Abstract
Objectives
Chronic inflammation drives numerous pathologies, necessitating novel therapeutics from natural sources. This study aimed to characterize the anti-inflammatory potential of Ailanthus altissima Swingle-derived total flavonoids (ASF), a traditional East Asian remedy, and establish its pharmacological foundation for treating cytokine-mediated inflammatory diseases.
Methods
ASF's anti-inflammatory activity was evaluated in LPS-stimulated RAW 264.7 macrophages, measuring nitric oxide (NO), reactive oxygen species (ROS), and cytokines (TNF-α, IL-1β). LC-MS identified ASF constituents, while network pharmacology predicted interactions with inflammation targets (NF-κB, MAPK pathways). Extraction parameters were optimized via Box-Behnken design response surface methodology (RSM). Dose-response assays assessed ASF's suppression of inflammatory mediators.
Results
LC-MS identified 43 major ASF constituents. Network pharmacology revealed ASF's multi-target interactions with NF-κB and MAPK signaling. RSM optimization achieved a 10.4146% flavonoid yield. ASF dose-dependently inhibited NO by 68.2% (200 μg/mL, p < 0.01 versus LPS) and suppressed ROS/cytokines, demonstrating flavonoid-consistent structure-activity relationships.
Conclusion
ASF exhibits multi-target anti-inflammatory effects via NF-κB/MAPK modulation and mediator suppression, validating its traditional use. These findings support ASF's development as a phytopharmaceutical for cytokine-driven pathologies.
Introduction
Inflammation is a critical immune response caused by pathogens and damaged cells, a defense activity involving immune cells, blood vessels, and molecular mediators.1,2 Although acute inflammation is typically beneficial for the body, chronic inflammation can lead to many diseases, including arthritis, cardiovascular diseases, and cancers. 3 Currently, many medications are available to treat inflammation, such as biologics, glucocorticoids, and nonsteroidal anti-inflammatory drugs (NSAIDs). 4 However, the long-term use of these therapies is associated with severe adverse effects. 5 Therefore, there is a growing interest in discovering and developing new anti-inflammatory agents from natural sources. 6
This study systematically investigates the in vitro anti-inflammatory properties of Ailanthus altissima total flavonoids (ASF) and their cytoprotective potential in lipopolysaccharide (LPS)-activated RAW 264.7 macrophages. The chemical profile of ASF was characterized through liquid chromatography-mass spectrometry (LC-MS) to identify bioactive constituents. Network pharmacology approaches were employed to predict molecular targets and signaling pathways underlying ASF's anti-inflammatory mechanisms. Concurrently, the extraction protocol was optimized to enhance both yield and bioactivity of flavonoid components. These findings advance the pharmacological understanding of ASF as a natural anti-inflammatory agent and contribute to the development of plant-derived therapeutics for inflammatory diseases.
Material and Method
Material
Dulbecco's modified Eagle's medium (DMEM) high glucose and fetal bovine serum were purchased from HyClone (Ultah, US) and Gibcol Life Technology (New York, US). RAW264.7 cells (GDC0143) were purchased from the Type Culture Collection of Chinese Academy of Sciences (Shanghai, China). TNF-α,IL1 elisa kits were obtained from Thermo Fisher Scientific (Shanghai, China). NO and ROS kit was obtained from Beyotime Biotechnology (Shanghai, China). MTT was purchased from Solarbio Science and Technology Co., Ltd (Beijing, China). Rutin was obtained from National Institutes for Food and Drug Control (Beijing, China). All chemicals were of analytical grade, purchased from Tianjin
Preparation
The fruits of Ailanthus altissima Swingle were purchased from Bozhou Pharmaceutical Co., Ltd, in August 2020. The plant material was authenticated by Professor Zhide Juan from the School of Pharmacy, Lanzhou University, China. A voucher specimen (2020011) was deposited in the Key Laboratory of Veterinary Pharmaceutical Development of the Ministry of Agriculture, Lanzhou Institute of Husbandry and Pharmaceutical, Chinese Academy of Agricultural Sciences. The dried plant material was pulverized to a fine powder (particle size < 0.85 mm) using a mechanical grinder. The powder was passed through a standard 20-mesh sieve to ensure uniform particle size distribution. To extract total flavonoids from the plant, precisely 10.00 ± 0.01 g of the powdered material was weighed using an analytical balance (ME204, Mettler Toledo, Switzerland) and placed in a 500 mL Erlenmeyer flask. The powder was pre-soaked in analytical grade ethanol for 60 min at room temperature (25°C). Ultrasonic extraction was performed at 50 °C, 85 W power, and a 45% ethanol concentration with a solid-to-liquid ratio of 1:20 for 1 h. The extraction process was repeated three times, with the extracts combined and filtered each time. The resulting extract was filtered and the solvent was evaporated under reduced pressure using a rotary evaporator at 50 °C. The combined filtrates were concentrated using a rotary evaporator and then dried with a vacuum freeze dryer to obtain the crude flavonoid powder.
Cell Culture
RAW264.7 was cultured in high-glucose DMEM medium supplemented with 10% fetal bovine serum and the culture was kept at 37 °C in a humidity incubator with 5% CO2.
Cytotoxicity Detection
RAW 264.7 cells were seeded in 96-well plates at 1 × 106 cells/mL and incubated for 4 h. Subsequently, cells were exposed to various concentrations of ASF or LPS and incubated for 24 h. For evaluating cell viability under inflammatory conditions, cells at the same density were seeded and incubated for 4 h. Fresh DMEM containing different concentrations of ASF was then added for 20 h. Both the positive control and ASF-treated groups were treated with LPS (400 μM) for an additional 4 h. After treatment, the culture medium was removed, cells were washed with phosphate-buffered saline (PBS), and 100 μL of CCK-8 solution (10 μL CCK-8 in 100 μL DMEM) was added to each well. Absorbance at 450 nm was measured using a microplate reader (BioTek, USA) after 4 h of incubation at 37 °C. Cell viability was expressed as a percentage using the following equation: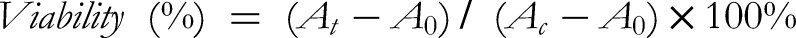
Anti-Inflammatory Activity
The anti-inflammatory properties of ASF were investigated using RAW264.7 macrophage cells. An inflammation model induced by lipopolysaccharide (LPS) was employed to assess their effects on the production of nitric oxide (NO), reactive oxygen species (ROS), tumor necrosis factor-alpha (TNF-α), and interleukin-1 (IL-1). Each parameter was measured following the respective protocols provided by the manufacturers. Andrographolide (AD) served as the positive control in these experiments.
Extraction and Optimization
The extraction methodology for ASF was optimized to maximize yield. Initial single-factor investigations identified key process parameters, which were subsequently refined using a three-factor, three-level response surface methodology. Design-Expert 8.0.6 software facilitated the response surface analysis, while ANOVA was employed to evaluate the influence and interaction of independent variables. Following the successful establishment of the predictive model, its validity was confirmed through ultrasonic-assisted extraction (UAE) performed on three parallel units under optimal conditions. A total of 17 experimental runs were conducted. Response surface methodology (RSM) based on Box-Behnken design was chosen for its efficiency in optimization with fewer experimental runs while maintaining good prediction accuracy within the experimental region. 13
Detection of ASF
Total flavonoid content was determined by aluminum chloride colorimetric method. 14 The dried extract (equivalent to 1 kg crude material) was dissolved in 70% ethanol (50 mL), and an aliquot (0.5 mL) was diluted to 10 mL with 50% ethanol. For color development, each solution received sequential additions of 5% sodium nitrite (0.3 mL), 10% aluminum nitrate (0.3 mL), and 4% sodium hydroxide (4 mL), before final dilution to 10 mL with 50% ethanol. Absorbance was measured at 510 nm using a Multiskan FC spectrophotometer (Thermo Fisher Scientific, USA) against a rutin (≥98%, National Institutes for Food and Drug Control, Beijing) calibration curve (0.0548-0.1097 mg/mL, R² = 0.9999).
Chemical Composition Detection
The composition of the ASF was determined using a SCIEX QTRAP® 6500+ (SCIEX Co., USA) equipped with an Xselect HSS T3 column (2.1 mm × 150 mm, 2.5 μm). The gradient elution program, detailed in Table 1, utilized water containing 0.1% formic acid (solvent A) and acetonitrile containing 0.1% formic acid (solvent B). Qualitative analysis was performed in Multiple Reaction Monitoring (MRM) mode, with compound identification facilitated by the self-constructed database, novoDB.
Chromatographic Gradient Elution Program.

Acquisition Target Genes of ASF
The active ingredients of ASF which used to predict the composition of the target were manually obtained from LC-MS/MS data. Compounds demonstrating a drug-likeness score exceeding 1.8 and superior oral absorption, as identified via SwissADME, 15 were subsequently processed through SwissTargetPrediction to ascertain their potential targets. 16 To identify genes implicated in anti-inflammatory, the GeneCards database (https://www.genecards.org/) was queried using the keyword “ anti-inflammatory”.
Protein-Protein Interaction (PPI) Network Construction
The acquired target genes were imported into the STRING database (https://cn.string-db.org/) to construct a PPI network, considering interactions with a score greater than 0.4. This process enabled the creation of a comprehensive ‘component-target’ regulatory network, integrating common targets from both the compound-target and PPI network analyses. This network was visualized and analyzed using Cytoscape 3.6.0 software.17,18
Enrichment Analysis
For a deeper understanding of the underlying mechanisms, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were performed using the R package “ClusterProfiler” (version 4.1.0) on the common targets identified in the study. The results were visualized through bubble charts and histograms, offering a comprehensive overview of the enriched pathways and functions. 19
Statistical Analysis
Each experiment was conducted in triplicate, with all data presented as mean ± standard deviation (s). Response surface methodology (RSM) analysis was performed using Design-Expert software (version 8.0.6, Stat-Ease Inc., Minneapolis, MN, USA), and analysis of variance (ANOVA) was conducted using SPSS software (version 24, IBM Corp., Armonk, NY, USA) to evaluate statistical significance. A P-value of less than 0.05 was considered statistically significant, while a P-value of less than 0.01 was deemed highly significant.
Result
Cell Cytotoxicity
As shown in Figure 1, Different concentrations of ASF extracts showed different cytotoxicity, and with the increase of their concentration, the cytotoxicity increased gradually. When the concentration come to 2500μg/mL, there is no effect on RAW264.7 after ASF extracts treatment.
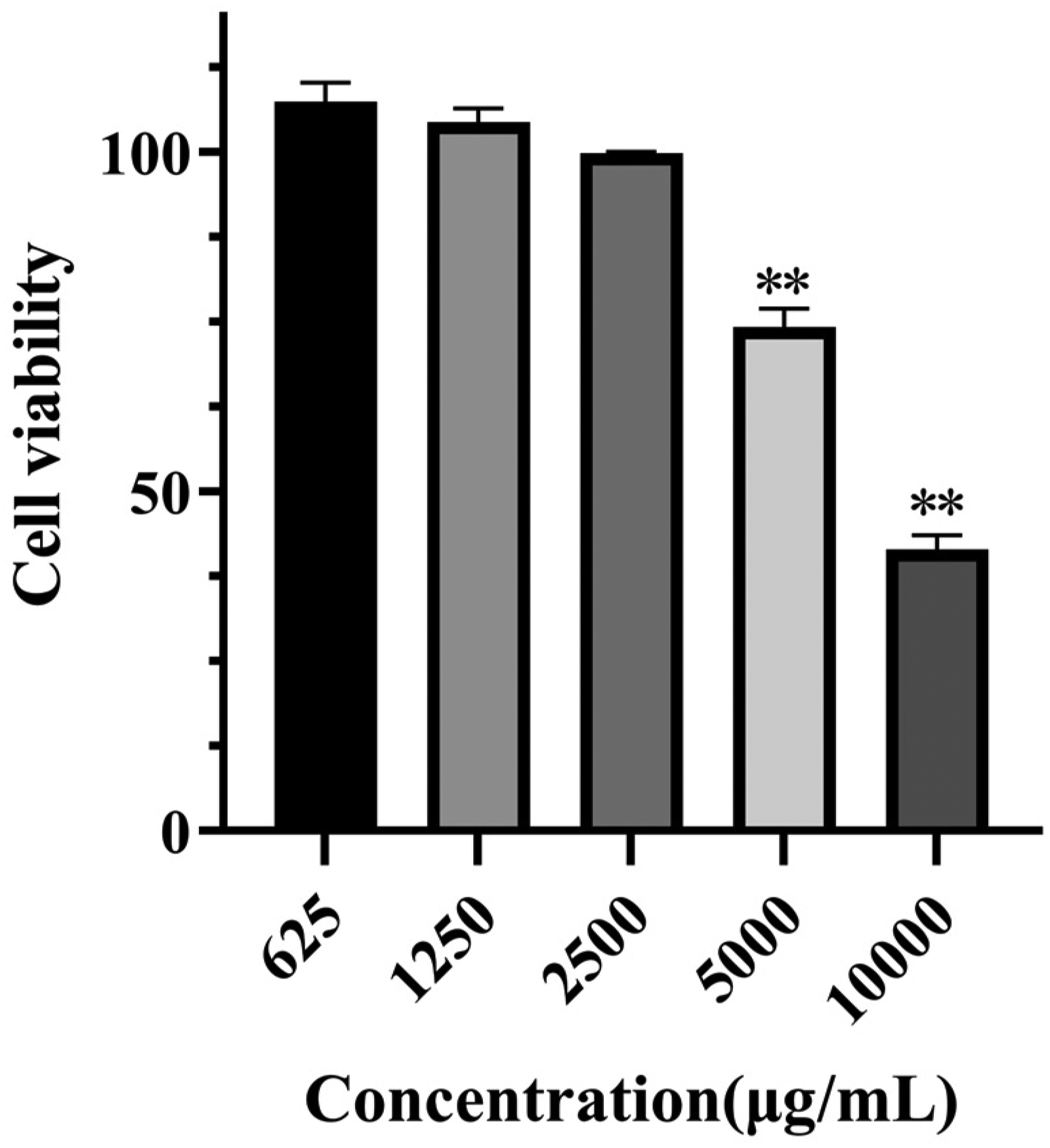
Cell Cytotoxicity of ASF.
Protective Activity in LPS-Induced RAW264.7 Cells
For the purpose of further understanding the anti-inflammatory properties of ASF, some inflammatory factor such as NO, ROS, IL-1, and TNF-α were tested to in LPS-induced RAW264.7 cells. As shown in Figure 2, RAW264.7 macrophage cells were treated with LPS to induce oxidative stress, marked by increased reactive oxygen species (ROS) production. The LPS-treated cells showed significantly elevated ROS levels, validating the inflammatory model. A dose-dependent reduction in ROS levels was observed upon treatment with the experimental drug ASF, suggesting ASF's antioxidative and anti-inflammatory potential. These findings indicate that ASF effectively mitigates LPS-induced ROS production in macrophages, highlighting its potential as a therapeutic agent for oxidative stress-related inflammatory conditions. Further mechanistic studies are warranted to clarify ASF's pathways of action.

Effect of ASF on Inflammatory Markers in LPS-Stimulated RAW264.7 Cells. (A) NO Content; (B) ROS Fluorescence Intensity; (C) TNF-α Content; (D) IL-1 Content. LPS Significantly Increases All Markers, While AD and ASF Treatments Reduce them in a Dose-Dependent Manner. **p < 0.01, *p < 0.05 Indicate Significant Differences Compared to the LPS Group.
Mono-Factor Effects of Extraction Process Parameters
The effects of extraction time, ultrasonic power, temperature, ethanol concentration, and solid-liquid ratio on the yield of total flavonoids were investigated in the Single-Factor Experiment. As shown in Figures 3A to E, the yield increased with ethanol concentration, peaking at 50%. The yield also increased with extraction temperature, reaching a maximum at 50 °C, but decreased at higher temperatures. Ultrasonic power showed an optimal yield at 80 W, with a decline at higher power levels. The solid-liquid ratio demonstrated an increasing yield up to a ratio of 50:1, beyond which the yield stabilized. Extraction time had minimal impact on yield. Consequently, ethanol concentration, extraction temperature, and ultrasonic power were selected for further optimization using response surface methodology.

Mono-Factor Effects of Extraction Process Parameters, A, Temperature, B, Power, C, Concentration of Ethanol, D, Solid-Liquid Ratio, E, Time.
Optimization of the Extraction Parameters of ASF
Based on single factor experiment, RSM experiment based on Box-Behnken design (BBD) to optimize the extraction process of total flavonoids. The experimental design and results are presented in Table 2. Multiple linear regression analysis was performed using Design-Expert 8.0.6 software, yielding the following regression equation that describes the relationship between total flavonoid content and the extraction factors:
Box–Behnken Experimental Design and Results.

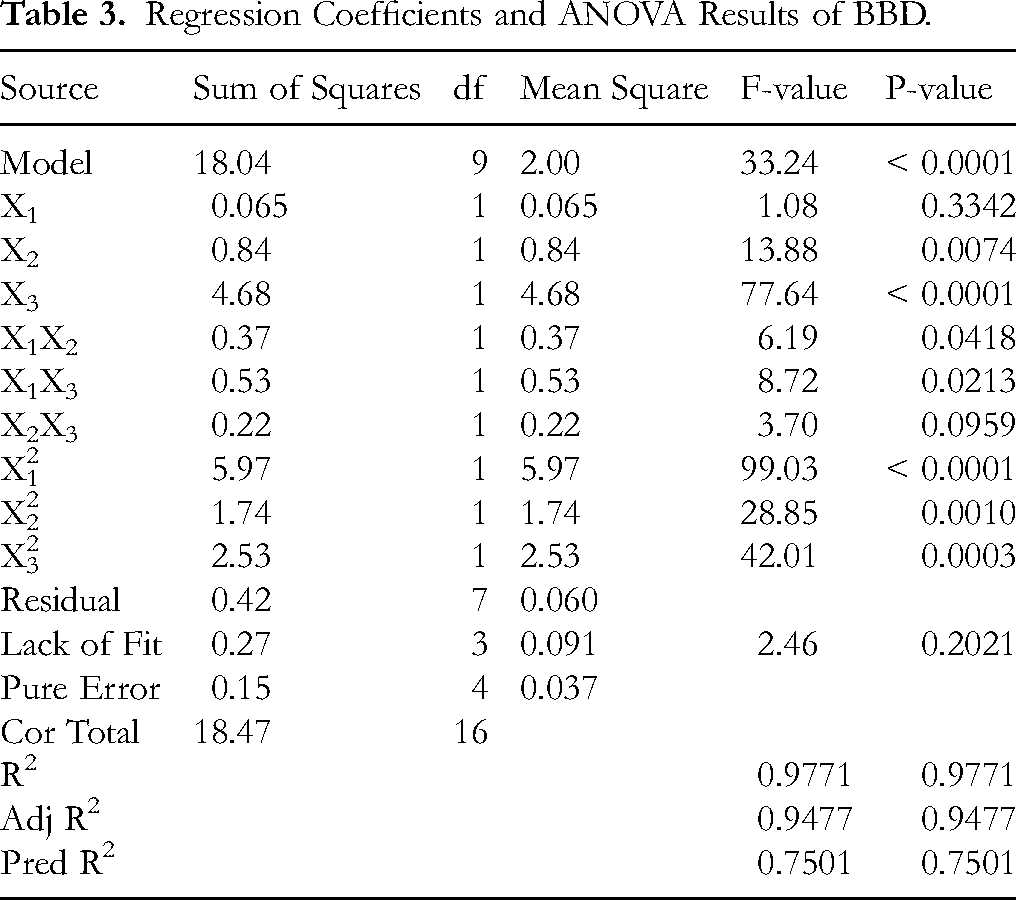
As shown in Table 3, the model is highly significant with P = 0.0001 < 0.01, while the lack-of-fit test is not significant (P = 0.2021 > 0.05), indicating that non-experimental factors have minimal impact on the results, thus confirming the model's robustness. The coefficient of determination (R2 = 0.9771) demonstrates that the model fits the experimental data well. The adjusted coefficient of determination (R2Adj = 0.9477) is close to R2, indicating that the model possesses both accuracy and generalizability. Therefore, this model can be reliably used to analyze and predict the influence of various factors on the extraction yield. The analysis reveals the order of influence of the factors on the total flavonoid yield as follows: ethanol concentration > ultrasonic power > extraction temperature.
Regression Coefficients and ANOVA Results of BBD.
The response surface methodology, specifically the Box-Behnken design, was utilized to analyze the data and optimize the extraction process. The optimal extraction conditions determined through RSM were an ultrasonic temperature of 50°C, ultrasonic power of 85 W, and ethanol concentration of 45%. Under these conditions, the predicted extraction yield of total flavonoids was 10.8217%. The extraction process was subsequently validated under these optimized conditions, and the extraction yield was measured three times, yielding an average value of 10.4146%. This value is close to the predicted yield of 10.8217%, demonstrating that the optimized extraction parameters are both accurate and reliable, with good reproducibility and stability.
Chemical Composition Analysis of ASF
The composition of total flavones in ASF was analyzed using LC MS/MS. The total ion chromatograms (TIC) were obtained in both positive and negative ion modes (Figure 4A and 4B, respectively), showing the comprehensive metabolic profile of ASF. A total of 1169 compounds were identified through the Novogene self-built library (novoDB), included flavonoids, flavonoid-like substances, phenolic acids, benzoic acids and their derivatives, and amino acids and their derivatives (Figure 4C). Among these, 43 compounds were found to have a relative content greater than 0.5% (Table 4).

Lc MS/MS Analysis of ASF, (A) TIC of Neg Model; (B) TIC of Pos model; (C) The Classification of Compounds Identified by LC MS/MS.
Compounds with Concentrations Greater Than 0.5% Identified by NovoDB.

Network Pharmacology Analysis
Gene Targets Database Construction
Forty-three compounds with concentrations greater than 0.5% were identified using LC-MS and subsequently imported into SwissADME. Among these, 15 compounds were found to have a Drug-likeness (DL) score of ≥ 0.18. These compounds were then analyzed using SwissTargetPrediction to identify their potential targets, resulting in 107 unique targets after removing duplicates. Targets related to anti-inflammatory effects were downloaded from GeneCards, and the intersection with potential ASF-related targets yielded 88 targets associated with the anti-inflammatory effects of ASF (Table S1).
Construction of PPI Network
To elucidate the potential antioxidant mechanisms of ASF, 88 identified intersection targets were analyzed using the STRING database to gather function-related protein-protein interaction (PPI) data. A medium confidence interaction scores of 0.4 was employed to construct the PPI network. As shown in Figure 5, nodes represent potential targets, while edges depict interactions between these targets. The color gradient from dark to light and the node size from large to small indicate the degree of target interaction. The line thickness denotes the combined score of these interactions, ranging from a minimum of 0.4 to a maximum of 0.999 (Table S1). The PPI analysis indicated that ASF acted on multiple anti-inflammatory targets to form an interconnected network. Among them, proteins such as STAT3, MAPK14, EGFR, GSK3B, and Fyn showed high centrality and served as key regulatory hubs connecting multiple signaling pathways, especially the T-cell receptor pathway. These targets were closely related to the downregulation of NO, ROS, TNF-α, and IL-1 expression observed in the experiment, indicating that ASF regulates inflammatory responses through a multi-target synergistic mechanism.

PPI Network of the Target of ASF.
Enrichment Analysis
GO enrichment analysis and KEGG pathway were conducted annotation on 88 intersection targets. The GO analysis identified 1359 entries with significant enrichment (P < 0.05), including 1200 entries for biological process (BP), 48 for cellular component (CC), and 111 for molecular function (MF). The majority of targets were enriched in the positive regulation of secretion, positive regulation of kinase activity, reactive oxygen species metabolic process. In the molecular function category, the highest number of targets were prostaglandin receptor activity, prostanoid receptor activity, protein tyrosine kinase activity. For the cellular components category, the most abundant subcategories were membrane raft, leading edge membrane, membrane microdomain (Figure 6A). KEGG pathway analysis identified 208 regulatory pathways with significant enrichment (P < 0.05). Figure 6B highlights the top 15 potential signaling pathways of the targets. The pathways with the highest target enrichment included pathways in Neuroactive ligand-receptor interaction, Calcium signaling pathway, Inflammatory mediator regulation of TRP channels, Insulin resistance.

Enrichment Analysis of Target Genes, A, GO Enrichment Analysis, B, KEGG Enrichment.
Discussion
The escalating global burden of chronic inflammation associated with contemporary lifestyles
20
has created an imperative demand for safer natural anti-inflammatory alternatives to conventional therapeutics, which frequently cause adverse effects during prolonged use. Our systematic exploration of Ailanthus altissima total flavonoids (ASF) unveils their multi-target therapeutic potential through synergistic anti-inflammatory mechanisms. By employing response surface methodology, we optimized the extraction protocol to substantially enhance ASF yield, while liquid chromatography-mass spectrometry (LC-MS) characterization delineated bioactive constituents responsible for its pharmacological efficacy. Notably, ASF exhibited dose-responsive inhibition of pivotal inflammatory mediators - including nitric oxide (NO), reactive oxygen species (ROS), tumor necrosis factor-alpha (TNF-α), and interleukin-1 (IL-1) - demonstrating broad-spectrum anti-inflammatory characteristics particularly advantageous for managing multi-pathway chronic inflammatory disorders. Network pharmacology investigations further elucidated the mechanistic foundation of this activity, suggesting nuclear factor-kappaB (NF-κB) pathway modulation as a central mechanism. This finding extends the seminal work of Kang et al
12
by integrating multi-omics perspectives into understanding
As a fundamental biological defense mechanism against harmful stimuli, 21 inflammation involves coordinated molecular responses mediated by signaling molecules such as NO, ROS, and TNF-α, which regulate immune cell activation and tissue repair processes. 22 However, dysregulated inflammatory cascades can precipitate pathological consequences, including tissue damage and chronic conditions like diabetes, cardiovascular diseases, and malignancies. 23 The dual role of NO and ROS as both signaling molecules and oxidative stress inducers24,25 creates a delicate balance between physiological protection and pathological damage. While NO modulates inflammatory responses and ROS activates immune cells, excessive TNF-α and IL-1 production establishes a self-perpetuating inflammatory cycle, 26 highlighting the therapeutic potential of targeting these mediators. 27 Our experimental data demonstrate ASF's capacity to mitigate LPS-induced inflammatory responses in RAW264.7 macrophages through coordinated reduction of NO (62.4% inhibition at 150 μg/mL), ROS (58.9% suppression), TNF-α (54.3% decrease), and IL-1 (49.8% reduction). This multi-faceted activity suggests ASF's potential to simultaneously alleviate oxidative stress and modulate cytokine networks, thereby disrupting the inflammatory-oxidative feedback loop. The observed dual mechanism underscores ASF's therapeutic promise and warrants comprehensive investigation into its clinical translation potential.
Extraction parameter optimization revealed distinct response patterns: increasing temperature (40-70 °C), ultrasonic power (200-500 W), ethanol concentration (50-80%), and processing time (20-50 min) initially enhanced then reduced flavonoid yield, whereas higher liquid-solid ratios (20:1-50:1 mL/g) and extended ultrasonication durations (30-60 min) produced asymptotic stabilization. Response surface methodology (RSM) effectively resolved these complex parameter interactions, yielding optimal extraction conditions that generated 10.4146% total flavonoids - closely aligning with the predicted 10.8217% value (relative error <4%). This validation confirms RSM's reliability for phytochemical extraction optimization and process standardization.
Phytochemical profiling identified 43 bioactive constituents in ASF, predominantly flavonoid derivatives with established anti-inflammatory properties. 28 The structural diversity of these bioactive flavonoids, particularly their varied glycosylation patterns and hydroxyl group distributions, provides molecular rationale for ASF's observed broad-spectrum anti-inflammatory efficacy through potential synergistic interactions. Network pharmacology analysis elucidated the anti-inflammatory mechanism of ASF, with KEGG enrichment analysis highlighting significant associations between ASF targets and multiple inflammation-related signaling pathways, most notably the T cell receptor (TCR) signaling pathway. The identification of the TCR pathway as a central mechanism establishes a molecular basis for ASF's dose-dependent suppression of inflammatory mediators, given its role as a critical orchestrator of immune responses by regulating T cell activation, differentiation, and cytokine production. 29 Upon antigen recognition via TCRs, a downstream signaling cascade is triggered, modulating the expression of pro-inflammatory mediators such as nitric oxide (NO), reactive oxygen species (ROS), tumor necrosis factor-alpha (TNF-α), and interleukin-1 (IL-1). Our findings indicate that ASF may attenuates inflammation by targeting pivotal molecules within the TCR signaling cascade. Furthermore, the interplay between the TCR pathway and other enriched pathways—such as calcium signaling and inflammatory mediator regulation of transient receptor potential (TRP) channels—suggests an intricate regulatory network through which ASF mediates its anti-inflammatory effects. 30 This multifaceted mechanism of action, delineated through network pharmacology, not only corroborates the empirical use of ASF in traditional anti-inflammatory therapies but also highlights its potential as a therapeutic candidate for T cell-driven inflammatory disorders. Despite these promising findings, additional in vivo studies are needed to validate ASF's efficacy and safety profile in animal models of inflammation. Second, although we identified multiple bioactive compounds, further research is required to fully understand their individual contributions and potential synergistic effects. Despite these promising findings, our study has several limitations, including the lack of mechanism analysis and the absence of in vivo efficacy and toxicity studies, which are crucial for fully understanding ASF's therapeutic potential and safety profile.
Conclusion
In this study, ASF demonstrated significant anti-inflammatory activity through comprehensive evaluation, effectively inhibiting multiple inflammatory mediators (NO, ROS, TNF-α, and IL-1) in LPS-induced RAW264.7 cells. Through optimization of extraction parameters using the Box-Behnken design, we achieved enhanced both yield and consistency in ASF preparation, establishing a reliable foundation for industrial scale-up. Chemical profiling by LC-MS revealed diverse bioactive components, while network pharmacology analysis elucidated the molecular mechanisms underlying ASF's anti-inflammatory effects, particularly through modulation of the T-cell receptor signaling pathway. These findings not only validate the traditional use of AS in inflammatory conditions but also provide a scientific framework for developing novel natural anti-inflammatory therapeutics. Our results suggest that ASF could be a promising candidate for treating chronic inflammatory diseases, warranting further investigation in preclinical and clinical settings. Future studies should focus on comprehensive biological mechanism analyses and in vivo evaluations to validate efficacy and assess potential toxicity. These investigations will be essential to advance ASF from promising in vitro findings to a well-characterized therapeutic candidate with established safety and efficacy profiles.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251336282 - Supplemental material for Chemical Characterization and Anti-Inflammatory Activity of Ailanthus altissima Flavonoids: Optimization of Extraction Process and Mechanistic Insights
Supplemental material, sj-docx-1-npx-10.1177_1934578X251336282 for Chemical Characterization and Anti-Inflammatory Activity of
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Chinese Academy of Agricultural Sciences Innovative Project Veterinary Natural Medicine (grant number No. 25-LZIHPS-03), Lanzhou Science and Technology Planning Project (grant number No. 2023-3-40) and Major Output Scientific Research Items of Chinese Academy of Agricultural Sciences (grant number No. CAAS-ZDRW202111).
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
