Abstract
Introduction
Knee osteoarthritis (KOA) is a prevalent chronic degenerative joint disease that primarily results in progressive damage to the articular cartilage, followed by osteoarthritis of the subchondral bone and the surrounding synovial structure.1,2 This condition is mainly characterized by knee pain, stiffness, limited function, and potential disability.3,4 It is estimated that KOA impacts around 250 million individuals globally, with 10.00%-16.00% of individuals over 60 years old experiencing symptomatic KOA.5,6 In 2018, There were 249 000 cases of knee replacement surgeries in China, amounting to about 12.948 billion yuan in costs. 7 As the global population continuous to age, the prevalence and incidence of KOA are increasing annually, making it a significant global public health concern. 8 Hence, KOA has become a major global public health problem, and effective treatment is significant. Invariably, the main purpose of KOA treatment is to eliminate or alleviate pain, improve knee joint function, and improve patients’ quality of life. Currently, international and national guidelines generally recommend non-steroidal anti-inflammatory drugs (NSAIDs) for KOA treatment.9,10 However, despite NSAIDs being commonly used to alleviate OA pain, many studies have found that long-term NSAIDs usage can lead to adverse effects, particularly an increased risk of gastrointestinal and cardiovascular diseases, posing a threaten to patients live and significantly impacting their quality of life.11,12 Recent guidelines in China and globally suggest topical medications as an alternative, adjuvant or even primary treatment for KOA. 13 Prior to oral administration, topical treatment is often recommended to alleviate mild or moderate knee arthritis pain due to their favorable safety profile. 14 Therefore, there is a growing interest in developing a topical medication that can effectively treat early to mid-stage KOA.
Traditional Chinese Medicine (TCM) has demonstrated significant therapeutic effects and is increasingly acknowledged. Various methods of Chinese herbal interventions for treating KOA by TCM, including fumigation, iontophoresis, hot compress, etc 15 TCM has a good effect on the treatment of symptomatic KOA, and its efficacy and safety have been confirmed by some experiments.16,17 Tongfu Guanjie Gao (TFGJG) (also known as Shujin Tongluo Gao) is a traditional Chinese medicine external preparation developed and refined by the Pharmacy Department of Gansu Provincial Hospital of Traditional Chinese Medicine, based on the hospital's in-house formulation Tongfu Heji. TFGJG comprises nine medicinal herbs: Lycopodii Herba (SJC), Phryma leptostachya L. (TGC), Clematidis Radix et Rhizoma (WLX), Periplocae Cortex (XJP), Chaenomelis Fructus (MG), Aconiti Radix (CW), Aconiti Kusnezoffii Radix (CAW), Carthami Flos (HH), and Piperis Fructus (HJ). It exhibits therapeutic effects of promoting blood circulation, relaxing tendons, unblocking meridians, alleviating pain, and dispelling wind-cold. The plaster has a high drug-loading capacity and excellent adhesion properties, effectively addressing common issues with external preparations such as easy detachment and skin irritation. Preliminary clinical trials have demonstrated that TFGJG shows significant efficacy in treating KOA, with patients reporting high levels of satisfaction. However, due to TCM's complex nature of multi-component, multi-target, and multi-link comprehensive action, the chemical components and mechanism present key research challenges. 18 Network pharmacology, including target prediction, gene ontology (GO), and pathway analysis, is widely used to exploring the molecular mechanisms of TCM therapy.19,20 In addition, Proteomics can elucidate the mechanisms of drug action and therapeutic targets through qualitative and quantitative analysis of proteins.
In summary, TFGJG can effectively alleviate the occurrence and development of KOA, but its targets and mechanisms for treating KOA are not fully understood. In this study, we established a rabbit model of KOA and combined network pharmacology and proteomics techniques to elucidate the specific mechanism of action of TFGJG. And our research indicates that TFGJG can alleviate the symptoms of KOA by improving the damage of articular cartilage in rabbits with KOA and reducing the levels of inflammatory factors in synovial fluid. Mechanism studies show that MAP2K1 and CRTAP proteins play significant roles in TFGJG's alleviation of inflammation and tissue damage in KOA.
Material and Methods
Experiment Animals
Thirty-six healthy and clean New Zealand white rabbits (half male and female, weighing 2.0 ± 0.2 kg) were free to drink water and feed under the same conditions (temperature at 23 ± 2 °C, relative humidity about 50 ± 10%, light and dark cycle of 12/12 h, and good ventilation). All animal experiments were provided by Shaanxi Junxing Biotechnology Co. (Quality Certificate No.: 61003200001229) and were approved by the Ethics Committee of Gansu University of Traditional Chinese Medicine (Ethics No.: 2022-67). After one week of adaptive feeding, six rabbits were selected as the control group (no modeling, routine feeding) by random number table method. The remaining 30 animals were divided into a model group (n = 6, conventional feeding, without any drug intervention), diclofenac sodium gel (DSG) treatment group (n = 6), and TFGJG treatment groups (low, middle, and high dose groups, with six rabbits in each group) after successful modeling.
Experimental Drugs and Reagents
The TFGJG was provided by the Department of Pharmacy of Gansu Provincial Hospital of Traditional Chinese Medicine, and it consists of 9 kinds of traditional Chinese herbs. Diclofenac sodium gel (DSG, 20 g/piece, H20058463) was purchased from Hainan Quanxing Pharmaceutical Co., Ltd Papainand L-homocysteine were purchased from Sigma-Aldrich (USA) pentobarbital sodium solution 4% histiocyte fixative solution, absolute ethanol, isopropanol, etc were purchased from Beijing Solarbio Science Technology Co., Ltd.
Preparation and Quality Control of TFGJG
The specific method is as follows: the Chinese herbal medicine including Carthami Flos and Lycopodii Herba, etc are placed in pure sesame oil and fried until their surfaces turn dark brown and their interiors turn dark yellow. The residue is removed at 220 °C. The residue is removed at 220 °C. The mixture is allowed to cool to 150 °C, and then the weighed white wax is added and stirred until it is completely melted. Select a 5 cm × 5 cm spunlace plaster cloth with an antiseepage ring and a circular sponge (diameter: 2 cm, thickness: 1 mm). Apply the ointment onto the antiseepage ring, and the final product is obtained. And the quality control of TFGJG refers to the results of previous studies. 21
Animal Experiments
Proteoglycans are important cartilage matrix to maintain the elasticity and compression resistance of articular cartilage. Papain can break down the proteoglycans in the cartilage matrix, thus weakening the protective effect of the cartilage matrix on the chondrocytes, leading to chondrocyte degeneration and cartilage degradation, and at the same time inducing the secretion of many inflammatory factors, and ultimately inducing osteoarthritis. In this experiment, the KOA model was constructed by injecting papain into the joint cavity.22,23 The control group was not modeled, while the other five groups were modeled. Specifically, the rabbits were first anesthetized by injecting 3% pentobarbital sodium solution (1 mL/kg). Then, a mixture of 4% papain solution and 0.03 mol/L L-homocysteine (0.1 ml/kg) was injected into the joint cavity of the rabbit (2:1 ratio of the two solutions). After injection, the right knee joint was flexed and stretched 30 times to fully infiltrate the entire joint cavity and to drive the rabbit to move for 30 min each day. Papain was injected respectively on the first, fourth and seventh day of modeling, respectively, respectively. Early KOA histomorphology would be shown at week 4 after papain injection and pharmacological intervention would be initiated.
According to the technical specification for Clinical Application of Traditional Chinese Medicine topical ointment (Draft), 24 an 11 cm × 11 cm skin area was treated with 1 g of ointment three times a day. Based on the human equivalent dose conversion, the daily dose at 4.65 g/kg in the TFGJG low-dose group (Clinical equivalent dose). The TFGJG medium-dose group and the DSG treatment group was at dosage of 9.30 g with medication externally to the right knee joint every day, and 18.60 g/kg in the TFGJG high-dose group, and to use an elastic bandage to wrap the joint once a day for 18 days. 25 Two days after the intervention ended, the rabbit serum and synovial fluid, intact knee joints, synovium, and articular cartilage were collected, and the samples were preserved according to the experimental needs. Furthermore, during the experimentation period, we measured the knee joint range of motion New Zealand rabbits and recorded the Lequesne MG behavioral score.
Hematoxylin & Eosin (H&E) Staining
The collected knee cartilage was fixed with 4% paraformaldehyde for 48 h. The paraffin-embedded tissue blocks were embedded after decalcification with 10% ethylenediaminetetraacetic acid disodium salt (EDTA-2Na). The embedded tissue sections were routinely dewaxed in water; hematoxylin staining (Solarbio, China) was performed for 5-10 min, rinsing with distilled water for 1 min, and differentiation fluid was applied for 30 s. Then, the samples were returned for blue eosin staining (Solarbio, China) for 1 min. The morphological structure of the cartilage and the subchondral bone tissue was observed with a digital microscope (IX-71, Olympus, Japan). The Mankin scale evaluated the HE-stained cartilage tissue morphology, with higher scores indicating more severe cartilage lesions in knee osteoarthritis.
ELISA for Synovial Fluid
The rabbit synovial fluid of the knee of each group was collected, referring to the assay instructions of the rabbit interleukin-1beta (IL-1β), interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), matrix metalloproteinase-13 (MMP-13), collagen type II (COL-2), and cyclooxygenase-2 (COX-2) ELISA kits, and synovial fluid was added. After incubation for two hours, the color development reagent was added, and the reaction plate was washed five times after color development termination. A microplate reader determined each well's absorbance value at 450 nm. The standard curves were fitted, and the levels of IL-1β, IL-6, TNF-α, MMP-13, COL-2, and COX-2 in the synovial fluid of each rabbit were calculated.
Screening of Active Ingredients for TFGJG
BATMAN-TCM (http://bionet.ncpsb.org/batman-tcm/) was used to search for the active ingredients of 9 TCM herbs in TFGJG, and the active compounds and their targets were searched according to the screening criteria of “Score cut off ≥20, Adjusted P-value <.05”. 26
KOA-Related Target Screening
Select “Knee Osteoarthritis” and “Osteoarthritis, Knee” as the keywords to search in the DisGeNet (http://www.disgenget.org/search) database and Gene Cards (https://www.genecards.org/Search/) database. 27 The retrieved potential targets of KOA were summarized, the duplicate targets were eliminated, and the relevant target genes were finally obtained.
Screening of TFGJG-KOA Common Targets
The retrieved KOA-related genes were mapped with the target genes of TFGJG to obtain the target of the treatment of KOA by the TFGJG. 28
Construction of Protein Interaction (PPI) Network
The TFGJG and KOA co-action targets were imported into the STRING database (https://cn.string-db.org/), the free proteins were hidden, and the appropriate PPI protein interaction map and its tsv file were selected and downloaded. The tsv file was imported into Cytoscape 3.8.0 to generate a visualization network, and the Network Analyzer plug-in analyzed the network to determine the core target proteins in the PPI network according to the degree value.
Enrichment Analysis of GO and KEGG Pathways
GO functional enrichment analysis and KEGG pathway enrichment analysis of proteins in the PPI network were performed using the STRING database, and a visual analysis of Cytoscape 3.8.0 was performed.
Protein Extraction and Enzymatic Hydrolysis
Knee joint cartilage tissue (20 mg) was ground in liquid nitrogen, homogenized with SDT lysis buffer, and transferred to an EP tube. The sample underwent a 3-min boiling water bath, 2-min ultrasonic disruption, and centrifugation at 16 000 g for 20 min. The supernatant was collected for BCA protein quantification. 15 μg of protein from each sample was used for SDS-PAGE electrophoresis. For FASP digestion, 200 μg of each sample was treated with 100 mM DTT for 5 min at 100 °C, followed by cooling to room temperature. UA buffer, IAA, NH4HCO3 buffer, Trypsin buffer, and 0.1% TFA were added sequentially for enzymatic digestion. The digested peptides were desalted using a C18 Cartridge, vacuum-dried, re-dissolved in 0.1% FA.
Peptide Fractionation and LC-MS/MS Analysis
The Agilent 1260 high-performance liquid chromatographer (HPLC) performed chromatographic separations in proteomics assays. Buffer A consisted of 10 mM ammonium formate with 90% water, and Buffer B consisted of 10 mM ammonium formate with 90% acetonitrile. LC-MS/MS analysis for data-independent acquisition (DIA) mass spectrometry was conducted using a Q-Exactive HF-X mass spectrometer (Thermo Scientific).
Database Retrieval
All mass spectrometry data were merged and analyzed using the software Spectronaut (version 17, Biognosys) to search the database and build a spectral library. The database was uniprot Oryctolagus cuniculus (Rabbit) [9986] 43523 20230410.fasta, sourced from the website https://www.uniprot.org/.29,30 The results were exported with < 1% FDR at peptide level. To analyze protein temporal changes with different groups, were transformed by Z-score, and the Hartigan-Wong method was used for k-means cluster analysis.
Targeted Protein Verified by Parallel Reaction Monitoring (PRM) Analysis
To confirm the protein expression levels obtained by label-free analysis, two proteins were further selected for validation using the technique of LC-PRM/MS. 31 We prepared the protein following the label-free protocol. The tryptic peptides were separated on an Easy nLC-1200 system (Thermo Scientific). PRM analysis was performed on a Q Exactive Plus mass spectrometer (Thermo Scientific). The mass spectrometer was operated in the positive ion mode, and the raw data were analyzed to obtain the signal intensities of individual peptide sequences using Skyline software 32 (https://skyline.ms/skyline.url). For the MS data from PRM analysis, each sample's average base peak intensity was extracted from the full scan acquisition using RawMeat 2.1 (VAST Scientific). Finally, the relative abundance of protein was defined as a certain peptide's intensity.
Statistical Analysis
Data analysis was performed using GraphPad Prism 8.0. software. All data are presented as mean ± standard deviation (SD). Statistical analysis was performed using one-way ANOVA Tukey post hoc test as a comparison between multiple groups. P < .05 was considered statistically significant. In the figure, “ns” means P > .05; *P < .05; **P < .01; ***P < .001.
Results
TFGJG Relieves the Symptoms of Rabbits with KOA
The results showed a gradually improvement in the activity and swelling of the knee joints in New Zealand white rabbits over time post-drug intervention (Tables 1 and 2). The TFGJG groups displayed superior efficacy compared to the DSG group, with the medium and high dosage groups, the most improvement.
Range of Motion of Rabbit's Right Knee Joint.

Compared with the Control group: #P < .01; compared with the Model group: *P < .01; compared with the DSG group: &P < .01.
The Lequesne MG Score of the Right Knee Joint of the Rabbit.

Compared with the Control group: #P < .01; compared with the Model group: *P < .01; compared with the DSG group: &P < .01.
To further ascertain TFGJG's role in ameliorating KOA, alterations in the knee cartilage tissue of rabbits were examined using H&E staining (Figure 1). The control group exhibited intact cartilage morphology with a smooth surface, normal chondrocytes count, ordinary matrix, and an unbroken tideline (Figure 1A). In contrast, the model group showed a damaged cartilage surface layer, disordered chondrocytes ring structure, missing tideline, and uneven matrix coloration (Figure 1B). In the DSG group, the articular cartilage surface structure remained largely undamaged, with the matrix coloration approximating normal, and the tideline essentially intact (Figure 1C). In the low, medium, and high dose TFGJG groups, there was a dose-dependent reduction in cartilage damage, normalization in chondrocytes morphology and count, and diminished tideline destruction (Figure 1D, E, and F). Compared to the control group, the Mankin scores of rabbits in the model group were significantly higher (P < .001), compared with the model group, the Mankin scores of all treatment groups significantly decreased (P < .001), the Mankin scores for the medium-dose TFGJG (TFGJG-MD) group and high-dose TFGJG (TFGJG-HD) groups were lower than thos of the DSG and low-dose TFGJG (TFGJG-LD) groups (P < .001). No statistically significant difference observed in Mankin scores between the TFGJG-MD and TFGJG-HM groups (P > .05) (Figure 2).

Articular cartilage of the rabbit knee with H&E staining (×100). Scale bar: 100 μm. (a) The control group. (b) The model group, the ↓ indicated cartilage structure was significantly disrupted. (c) The diclofenac sodium gel (DSG) group, the ↓ indicated surface damage and loss of cartilage. (d) The low-dose TFGJG (TFGJG-LD) group. (e) The medium-dose TFGJG (TFGJG-MD) group. (f) The high-dose TFGJG (TFGJG-HD) group. The ↓ in the D, E, and F figures indicated a gradual increase in knee joint cartilage thickness.

Mankin scale of the knee articular cartilage of rabbits. Data were presented as mean ± SD, ### P < .001 versus the Control group; *** P < .001 versus the Model group.
TFGJG Suppresses the Knee Synovial Fluid Levels of Inflammation
The findings indicated an elevated level of synovial fluid inflammatory cytokines, including TNF-α, IL-1β, IL-6, COX-2, COL-2, and MMP-13, in the model group as compared to the control group (Figure 3). Contrasting with the model group, the TFGJG treatment group and the DSG group exhibited significantly reduction in TNF-α, IL-1β, IL-6, COX-2, COL-2 and MMP-13 levels. Furthermore, the levels of TNF-α, COL-2, and MMP-13 in the synovial fluid of rabbits in the TFGJG-MD group were significantly lower than those in the DSG treatment group. The above results indicate that TFGJG treatment can significantly improve the joint inflammation of rabbits with KOA.
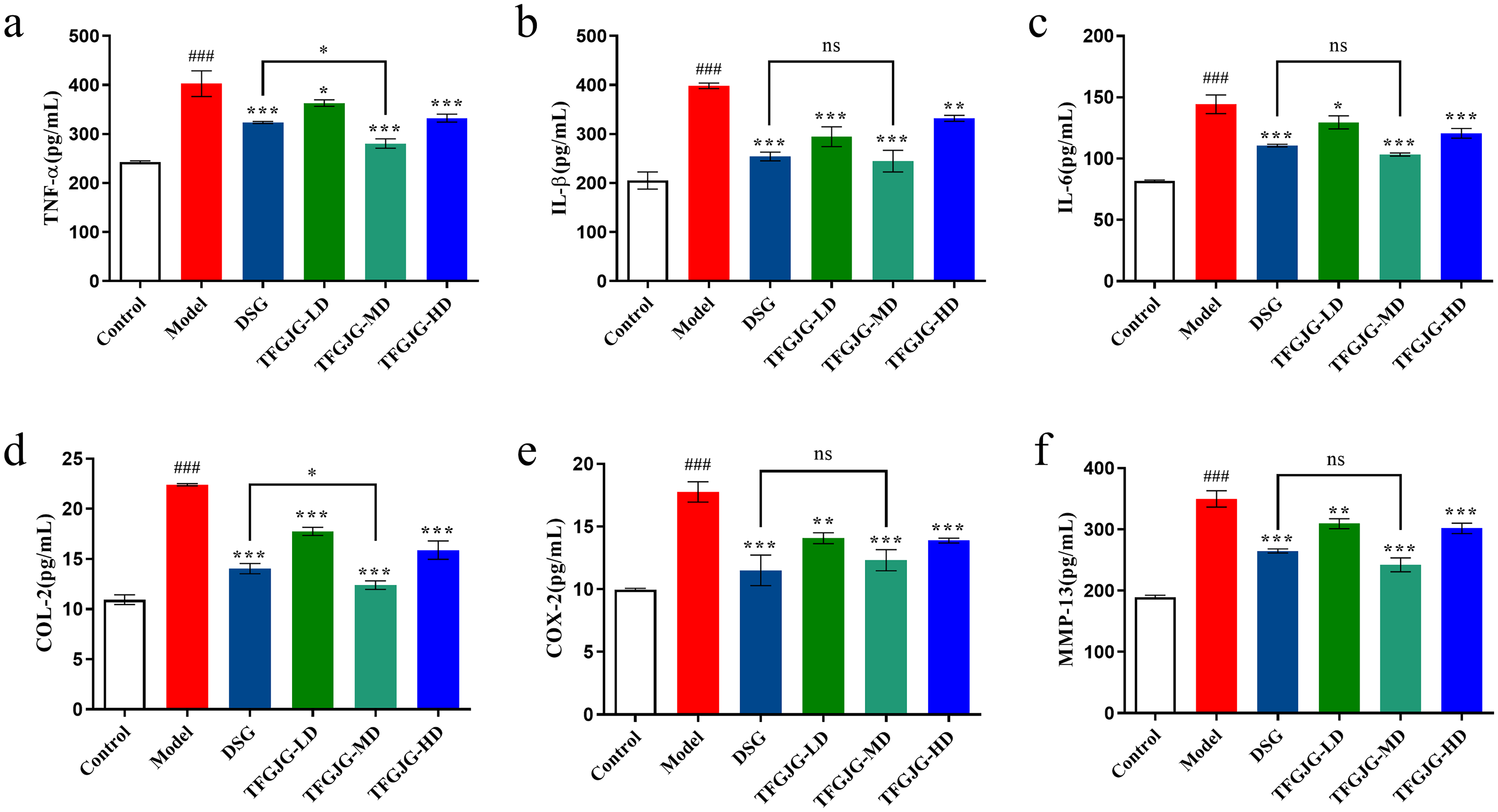
The knee synovial fluid ELISA analysis of rabbits in each group. (a) TNF-α, tumor necrosis factor-alpha. (b) IL-1β, interleukin-1beta. (c) IL-6, interleukin-6. (d) COL-2, collagen type II. (e) COX-2, cyclooxygenase-2. (f) MMP-13, matrix metalloproteinase-13. Data were presented as mean ± SD, ### P < .001 versus the Control group; ns P > .05; * P < .05; ** P < .01; *** P < .001 versus the Model group.
Network Pharmacology Analysis of TFGJG
The construction of the TFGJG -active ingredients-targets network is presented in Figure 4A. In this study, TFGJG was composed of 9 kinds of Chinese herbal medicines, including Lycopodii Herba (named Shenjingcao in Chinese, SJC), Phryma leptostachya L. (named Tougucao in Chinese, TGC), Clematidis Radix et Rhizoma (named Weilingxian in Chinese, WLX), Periplocae Cortex (named Xiangjiapi in Chinese, XJP), Chaenomelis Fructus (named Mugua in Chinese, MG), Aconiti Radix (named Chuanwu in Chinese, CW), Aconiti Kusnezoffii Radix (named Caowu in Chinese, CAW), Carthami Flos (named Honghua in Chinese, HH), and Piperis Fructus (named Hujiao in Chinese, HJ). We retrieved 135 active compounds related to the 9 herbs of TFGJG from the BATMAN-TCM database. For example, we screened and obtained 43 effective components of Clematidis Radix et Rhizoma, including Alpha-Terpineol, Linalool, Methyl Eugenol, etc Piperis Fructus. screened 28 active ingredients, such as Beta-Amyrin, Anethole, Citral, etc; Carthami Flos. selected 22 active ingredients, like Lauric Aldehyde, Quercetin, Kaempferol, among others. At the same time, a total of 1326 potential targets were screened for these 135 active ingredients.

The study used network pharmacology analysis of TFGJG on KOA. (a) The construction of the TFGJG-active ingredients-targets network. (b) Potential targets of the KOA and active ingredients of the TFGJG. There were 218 overlapping proteins between TFGJG and KOA. (c) The top Fifteen common targets. (d) GO analysis of intersecting targets. (e) KEGG analysis of intersecting targets. BP, biological processes; MF, molecular functions; CC, cellular components.
Potential targets for KOA were identified via DisGeNet and Gene Cards databases, resulting in the selection of 1212 targets. The KOA-related target genes retrieved and TFGJG target genes were mapped, yielding 218 TFGJG target genes for KOA treatment (Figure 4B). Analysis was conducted using the cytoHubba plugin of Cytoscape 3.8.0. Five algorithms, namely MCC, MNC, DMNC, Degree, and EPC, were employed to screen hub genes. The intersection of the top 15 genes of the five algorithms was taken, and as a result, hub genes were obtained. The top Fifteen common targets include AKT1, IL6, TNF, IL1B, INS, TP53, STAT3, TGFB1, CTNNB1, PPARG, HIF1A, ESR1, COL1A1, MAP2K1, and CRTAP (Figure 4C).
The GO function enrichment analysis was carried out by DAVID online software to obtain 3395 items of biological processes (BP) (P < .01), 260 items of molecular function (MF) (P < .01), 114 items of cellular component (CC) (P < .01), the top 10 BP/MF/CC items were shown in Figure 4D. BP enrichment mainly involves processes like epithelial cell proliferation, gland development, ossification, response to nutrient levels, regulation of inflammatory response, and other. MF enrichment mainly involves receptor ligand activity, signaling receptor activator activity, DNA-binding transcription factor binding, cytokine activity RNA polymerase II-specific, cytokine receptor binding, and so forth. CC enrichment mainly involves areas like the endoplasmic reticulum lumen, collagen-containing extracellular matrix, external side of the plasma membrane, cytoplasmic vesicle lumen, membrane raft, and more. Furthermore, 212 pathways were screened from KEGG pathway enrichment (P < .01) by using DAVID online software. By means of the DAVID online website, the gene EntrezID was obtained. Visualization was carried out using R 4.2.3, and in its console, “showNum = 30” was set to display the Top 30 signaling pathways. The top 30 pathways are presented in Figure 4E. Target genes were enriched in these pathways, 6 of which were closely related to KOA, including the TNF signaling pathway, FoxO signaling pathway, IL-17 signaling pathway, PI3K-Akt signaling pathway, Cellular senescence, and MAPK signaling pathway. The results revealed that TFGJG might have therapeutic effects on KOA through anti-inflammation, anti-apoptosis, and the regulating of hormone biosynthesis and secretion.
Proteomics Analysis of TFGJG
In this study, 2608 proteins were Quantified. In the analysis of the T-test method combined with Fold change (FC, ratio of mean expression between two groups) was employed to identify significantly different proteins (usually P-value < .05, FC > 1.5 or <1/1.5). There were 203 up-regulated proteins and 635 down-regulated proteins between the KOA model group and the control group (Figure 5). Between the TFGJG treatment group (the medium dose TFGJG group) and the control group, there were 67 up-regulated proteins and 660 down-regulated proteins. Between the TFGJG treatment group and the model group, there were 26 up-regulated proteins and 124 down-regulated proteins (Figure 5A and B).

Identification and bioinformatics analysis of differential proteins. (a) The volcano plot depicts the differential proteins between the TFGJG treatment group (the medium-dose TFGJG group) and the control group. (b) The volcano plot depicts the differential proteins between the TFGJG treatment and the model group. (c) GO analysis of differential proteins between the TFGJG treatment and the control groups. (d) GO analysis of differential proteins between the TFGJG treatment group and the model group. (e) Top 10 of KEGG pathway analysis between the TFGJG treatment and the control groups. (f) Top 10 of KEGG pathway analysis between the TFGJG treatment group and the model group.
The GO analysis of differential proteins between TFGJG treatment group and control group demonstrated that these proteins were involved in biological processes like cellular process, metabolic process, localization, biological regulation, response to stimulus. Cellular components like cellular anatomical entity and protein-containing complex were enriched, and the differential proteins were enriched in molecular functions such as binding, catalytic activity, structural molecule activity, ATP-dependent activity and molecular function regulator activity (Figure 5C). In addition, the GO analysis of differential proteins between the TFGJG treatment group and the model group showed that these proteins were involved in biological processes like cellular process, metabolic process, response to stimulus, localization, immune system process. Cellular components such as cellular anatomical entity and protein-containing complex were enriched, and the differential proteins were enriched in molecular functions such as binding, catalytic activity, molecular function regulator activity, structural molecule activity and transporter activity (Figure 5D).
The KEGG pathway enrichment analysis between the TFGJG treatment group and the control group showed that 305 pathways were enriched with differential proteins Figure 5E showed the top 10 pathways with significant enrichment. These including Amyotrophic lateral sclerosis, Carbon metabolism, Complement and coagulation cascades, Glutathione metabolism, Lysosome, Phagosome, Protein processing in the endoplasmic reticulum, Spliceosome, Staphylococcus aureus infection, Valine, leucine and isoleucine degradation. Additionally, the KEGG pathway enrichment analysis between the TFGJG treatment group and the KOA model group showed that 211 pathways were enriched with differential proteins. Figure 5F showed the top 10 pathways with significant enrichment. These including 2-monocarboxylic acid metabolism, Amino sugar and nucleotide sugar metabolism, Amyotrophic lateral sclerosis, Arginine and proline metabolism, Biosynthesis of nucleotide sugars, Complement and coagulation cascades, Phagosome, Prion disease, Proteasome, Tuberculosis.
Integrative Analysis of Network Pharmacology and Proteomics to Identify Key Targets of TFGJG for KOA
The 796 differential proteins (P < .05) were targeted, and their expression values in the TFGJG treatment group, the model group, and the control group were analyzed. K-means clustering analysis was performed on these 796 differential proteins, and they were classified into four types based on expression patterns (Figure 6A). The number of proteins in cluster 1 to cluster 4 are 132, 65, 89, and 510, respectively. Among the four clusters, the differential proteins of cluster 3 were of primary interest to us. This is because the differential proteins of the model group were up-regulated, while the differential proteins of the TFGJG treatment group and the control group remained consistently down-regulated. Furthermore, we combined 218 target proteins of TFGJG for KOA from obtained from network pharmacology with the differential proteins of cluster 3. This allowed us to obtain two common proteins MAP2K1 and CRTAP (Figure 6B).

Comprehensive analysis of network pharmacological and proteomic results. (a) Four clusters of the 796 differential proteins in the three groups. (b) The target protein of TFGJG for KOA from network pharmacology was combined with the differential protein of cluster 3.
The Validation of MAP2K1 and CRTAP by PRM Analysis
To validate the reliability of the DIA proteomic data, we conducted a high-throughput technology PRM analysis. Based on the quantitative results from both proteomics and network pharmacology analyses, two proteins of interest, MAP2K1 and CRTAP, were selected for further analysis. Figures 6A and 7C show the secondary mass spectra of the target peptides DVKPSNILVNSR and SYESLFIR of MAP2K1 and CRTAP, respectively. The results indicated that the expression levels of MAP2K1 and CRTAP in the model group were significantly higher than those in the control group. However, the expression levels of MAP2K1 and CRTAP in the TFGJG treatment group were significantly lower than those in the model group and no significant difference from the control group (Figure 7B and D). Most importantly, the PRM results for these key proteins were consistent with the experimental data from the proteome. This consistency further validates the accuracy of our findings.

Validation of MAP2K1 and CRTAP by PRM analysis. The Skyline analysis of target peptides of (a) MAP2K1 and (c) CRTAP. The Relative abundance of (b) MAP2K1 and (d) CRTAP in each group. Data were presented as mean ± SD, * P < .05.
Discussion
KOA is a complex joint disorder that involves pathological changes in various components of the knee, including the articular cartilage, subchondral bone, synovium, meniscus, ligaments, and surrounding muscles.33,34 Common clinical manifestations of KOA include knee joint pain, swelling, stiffness, and limited mobility. Many studies have shown that the pathological mechanisms of KOA are closely related to inflammation, metabolism, ageing, apoptosis, and cellular autophagy.35,36 Topical drugs are often recommended as the first choice for the treatment of KOA. 37 Although NSAIDs significantly alleviate KOA symptoms, some studies suggest that long-term use of NSAIDs may exacerbate KOA symptoms and accelerate the process of knee joint replacement surgery. 38 The use of traditional Chinese medicine externally for the treatment of KOA has a long history, but its specific mechanism of action remains unclear. In this study, we established an animal model of KOA via intra-articular injection of papain. The mechanism of action and potential therapeutic targets of TFGJG in the treatment of KOA were studied mainly by network pharmacological analysis and proteomics.
Firstly, our observation revealed that TFGJG can significantly improve knee joint flexibility and reduce swelling. Hematoxylin and Eosin staining showed that TFGJG, particularly in medium and high-dose groups, can alleviate cartilage damage, and the morphology of cartilage cells gradually becomes regular. Therefore, we believe TFGJG has certain therapeutic effects on KOA. Previous studies have found that the pathogenesis of KOA is closely related to inflammation,39,40 immunity, cellular senescence, cellular apoptosis, and autophagy. 41 In our study, the levels of TNF-α, IL-1β, IL-6, COX-2, COL-2, and MMP-13 in synovial fluid, as detected by ELISA showed some changes in both TFGJG and DSG groups. The changes in medium dose TFGJG group were most significant. Therefore, we consider that TFGJG treats KOA mainly through its anti-inflammatory and cartilage morphology-improving effects.
To further investigate the mechanism of action of TFGJG on KOA, we used Network pharmacology to reveal its potential targets and pathways of action. In the current study, we identified 135 active ingredients of 9 kinds of Chinese medicines in TFGJG, and 1326 potential targets of active ingredients, 1212 potential targets of KOA, and 218 target genes of TFGJG for KOA. The top fifteen common targets include AKT1, IL6, TNF, IL1B, INS, TP53, STAT3, TGFB1, CTNNB1, and PPARG, HIF1A, ESR1, COL1A1, MAP2K1, and CRTAP. These target genes were enriched in several pathways, 6 of which were closely related to KOA, including the TNF signaling pathway, FoxO signaling pathway, IL-17 signaling pathway, PI3K-Akt signaling pathway, Cellular senescence, and MAPK signaling pathway. Within these signaling pathways, TNF-α forms a complex upon binding to the TNF receptor (TNFR), thereby activating the MAPK and NF-κB signaling pathways and resulting in the expression of pro-inflammatory genes such as IL-1β, IL-6, and COX-2. In the present study, after TFGJG treatment, the levels of IL-1β, IL-6, and COX-2 were significantly decreased, suggesting that TFGJG can suppress joint inflammation in rabbits. Furthermore, once the inflammatory factors increase, the catabolism of chondrocytes is accelerated. In the present study, after TFGJG treatment of KOA, the expressions of COL-2 and MMP-13 were both suppressed, suggesting that it can protect cartilage from degeneration. These results revealed that TFGJG might have therapeutic effects on KOA through anti-inflammation, anti-apoptosis, and regulating hormone biosynthesis and secretion.
Subsequently, we identified 2608 proteins by proteomic analysis using DIA technology. Twenty-six proteins were up-regulated and 124 proteins were down-regulated between the TFGJG treatment group and the model group, respectively (P < .05). Moreover, we combined 218 target proteins of TFGJG for KOA from Network pharmacology with differential proteins of cluster 3 to obtain two potential targets, including MAP2K1 and CRTAP. MAP2K1 lies upstream of MAP kinases and stimulates the enzymatic activity of MAP kinases in response to a wide variety of extra- and intracellular signals. As an essential component of the MAP kinase signal transduction pathway, this kinase is involved in numerous cellular processes, such as proliferation, differentiation, transcription regulation, and development. MAP2K1 has been shown to participate in the pathogenesis, pathological process, and related treatment of Rheumatoid Arthritis (RA) and osteoporosis in patients or induced RA and osteoporosis cell models and animal models.42,43 The relevant signaling pathways involved in MAP2K1 include the PI3K/Akt and MAPK pathways. Moreover, it has been discovered that targeting MAP2K1 can block the MAPK signaling pathway and mitigate the damage to chondrocytes inflicted by IL-1β. CRTAP is a scaffolding protein that may influence the activity of at least one member of the cytohesin/ARNO family in response to specific cellular stimuli. Defects in this gene are associated with osteogenesis imperfecta, a connective tissue disorder characterized by severe osteoporosis and low bone mass. CRTAP protein can form a complex with P3H1 and CYPB (cyclophilin B), and Crtap−/− bone and cartilage collagen exhibit reduced levels of proline 3 hydroxylation. 44 CRTAP exerts a crucial role in bone metabolism. The lack of CRTAP can result in chondrodysplasia, a decrease in bone matrix, and functional defects of osteoblasts, etc Therefore, we speculated that TFGJG may exert an anti-KOA effect by regulating MAP2K1 and CRTAP.
Finally, we further validated these two targets using PRM targeted proteomics techniques. 45 The expression levels of MAP2K1 and CRTAP in the TFGJG-treated group were significantly lower than those in the model group, and were not significantly different from those in the control group. There are limited reports in the literature on the mechanism of interaction between MAP2K1 and CRATP proteins with KOA. In this part, we opted to use PRM to validate target proteins due to its antibody-independent, reproducibility, and high sensitivity. However, it is the first time that we have studied TFGJG on KOA effect, and its mechanism of action needs to be further explored.
This study acknowledges several limitations. At present, there are few and insufficiently in-depth studies on the improvement of KOA by TFGJG, which has thus restricted the research in this paper. While we have conducted an in-depth investigation into the mechanisms by which TFGJG affects KOA, a comprehensive understanding of its therapeutic mechanisms remains elusive. Additionally, TFGJG is predominantly prepared manually by experienced traditional Chinese medicine pharmacists, and large-scale mechanized production has not yet been achieved. This manual preparation process may introduce batch-to-batch variability, potentially impacting the consistency and reproducibility of experimental outcomes. Furthermore, due to constraints in time and funding, the animal model used in this study was established via intra-articular injection of papain. Alternative modeling techniques that more closely mimic degenerative knee osteoarthritis could provide more robust and clinically relevant results.
This study elucidated the mechanisms and therapeutic targets of TFGJG in treating KOA through an integrated approach combining network pharmacology and proteomics. TFGJG significantly reduced levels of pro-inflammatory factors, including TNF-α, IL-1β, IL-6, COX-2, COL-2, and MMP-13, in the synovial fluid of KOA rabbits, thereby improving cartilage damage. MAP2K1 and CRTAP were identified as key targets and validated using PRM proteomics. Our findings enhance the understanding of TFGJG's mechanism of action and provide novel insights into potential therapeutic targets. Future research will employ metabolomics to further investigate TFGJG's effects and conduct larger-scale clinical randomized controlled trials to provide robust evidence for its therapeutic efficacy.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251329454 - Supplemental material for Integrated Network Pharmacology and Proteomics to Elucidate the Mechanism and Potential Targets of Tongfu Guanjie Gao on KOA in a Papain-Induced Rabbit Model
Supplemental material, sj-docx-1-npx-10.1177_1934578X251329454 for Integrated Network Pharmacology and Proteomics to Elucidate the Mechanism and Potential Targets of Tongfu Guanjie Gao on KOA in a Papain-Induced Rabbit Model by Hong Shao, Zhiwei Chen, Licun Zhang, Wenjuan Ye, Xixiang Li, Qingjie Huang, Liang Pei and Jirong Zhao in Natural Product Communications
Footnotes
Acknowledgements
We thank the researcher and clinicians in this group for their perseverance and dedication, and the editors for their comments on this article.
Author Contributions
Hong Shao, Zhiwei Chen, Licun Zhang, Liang Pei wrote and conducted experiments, Wenjuan Ye, Xixiang Li, Qingjie Huang conducted data analysis and revised the manuscript. Jirong Zhao participated in the design of the study and was responsible for the overall planning of the manuscript. All authors have read and approved the final draft.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The Gansu University of Chinese Medicine approved the study.
Funding
The author discloses that the research, authorship and/or publication of this article has received the following financial support: This study is funded by the Traditional Chinese medicine characteristic technology inheritance and protection center and talent training base project (202117).
Statement Informed Consent
There are no human participants in this article and informed consent is not required.
Trial Registration Number
Not applicable.
Grant Number
Not applicable.
Statement of Animal Rights
All animal experiments were approved by the Ethics Committee of Gansu University of Chinese Medicine (No. 35, Dingxi East Road, Chengguan District, Lanzhou City, China) (Ethics Number: 2022-67, Approval Date: January 19, 2022).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
