Abstract
Background
Senotherapeutics, agents targeting cellular senescence, have recently shown great promise in anti-aging applications by removing senescent cells or attenuating the senescence-associated secretory phenotype (SASP). Ginseng and its bioactive component, ginsenosides, are widely recognized in Asia for their anti-aging effects. In this study, we investigated the senotherapeutic effects of wild and cultivated ginseng, and ginsenosides on fibroblasts.
Methods
Using a human dermal fibroblast cell line, we induced senescence via UV exposure and cultured the cells with ginseng extracts and ginsenosides. Senolytic effects were evaluated by flow cytometry quantifying apoptotic cells, and senomorphic effects were evaluated by measuring gene expression related to SASP.
Results
Wild ginseng exhibited a stronger senolytic effect than cultivated ginseng. We then focused on ginsenosides unique to wild ginseng, namely C-Mc1 and C-O. We demonstrated that these two ginsenosides are senolytic agents that selectively target senescent cells modulating expression of BCL2 family. Moreover, these ginsenosides also alleviated paracrine senescence by suppressing SASP factors in senescent cells.
Conclusion
Wild ginseng and its ginsenosides C-Mc1 and C-O are potential senotherapeutic agents that can prevent the accumulation of senescent cells and the spread of senescence into neighboring cells.
Introduction
Cellular senescence is a multifactorial process triggered by various stimuli, such as DNA damage and telomere dysfunction, and is associated with the aging of organisms. Senescent cells exhibit resistance to apoptosis due to the increased activation of senescent-cell anti-apoptotic pathways (SCAPs), such as the BCL2 family network, p53/p21/serpine, PI3 K/AKT, HIF-1α and HSP-90, along with the down-regulation of key apoptotic mediators.1–3 Senescent cells also develop the senescence-associated secretory phenotype (SASP) profile comprising pro-inflammatory cytokines, chemokines, proteases and other factors which drive senescence spreading to neighboring cells via paracrine signaling.
To mitigate the pro-aging roles of senescent cells, recent studies have emphasized the importance of developing senotherapeutic approaches.4–6 Senotherapeutics encompass two classes: senolytics and senomorphics. Senolytic agents can suppress the accumulation of senescent cells by inhibiting SCAPs and thus triggering apoptosis specifically in the senescent cells. This targeted elimination of senescent cells ensures that neighboring normal cells remain unaffected. On the other hand, senomorphics are agents that promote the SASP blockade and thus inhibit accelerated senescence of neighboring cells. Several studies have demonstrated that natural compounds, including quercetin, dasatinib, and fisetin, exhibit significant efficacy as senotherapeutic agents.7–9
Ginsenosides are triterpene saponins with a four-ring skeleton that are unique to the ginseng. Ginsenosides are the major active components of ginseng, and exert a wide range of biological activities; the quality of ginseng is determined by ginsenosides. 10 More than 150 naturally occurring ginsenosides have been identified to date. Among them, ginsenosides Rb1, Rg1, Rg3, Re, and Rd are the most frequently studied.10,11 The potential health benefits of ginsenosides include anti-carcinogenic, anti-inflammatory, anti-stress, anti-oxidative, and anti-aging effects.12–14
Ginseng has been widely utilized in traditional oriental medicine in Asia for thousands of years and is known for its ability to enhance physical performance and slow aging. 15 Ginseng can be classified as either cultivated or wild based on its distinct cultivation methodologies. Cultivated ginseng is farmed on farmlands and attains maximum growth within 4–5 years. However, after 6 years of growth, the epidermal tissue of cultivated ginseng starts to keratinize, with an increase in cavities within, resulting in a decline in quality. 16 In contrast, wild ginseng grows naturally in the forest and is distinguished by its significantly slower growth rate compared with cultivated ginseng. Under optimal natural conditions, wild ginseng exhibits long-term survival, with a lifespan exceeding 20–30 years without a deterioration in quality, and potentially extending up to a century.17,18
In Korea and China, the pharmacological activity of wild ginseng has been found to be greater than that of cultivated ginseng.17,19,20 However, comparative studies on the differences in efficacy between cultivated and wild ginseng and their underlying causes have been limited. One hypothesis is that the differences in efficacy are owing to the variation in the active compounds between cultivated and wild ginseng. 20 Based on previous studies, ginsenoside content varies owing to age of the plant and differences in growing conditions, such as soil fertility, light, and temperature.19,21,22
In this study, we compared the senotherapeutic activities of cultivated and wild ginseng, and demonstrated the potential of ginsenosides exclusively found in wild ginseng as senolytic and senomorphic agents.
Materials and Method
Cell Culture and Treatment
The human dermal fibroblast cell line Hs68 (ATCC, Manassas, VA, USA) was cultured in Dulbecco's modified Eagle medium (DMEM; Gibco, Waltham, MA, USA) supplemented with 10% (v/v) fetal bovine serum (FBS; Gibco, Waltham, MA, USA) and 1% (v/v) penicillin–streptomycin (PS; Gibco, Waltham, MA, USA) and incubated at 37 °C with 5% CO2. To induce senescence, fibroblasts were irradiated once with UV-B light (30 Watt, 312 nm) at an intensity of 20 mJ/cm2 using BIO-SUN irradiation system (Vilber Lourmat, Marne-la-Vallée, France). The irradiation time was about 10–15 s and irradiation distance was 25 nm.
In this study, a vehicle treatment and a combination of 20 μM of quercetin and 500 nM of dasatinib were utilized as negative control and positive control, respectively. Extracts of wild ginseng and cultivated ginseng were obtained as described in previous study. 23 Ginsenosides C-Mc1 and C-O were purchased from Ambo Institute (Daejeon, Korea). To inhibit BAX activation, fibroblast cells were pre-incubated with 2 μM BAI1 (MedChemExpress, China) for 2 h.
Cell Viability
To investigate the cell viability, fibroblasts were seeded at a density of 1 × 104 cells/well in a 96-well plate. Young and senescent cells were incubated with the test materials for 24 h. After incubation, the cells were treated with CCK-8 solution (Dojindo, Japan) at a 1:10 dilution and incubated for 2 h. After this period, the optical density was measured at 450 nm using a plate reader.
Annexin V Apoptosis Assay
For Annexin V/Propidium iodide (PI) staining, fibroblasts were seeded at a density of 1 × 105 cells/well in a 6-well plate, and senescence was induced as described in
Collection of Conditioned Media
To investigate the paracrine effects of senescent fibroblasts on normal cells, conditioned media (CM) was collected and employed as previously described.
3
Fibroblasts were seeded at a density of 1 × 105 cells/well on a 6-well plate and senescence was induced as described in
Real-Time Quantitative PCR (RT-qPCR)
RNA was extracted from cells using AccuPrep® Universal RNA Extraction Kit (Bioneer, Korea). The isolated RNA was reverse transcribed to cDNA using a cDNA synthesis kit (PhileKorea, Seoul, Korea) following the manufacturer's instructions using a Veriti 96 Well Thermal Cycler (Applied Biosystems, USA). Relative gene expression analysis was performed using a StepOnePlus RT-PCR system (Applied Biosystems, USA). Briefly, 20 μL of reaction mixture including each primer (Bionics, Korea) and SYBR green PCR master mix (Applied Biosystems, USA) was subjected to 40 cycles of RT-qPCR. The mRNA expression level was normalized against GAPDH and calculated using the 2−(△△Ct) method. The primers used for RT-qPCR are listed in
Primer Sequences Used for RT-qPCR.

Statistical Analysis
Data are expressed as mean ± SD (standard deviation) and all experiments were conducted in triplicate. Statistical analysis was performed using one-way analysis of variance (ANOVA) through GraphPad Prism 9 software (San Diego, CA, USA).
Results and Discussion
Comparing Senolytic Effects of Cultivated and Wild Ginseng
To evaluate the senolytic effects of the cultivated and wild ginseng extracts on senescent fibroblasts, we assessed the percentage of Annexin V-FITC-positive cells using flow cytometry. Cells were treated with extracts derived from the leaves, stems, roots, and fruits of both wild and cultivated ginseng at a concentration of 0.1% for 24 h. As illustrated in
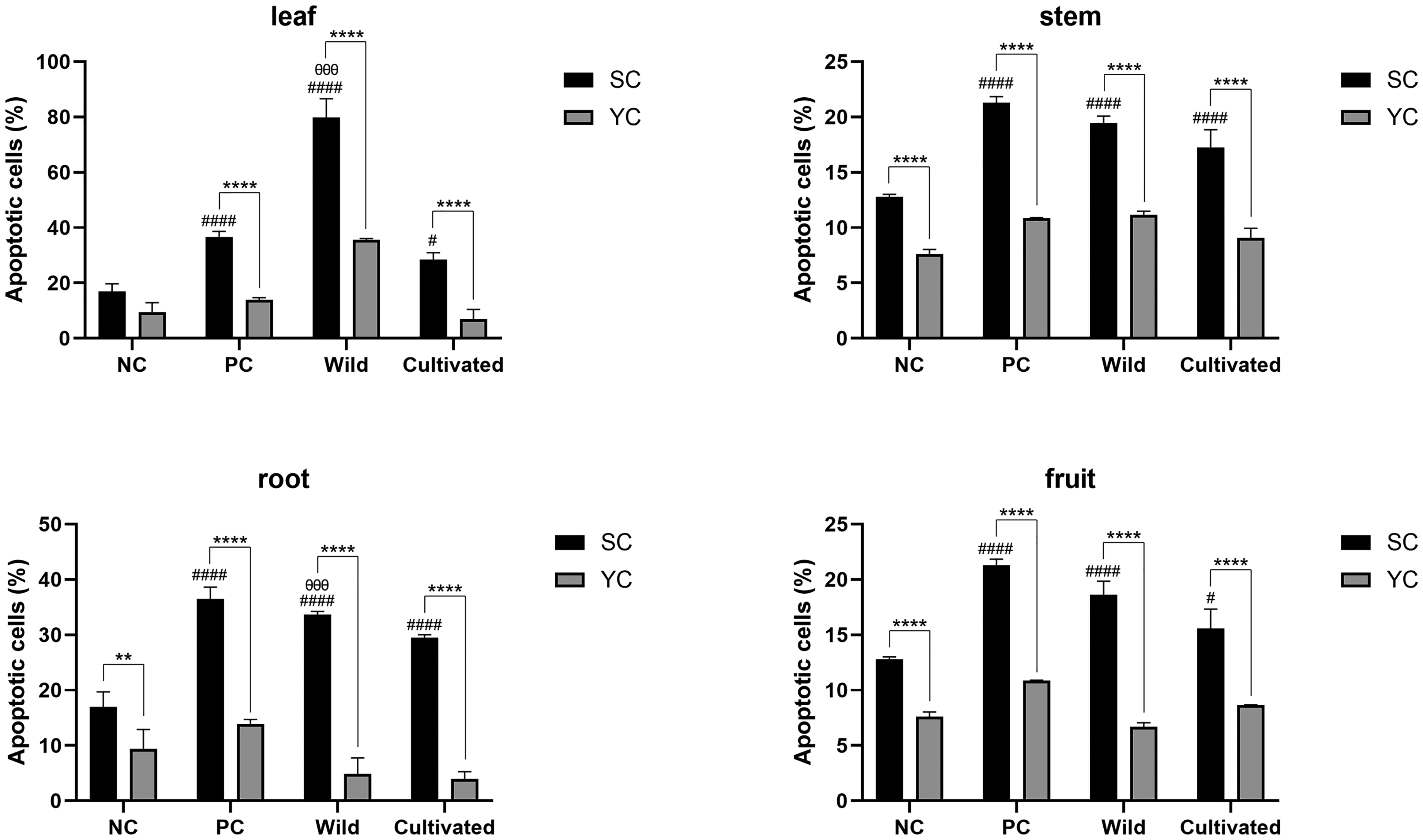
Senolytic Effect of Wild and Cultivated Ginseng Extracts Studied Using the UV-Irradiated Senescence Model (n=3). The Figure Shows the Comparison of Apoptotic Cell Percentages Between Young Cells (YC), Senescent Cells (SC), and Cells Treated with Wild or Cultivated Ginseng Extracts. * Denotes a Statistically Significant Difference Between Young and Senescent Cells, whereas # Indicates a Significant Difference Relative to the Negative Control (Vehicle Treatment) of Senescent Cells. θ Indicates a Significant Difference Relative to Cultivated Ginseng. The Significant Differences are Represented by p < 0.05 (#), p < 0.01 (**), p < 0.001 (θθθ) and p < 0.0001 (****, ####). SC, Senescent Cell; YC, Young Cell; NC, Negative Control; PC, Positive Control.
The wild ginseng exhibited superior senolytic efficacy compared to cultivated ginseng. Specifically, the leaf and root extracts from wild ginseng showed significantly higher portion of apoptotic cells than those from cultivated ginseng (p < 0.001). We also observed that the rate of apoptosis induction was highest in cells treated with extracts from the leaves of wild ginseng (79.6 ± 6.7%), which was 51% higher than that of cells treated with extracts from cultivated ginseng leaves. This indicates that the difference in senolytic efficacy is most pronounced in leaf extracts. Previous studies have revealed that the aerial part of wild ginseng contains higher levels of ginsenosides C-Mc1 and C-O than the underground part. 24 Consequently, we hypothesized that the enhanced efficacy of wild ginseng would be owing to differences in the content of ginsenosides C-Mc1 and C-O. Therefore, we focused on exploring the therapeutic effects of these two ginsenosides.
Senolytic Effects of Ginsenosides C-Mc1 and C-O
To investigate the senolytic effect of the two ginsenosides, we determined the percentage of apoptotic cells using flow cytometry with Annexin V/PI staining. As shown in
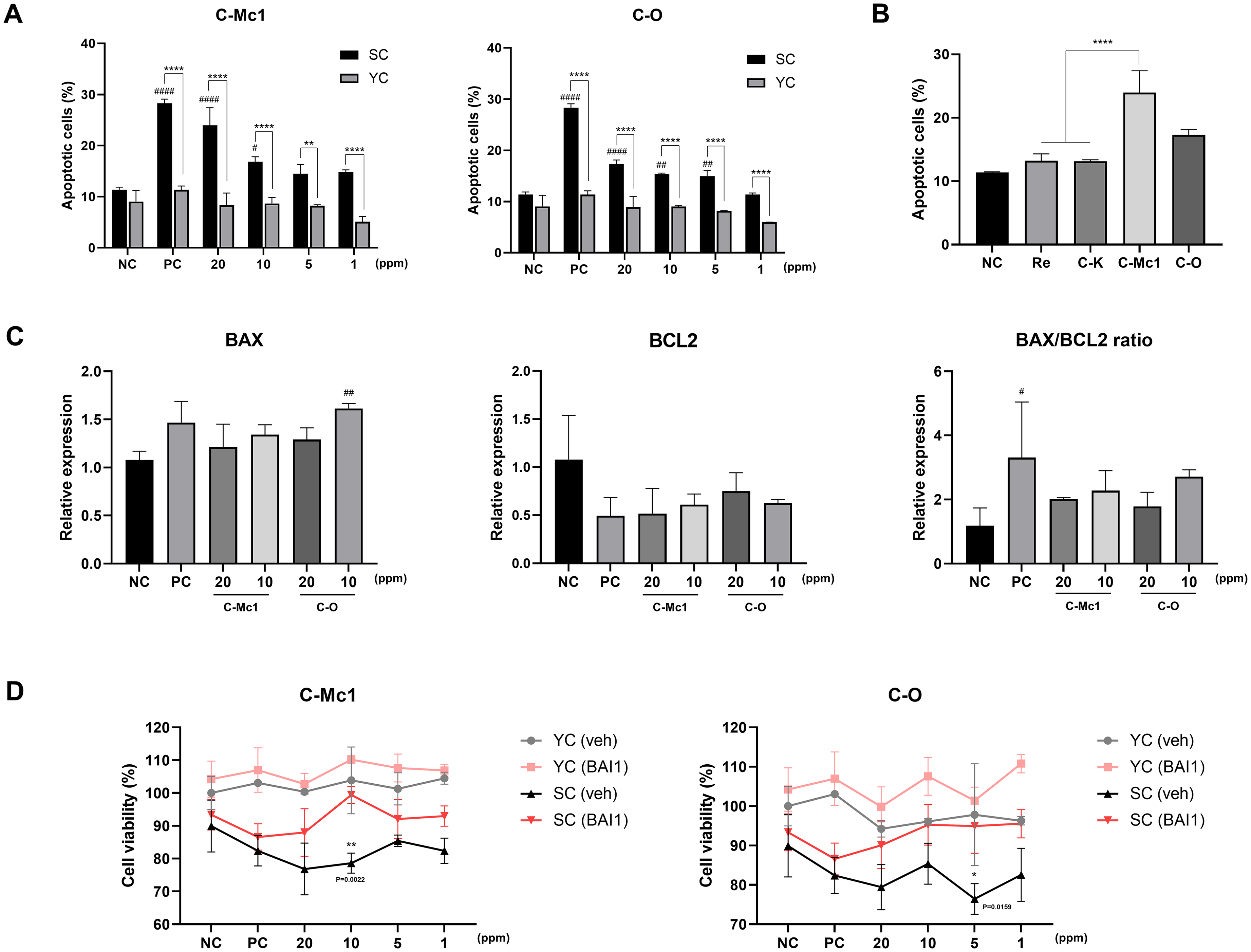
Senolytic Effect of Ginsenosides C-Mc1 and C-O in Senescent Cells. (A) Quantification of the Percentage of the Percentage of Apoptotic Cells 24 h after Ginsenosdies Treatment (n=3). (B) Comparison of Senolytic Effects of Ginsenosides Re, C-K, C-Mc1 and C-O on Senescent Cells. (n = 3) (C) Relative mRNA Expression Levels of BAX and BCL2 in Senescent Cells, Measured by RT-qPCR to Assess Apoptosis Regulation (n =3). (D) Cell Viability of Vehicle- or BAI1-Treated Young and Senescent Cells after Ginsenoside Treatment at the Concentrations Indicated (n=3). * Denotes a Statistically Significant Difference Between Young Cells and Senescent Cells, # Indicates a Significant Difference Relative to the Negative Control of Senescent cells. * in Panel D Denotes a Significant Difference Relative to Senescent Cells Pre-Treated with BAI1. The Significant Differences are Represented by p < 0.05 (*, #), p < 0.01 (**, ##) and p < 0.0001 (****, ####). SC, Senescent Cell; YC, Young Cell; NC, Negative Control; PC, Positive Control.
We also compared the senolytic effect of the two ginsenosides with other well-known ginsenosides (
BCL2 protein family is known for playing a crucial role in regulating apoptosis. BAX is a pro-apoptotic gene that triggers and promotes apoptosis, whereas BCL2 is an anti-apoptotic gene that inhibits programed cell death.9,29 A low BAX/BCL2 ratio is characteristic of resistant cells, whereas a high BAX/BCL2 ratio is indicative of sensitive cells undergoing apoptosis. 30 Senolytic agents such as navitoclax and combination of dastatinib and quercetin target BCL2 family to induce cell death in senescent cells.29,31 Therefore, we measured the mRNA expression levels of BAX and BCL2 using RT-qPCR to investigate whether ginsenosides C-Mc1 and C-O also target BCL2 family members.
As shown in
To further confirm the role of BAX in ginsenoside-induced apoptosis, we investigated the impact of inhibiting BAX activation. Fibroblasts were pre-incubated with 2 μM BAX inhibitor (BAI1) for 2 h to block BAX activation. Subsequently, the cells were treated with ginsenosides C-Mc1 and C-O for 24 h, and cell viability was assessed using the CCK-8 assay. As shown in
Senomorphic Effects of Ginsenosides C-Mc1 and C-O
Senescent cells exhibit elevated expression of the SASP, including inflammatory cytokines and metalloproteases, which propagate senescence through paracrine mechanisms. Recent studies have sought to elucidate the relationship between SASP factors and the acceleration of senescence via paracrine signaling.32–35 For example, senescent melanocytes that accumulate in human skin with aging have been shown to induce paracrine telomere damage in adjacent cells. 32 Specifically, keratinocytes surrounding senescent melanocytes in 3D epidermal equivalents exhibit a significant increase in telomere damage. Furthermore, it was observed that CM derived from senescent melanocytes stimulates the formation of telomere-associated foci (TAF) in dermal fibroblasts by triggering an elevation in the mitochondrial reactive oxygen species (ROS) production, suggesting that the paracrine induction of TAF is mediated by soluble factors released from senescent melanocytes.
Senomorphics can function as SASP inhibitors, thereby indirectly inhibiting senescence. In this study, we aimed to assess the potential of ginsenosides C-Mc1 and C-O as senomorphic agents. Three types of CM were collected as described in

Senomorphic Effect of Ginsenoside C-Mc1 and C-O in Senescent Cells. (A) Schematic Illustration Showing the Collection of Conditioned Media for Subsequent Analysis. (B) RT-qPCR Validations of SASP Factors in Fibroblasts Treated with CM for 24 h (n=3). (C) Gene Expression of NF-κB in Senescent Fibroblasts Treated with Ginsenosides. The Gene Expression was Normalized with Negative Control (Young Cells) (n =3). * and # Denote Significant Difference Compared with Senescent Cell Control and Young Cell Control, Respectively. The Significant Differences are Represented by p < 0.05 (*, #), p < 0.01 (**, ##), p < 0.001 (***) and p < 0.0001 (****, ####). SC, Senescent Cell; YC, Young Cell; NC, Negative Control; PC, Positive YC, Young Cell; SC, Senescent Cell; CM, Conditioned Media.
To investigate the mechanism underlying the inhibition of SASP, we examined the regulation of the NF-κB signaling pathway following ginsenoside treatment using RT-qPCR analysis (
Collectively, these results indicate that ginsenosides C-Mc1 and C-O act as senomorphic agents, reducing SASP by targeting the NF-κB signaling pathway.
Our study offers novel insights into the senotherapeutic efficacy of Ginsenosides C-Mc1 and C-O, which have received less attention compared to ginsenosides Re and C-K. However, several limitations should be considered. This study provides insight into the underlying mechanisms, including the modulation of the BCL2 family and the NF-κB pathway, which are central to the senolytic and senomorphic processes. However, given the complexity of cellular mechanisms, further research is required to fully elucidate the pathways involved in senolytic and senomorphic processes. Additionally, the results are based on in vitro experiments, so validation in in vivo models is necessary to assess the systemic effects and therapeutic potential of Ginsenosides C-Mc1 and C-O. Moreover, this study primarily focuses on a single cell line. Testing across different cell types, such as keratinocytes and adipocytes, would provide further insight into the broader applicability of these compounds. Despite these limitations, the findings offer a strong foundation for future research into Ginsenosides C-Mc1 and C-O as promising senotherapeutic agents.
Conclusion
Wild ginseng extracts showed higher senolytic efficacy than cultivated ginseng extracts. Based on a previous study indicating that the aerial part of wild ginseng contains high levels of ginsenosides C-Mc1 and C-O, further research on these compounds was conducted. The current investigation demonstrated that ginsenosides C-Mc1 and C-O, the key bioactive constituents of wild ginseng, selectively trigger apoptosis in senescent cells by modulating the BCL2 protein family. Additionally, these ginsenosides demonstrate senomorphic activity by attenuating the expression of SASP-related genes. Thus, ginsenosides C-Mc1 and C-O exhibit senotherapeutic properties and hold promise as potential anti-aging agents, effectively regulating cellular senescence and preventing the accumulation of senescent cells.
Footnotes
Ethical Considerations
Ethical Approval is not applicable for this article.
Author Contributions
Dohyun Kim: Methodology, Investigation, Validation, Formal analysis, Data curation, Writing – Original Draft. Seongsu Kang: Conceptualization, Methodology, Writing – Review & Editing. Seung-Hyun Jun: Conceptualization, Writing – review & editing, Supervision, Project administration. Nae-Gyu Kang: Writing – review & editing, Supervision, Project administration
Funding
All research in this article was funded by LG H&H. This research received no external funding.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
