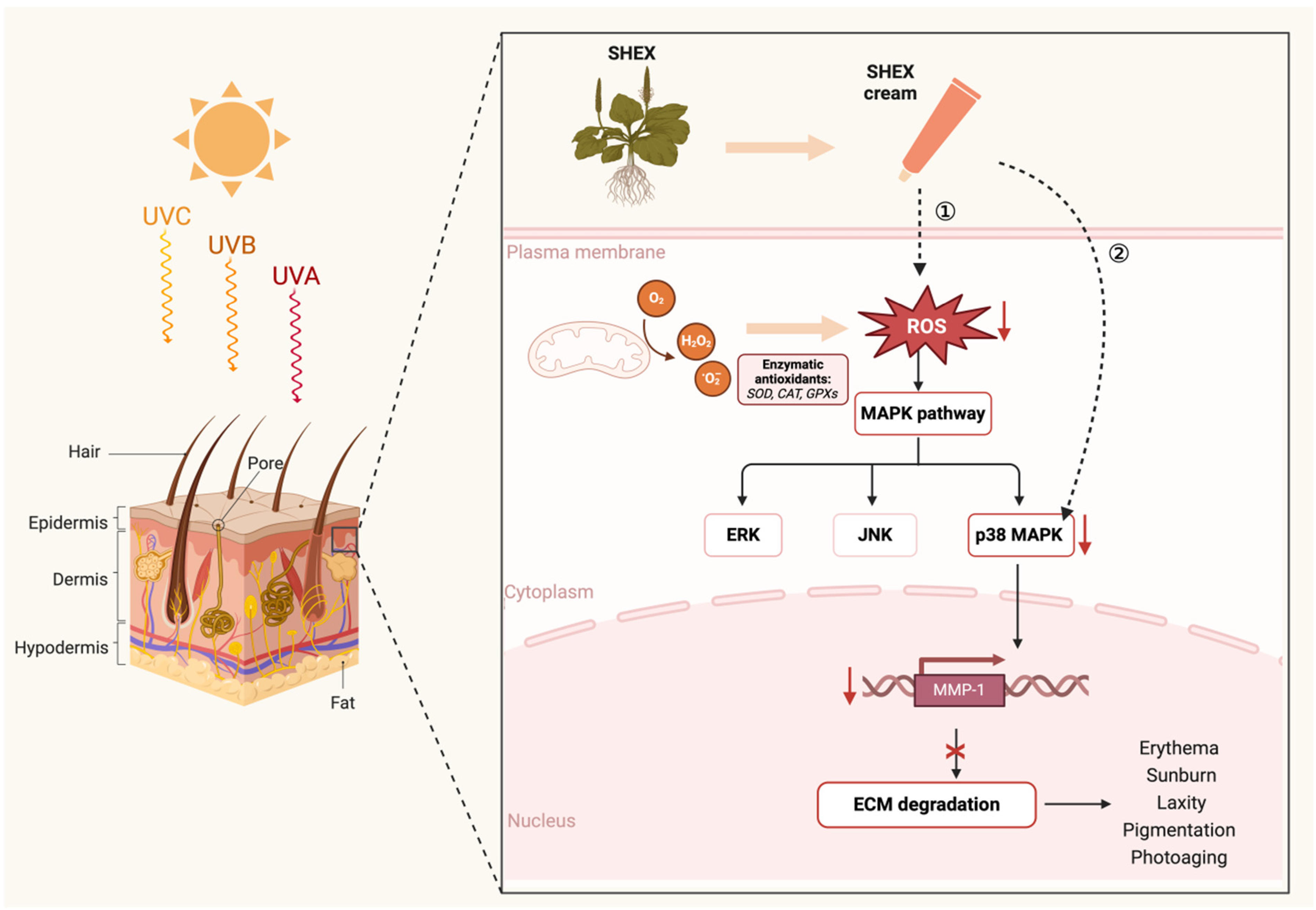
Schematic Representation of the Mechanism of SHEX Against UV-Induced Skin Damage Diseases. (Created in BioRender (https://BioRender.com/), Creation Date: February 23rd, 2025, Agreement Number: AD27Y49IP3).
Nonetheless, this study has many limitations: 1) The target proteins of the MAPK pathway were chosen for validation in this study based on network pharmacology and existing literature; however, the expected expression of other relevant core target proteins, as well as the interactions between drugs and these target proteins, requires further investigation and validation, particularly for collagen. Collagen in the skin, especially collagen type I (COL-1) and collagen type III (COL-3), is essential for the skin to withstand tensile forces and maintain skin plumpness, so assessing the expression levels of collagen-producing genes may provide deeper insights into the mechanisms of SHEX against UV-mediated skin damage. Our initial studies focused on assessing SHEX's direct reparative effects on UV-induced skin damage, such as appearance performance, histological changes, and oxidative stress, but did not directly assess collagen-producing gene expression levels. Therefore, further investigation of collagenogenic genes and other related mechanisms, such as inhibition of the transforming growth factor (TGF)-β/small mothers against decapentaplegic (Smad) pathway, mitochondrial dysfunction, and inflammatory responses, will be an important direction for future research. 2) Although the active ingredients in SHEX were screened by network pharmacology in this study, due to the complexity of the active ingredients in TCM compounding, the composition and content of the active ingredients of SHEX have not yet been determined in this study; consequently, we will persist in conducting qualitative and quantitative analyses of the biologically active components of SHEX to further elucidate its role in the treatment of UV radiation damage. 3) Regarding model design, the absence of standardized UV damage modeling techniques and apparatus in the present study resulted in discrepancies during in vivo experiments. Although the high-dose UVA combined with low-dose UVB modeling aimed to replicate the composition of environmental UV rays as closely as possible, it still exhibited some inconsistencies with the actual irradiation dose. In the natural environment, the UVA: UVB ratio reaching the ground is approximately 20: 1, whereas this study utilized a ratio of 4: 1, thus failing to fully replicate the UV-induced skin damage observed in real-world conditions. Furthermore, the in vitro experiments only used a single UVB-induced damage model involving fibroblasts while ignoring a UVA-induced cellular damage model. This limitation constrains the study's findings. Consequently, future research must incorporate a broader array of experimental models and clinical studies to comprehensively explore the role of SHEX in the treatment of UV-induced skin damage.
Conclusion
In conclusion, this study using network pharmacology and in vivo and in vitro experiments demonstrated that SHEX has an anti-UV-induced skin damage effect, the mechanism of which may be related to antioxidant effects and inhibition of p38 MAPK and MMP-1 release. It not only provides a new treatment approach for UV-associated diseases but also provides theoretical support for the development and clinical application of SHEX.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251345699 - Supplemental material for Potential Pharmacological Effects of Sanhuang Erxiang Powder in the Treatment of Ultraviolet-induced Skin Damage: Insights from Network Pharmacology and Experimental Validation
Supplemental material, sj-docx-1-npx-10.1177_1934578X251345699 for Potential Pharmacological Effects of Sanhuang Erxiang Powder in the Treatment of Ultraviolet-induced Skin Damage: Insights from Network Pharmacology and Experimental Validation by Qian Wang, Cairang Nima, Linli La, Guiqin Liu, Delong Duo, Junbo Zhu, Junjun Han, Xiangyang Li and Ning Qu in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank the Department of Pharmacy, Qinghai University, and the State Key Laboratory of Plateau Ecology and Agriculture for their support and assistance.
Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Qinghai Basic Research Programme Project, (grant number 2024-ZJ-724).
Author Contributions
Qian Wang designed, developed, and optimized the assays and wrote the manuscript. Cairang Nima analyzed and interpreted the data. Linli La collated data. Guiqin Liu and Delong Duo collected samples and provided scientific guidance. Junbo Zhu analyzed samples and performed the validation experiments. Junjun Han provided support to the assay development. Xiangyang Li critically reviewed the manuscript and provided financial support. Ning Qu proposed an assay strategy and supervised overall assay development.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals, and the protocol was approved by the Animal Ethics Committee of Qinghai University (Approval No. PJ-202302-12).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
