Abstract
Introduction
Coronavirus disease 2019 (COVID-19) is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). 1 Due to its strong infectivity and highly pathogenic characteristics, COVID-19 is rapidly spreading around the world and thus seriously affecting the life and health of people worldwide. The treatment and elimination of COVID-19 have become a common problem facing all mankind. Since ancient times, many epidemics of plagues have been recorded in China, and TCM has made great contributions to the prevention and treatment of epidemics. 2 In view of the characteristics of “dampness, heat, toxicity, blood stasis, and deficiency” presented by this epidemic, a series of TCM treatment schemes have been successively introduced, and these include Qingfei detoxification and Maxing Shigan and Shegan Ephedra decoctions, which have achieved clear results.3,4
Chaiyin particles (CYPs), which are prepared from Chaigu Jieyi decoction and Yinqiao powder, have the effect of clearing heat and detoxification and thus supporting the pharynx and relieving cough. These particles are commonly used in Chinese medicine to relieve sweating and reduce fever in clinical practice and exert definite curative effects on fever, wind-cold, headache, sore throat, sweating, and cough.5,6 The CYPs formulas include Bupleurum chinense DC. (BC), Lonicera japonica Thunb. (LJT), Forsythia suspensa (Thunb.) Vahl (FS), Houttuynia cordata Thunb. (HCT), Scutellaria baicalensis Georgi. (SBG), Pueraria lobata (Willd.) Ohwi (PLO), Schizonepeta tenuifolia Briq. (STB), Artemisia annua L. (AAL), Mentha haplocalyx Briq. (MHB), Semen Armeniacae Amarum [SAA, including Prunus armeniaca L. var. ansu Maxim., Prunus sibirica L., Prunus mandshurica (Maxim.) Koehne or Prunus armeniaca L.], and Platycodon Grandiflorum (Jacq.) A.DC. (PGA). The primary and secondary components interact and complement each other. Bao Yanyan constructed a mouse model of human coronavirus-associated acute respiratory distress syndrome combined with pneumonia and simulated typical COVID-19 symptoms, and the results showed that CYPs can enhance the body's immunity and reduce the expression of inflammatory factors in the lung, which indicates a clear therapeutic effect on typical COVID-19 symptoms. 7 However, the mechanism of CYPs in the treatment of COVID-19 has not been thoroughly studied due to the complex composition of TCMs and their interactions with each other, which have restricted the development and widespread utilization of CYPs.
Network pharmacology is based on the “drug-target-disease” interaction network, and the research strategy is integrated and systematic, consistent with the advantages of the multicomponent, multitarget, and multipathway synergistic effects of TCM. In recent years, the development and improvement of network pharmacology has provided a new perspective for the systematic study of complex components of TCMs. 8 To explore the potential effects of CYPs treatment on COVID-19 symptoms and the underlying mechanism of action and to make CYPs safer and more effective for the clinical treatment of COVID-19, we used pharmacological and molecular docking methods and network analysis to perform a more in-depth exploration of the potential active components of CYPs and the interactions among the targets, functions, and pathways. After performing a systematic analysis and screening for components that may bind to key SARS-CoV-2 target proteins, we verified the active component of CYPs and their potential efficacy experimentally; the workflow is shown in Figure 1.
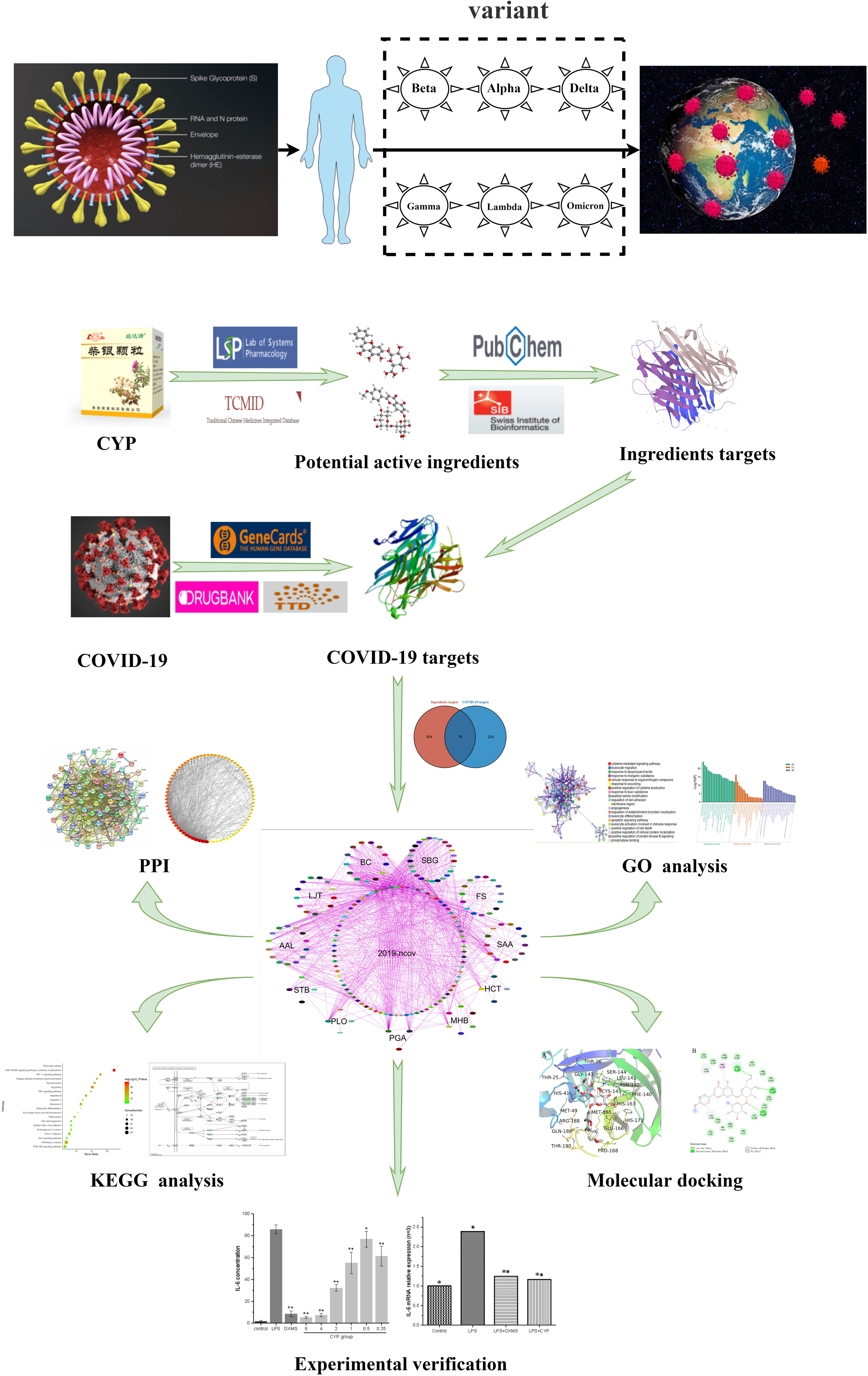
Research and analysis flow chart.
Materials and Methods
Analysis of the Active Components of CYPs
The TCMSP database (http://tcmspw.com/index.php) was used to collect the active components of CYPs. 9 The collected components were screened based on the criteria OB ≥30% and DL ≥0.18 to obtain potential active components that could be easily absorbed into the blood in the human body.
Potential Target Mining and Multinetwork Construction
The PubChem database (https://pubchem.ncbi.nlm.nih.gov/) and SwissTargetPrediction database (http://www.swisstargetprediction.ch/) were used to predict potential active components. “COVID-19,” “2019-nCoV,” and “novel coronavirus pneumonia” were used as keywords, and the GeneCards database (https://www.genecards.org/), DrugBank database (https://go.drugbank.com/), and TTD database (http://db.idrblab.net/ttd/) were used to retrieve disease targets associated with COVID-19. The UniProt database was used to standardize the targets, and the 2D and 3D structures of the targets were downloaded.
The potential targets in the treatment of COVID-19 by CYPs were obtained by repeated screening of disease targets and predicted targets of the potential active components. The potential targets and their related components and TCMs were imported into Cytoscape 3.7.1 software, and a multivariate network was constructed to analyze the relationships among the sets.
Protein–Protein Interaction Analysis and Acquisition of key Potential Targets
The STRING database (https://string-db.org/) 10 contains a large number of protein–protein interaction (PPI) relationships, and the potential targets were imported into the STRING database to obtain comprehensive PPI data. To further analyze the interaction relationship between targets and screen the key potential targets, the PPI data obtained were imported into Cytoscape 3.7.1 software to build a network, and the related topological parameters of the network were analyzed.
Gene Ontology Analysis and Kyoto Encyclopedia of Genes and Genomes Pathway Analysis
Gene ontology (GO) analysis and kyoto encyclopedia of genes and genomes (KEGG) pathway analysis of potential targets were performed with the Metascape website (http://metascape.org) 11 and KEGG database (https://www.kegg.jp/), 12 and the results were processed and visualized.
Molecular Docking
Elucidation of the mechanism of SARS-CoV-2 invasion into the human body revealed the key pathogenic targets of SARS-CoV-2. As a functional protein that mediates the hydrolysis of replicase peptides 1a and 1ab, 3-chymotrypsin-like protease (3CLpro) is involved in the replication and proliferation of SARS-CoV-2 and is one of the key proteases needed for viral replication.13,14 The S protein of SARS-CoV-2 invades the human body by binding to the angiotensin converting enzyme II (ACE2) receptor, and excessive ACE2 consumption causes overactivation of the renin-angiotensin system and eventually leads to the occurrence of disease.15,16 Therefore, 3CLpro and ACE2 can be used as key target proteins to find drugs for COVID-19. 17 In addition, RNA-dependent RNA polymerase (RdRp) and papain-like protease (PLP) are two key target proteins for anti-COVID-19 drug screening.18,19
Data for 3CLpro (PDB ID: 6LU7), ACE2 (PDB ID: 1R42), RdRp (PDB ID: 7D1M), and PLP (PDB ID: 4OVZ) were downloaded from the RSCB database (https://www.rcsb.org/). The 3D structures of four important target proteins were extracted using PyMOL 1.7.2.1 software. Water molecules, redundant ligands and residues were removed, and the structures were optimized using AutoDock Tools 1.5.6 software and then stored in PDBQT format. The 2D structure of the ligand compound was downloaded from the TCMSP database, and the energy minimization of the ligand compound was performed using ChemOffice 2016 software. Hydrogenation and other optimization processes were conducted using AutoDock Tools 1.5.6 software. All the flexible bonds of the ligand compound were rotatable by default. The grid box for molecular docking was set to all surrounding residues centered on the primary ligand of the target protein. AutoDock Vina 1.1.2 and Python scripts were used for the molecular docking simulations.
As protease inhibitors, lopinavir and ritonavir can significantly reduce the viral load of β-coronavirus and exert a clear therapeutic effect on COVID-19. 20 Therefore, the binding energies of lopinavir and ritonavir with the four target proteins were selected as positive references to sort and screen the optimal binding energies of the obtained ligand compounds. The Molecular Mechanics-Poisson Boltzman Surface Area (MM/PBSA) combined free energy prediction was obtained using the open-source software Gromacs v2019.
Experimental Verification
Instruments
A carbon dioxide incubator (Thermo 371), an inverted microscope (Olympus CKX41), an enzyme plate instrument (Bio-Rad Imark), an Agilent 1290 UPLC, and an Agilent 6230 TOF LC/MS four-stage rod time-of-flight mass spectrometer (Agilent, USA) were used.
Reagents
A549 cells were kindly provided by the Stem Cell Bank of the Chinese Academy of Sciences (Serial, SCSP-503). PBS buffer (SolarBio, P1020-500), a programmed temperature box (Ecosio), Ham's F-12 K medium (ProCell, PM150910), Australian fetal bovine serum (Ecosio, FND500), penicillin streptomycin solution (Sciencell, 0503), trypsin-EDTA digestive solution (SolarBio, T1320), lipopolysaccharide (LPS, SolarBio, L8880), a Human IL-6 ELISA Kit (EH004-96), a Human TNF-α ELISA Kit (EH009-96), CCK8, CYPs, linarin, eicosadienoic acid (Sigma, E3127), baicalin daidzein-4,7-diglucoside (B52554), robinin (B50946), fortunellin (B27528), dexamethasone (SolarBio, D8040), TRIzol reagent (Life Technologies, 15596-026), DEPC water (SolarBio, R1600-500), nucleic acid precipitant (Biyuntian, B50946) SP SD 0812), reverse transcription reagent (TAKARA, RR047A), and 2*HotStart SYBR qPCR Master Mix (Exaxel, MB000-3073) were used in this study.
Detection of Cell Viability by the CCK-8 Assay
Different concentrations were used in the CYPs p and monomer drug groups. The CK group was established, and the testing of each group was repeated three times. CCK-8 reagent (10 μL) was added to each well at 24 h, 48 h, and 72 h. After incubation at 37 °C for 2-3 h, the 50% cytotoxicity concentration (IC50) was calculated based on the absorbance at 450 nm.
Measurement of the Expression of Inflammatory Cytokines by ELISA
A549 cells in the logarithmic growth phase were inoculated into 96-well plates at a density of 1 × 104 cells per well. When the cells reached 50% confluence, the A549 cells were damaged for 3 h by replacing the medium with fresh medium containing 10 µg/mL LPS. The A549 cell injury model was prepared, and the CK group was established. Three hours later, the old medium was removed, new medium was added, and drugs were added to the corresponding groups. The expression of inflammatory factors was detected using ELISA kits according to the instructions.
Detection of the Effect of CYPs on Gene Expression
The CK group, LPS group (LPS concentration of 10 μg/mL), positive dexamethasone (DXMS) group and CYPs group were established. On the second day, the old medium in all the groups with the exception of the CK group was removed, and medium containing 10 μg/mL LPS was added to damage the A549 cells for 3 h and thus prepare the A549 cell injury model. After 3 h, the medium was changed, and drugs were added (the same volume of DMSO was added to the CK and LPS groups). RNA was extracted by the TRIzol method, and cDNA was obtained via the reverse transcription of mRNA using a reverse transcription kit. The expression of key target genes was detected by real-time quantitative polymerase chain reaction (qPCR) and calculated using the 2−ΔΔCt method. 21 The primer sequences are shown in Table 1. The 25-µL PCR mixture consisted of 12.5 µL of 2*HotStart SYBR qPCR Master Mix, 1 µL of each of the forward and reverse primers, 1 µL of cDNA, and 9.5 µL of DEPC water. The cycling conditions included initial denaturation at 95 °C for 10 min followed by 40 cycles of 95 °C for 20 s, *Tm for 30 s, and 72 °C for 20 s.
Primer Sequences in Polymerase Chain Reaction.

Six Target Components Were Identified
To avoid the occurrence of false positives, CYPs were investigated by UPLC-TOF-LC/MS liquid mass spectrometry, and the main active components were qualitatively analyzed.
Statistical Analysis
The experiments were performed in triplicate. The data obtained in the study are presented as the means ± standard deviations (SDs), and statistical analysis of variance was performed with SPSS software (SPSS 18.0 for Windows; SPSS Inc, Chicago, IL, USA). In the one-way analysis of variance among multiple groups, the differences were considered statistically significant at the P < 0.05 level. The post hoc test was applied in the comparison among test groups.
Results
Analysis of the Active Components of CYPs
In total, 141 active components of CYPs were collected and screened from the TCMSP database (some components appeared simultaneously in multiple Chinese medicines). These active components included 17 BC, 23 LJT, 23 FS, 7 HCT, 36 SBG, 4 PLO, 11 STB, 22 AAL, 10 MHB, 19 SAA, and 7 PGA. The active components common to many TCMs are summarized in Table 2.
Common Active Ingredients Information of Chaiyin particles’ Various Chinese Medicines.

Potential Target Mining and Multinetwork Construction
Overall, 302 COVID-19-related disease targets (as of November 03, 2020) were retrieved from the GeneCards database, DrugBank database, and TDD database, and 902 predicted targets of potential active components of CYPs were obtained. After repeated screening of the two types of targets, 78 potential targets were obtained, as shown in Figure 2.

Venn diagram of components prediction targets and disease targets.
By taking the active components directly related to potential targets and TCMs as nodes, a multicomponent network was constructed, as shown in Figure 3. At least four active components of each TCM could act on the target of COVID-19, and the number of active components was roughly consistent with the primary and secondary components of CYPs. A total of 114 components are included in the figure; these components can act directly on COVID-19 disease targets and are referred to as key potential components.

PPI Analysis and Acquisition of key Potential Targets
After analysis of 78 potential targets in the STRING database, a comprehensive PPI relationship network was obtained, as shown in Figure 4. The lines between nodes in the figure reflect the interaction relationships between targets, and the lines of different colors reflect different interactions. Among these interactions, we focus on the lines shown in light blue (curated in databases) and pink (experimentally detected) because the relationships between targets represented by these lines are more reliable.

Protein–protein interaction (PPI) comprehensive relationship network of potential targets.
To further clarify the interactions between targets in the PPI network, PPI data with scores ≥0.5 were screened and imported into Cytoscape 3.7.1 software. The constructed potential target network is shown in Figure 5. The figure contains a total of 77 nodes and 938 edges. The interactions between nodes reflect the correlations and mutual impacts among potential targets (not reflected in TK1). The degree of a node represents the number of nodes that directly interact with the node in the network. In general, nodes with a higher degree value perform more functions in the network. In the figure, the color and size of a node reflect the degree value of the potential target. Larger degree values are redder in color and larger in the graph; that is, the degree value of the potential target decreases clockwise from MAPK3.

Topological parameter analysis network of potential targets.
The betweenness centrality reflects the importance of a node in information transmission through the network. A greater number of nodes indicates that the node has higher importance in the network. The closeness (closeness centrality) reflects the degree of closeness between a node and other nodes in the network, and a greater closeness centrality is indicated by closer proximity to other nodes. We analyzed the topological parameters of the nodes involved in the network and selected 11 key potential targets according to the screening principles of intermediate number, proximity centrality, and degree greater than the respective median, as shown in Table 3.
Key Potential Target Information.

GO Analysis
A GO cluster analysis was performed using 78 potential targets, which mainly focused on biological process (BP), molecular function (MF) and cell composition (CC). The results are shown in Figure 6. The GO enrichment analysis results were screened according to the P value and then visualized. As shown in Figure 7, BP is mainly involved in leukocyte migration and the response to wounding. CC mainly involves the membrane region and side of the membrane, and MF mainly involves phosphatase binding and phosphotransferase activity, in which the alcohol group serves as the acceptor.
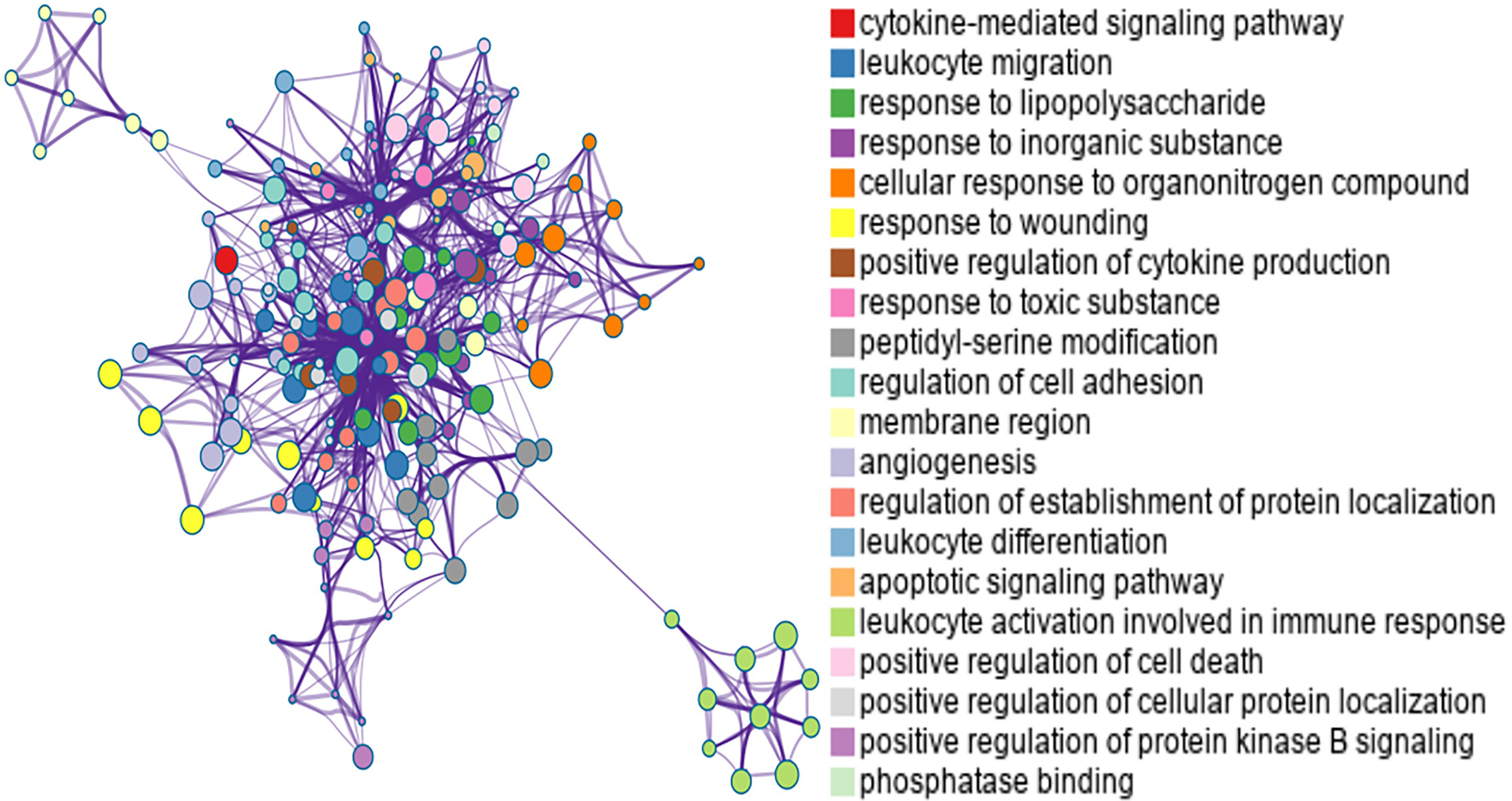
Gene ontology (GO) cluster analysis diagram.

Gene ontology (GO) enrichment analysis diagram.
KEGG Pathway Analysis
In total, 113 pathways were obtained from 78 potential targets. Using the gene number, gene ratio and P value as the investigation parameters, the top 20 pathways were selected for visual analysis, and the results are shown in Figure 8. Among these pathways, the AGE-RAGE signaling pathway, TNF signaling pathway, PI3K-Akt signaling pathway and HIF-1 signaling pathway are related to inflammatory hypoxia. Pathways associated with virus infection include the hepatitis B, hepatitis C, influenza A, and HTLV-1 infection signaling pathways, viral carcinogenesis, and Epstein–Barr virus infection. The pathways involved in immune system differentiation include apoptosis and osteoclast differentiation.

Kyoto encyclopedia of genes and genomes (KEGG) pathway enrichment analysis bubble chart.
We built the relationships between key pathways and the key potential target network and further clarified the pathways and relationships between the targets. Each key target in the key pathways covering key targets is involved in several key pathways, as shown in Figure 9. Thus, to further prove the role of CYPs in these pathways, we can prove the correlation between CYPs and key targets.

“Critical path-key potential target” relationship network.
Based on the combination of gene number, gene ratio, and P value, the AGE-RAGE signaling pathway was selected as the key pathway, and a labeled chart of potential targets in the AGE-RAGE signaling pathway was generated, as shown in Figure 10. The green nodes in the figure represent potential targets in this pathway. As shown in the figure, CYPs have a great influence on this pathway, which involves 17 potential targets, and this finding suggests that CYPs may mainly play a role in the treatment of COVID-19 through this pathway.

Annotated map of potential targets on the AGE-RAGE signaling pathway.
Molecular Docking
The 114 potential active components of CYPs were subjected to molecular docking with the four target proteins. A binding energy lower than 0 indicated that the ligand compound could spontaneously bind to the receptor protein; a lower binding energy indicated a greater possibility of its action. The results of the molecular docking simulations showed that the binding energies of the 114 potential active components with 3CLpro were all lower than 0; in addition, four of these energies were better than that of lopinavir, and 32 of these energies were better than that of ritonavir. The binding energies with ACE2 were all lower than 0 with the exception of that of MOL003062; in addition, 46 components were superior to lopinavir, and 86 components were superior to ritonavir. The binding energies with RdRp were all lower than 0 with the exception of MOL003062. Ten components were superior to lopinavir, and 51 components were superior to ritonavir. The binding energies with PLP were all lower than 0 with the exception of MOL003062. Thirty-four components were superior to lopinavir, and 49 components were superior to ritonavir.
Fortunellin and 3CLpro exhibited the best binding energy, with a value of −8.8, whereas for ACE2 binding, baicalin and ACE2 presented the best binding energy, with a value of −6.9. The binding energy between bicuculline and RdRp was −9.4. Liquiritin had the best docking and binding energy with PLP, with a value of −10.3. The docking results for the above four components and their corresponding target proteins are shown in Figure 11. The MM/PBSA binding free energy is shown in Table 4. The binding site of the potential active component and the amino acid residues of the target protein mainly undergo hydrophobic binding via pi-alkyl, pi-pi-T-shaped, pi-anion noncovalent, pi-pi-stacked, pi-donor hydrogen bond and hydrogen bond interactions, which increase the binding between the active component and the target protein. Taking the combination of fortunellin and 3CLpro as an example, fortunellin formed conventional hydrogen bonds with the amino acid residues GLU A:166, SER A:144 and CYS A:145; formed carbon hydrogen bonds with PRO A:168, HIS A:41 and GLN A:189; and formed pi-alkyl with the amino acid residue MET A:165.

3clpro and fortunellin molecular docking 2D and 3D images (A, B), ACE2 and baicalin molecular docking 2D and 3D images (C, D), RdRp and bicuculline molecular docking 2D and 3D images (E, F), and PLP and liquiritin molecules docking 2D and 3D images (G, H).
MM/PBSA Binding Free Energy and of Four Components.
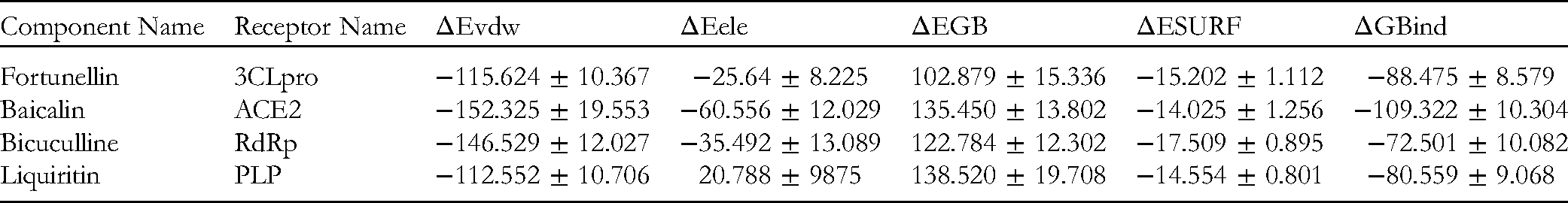
Experimental Verification
COVID-19 patients usually experience fever, fatigue and dry cough in the initial stage of infection; in severe cases, acute lung injury (ALI), acute respiratory distress syndrome and respiratory failure may occur, and most patients die due to the above three complications.22,23 Therefore, ALI, a complication of COVID-19, was selected as the pathological model for our experimental verification. The progression of COVID-19 to ALI is mainly related to a cytokine storm caused by an excessive immune response involving interleukin and tumor necrosis factor-α (TNF-α) production, and various studies have shown that a reduced level of interleukin-6 (IL-6) is associated with a reduced inflammatory response in ALI.24,25 Through a network pharmacological analysis of CYPs, we found that the key targets of TNF and IL-6, which ranked among the top targets, were closely related to the inflammatory cytokines TNF-α and IL-6; thus, the components that directly acted on these two key targets were selected as the target components for experimental verification. The data and structures of the target components are shown in Table 5 and Figure 12. In addition, DXMS is recognized as a drug with significant effects and was thus used as a control.26,27

Target components structure.
Target Ingredients Information.

Six Target Components Were Determined
Figure 13 shows the total ion flow diagram of CYPs in the negative ion mode. We found that all six target components used for experimental verification could be located on the map. Among them, the baicalin (third peak) content was relatively high, which was related to the high content of baicalin in Scutellariae Radix. In addition, this component played an important role in our network pharmacological analysis and experimental verification. Therefore, the high content of baicalin in CYPs may be one of the main reasons explaining CYPs play a role in the treatment of COVID-19.

Total ion current diagram of CYP negative ion mode (note: peak 1: linarin, peak 2: 11,14-eicosadienoic acid, peak 3: baicalin, peak 4: daidzein-4,7-diglucoside, peak 5: robinin, and peak 6: fortunellin.).
Groups
We established the following groups: CK group, CYPs group, DXMS group, LPS group, six monomer groups, namely, G1 (linarin), G2 (11,14-eicosadienoic acid), G3 (baicalin), G4 (daidzein-4,7-diglucoside), G5 (robinin), and G6 (fortunellin), and G7 (mixture of six monomeric drugs).
Cytotoxicity Test
The concentrations of the CYPs group were set to 0.5, 1, 2, 4, 8, and 16 mg/mL (filtered in advance with a 0.22-μm sterile membrane), and the concentrations of G1-G6 were set to 0.02, 0.1, 0.5, 2.5, 12.5, and 62.5 μmol/L. The results showed that different concentrations of CYPs and the six monomeric drugs had significant inhibitory effects on the proliferation of A549 cells in a dose- and time-dependent manner. After a comprehensive analysis of various experimental factors, 48 h was selected as the time of cell administration. After 48 h of treatment, the IC50 values of the CYPs group and G1-G6 groups were 12.73 mg/mL and 55.36, 58.73, 41.01, 45.90, 55.56, and 50.30 μmol/L, respectively (the data from the cytotoxicity experiment are not shown).
Expression Levels of the Inflammatory Cytokines IL-6 and TNF-α Detected by ELISA
The concentrations of the CYPs group were set to 0.5, 1, 2, 4, 8, and 16 mg/mL, and the concentrations of the G1-G6 groups and DXMS group were set to 0.02, 0.1, 0.5, 2.5, 12.5, and 62.5 μmol/L. The concentrations of group G7 were set according to the results for the G1-G6 groups; that is, the concentrations of each monomeric drug in the G7 group were the best concentrations obtained from the other groups.
The analysis of the effect of LPS on A549 cells revealed that the levels of IL-6 and TNF-α were significantly after treatments than in the control group. The administration of different concentrations of CYPs significantly decreased the levels of IL-6 and TNF-α in cells compared with those in the LPS group. To more clearly reflect the different concentrations of CYPs used to inhibit IL-6 and TNF-α production, we visualized the results as shown in Figure 14. As shown in the figure, the strongest inhibitory effect of CYPs on the two inflammatory factors was obtained with a CYPs concentration of 8 mg/mL, and the inhibitory effect of CYPs on IL-6 at this concentration was even better than that of DXMS.

Chaiyin particles (CYPs) inhibit interleukin-6 (IL-6) (A) and tumor necrosis factor-α (TNF-α) (B) (note: compared with the control group, *P < 0.05, **P < 0.01; compared with the model group, #P < 0.05, ##P < 0.01).
We simultaneously explored the inhibitory effects of monomeric drugs at different concentrations on the two inflammatory factors, but considering the brevity of this manuscript, not all the data are shown; only the optimal concentrations of CYPs and G1-G7 were selected (Figure 15). The inhibition of IL-6 and TNF-α was more significant in the CYPs group than in the monomeric drug groups. The inhibitory effect of the G7 treatment on the two inflammatory factors was worse than that of CYPs but better than that of the G1-G6 treatments (comparison of G7 with G1-G6: P = 0.022 < 0.05). Among the six monomeric drugs, G5 was superior to the other monomeric drugs in inhibiting IL-6 and TNF-α production (comparison of G7 with G1-G4 and G6: P = 0.038 < 0.05).

The optimal concentration of Chaiyin particles (CYPs) and monomer drugs inhibits interleukin-6 (IL-6) (A) and tumor necrosis factor-α (TNF-α) (B) (note: compared with the control group, *P < 0.05, **P < 0.01; compared with the model group, #P < 0.05, ##P < 0.01; compared with the control group, △P < 0.05, △△P < 0.01).
Expression of Key Target Genes of IL-6, TNF-α, VEGFA, EGFR, and MAPK1 Detected by Real-Time qPCR
Based on the results described in section 2.7.3, we selected 8 mg/mL CYPs for subsequent experiments and used DXMS as the positive control drug.28-30 The real-time qPCR results are shown in Figure 16. The mRNA expression levels of five key targets after CYPs intervention were significantly lower than those found for the LPS group (P < 0.01), and the mRNA expression levels of IL-6 and EGFR after CYP treatment were lower than those of the DXMS group, which indicated that CYPs exert a stronger inhibitory effect on TNF-α and EGFR expression than DXMS. The inhibitory effect of CYPs on TNF-α, VEGFA and MAPK1 mRNA expression was weaker than that of DXMS but still significant.

The effect of Chaiyin particles (CYPs) on the expression of five key target genes of interleukin-6 (IL-6) (A), tumor necrosis factor-α (TNF-α) (B), VEGFA (C), EGFR (D), and MAPK1 (E) (note: compared with the control group, *P < 0.05, **P < 0.01; compared with the model group, #P < 0.05, ##P < 0.01).
Discussion
With the release of the COVID-19 vaccine, control of the global COVID-19 epidemic has reached an inflection point. However, even if the vaccine is effective in reducing the death rate of those who get sick, zero infection cannot be achieved among the vaccinated population. In addition, there is a minimum age for vaccination, which prevents the whole population from becoming immunized. Therefore, other approaches in addition to vaccines are needed for the prevention, control and treatment of the epidemic. In China, more than 91.5% of infected patients have received TCM treatment, and the results show that the total effective rate of TCM treatment for COVID-19 is over 90%.31,32 In addition, TCM has a significant effect on both the prevention of COVID-19 and the treatment of patients with mild, moderate and severe disease. Therefore, in the context of SARS-CoV-2 mutations and epidemic rebound, TCM must be taken seriously and used rationally.
TCM is an excellent cultural treasure in China that has been passed down for thousands of years and continuous to be developed and innovated. TCM has a unique advantage in the treatment of COVID-19. In particular, it exhibits outstanding clinical performance in terms of anti-infection and antipyretic activities, shortening the course of the disease and improving the curative effect. On the one hand, TCM helps the body rapidly clear the pathogenic virus, improves the immunosuppressive state, and regulates the body's environment.33-35 On the other hand, TCM regulates the balance of the immune system, prevents the occurrence of cytokine storms and the potential damage to normal cells of the immune system, plays a two-way regulatory role, balances the immune system of the human body, and reduces mortality.36,37 Based on an analysis of the main active components of CYPs, we used network pharmacology and molecular docking technology to search for targets, identify the pathways of the targets, and explain the mechanisms based on the pathways. The obtained theoretical results were verified through molecular docking and cellular experiments, and the potential active components and related mechanisms of CYPs in the treatment of COVID-19 were explored in depth. By constructing a network of “traditional Chinese medicine, key potential components and potential targets,” 114 key potential active components directly acting on COVID-19 targets were obtained, and these may be the key components involved in the efficacy of CYPs. Luteolin and quercetin were found in 6 types of TCM compounds and acted on 36 targets, which fully reflects the multitarget characteristics of TCMs. Studies have shown that luteolin can inhibit the activation of the NF-κB signaling pathway, the production of the inflammatory factors IL-6 and TNF-α, and the activation of Akt; increase vascular permeability; induce the induction of nitric oxide synthase and cyclooxygenase-2; and exert a clear protective effect on LPS-induced ALI in mice.38-40 Quercetin may downregulate the expression of NF-kB/p65 and ICAM-1 by affecting antioxidant proteins in the Nrf-2/ARE signaling pathway and ultimately reduce the release of proinflammatory mediators, thus achieving a protective effect on LPS-induced ALI in rats. 41 Quercetin also improves gas exchange function, inhibits the release of inflammatory factors, and reduces endotoxin-induced ALI by inhibiting the activation of the JAK2/STAT3 signaling pathway. 42
After PPI analysis and topological parameter screening of 78 potential targets, 11 key potential targets were obtained. As an important cytokine in inflammation and immunity, IL-6 is involved in the TNF signaling pathway. Some researchers have found that IL-6 deletion in rats can improve ALI caused by intestinal ischemia/reperfusion. 43 TNF-α is an important proinflammatory factor that is released early during the inflammatory response of ALI and can promote the release of other cytokines. 44 Studies have shown that various inflammatory factors are involved in the occurrence and development of severe pneumonia, and among these, IL-6 and TNF-α are two major inflammatory factors. 45 An increase in the plasma IL-6 and TNF-α levels can be an important indicator of pulmonary infection, and the expression levels of these factors are closely related to the severity of the disease. These two inflammatory factors can also lead to hypotension and multiple organ failure; for example, higher plasma IL-6 and TNF-α levels are associated with more severe pulmonary infection in patients with severe pneumonia.46,47 In addition, anti-cytokine (IL-6 and IL-1) therapy can effectively inhibit excessive inflammatory responses in COVID-19 patients during treatment. 48 Therefore, if the expression levels of the inflammatory cytokines IL-6 and TNF-α can be inhibited, the inflammatory response of patients can be suppressed to a certain extent. Among the results obtained in our analysis, TNF and IL6 were the first and second key potential targets, respectively, suggesting that CYPs may prevent the occurrence and progression of COVID-19 and its complications by inhibiting the expression of core inflammatory factors.
The results of GO BP analysis revealed that the cytokine-mediated signaling pathway (the signaling pathway mediated by cytokines) exhibited the greatest enrichment. Intracellular signal transmission affects the biological effects of external stimuli on cells, including the reaction of cells to inflammatory factors such as IL-6, which can affect the occurrence and development of diseases. 40 Phosphatase binding (phosphatase binding) showed the greatest enrichment in the GO MF analysis. Phosphatases are key components of many signal transduction pathways and can activate signaling pathways to change protein functions by removing phosphate groups from specific proteins. 49 The GO CCs were mainly enriched in the membrane region, and receptors on the membrane can play a role in signal transduction. When this process is blocked, abnormal growth, differentiation, metabolism and biological characteristics occur, leading to various diseases. 50
The AGE-RAGE signaling pathway (AGE-RAGE) and HIF-1 signaling pathway obtained from the KEGG analysis are closely related to the occurrence and development of COVID-19. Studies have shown that the mechanisms of the AGE-RAGE pathway mainly include activation of nuclear factor-kappaB (NF-κB), stimulation of vascular endothelial growth factor (VEGF) and transforming growth factor (TGF-β1) production, induction and enhancement of the expression of monocyte chemotactic protein (MCP-1), and activation of nicotinamide adenine dinucleotide phosphate oxidase to induce oxidative stress. 51 NF-κB is an important signaling pathway in inflammation, and the levels of inflammatory factors play an important role in its regulation. The inhibition of NF-κB can block the expression of inflammatory cytokines in the lung, significantly reduce inflammation in the lungs, and improve the survival rate. ALI is induced by LPS in mice.52,53 In addition, this pathway is also involved in LPS stimulation of alveolar type II epithelial cells and the expression of blood coagulation and fibrinolytic factors to avoid abnormal blood coagulation and fibrinolysis in the alveoli of patients. 54 Studies have shown that the serum VEGF content is correlated with the severity of lung injury. 55 Therefore, once the AGE-RAGE signaling pathway is activated, it promotes NF-κB activation to produce a large number of inflammatory factors, such as VEGF, thus aggravating the inflammatory response. If this process is inhibited, inflammation can be indirectly suppressed; that is, such inhibition exerts a clear therapeutic effect on COVID-19. Studies have shown that HIF-1α inhibition can inhibit the expression of inflammatory cytokines, including interleukins and TNF-α, which suggests that HIF-1α plays an important role in the secretion of inflammatory cytokines. 56 In the normal or physiological state, RAGE is expressed at low levels in tissues and cells, and when the body is in a state of inflammation and trauma, such as during exposure to stress, RAGE is expressed at abnormally high levels. Furthermore, activation from short pulses into a continuous cascade activates downstream transcription factors, such as NF-κB, and abnormally high expression triggers a waterfall inflammatory reaction, causing IL-6, TNF-α and other inflammatory cytokines to be released at high levels, which eventually leads to tissue cell dysfunction and structural damage caused by a variety of pathological changes. 57 Studies have noted that reducing NF-κB activation reduces cytokine levels. Thus, this strategy could be helpful in combating the cytokine storm associated with COVID-19. 58
Through molecular docking, we identified a variety of components that exhibit good binding with the key target proteins of COVID-19, and the forms of binding observed for these four proteins could also be intuitively observed through 2D and 3D diagrams. Among these components, fortunellin exhibited the best binding with 3CLpro, and studies have shown that fortunellin can protect against fructose-induced inflammation and oxidative stress by activating the AMPK/Nrf2 pathway. 59 Baicalin showed the best binding with ACE2, and studies have found that baicalin can inhibit the TLR4-NF-κBp65 and JNK-ERK signaling pathways and the pulmonary inflammatory response and thereby reduces the degree of lung tissue damage in ALI mouse models. In addition, baicalin exerts clear inhibitory effects on pulmonary fibrosis and pneumonia.60-62 γ-Aminobutyric acid A receptor (GABAAR) can aggravate inflammatory response syndrome and oxidative stress in the lung, whereas bicuculline can block the GABAAR receptor, thereby reduces the expression of the inflammatory cytokines TNF-α and IL-6 and thus plays a protective role against LPS-induced ALI. 63 Transient receptor potential vanilloid receptor (TRPV1) and transient receptor potential anchor protein (TRPA1) are cation channels that play a key role in inflammatory signaling pathways; they can be expressed and exert synergistic effects and cause inflammation and cough. Liquiritin inhibits LPS-induced inflammation in bronchoalveolar lavage fluid. TRPV1 and TRPA1 increase protein expression, and TRPV1 and TRPA1 mRNA expression protects against ALI induced by LPS. 64 These components are expected to be potential anti-COVID-19 drugs. According to previous studies, rutin and quercetin contained in CYPs could be used as SARS-CoV-2 main protease (Mpro) inhibitors.65,66 In addition, many studies have investigated whether traditional medicinal plant extracts exhibit anti-SARS-CoV-2 activity.67-70
Our experiments investigated the effects of CYP and monomer drugs on the proliferation ability of A549 cells at different times and found that longer treatment times and higher drug concentrations have a positive effect on the inhibition of A549 cell proliferation. With experimental durations of 24 h and 48 h, the IC50 values of CYP in A549 cells were 21.06 mg/mL and 12.73 mg/mL, respectively. However, after 72 h of treatment, a significant difference was observed in the inhibition rate of CYP on A549 cells, with an IC50 of 3.88 mg/mL. At a concentration of 8 mg/mL, the cell inhibition rate after 72 h of treatment was 1.80 times and 1.12 times higher than that after 24 h and 48 h of treatment, respectively. In the 24 h and 48 h treatments, the cell inhibition rate was mainly affected by the drug concentration. However, the influence of time became the main factor in the inhibition of cell proliferation after 72 h of treatment, and at 72 h, all concentrations of CYP exerted inhibitory effects on A459 cells. To prevent the inhibition of cell proliferation from affecting the experimental results when measuring the expression levels of TNF-α and IL-6, we comprehensively analyzed various factors and selected 48 h as the experimental time.
IL-6 and TNF are the key potential targets and the two targets with the closest relationships with ALI and inflammatory factors. Through a cell-based experiment, we found that CYPs and six types of monomers could significantly inhibit the release of the inflammatory cytokines IL-6 and TNF-α. The efficacy of the tested compounds followed the order monomer < six monomer mixture < CYPs, which further proved that these six components of CYPs, including baicalin, were the main components involved in its efficacy. These results were consistent with the TCM theory “1 + 1 > 2,” that is, the combined use of multiple TCMs (components) has better efficacy than the use of a single TCM (component). In addition, fortunellin and baicalin were the two components that showed the best docking with the two key proteins 3CLpro and ACE2, respectively, which again suggests the importance of these two components. Through real-time qPCR and using DXMS as the positive control, we verified the influence of CYPs on the expression of five key target genes and found that CYPs exerted a better inhibitory effect on IL-6 and EGFR expression than DXMS, which indirectly indicated that CYPs have a better inhibitory effect on such targets than specific drugs.
We believe that the most basic principles for the use of TCM compounds are their nontoxicity and effectiveness. However, our group and other researchers have proven that the synergistic effects of the various herbs in CYPs make them more effective and endow them with a unique curative effect, including a clear therapeutic effect on COVID-19. In addition, this compound has been strictly approved by the State Food and Drug Administration of China and has been on the market in China for many years, which guarantees its safety. Therefore, CYPs can be used for the prevention and treatment of COVID-19 and thus play an important role in the prevention and control of the pandemic and the prevention of epidemic rebound.
Conclusion
The study showed that 114 components of CYPs could be regarded as potential active components for the treatment of COVID-19, and linarin, 11,14-eicosadienoic acid, baicalin, daidzein-4,7-diglucoside, robin, and fortunellin were the main active components through which CYPs blocked the entry of SARS-CoV-2 into cells and exerted anti-inflammatory and immunomodulatory effects. IL-6 and TNF are the key target genes through which CYPs exert anti-inflammatory effects, and the AGE-RAGE signaling pathway is the most important signaling pathway involved in the activity of CYPs. This study provides theoretical support and a pharmacological basis for the further development and utilization of CYPs for the prevention and treatment of COVID-19. The results provide important insights for future studies of TCM modernization and a reference for the use of TCMs for the prevention and treatment of new sudden acute infectious viral diseases in the future due to their characteristics and advantages.
Footnotes
Authors’ Contributions
LX and YL: study concept and design; analysis and interpretation of data; prepared figures; wrote the manuscript text. HZ and YW: acquisition of data. LZ and YZ: critical revision of the manuscript for important intellectual content; obtained funding. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The data of the cytotoxicity experiment and different concentrations efficacy experiments of monomer drugs were not shown. The datasets used during the current study are available from the corresponding author on reasonable request.
Ethics Approval and Consent to Participate
Not applicable.
Patient Consent for Publication
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Natural Science Foundation, the major scientific and technological innovation projects in Shandong Province, and National Key R&D Program of China (grant numbers: ZR2021QH238, 2019JZZY011020, and 2017YFC1701503).
