Abstract
Objective
The aim of this study was to investigate the mechanism of Qingjieyifei Miao Fang (QJYFMF) in combination with gefitinib (GE) against non-small cell lung cancer (NSCLC).
Methods
Network pharmacology was used to identify the key targets and signaling pathways of QJYFMF in the treatment of NSCLC. Both
Results
A total of 98 active components and 901 targets of QJYFMF were identified, with the majority involved in the PI3K-AKT, MAPK, and apoptosis signaling pathways. Compared with the control group, QJYFMF significantly inhibited A549 cell proliferation, promoted apoptosis, and reduced cell migration and invasion abilities.
Conclusion
QJYFMF significantly inhibited NSCLC proliferation, migration, and invasion while promoting apoptosis. The combination of GE and QJYFMF enhances the therapeutic effects of GE, potentially through the modulation of the PTEN/PI3 K/AKT signaling pathway.
Keywords
Introduction
Lung cancer is one of the most prevalent and burdensome malignancies worldwide. According to GLOBOCAN estimates, in 2022, there were 19.965 million new cases of malignant tumors and 9.737 million deaths globally, with lung cancer accounting for 12.4% of new cases (approximately 2.48 million) and 18.7% of deaths (approximately 1.817 million). 1 Non-small cell lung cancer (NSCLC) is the most common type of lung cancer, representing about 85% of all cases. 2 Over 70% of NSCLC patients are diagnosed at stages IIIB-IV, with distant metastasis, and thus are no longer candidates for surgical resection. 3 Gefitinib, an epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI), is a first-line treatment for advanced NSCLC and offers superior clinical efficacy compared to chemotherapy.3,4 However, drug resistance to gefitinib has emerged as a major challenge in clinical treatment. Not only does primary resistance affect approximately 20% of patients, but secondary resistance typically develops within 10 months in patients who initially respond, significantly limiting the clinical benefits of gefitinib. 4 As a result, finding strategies to prevent or overcome gefitinib resistance has become a key focus in the development of new therapeutic approaches.
Clinical studies have confirmed that for patients with advanced lung cancer, the application of traditional Chinese medicine combined with EGFR-TKI has fewer side effects, prolongs progression-free survival, and enhances the therapeutic effects. 5 Studies have found that when gefitinib and Fuzhengkangai (FZKA) decoction are used in the treatment of NSCLC, FZKA can prolong the progression-free survival of NSCLC patients and reduce the toxic effects of gefitinib,6,7 The combination of traditional Chinese medicine and gefitinib can improve gefitinib efficacy and reduce its toxicity, 8 but the associated mechanisms need further research.
Qingjie Yifei Miao Fang (QJYFMF) was developed by Professor Liu Ziping, a distinguished academic inheritor and mentor from the sixth batch of renowned traditional Chinese medicine experts in China, with over 60 years of experience. It was based on the clinical trial of Qingjie Yifei Tang (QJYFT). QJYFMF is composed of 12 traditional Chinese medicines, including
Network pharmacology integrates systems biology and computational technology to construct a multi-dimensional ‘component-target-pathway’ network, offering distinct advantages in the study of traditional Chinese medicine (TCM) compounds. It provides new insights into the pharmacological effects of TCM and the molecular mechanisms underlying disease treatment.17,18 In this study, potential targets and key signaling pathways of QJYFMF in the treatment of NSCLC were analyzed through network pharmacology. The effects of QJYFMF on NSCLC cell proliferation and apoptosis were investigated at the animal, cellular, and molecular levels. Additionally, the efficacy and mechanisms of combining QJYFMF with gefitinib for NSCLC treatment were explored. This research offers modern pharmacological evidence supporting the use of Miao medicine in lung cancer treatment.
Materials and Methods
Drug Preparation
The QJYFMF formula was composed of
Gefitinib (GE) suspension: One tablet of gefitinib (250 mg/tablet, purchased from Astrazeneca Pharmaceutical Co., Ltd, JiangSu, China) was ground into powder, distilled water was added and mixed well to form a suspension, and the volume was made up to 50 ml, with a final drug concentration of 5.0 mg/ml. This was stored at 4 °C for later use.
Network Pharmacology
Acquisition of Active Ingredients and Targets
A literature search and the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, https://old.tcmsp-e.com/tcmsp.php) were utilized to identify the active chemical components of each traditional Chinese medicine in QJYFMF. Candidate active ingredients were determined based on the use of two ADME (absorption, distribution, metabolism, or excretion) indicators: bioavailability (OB ≥ 30%) and drug-like properties (DL ≥ 0.18), as well as relevant literature. The CAS number of each active ingredient was obtained from the ChemSrc website (https://www.chemsrc.com/) and imported into the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) to retrieve its 2D structure. Subsequently, target prediction was conducted using the SwissTargetPrediction database (http://www.swisstargetprediction.ch/). The predicted targets for each traditional Chinese medicine were collected following the aforementioned methods, then systematically organized and summarized. In the GeneCards (https://previous.genecards.org/) database, the Online Mendelian Inheritance in Man database (OMIM, https://www.omim.org/) was applied to identify the targets related to NSCLC. Venny 2.1 (https://bioinfogp.cnb.csic.es/tools/venny/) was used to obtain the potential targets for QJYFMF treatment of NSCLC.
Construction of Compound Network
Organize the relevant targets and active ingredients collected in section 2.2.1 into a table. An “Active Compound - Target” network of the QJYFMF prescription was constructed using Cytoscape (3.9.1, https//cytoscape.org/) software.
Protein-Protein Interaction (PPI) Function Analysis and Network Construction
The intersection of QJYFMF prediction targets and NSCLC targets were determined to obtain a common target, which is a potential target of QJYFMF treatment of NSCLC. The target PPI network was obtained using the String database (https://string-db.org/). The results were imported into the CytoHubba plugin in Cytoscape software (Version 3.9.1) to obtain the key hub gene of the network as the core network for QJYFMF treatment of NSCLC.
Enrichment Analysis of the GO and KEGG Pathways
Using the DAVID database (https://david.ncifcrf.gov/summary.jsp), bioinformatics enrichment analysis on targets was performed, including GO analysis of biological processes (BP), molecular functions (MF), cellular components (CC), and KEGG pathways. Each analysis result was sorted from the smallest to the largest
Preparation of Drug-Containing serum from Rats
Thirty SPF grade male SD rats weighing 180 ± 20 g and aged 3-4 weeks were purchased from Hubei Academy of Preventive Medicine (license No.: SCKX(Hubei)2020-0018) and randomly assigned to a control or treatment group, with 15 animals in both groups. After 1 week of adaptive feeding, rats in the treatment group were poured into stomach through mouth 17.37 g/kg/d (equivalent to the dose of normal adults) of QJYFMF. The control group was given the same dose of normal saline once daily for 7 days. After the last gavage for 2 h, the rats were anesthetized with 10% pentobarbital sodium solution (0.4 ml/100 g body weight), after which abdominal aortic blood was obtained, centrifuged at 3000 ×g for 15 min, and the serum stored at −80 °C for later use.
Cell Culture and Grouping
The human A549 lung adenocarcinoma cell line (which was completely matched to A549 cells by STR genotype detection, and originated from Shanghai Zhongqiao Xinzhou Biotechnology Co., Ltd) was cultured in a growth medium of RPMI-1640 (with 10% fetal bovine serum (FBS) + 1% double antibody) at 37 °C under100% saturated humidity and 5% CO2. Change the culture medium daily, and cells grow in a logarithmic manner. Cells displaying logarithmic growth were divided into control (CG), GE, QJYFMF, and GE + QJYFMF groups, cultured at 37 °C under 5% CO2 for 24 h, and subsequently cultured with different concentrations of drug-containing serum of the QJYFMF and/or GE suspension.
A549 Cell Proliferation Inhibition Rate was Determined by the CCK-8 Method
To investigate the effects of gefitinib (GE) and QJYFMF on A549 cells, the cells were seeded in 96-well plates at a density of 3 × 10³ cells per well. In the GE group, GE was administered at concentrations of 10, 20, 40, 60, 80, and 100 μM. In the QJYFMF group, drug-containing serum was applied at concentrations of 0%, 2.5%, 5%, 10%, 15%, 20%, and 25%. Three compound pores were set for each drug concentration, and after incubation with 100 μL of the corresponding drug concentration in each well for 24, 48, or 72 h, the cells were placed in 10 μL CCK-8 solution (MCE, HY-K0301, China) and incubated at 37 °C for approximately 2 h. The optical density value of each hole was determined at 450 nm using an enzyme-labeled instrument.
AnnexinV-FITC/PI Flow Cytometry was Performed to Detect the A549 Cell Apoptosis Rate in Each Group
The cell culture medium was aspirated from each six-well plate into a centrifuge tube, and the adherent cells were gently washed twice with phosphate-buffered saline (PBS) solution, digested with trypsin, transferred to a centrifuge tube to collect cell culture medium, mixed slightly, transferred to a new centrifuge tube, and centrifuged 1200 × g for 5 min. The supernatant was removed and the cells resuspend with PBS, rinsed twice with PBS, and centrifuged at 1200 × g for 5 min. Apoptosis was detected using the AnnexinV-APC/7-AAD apoptosis detection kit (Keygen Biotech, KGA108, China), and on-machine analysis was performed by flow cytometry (Beckman Coulter, cytoFLEX, USA).
Cell Migration and Invasion
Cell migration was determined using stromate-free inserts (Corning, 3422, USA) and cell invasion was determined using stromate-containing inserts (BD Biosciences, 356234, USA). Treatment cells (1 × 104/ well) from 100 μL non-FBS medium were inoculated into the insert and the lower compartment was filled with 10% FBS-enhanced medium. After 24 h of reaction, the cells at the top of the compartment were wiped with a cotton swab, whereas the cells in the lower compartment were fixed in 4% paraformaldehyde (Leagene, DF 0135, China) for 15 min and stained with 0.1% crystal purple (Qiangshun, 548-62-9, China) for 30 min. Finally, optical microscopy (Motic, AE 2000, Germany) was performed to obtain images of the bottom side of the membrane. The number of migrating and invading cells was quantified using the ImageJ 1.52a software (National Institutes of Health). Cells at a concentration of 5 × 105/well were inoculated into six-well plates overnight at 37 °C under 5% CO2. The following day, a gap was created vertically with a yellow pipette tip. After culture at 37 °C under 5% CO2 for 48 h, the results were observed. Images were obtained in an optical microscope at 0 and 48 h.
Animal Grouping and Model Establishment
A total of 80 BALB/c-Nu naked female mice that were healthy with SPF grade and weighing 18–22 g were purchased from Jiangsu Jicui Yaokang Biotechnology Co., Ltd (Animal certificate No.: SCXK (Su) 2020-008) and maintained in an SPF grade animal room of the Clinical Research Center of Affiliated Hospital of Guizhou Medical University. All animal experiments were approved by the Experimental Animal Ethics Committee of Guizhou University of Traditional Chinese Medicine (approval number: 20230182). A549 cells (concentration: 1 × 107/mL, dosage: 200 μL/mouse) were injected subcutaneously under the armpit of the right forelimb to prepare a mouse model of lung cancer transplantation. Excluding dead and unsuccessful nude mice, only nude mice with a subcutaneous tumor diameter (long diameter) of 3–4 mm were selected and assigned to the control (CG), gefitinib (GE), Qingjieyifei decoction (QJYFMF), and Qingjieyifei decoction + gefitinib (QJYFMF + GE) groups with ten mice per group by the random number table method. The CG group was given normal saline by intragastric administration (0.2 mL/ animal), the GE group received gefitinib suspension by intragastric administration (32.5 mg/kg/d), the QJYFMF group was given Qingjieyifi decoction by intragastric administration (25.09 g/kg/d), and the QJYFMF + GE group received gefitinib suspension and Qingjieyifi decoction by intragastric administration at the same time. After 3 weeks of intragastric administration, the mice were euthanized by cervical vertebrae dislocation, and the subcutaneous tumor was immediately removed and stored at −80 °C for later use.
Volume Changes of Transplanted Tumor Bodies in Each Group
During the experiment, tumor volumes from all groups of mice were measured and calculated every 3 days until day 28. The tumor volume (V) was calculated using the following formula: V = 0.5 × W∼2 × L (W: tumor width; L: tumor length).
qRT-PCR
RNA was obtained from tissues using Trizol reagent (Aidlab, Lot:252250AX, Beijing), and was reverse-transcribed into cDNA (reaction conditions :25 °C for 5 min, 50 °C for 15 min, 85 °C for 5 min, 4 °C for 10 min). Real-time fluorescence quantitative PCR detection was subsequently performed (reaction conditions: 2 min at 50 °C, 10 min at 95 °C; 95 °C for 30 s, 60 °C for 30 s, 40 cycles), and the relative mRNA expression levels of
Western Blot
A small number of tumor tissue blocks and cells were placed in cracking solution containing PMSF (Aladin, P105539, Hangzhou, China) and homogenized in a manual homogenizer. After cracking for 30 min, the cracking solution was centrifuged at 12000 × g at 4 °C for 5 min and the supernatant was transferred to a centrifuge tube for storage at −20 °C. The total protein was obtained using RIPA buffer containing PMSF (Biyuntian, P0013B) and quantified using a BCA kit (Biyuntian, P0010, Nanjing). After denaturation and electrophoresis, the proteins were transferred to a nitrate cellulose membrane. After closure, the cellulose nitrate membrane was treated at 4 °C with the following primary antibodies: GAPDH (1:1000, Hangzhou Xianzhi Biological Co., Ltd, AB-P-R 001), PTEN (1:1000, Abcam, Ab32199), PI3 K (1:1000, CST, 4292), p-PI3 K (1:1000, CST, 4228), AKT (1:1000, Affinity, AF6261), p-AKT (1:1000, Affinity, AF0016), p-BAD (1:1000, Affinity, AF7424), BAD (1: 1000, Affinity, AF6471), and caspase-9 (1:1000, Affinity, AF6348) overnight. On the second day, the bound antibodies were treated at 37 °C for 2 h with TBST-diluted HRP-labeled sheep anti-rabbit secondary antibody (1:50,000 dilution, BA1054, Wuhan BDE Bioengineering Co., Ltd). The working liquid was added to a PVDF film, and when the fluorescence band was obvious, the film was covered with cling film and an x-ray film was pressed on it, which was subsequently placed in the developer (Tianjin Hanzhong Photography Materials Factory) for development and fixing. The film was rinsed, dried, scanned, and the gray value of the film was analyzed using BandScan software (Invitrogen).
Statistical Analysis
All the graphs in this paper were generated using software GraphPad Prism 9, Excel 2016, or Origin 2021. Data were analyzed using SPSS 27.0 software (IBM) and presented as mean ± standard deviation from three independent replicates. One-way analysis of variance (ANOVA) was performed to compare the mean values among groups. If a significant difference was detected, the least significant difference (LSD) test was conducted for pairwise comparisons. Statistical significance was determined based on the
Results
Network Pharmacological Analysis of QJYFMF in the Treatment of NSCLC
Through TCMSP retrieval, 98 active ingredients of the QJYFMF formula were obtained and 901 targets were identified from the OB and DL values. The targets of QJYFMF were crossed with the NSCLC targets downloaded from the Genecards and OMIM databases (the first 1500). Using the Venn diagram, a total of 312 common targets were obtained (Figure S1), and an “Active Compound - Target” network was constructed (Figure 1A), Protein interaction analysis was performed (Figure S2). CytoHubb plug-in in
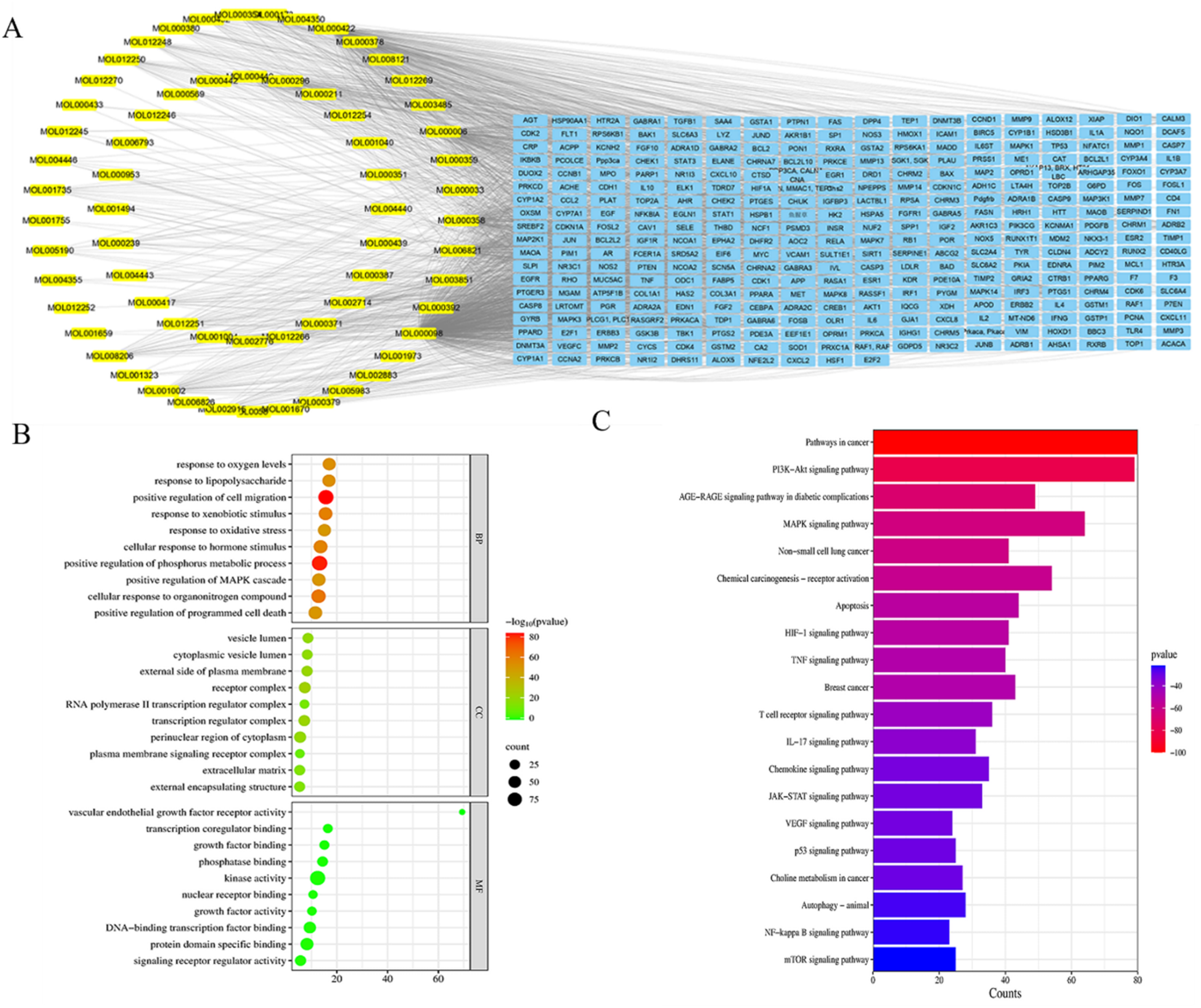
Network Pharmacology Results of “QJYFMF-NNSCLC”. A: Main Active Compounds-target Network, B: GO Functional Analysis, C: KEGG Enrichment Analysis.
GO enrichment analysis included 2941 BP, 141 CC, and 308 MF (Figure 1B). The treatment of NSCLC with QJYFMF mainly involves biological processes such as the response to oxidative stress and exogenous biological stimuli. Enrichment analysis of the KEGG pathway showed that a total of 211 signaling pathways were enriched, including the PI3K-AKT and MAPK signaling pathways and apoptosis, etc (Figure 1C, Table S2).
A549 Cell Proliferation Rate Detection by the CCK-8 Assay
Compared with the initial concentration, different times and concentrations of GE and QJYFMF drug-containing serum had significant inhibitory effects on A549 cells (

CCK-8 Detection of the Proliferation Rate of A549 Cells. (* vs CG Group, *
A549 Cell Apoptosis Rate Detection by Flow Cytometry
The apoptosis rate of each group was determined by flow cytometry, as shown in Figure 3. After drug intervention, the apoptosis rates of the GE and QJYFMF groups were significantly increased compared with the CG group (
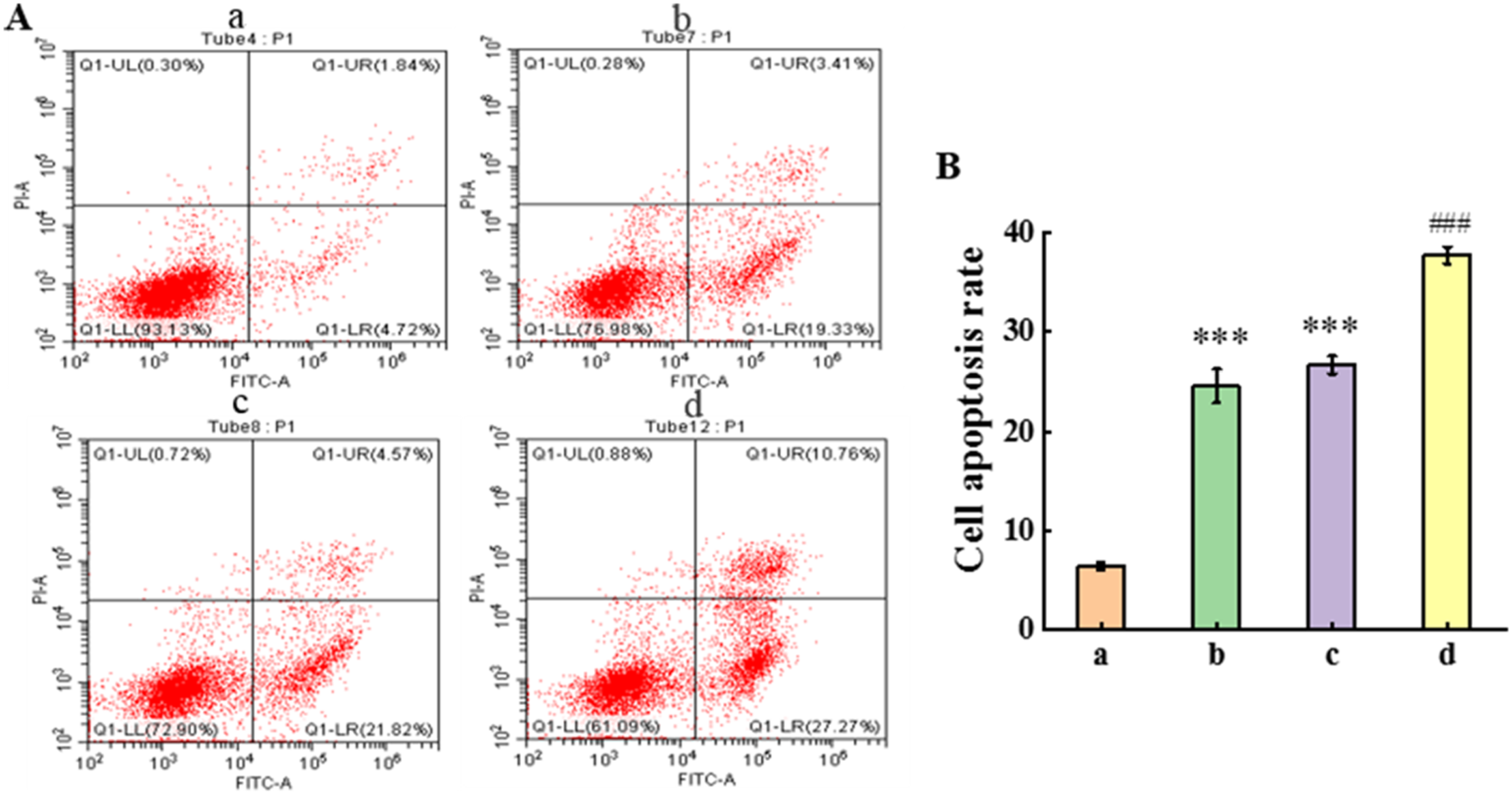
Detection of Apoptosis Rate of A549 Cells Using Flow Cytometry. (a: CG, b: GE, c: QJYFMF, d: GE + QJYFMF Group. The Same as Figure 4-7).
A549 Cell Migration and Invasion
A549 cell migration and invasion are presented in Figure 4. Wound healing results showed that cells migrated significantly after 24 h compared with 0 h, and the migration ability of the GE and QJYFMF groups was significantly diminished compared with the CG group (

Representative Microscopic Images (200×) of Wound Healing (A) and Transwell Assay (B) in A594 cells, C and D Represent their Respective Quantitative Results. (a: CG, b: GE, c: QJYFMF, d: GE + QJYFMF Group).
Tumor Volume Changes
The changes in tumor volume in each group are shown in Figure 5. A decreasing trend in the tumor volume of all the drug groups was shown compared with the CG group (Figure 5, A). After 28 days of drug intervention, the tumor volume and weight in the GE and QJYFMF groups were significantly decreased compared with the CG group (

The Effect of Transplanted Tumor Volume and Body Weight in Mice. A: The Volume Changes of 28 Days in Tumors, B: Tumors, C: Tumors Volume; D: Tumors Weight (a: CG, b: GE, c: QJYFMF, d: GE + QJYFMF Group).
Western Blotting Detection of the Expression Level of Proteins Associated with the PTEN/PI3 K/AKT Signaling Pathway
The
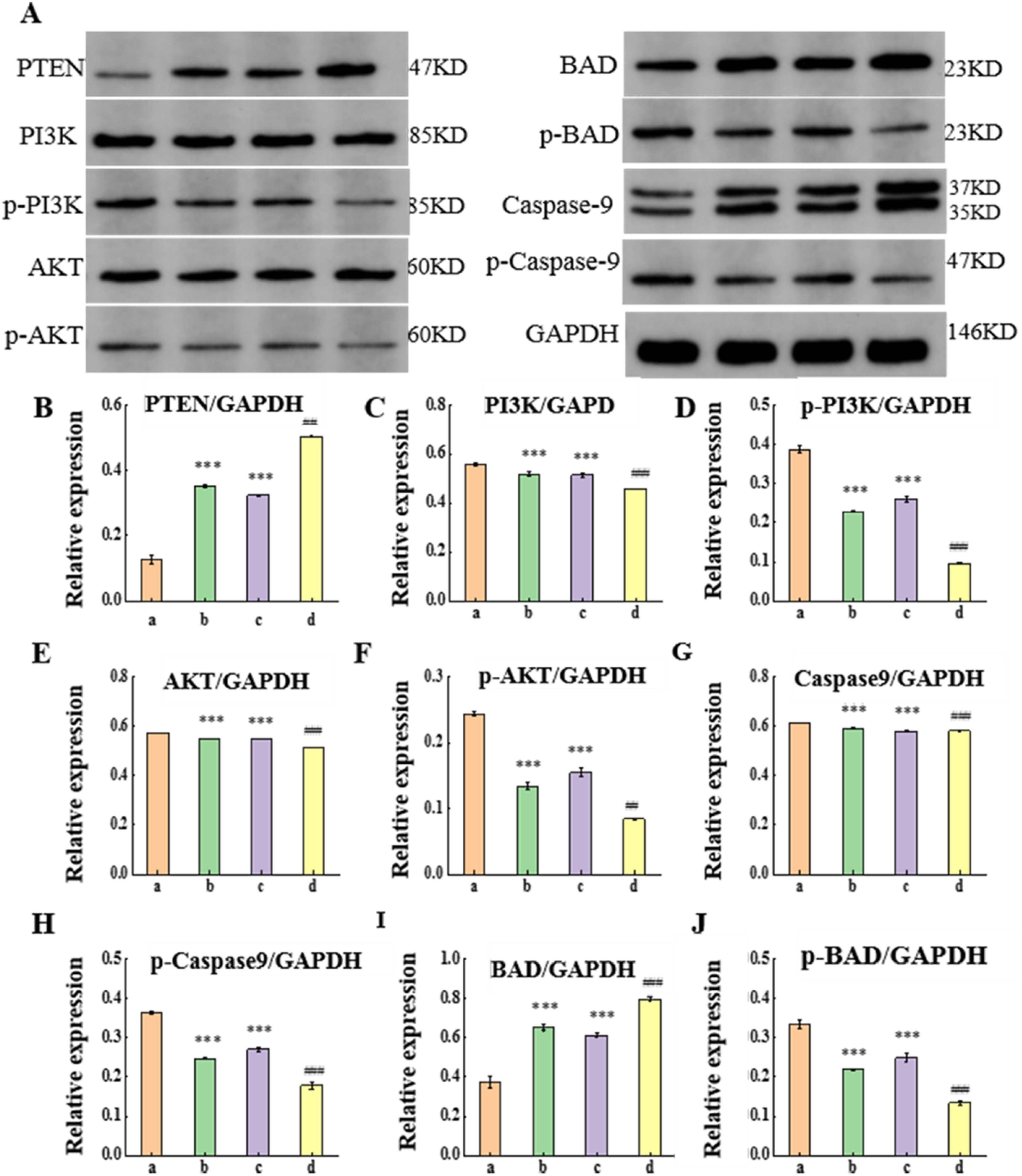
Expression Levels of PTEN/PI3 K/AKT Signaling Pathway-Related Proteins in Mouse Tissues. (a: CG, b: GE, c: QJYFMF, d: GE + QJYFMF Group).
BAD and cCaspase-9 mRNA Expression Levels
In

The mRNA Expression Levels of
Discussion
Lung cancer is primarily classified into small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC). The progression of SCLC is influenced by inflammatory responses, and it may exhibit a better response to first-line immunotherapy. 19 NSCLC is one of the most common lung cancer types and is globally the malignant tumor with the highest incidence and mortality. 20 Even individuals with a low genetic risk may face an increased likelihood of illness due to unhealthy lifestyle habits, such as smoking. 21 Tumor immunity plays a crucial role in the treatment of NSCLC, as the number and characteristics of macrophage infiltration in the tumor immune microenvironment (TEM) can influence tumor progression.22,23 However, in cases of multiple primary lung cancers, PD-L1 and CD8 levels are significantly lower, rendering patients less suitable for immunotherapy.24,25 The current popular treatment methods include targeted therapy, surgery, etc But the limitations of these treatment methods have gradually emerged over time. Therefore, it is necessary to find effective, non - toxic, and easy - to - obtain treatments.
Traditional Chinese medicine has been widely used in tumor treatment because of its accurate efficacy, strong synergistic effect, and limited adverse reactions. 26 Studies have identified and purified an active ingredient (EPPA) from the Chinese herb Echinacea and shown that it can treat tumors by modulating M1 macrophage function. 27 Clinically, acquired resistance to gefitinib (GE) remains a major challenge in the treatment of NSCLC patients. A randomized clinical trial involving 159 NSCLC patients demonstrated that those treated with Chinese medicine alone or in combination with gefitinib had significantly extended progression-free survival (PFS) and median overall survival (OS) in the combined treatment group. 28 Additionally, studies have shown that Sanren Decoction and other well-known formulations, when combined with gefitinib or erlotinib, can prolong PFS in patients with EGFR-mutant NSCLC and enhance therapeutic efficacy. 5 Moreover, Chinese medicine can help overcome gefitinib resistance. For instance, Jin Fu Kang Decoction (JFKD) combined with gefitinib has been shown to significantly inhibit tumor growth by targeting drug-resistant cells. 29 Therefore, elucidating the molecular mechanism of the synergistic effect of QJYFMF on GE is of great significance for investigating the individualized treatment of NSCLC patients, further prolonging their survival cycle and improving their quality of life.
The A549 cell line, derived from human lung adenocarcinoma, is a widely used non-small cell lung cancer (NSCLC) model due to its epithelial morphology and stable characteristics. When cultured
The characteristics of tumor cells, such as proliferation, anti-apoptosis, cell cycle instability, invasion, and metastasis, form the basis of the biological behavior associated with malignant tumor initiation and progression. These characteristics also represent key points of intervention for anti-tumor drugs.
35
The CCK-8 cell proliferation inhibition assay demonstrated that both QJYFMF and GE inhibited A549 cell proliferation in a dose- and time-dependent manner, with the combined treatment further enhancing this inhibitory effect. Transwell and wound healing assays revealed that QJYFMF effectively inhibited A549 cell invasion and migration. When combined with GE, it improved GE's effects on lung cancer cell invasion and migration, suggesting that QJYFMF can effectively inhibit acquired drug resistance in lung cancer cells. Similar results were observed in flow cytometry experiments, where QJYFMF induced A549 cell apoptosis, and the combination of QJYFMF and GE enhanced this apoptotic effect. Furthermore,
Network pharmacology is a powerful approach for investigating the relationships between drugs, targets, and diseases, particularly for studying the mechanisms of traditional Chinese medicine (TCM), which is characterized by multi-component, multi-target, and multi-channel therapeutic effects.
37
In our study, PTEN, the central target of QJYFMF in the treatment of non-small cell lung cancer (NSCLC), and the PI3K-AKT pathway, a key signaling pathway, were selected through network pharmacology for further experimental investigation. PTEN is a critical tumor suppressor gene involved in the development and inhibition of various cancers, including thyroid cancer,
38
osteosarcoma,
39
gastric cancer,
40
and myeloma.
41
Studies have shown that PTEN loss may have a poor prognostic impact on the overall survival of NSCLC patients.
42
Increased PTEN expression inhibits PI3 K/AKT pathway activation, reduces tumor cell growth, and promotes apoptosis.43-45 In our study, both
AKT, a potent apoptosis inhibitor, plays a crucial role in regulating apoptosis. Upon activation, AKT phosphorylates its downstream target proteins, including BAD, caspase-9, and mTOR, thereby influencing cell proliferation, differentiation, apoptosis, migration, and other vital cellular processes.
50
BAD (Bcl-2/Bcl-xL-associated death promoter), a pro-apoptotic protein belonging to the Bcl-2 family, promotes apoptosis by binding to the pro-survival proteins Bcl-2 and Bcl-xL, inhibiting their protective effects. Continuous AKT activation phosphorylates BAD, causing it to dissociate from Bcl-2, which then exerts its anti-apoptotic function.51-53 Therefore, the induction of apoptosis depends on the Bax/Bcl-2 ratio. The caspase family, a group of proteases, plays a pivotal role in controlling apoptosis. Their expression promotes programmed cell death and suppresses tumor progression. Studies have shown that activation of caspase-3 leads to the upregulation of Bax and the downregulation of Bcl-2, thereby increasing the Bax/Bcl-2 ratio and triggering apoptosis.
54
In the final stages of apoptosis, caspase activation results in the disintegration of apoptotic cells, with caspase-12 activation marking the initiation of the process. As a key inducer of apoptosis, caspase-9 plays a central role in the progression of apoptosis and can serve as a reliable marker for determining the success of the process.
55
In our study, both
However, there are still many limitations in the research content of this article. We did not use drugs or RNA interference to block or activate signaling pathways, and we did not study the pharmacodynamic material basis of QJYFMF. In future research, we will further explore the activation of the PTEN/PI3 K/AKT signaling pathway upstream and downstream, providing rich theoretical basis for QJYFMF targeted therapy of NSCLC.
Conclusion
In conclusion, GE combined with QJYFMF showed significant synergistic effects both
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251334731 - Supplemental material for Investigating the Mechanism of Qingjie Yifei Miao Fang in Synergistic Treatment of Non-Small Cell Lung Cancer with Gefitinib Based on Network Pharmacology, In Vitro and In Vivo Experiments
Supplemental material, sj-docx-1-npx-10.1177_1934578X251334731 for Investigating the Mechanism of Qingjie Yifei Miao Fang in Synergistic Treatment of Non-Small Cell Lung Cancer with Gefitinib Based on Network Pharmacology,
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251334731 - Supplemental material for Investigating the Mechanism of Qingjie Yifei Miao Fang in Synergistic Treatment of Non-Small Cell Lung Cancer with Gefitinib Based on Network Pharmacology, In Vitro and In Vivo Experiments
Supplemental material, sj-docx-2-npx-10.1177_1934578X251334731 for Investigating the Mechanism of Qingjie Yifei Miao Fang in Synergistic Treatment of Non-Small Cell Lung Cancer with Gefitinib Based on Network Pharmacology,
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251334731 - Supplemental material for Investigating the Mechanism of Qingjie Yifei Miao Fang in Synergistic Treatment of Non-Small Cell Lung Cancer with Gefitinib Based on Network Pharmacology, In Vitro and In Vivo Experiments
Supplemental material, sj-docx-3-npx-10.1177_1934578X251334731 for Investigating the Mechanism of Qingjie Yifei Miao Fang in Synergistic Treatment of Non-Small Cell Lung Cancer with Gefitinib Based on Network Pharmacology,
Footnotes
Acknowledgements
We thank International Science Editing for language editing. The authors would also like to thank the editors and anonymous reviewers for their valuable comments and suggestions.
Statement of Human and Animal Rights
All animal experiments were approved by the Experimental Animal Ethics Committee of Guizhou University of Traditional Chinese Medicine (approval number: 20210086), Guizhou Province, China.
Ethical Considerations
This study was approved by the Experimental Animal Ethics Committee, Guizhou Province, China.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82160952) and the Basic Research Program of Guizhou Provincial Department of Science and Technology (qiankehe jichu [2020]1Y364).
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
