Abstract
Keywords
Introduction
The new concept of quality control of traditional Chinese medicine (TCM) Q-marker was put forward by Dr. Chang-xiao Liu in 2016 6 based on the “5 principles” of effectiveness, peculiarity, transmission and traceability, measurability, and prescription compatibility. It has been widely used in the quality research of Chinese herbal medicines, decoctions, and compound prescriptions, which is helpful to improve the quality control of Chinese herbal medicines. 7 The fingerprint of TCM has the characteristics of integrity and fuzziness, which can reflect the stability and uniformity of the quality of Chinese herbal medicines,8-10 especially compound TCM prescriptions containing multicomponents, multitargets, and multipathways. Network pharmacology can promote the understanding of the mechanism of drug actions and find out biomarkers and potential targets of drug action accurately and quickly.11-13 At present, there have been few reports on the systematic study of FYD. In this study, we used the fingerprint and network pharmacology methods to predict and analyze the potential Q-marker of rhubarb in FYD qualitatively and quantitatively based on the Q-marker theory, hoping that the results could provide a reference for the quality control of FYD and the development of subsequent preparations, and lay a theoretical foundation for study of the action mechanism of FYD. In addition, our study may provide a novel idea for the study of Q-marker of classical compound TCMs.
Materials and Methods
Preparation of FYD and Standard Solutions
Sample Preparation
Rhubarb (Lot No. S1-S15) from Gansu Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-028; Dried ginger (Lot No. 190501) from Sichuan Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-041; Cinnamon twig (Lot No. 190401) from Guangxi Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-454; Licorice (Lot No. 190401) from Xinjiang Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-042; Keel (Lot No. 190501) from Ningxia Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-556; Oyster (Lot No. 190601) from Zhejiang Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-522; Calcitum (Lot No. 190101) from Zhejiang Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-560; Talc (Lot No. 181101) from Shandong Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-562; Red halloysite (Lot No. 190401) from Shanxi Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-558; Fluoritum (Lot No. 190401) from Gansu Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-568; and Gypsum fibrosum (Lot No. 190401) from Guangdong Province of China on the inspection basis of the Chinese Pharmacopoeia 2015 Edition and Enterprise Standard TS-ZL-574 were purchased from Zhejiang University of Traditional Chinese Pharmaceuticals Co., Ltd (http://www.zjzyyp.com/Default.aspx).
On the basis of the prescription dose and the best extraction process determined in the previous experiment, 24.5 g medicinal materials were weighed in a proportion of rhubarb:dried ginger:keel:cinnamon twig:licorice:oyster:calcitum:talc:red halloysite:fluoritum:gypsum fibrosum = 2:2:2:1.5:1:1:3:3:3:3:3. The sample of FYD was obtained by adding 75% ethanol solution at 1:10, soaking for 30 min, boiling in a water bath at 80 °C for 30 min, filtration, rotary evaporation, and making to a fixed volume. According to the above extraction process of FYD, 15 batches of FYD and rhubarb samples, FYD samples with anhydrous ethanol as an extraction solvent, FYD samples with 50% ethanol as an extraction solvent, FYD samples with water as an extraction solvent, and negative control samples without rhubarb, dried ginger, cinnamon twig, and licorice were prepared.
Preparation of the FYD Extracts as the Test Solutions
The FYD sample solution prepared in the “Sample preparation” section was diluted 5 times with 85% methanol (TEDIA). The diluted solution was mixed by ultrasound and through a 0.22 μm organic micropore film for analysis.
Preparation of Standard Solutions
Aloe-emodin (Lot No. Y02M11Y16995), rhein (Lot No. T30A8F42628), chrysophanol (Lot No. T29D10F107203), cinnamaldehyde (Lot No. H02M6Q1), and physcion (Lot No. T18O10F100404) were placed in a 25 mL volumetric flask. Glycyrrhizic acid ammonium salt (Lot No. P13A9L67602), emodin (Lot No. T17A10F95418), and 6-gingerol (Lot No. P17M10F88628) (all purchased from Shanghai Yuanye Bio-Technology Co., Ltd) were weighed accurately in a 10 mL volumetric flask and dissolved with methanol ultrasonically. Glycyrrhizic acid ammonium salt was dissolved in 85% methanol ultrasonically, and an appropriate amount of each standard mother solution was accurately drawn to obtain aloe-emodin (2.43 μg/mL), ammonium glycyrrhizinate (22.26 μg/mL), rhein (10.02 μg/mL), emodin (13.91 μg/mL), chrysophanol (9.65 μg/mL), physcion (6.57 μg/mL), cinnamaldehyde (55.62 μg/mL), and 6-gingerol (25.93 μg/mL) reference substance mixed solution, and stored in a refrigerator at 4 °C for later use.
Ultra Performance Liquid Chromatography Analysis
Chromatographic Conditions
Liquid chromatography (LC) was performed on a Waters H-class Ultra Performance Liquid Chromatography (UPLC) system (Waters Co.) equipped with a photo diode array detector and controlled with Empower chromatography data software. The chromatographic separation was performed on a Waters ACQUITY UPLC BEH C18 column (2.1 mm × 50 mm, 1.7 μm) at a column temperature of 30 °C. The mobile phase consisted of acetonitrile (A, Merck) and 0.1% formic acid solution (B, Aladdin). The gradient elution program was set as follows: 0 to 7.9 min, 10.0%→25.0% A; 7.9 to 8.0 min, 25.0%→30.0% A; 8.0 to 12.4 min, 30.0%→30.0% A; 12.4 to 12.5 min, 30.0%→38.0% A; 12.5 to 15.0 min, 38.0%→38.0% A; 15.0 to 15.1 min, 38.0%→43.0% A; 15.1 to 17.0 min, 43.0%→50.0% A; 17.0 to 21.3 min, 50.0%→62.0% A; 21.3 to 27.0 min, 62.0%→90.0% A; 27.0 to 30.0 min, 90.0%→10.0% A; and 30.0 to 32.0 min, 10% A. The detection wavelength was 254 and 280 nm (cinnamaldehyde, 6-gingerol). The volume flow was maintained at 0.3 mL/min. The injection volume was 2 µL.
Validation of the Fingerprint Method
The chromatographic results showed a high chrysophanol content with a good and stable chromatographic peak, indicating that chrysophanol was the main component of rhubarb in FYD. Therefore, chrysophanol was selected as the reference peak to calculate the relative retention time and relative peak area of each common peak.
The same batch of FYD (S1) was injected continuously, 6 times, and the chromatograms were recorded. The results showed that the relative standard deviation (RSD) of the relative retention time of each common peak of FYD was <0.10%, and the RSD of the relative peak area was <1.21%, demonstrating good precision of the instrument.
Six samples of the same batch of FYD (S1) were prepared in parallel according to the method in the “Preparation of the FYD Extracts as the Test Solutions” section, and the samples were detected. The RSD of relative retention time of each common peak of FYD was <0.38%, and the RSD of relative peak area was <3.07%, demonstrating good reproducibility of the method.
The same batch of FYD (S1) was prepared according to the method in the “Preparation of Standard Solutions” section, and the samples were injected and detected at 0, 2, 4, 8, 12, and 24 h. The RSD of relative retention time of each common peak of FYD was <1.18%, and the RSD of relative peak area was <2.15%, demonstrating good stability of the sample solution within 24 h.
Validation of the Method in Determining the Active Ingredients in FYD
The mixed reference substance of a series of concentrations was diluted step by step according to the multiple ratios. The standard curve was drawn by linear regression with the mass concentration of each component as the abscissa (
Linear Regression Results of 8 Active Ingredients of

The RSDs of the peak area of aloe-emodin, glycyrrhizic acid ammonium salt, rhein, emodin, chrysophanol, physcion, cinnamaldehyde, and 6-gingerol were 0.15%, 0.42%, 0.26%, 0.50%, 0.25%, 0.53%, 0.14%, and 0.31%, respectively, indicating that the precision of the instrument was good.
The RSDs of the peak area of aloe-emodin, glycyrrhizic acid ammonium salt, rhein, emodin, chrysophanol, physcion, cinnamaldehyde, and 6-gingerol were 1.76%, 1.29%, 2.39%, 2.33%, 2.21%, 1.65%, 2.09%, and 2.42%, respectively, indicating that the method had good repeatability.
The RSDs of the peak areas of aloe-emodin, glycyrrhizic acid ammonium salt, rhein, emodin, chrysophanol, physcion, cinnamaldehyde, and 6-gingerol were 2.12%, 0.95%, 0.42%, 0.36%, 0.13%, 0.85%, 0.40%, and 0.49%, respectively, indicating that the FYD solution was stable within 24 h.
After accurately removing 6 samples of FYD1 (1 mL each) with known contents, aloe-emodin 0.32 mL (0.028 mg/mL), rhein 0.54 mL (0.0576 mg/mL), glycyrrhizic acid ammonium salt 0.65 mL (0.128 mg/mL), emodin 0.12 mL (0.16 mg/mL), chrysophanol 1 mL (0.0444 mg/mL), physcion 0.55 mL (0.0504 mg/mL), cinnamaldehyde 0.03 mL (0.8 mg/mL), and 6-gingerol 0.23 mL (0.426 mg/mL) were added precisely. They were prepared in parallel according to the method in the “Preparation of the FYD Extracts as the Test Solutions” section. The results showed that the recovery rate of aloe-emodin, glycyrrhizic acid ammonium salt, rhein, emodin, chrysophanol, physcion, cinnamaldehyde, and 6-gingerol averaged 101.12%, 101.64%, 102.75%, 102.42%, 102.46%, 101.98%, 102.48%, and 100.73%, respectively. The RSD of peak areas was 2.68%, 2.90%, 2.08%, 2.76%, 1.70%, 3.02%, 2.54%, and 2.80%, respectively, indicating that the recovery rate of the method was good.
Results and Discussion
Fingerprint of FYD
Fifteen batches of FYD and 15 batches of rhubarb samples were detected according to the chromatographic conditions, and each chromatogram was recorded. The chromatographic data were sequentially input into the similarity evaluation software issued by the State Pharmacopoeia Committee, “similarity Evaluation system of chromatographic fingerprint of traditional Chinese Medicine” (2012.130723 edition), with the fingerprint of S1 as the reference atlas, using the average method. The width of the time window was 0.1, multi-point correction and chromatographic peak matching were carried out, the common pattern of fingerprint was automatically generated, and the similarity was calculated. The results showed that the similarity of the 15 batches of FYD was 0.948 to 0.999, and the liquid phase similarity of the 15 batches of rhubarb was 0.907 to 0.999, indicating that the 15 batches of FYD and the 15 batches of rhubarb had good consistency and stable quality. The fingerprint of the 15 batches of FYD and the 15 batches of rhubarb are shown in Supplemental Figures S1 and S2; the chromatogram of the mixed standard and the pattern diagram of the common peak of the sample are shown in Figure 1, and the similarity is shown in Supplemental Table S1.

(A) Common patterns of
Twenty-eight common peaks were identified by FYD fingerprint and 19 common peaks were determined by rhubarb fingerprint. By comparing the fingerprints of FYD, lack of rhubarb, lack of cinnamon twig, lack of dried ginger, lack of licorice, and mixed standard fingerprints, it could be determined that peak 7 (cinnamaldehyde), peak 15 (aloe-emodin), peak 16 (rhein), peak 17 (glycyrrhizic acid ammonium salt), peak 18 (6-gingerol), peak 22 (emodin), peak 25 (chrysophanol), peak 26 (physcion), and peak 7 (cinnamaldehyde) had some interference in the negative samples lacking rhubarb.
According to the sample preparation method, 3 different extraction solvents of FYD were prepared by using anhydrous ethanol, 50% ethanol, and pure water, respectively. Combined with the results of the previous pharmacodynamic study of the research group, the antiepileptic effect of FYD extracted by anhydrous ethanol was the best, followed by 50% ethanol. A comparison of the FYD chromatographic peaks of the 3 extraction methods showed that the content of each component of rhubarb increased with the increased concentration of ethanol. Peaks 15 (chrysophanol), 14 (emodin), 11 (rhein), 10 (aloe-emodin), and 16 (physcion) were more significant. Among them, peaks 2, 3, 4, 6, 7, 8, 9, 10, 14, 15, 16, and 17 could hardly be detected in the water extract, indicating that these components may be the potential material basis of pharmacodynamics. Rhubarb was the monarch medicine of FYD. In this study, we performed an in-depth analysis of the components of FYD from the microscopic viewpoint and found that the main chemical component of FYD was rhubarb, which also validated the TCM theory that rhubarb is the monarch medicine and plays a major role in TCM.
Sample Determination
The FYD samples extracted by 3 extracting agents were detected, and the contents of aloe-emodin, glycyrrhizic acid ammonium salt, rhein, emodin, chrysophanol, physcion, cinnamaldehyde, and 6-gingerol were calculated. The results are shown in Figure 2 and Table 2.

Chromatograms of different extracts of
Results of Different Extraction Methods of 8 Active Ingredients in
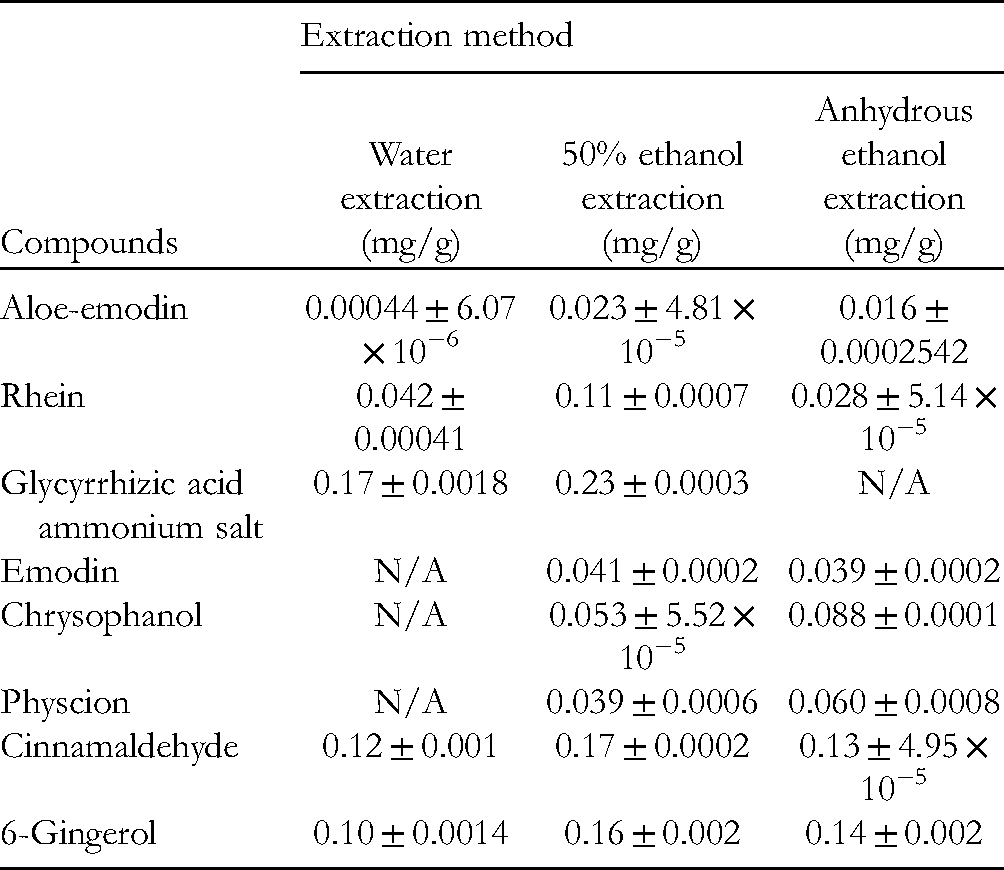
The results showed that there were significant differences in the contents of 8 components in FYD with different extraction methods, and the anthraquinone content of rhubarb in ethanol-extracted FYD increased significantly, in which the content of chrysophanol was the highest, followed by physcion. In water-extracted FYD, emodin, chrysophanol, and physcion were all lower than the detection limit, so the content could not be calculated, while in anhydrous ethanol-extracted FYD, glycyrrhizic acid ammonium salt was lower than the detection limit. Combined with the previous antiepileptic efficacy evaluation results in the laboratory, rhubarb anthraquinones may be the potential main antiepileptic active components, in which chrysophanol was particularly important; cinnamaldehyde and 6-gingerol were the main active components in cassia twig and dried ginger, and the content of ethanol extraction was higher than that of water extraction. This study started from the “5 principles” of Q-marker (effectiveness, specificity, transmission and traceability, measurability, and prescription compatibility) put forward by Chinese Academician Liu Chang-xiao. Studies in the literature showed that rhubarb anthraquinones were the main active ingredients of rhubarb. Our research group verified the antiepileptic pharmacodynamics of the 3 extraction methods of FYD in the early stage, which combined with network pharmacology to reflect the effectiveness of the predicted rhubarb Q-marker. The 5 anthraquinones of rhubarb were not detected in the negative control samples of rhubarb, indicating the specificity of the predicted rhubarb Q-marker. Eight chemical components of FYD and 5 anthraquinones in rhubarb were identified by fingerprint, and the contents of 8 active components were quantitatively analyzed, indicating the transitivity, traceability, and testability of the effective components of TCM. Predicting the Q-marker of rhubarb in the compound compatibility environment of FYD is helpful to find the Q-marker of rhubarb more accurately and efficiently.
Network Pharmacology Research
Target Prediction of Candidate Compounds
By prediction of the candidate compound targets by referring to the pharmacological database of the TCM system (TCMSP, http://tcmspw.com/tcmsp.php), PubChem Compound database (https://www.ncbi.nlm.nih.gov/pubmed/), and Swiss Target Prediction database (http://www.swisstargetprediction.ch/), with oral bioavailability ≥ 30% and drug likeliness ≥0.18 as screening conditions, 16 compounds were extracted: eupatin, mutatochrome, physciondiglucoside, procyanidin B-5,3′-
Analysis of the Target Protein–Protein Interaction Network
The selected 201 target proteins were uploaded to STRING 11.0 online software (https://stringdb.org). The species was selected as “homo sapiens”, and >0.9 was set as the highest confidence protein interaction parameter score. A single node in the network was hidden, and the protein–protein interaction (PPI) network was obtained (Figure 3). A total of 473 edges were obtained, and the PPI data were introduced into Cytoscape 3.7.2, and the “Network Analyzer” function was used for topological feature analysis. Three topological parameters (degree, betweenness centrality, and closeness centrality) were greater than the median. Using degree >10 as the core target, a total of 22 core targets were selected (Supplemental Table S2). The result showed that 22 targets were related to 12 components, including aloe-emodin, emodin, eupatin, palmidin A, procyanidin B-5,3′-

The protein–protein interaction network diagram of core targets of rhubarb (the interaction of 201 targets of rhubarb).
Functional Enrichment of Gene Ontology and Analysis of Kyoto Encyclopedia of Genes and Genomes Pathway
The gene ontology (GO) function and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment of 22 potential core target proteins were analyzed by DAVID6.8 database (https://david.ncifcrf.gov/). GO function analysis included the biological process (BP), molecular function (MF), and cellular component (CC), while KEGG enrichment analysis could obtain the signal pathway of the core target.
A total of 215 items were obtained by GO enrichment analysis, of which BP accounted for 151, 43 for MF, and 21 for CC. Twenty-two items were selected according to a
Enriched Gene Ontology Terms for Biological Processes of Potential Core Targets of the Main Active Ingredients of Rhubarb.

Abbreviations: FDR, false discovery rate; BP, biological process; CC, cellular component; MF, molecular function; ERBB2, erythroblastic oncogene B2.
Seventy-three pathways were obtained by KEGG enrichment analysis, and 21 related pathways were screened according to a
Enriched Kyoto Encyclopedia of Genes and Genomes Pathways of Potential Core Targets of the Main Active Ingredients of Rhubarb.

Abbreviations: FDR, false discovery rate; PI3K, phosphoinositide 3-kinase; Akt, protein kinase B; MAPK, mitogen-activated protein kinase; ERBB, erythroblastic oncogene B.
Construction of “Components–Targets–Pathways” Network
According to the screening of 19 core targets (among which 3 targets whose

“Compounds–targets–pathways” network diagram of rhubarb (19 core targets related to 12 components and 21 pathways as nodes).
Based on the degree size, the items whose degree was no less than the median value of 12 were screened. It was found that the connectivity of compounds such as aloe-emodin (degree = 45), emodin (degree = 31), β-sitosterol (degree = 27), physcion (degree = 24), emodin-1-
Rhubarb contains anthraquinones, anthrones, stilbenes, organic acids, tannin, and other components.16-18 At present, most studies have focused on free anthraquinones of rhubarb, which are the material basis of rhubarb, including aloe-emodin, rhein, emodin, chrysophanol, and physcion. Anthrones are also the main active components of rhubarb, such as rhubarb dianthrone A–C, palmleaf dianthrone A–C, and sennoside A–F. Previous studies have found that rhubarb anthraquinone in rhubarb is the main ingredient in blood, and it is easy to conduct qualitative and quantitative analyses. In the pharmacokinetic study of raw rhubarb and processed rhubarb, significant differences in rhein-8-
We found by Bioinformatics Analysis Tool for Molecular Mechanism of TCM that FYD may regulate neuroactive ligand–receptor interaction, arachidonic acid metabolism, serotonergic synapse, ovarian steroidogenesis, retrograde endocannabinoid signaling, thyroid hormone signaling pathway, oocyte meiosis, oxytocin signaling pathway, Parkinson's disease, dopaminergic synapse, and estrogen signaling pathway to treat analgesia, unspecified pain, breast cancer, depression, neurodegenerative diseases, cocaine dependence, cardiovascular disease, brain injury, Alzheimer’s disease, migraine, erectile dysfunction, drug dependence, and schizophrenia. A comparison of the fingerprints of FYD with different extraction methods shows that some potential pharmacodynamic components remain unclear and need to be further confirmed by LC–mass spectrometry (MS)/MS, metabolomics, and molecular probing.
Conclusion
In this study, we identified 8 compounds (aloe-emodin, rhein, emodin, chrysophanol, physcion, glycyrrhizic acid ammonium salt, cinnamaldehyde, and 6-gingerol) in FYD and 5 anthraquinones in rhubarb by fingerprints, and screened 19 targets and 21 pathways of 12 core components in rhubarb by network pharmacology. Based on the “5 principles” and requirements of Q-marker, combined with fingerprint, network pharmacology, and above analysis, we preliminarily predicted that rhubarb anthraquinone is the potential Q-marker of rhubarb.
As the composition of TCM compound preparations is complex, it is relatively difficult to clarify their action mechanism and control the quality. The fingerprint of TCM has the characteristics of integrity and therefore can better reflect the correlation between the ingredients in the compound and the curative effect. Network pharmacology can preliminarily clarify the action mechanism of Chinese herbal medicines through the establishment of a network of interactions among ingredients, targets, pathways, and diseases, and provide a theoretical basis for further cell or animal verification experiments. This study preliminarily predicted the Q-marker of rhubarb in FYD.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211038792 - Supplemental material for Q-marker Prediction Analysis of Rhubarb in Fengyin Decoction Based on Fingerprint and Network Pharmacology
Supplemental material, sj-docx-1-npx-10.1177_1934578X211038792 for Q-marker Prediction Analysis of Rhubarb in
Footnotes
Acknowledgments
We appreciate the great help from the Public Platform of Pharmaceutical Research Center, Academy of Chinese Medical Science, Zhejiang Chinese Medical University.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (81641139) and the Zhejiang Provincial Science Foundation Project (LY20H280004).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
