Abstract
Introduction
Cancer is the second cause of death worldwide and accounted for 20 million new cases and 9.7 million deaths in 2022.1,2 Leukemia is the 13th most frequently diagnosed cancer, and the 10th leading cause of cancer-related death worldwide and frequently affects people under 20 years of age.2,3 The main drugs for the treatment of leukemia, such as dasatinib and imatinib, cause side effects and face challenges such as toxicity, development of cellular resistance mechanisms, and disease recurrence.4–6 Therefore, it is necessary to search for more effective and selective new drugs against leukemia cells.
One of the most important alternatives in the search for new anticancer drugs are natural products. It is estimated that 40% of the drugs for the treatment of cancer have been inspired by natural products or semi-synthesized from a compound isolated from a natural source. 7 For this reason, numerous studies are aimed at searching for new bioactive compounds from different natural sources, as well as in the development of structural analogues of these compounds to improve their biological activity and reduce side effects.7,8
Pentacyclic triterpenes (PT) are compounds isolated from natural sources that exhibit various biological activities.9–12 Although they have received significant attention in cancer research due to their antineoplastic properties, 13 their therapeutic use is limited by their low bioavailability, which results from their poor water solubility. 14
To improve the bioavailability of triterpenes, derivatives can be synthesized by binding them to cations, nitrogen compounds, counterions or by forming complexes with cyclodextrins, liposomes, carbon nanotubes and gold nanoparticles.15–18 In addition, the introduction of functional groups such as glycosides, esters, amides, phosphates and ammonium salts has been shown to optimize both their bioavailability and biological activity.16–18
Additionally, PT offer promising sites for obtaining new derivatives with improved biological activity. For example, the introduction of a propynoyl group at C-28 of betulin has demonstrated a strong cytotoxic effect on acute lymphoblastic leukemia (CCRF-CEM) and murine leukemia (P388) cells. 19 Also, the combination of hydroxyl oxidation at C-3 and the incorporation of an acetylenic group at C-28 in betulin has enhanced its activity against the acute promyelocytic leukemia cell line (HL-60). 19 Additional modifications at C-3, such as the addition of a 4-fluorophenylhydrazone molecule in betulin, have generated compounds with significant cytotoxic activity in the acute lymphoblastic leukemia cell line (MOLT-4). 20 On the other hand, the formation of betulinic and betulonic acid methyl esters has shown increased activity in chronic myeloid leukemia (K562).21–23 In addition, the introduction of heterocycles such as 12,3-triazole at C-3 or at C-28 of betulinic acid has generated derivatives with enhanced cytotoxic activity in HL-60 and acute myeloid leukemia (THP-1) cell lines.13,24
Moreover, other structural modifications of lupane-type triterpenes have also been performed at C-30. For example, oxidation at C-30 of lupeol leads to the formation of an α,β-unsaturated aldehyde on the isopropylidene fragment, generating a compound with a more cytotoxic profile in acute lymphoblastic leukemia (JURKAT) and K562 cell lines. 4 Therefore, the presence of the formyl group, in the form of the α,β-unsaturated aldehyde, has contributed to more active compounds against leukemic cells compared to its non-functionalized analogue.25–27
PT also have become crucial compounds in medicinal chemistry due to their versatility and broad compatibility with various biological structures and targets. 18 This highlights their importance in the research and development of new antineoplastic agents. 28
In previous studies, the pentacyclic triterpenes 3α,23-O-isopropylidenyl-3α,23-dihydroxylup-20(29)-en-28-oic (
Materials and Methods
General Experimental Procedures
NMR spectra were recorded in CDCl3 on a Bruker Avance 400 spectrometer. The chemical shifts are given in δ (ppm) with residual deuterated solvent as internal reference and coupling constants in Hz.
Precoated TLC silica gel 60 F254 aluminum sheets from Sigma-Aldrich were used for thin-layer chromatography (0.25 and 0.5 mm layer thickness for analytical and preparative TLC, respectively) and visualized under short (254 nm) and long (366 nm) wave-length UV light, or a spray reagent (H2SO4-AcOH-H2O, 1:20:4). Column chromatography (CC) was conducted using silica gel 60 (63-200 or 40-63, particle size), TLC (2-25 µm, particle size) or Sephadex LH-20 from Sigma-Aldrich (Saint Louis, MO, USA).
The bioassays were performed inside a laminar flow hood, brand NuAire, class II, type A2. The cells were kept inside a CO2 incubator with a water jacket and HEPA filter, brand NuAire.
Plant Material
Aerial parts of Phoradendron wattii Krug & Urb. (Santalaceae) (synonym: Phoradendron vernicosum Greenm.) were collected in March 2019 on the Hunucmá-Sisal highway (21°05038.0” N, 89°58021.4” W), Yucatán (México). Plant material was identified by Tech. Paulino Sima-Polanco (CICY). A voucher sample (PS 3220) was deposited at the U Najil Tikin Xiu herbarium of CICY. The aerial parts were dried under artificial light (50-60 °C) for three days and then ground. Finally, after having the plant material dried, it was crushed using a blade mill without cooling (Pagani, model 2030) until 33.5 kg of powdered material of a particle size # 7 were obtained.
Extraction and Isolation
The compounds 3α,23-O-isopropylidenyl-3α,23-dihydroxylup-20(29)-en-28-oic acid (
Preparation of Compound T2a from T2
A solution of 50 mg (0.10 mmol) of compound
3α,23-O-Isopropylidenyl-3α,23-dihydroxylup-20(29)-en-28-ol (
Preparation of Compound T2b from T2a
An amount of 23 mg (0.05 mmol) of compound
3α,23-O-Isopropylidenyl-3α,23-dihydroxylup-20(29)-en-28-methylene acetate (
Preparation of Compound T2c from T2
Compound
3α,23-O-Isopropylidenyl-3α,23-dihydroxy-30-oxolup-20(29)-en-28-oic acid (
Preparation of Compound T2d and T2e from T2a
Compound
3α,23-O-Isopropylidenyl-3α,23-dihydroxy-30-oxolup-20 (29)-en-28-ol (
3α,23-Dihydroxy-30-oxolup-20(29)-en-28-ol (
Preparation of Compound T2f from T2
An amount of 15.3 mg (0.03 mmol) of compound
Methyl ester of 3α,23-O-isopropylidenyl-3α,23-dihydroxylup-20(29)-en-28-oic acid (
Leukemia Cell Lines
Acute myeloid leukemia cell line (THP-1, ATCC TIB-202) and acute lymphoblastic leukemia cell lines (CCRF-CEM, ATCC CCL-19; JURKAT CLONE E6-1, ATCC TIB-152; MOLT-4, ATCC CRL-1582; REH, ATCC CRL-8286) from the American Type Culture Collection (ATCC) were provided by María Antonieta Chávez González, from Unidad de Investigación Médica en Enfermedades Oncológias, UMAE Hospital de Oncología, Centro Médico Nacional Siglo XXI, IMSS. All procedures were approved by the Ethics and Scientific Committee at IMSS. Acute lymphoblastic leukemia cell lines and THP-1 cell line were maintained with RPMI 1640 medium (Roswell Park Memorial Institute medium) at 10% FBS (fetal bovine serum); 1% penicillin-streptomycin was added and incubated at an atmosphere of culture with 95% humidity and 5% CO2 at 37 °C. All evaluations were performed between 3 and 4 cell passages.
Compounds and Controls
The compounds were dissolved in dimethyl sulfoxide (DMSO) at a concentration of 20 mg/mL. In all cases, medium with DMSO (0.1%) was used as a negative control, and dasatinib was used as positive control. Normal mononuclear cells (MNC) were used as normal cell control. All tests were performed in triplicate.
Bioassay of Viability in Leukemic Cell Lines
A total of 1 × 105 cells/well were cultured into 96-well plates and treated with different concentrations of compounds (0.01, 0.1, 1.0, and 10 µg/mL) for 48 h. At the end of this time, the cells were collected and stained with 4’,6-diamidino-2-phenylindole dihydrochloride (DAPI) at 500 ng/mL for 15 min in darkness, according to manufacturer instructions. The samples were analyzed by flow cytometry with a FACSVerse flow cytometer (BD Bioscience, San Jose, CA, USA). The IC50 obtained in this analysis (µg/mL) was changed to µM.
Viability Test in Normal Mononuclear Cells (MNC)
Normal mononuclear cells were obtained from normal human peripheral blood monocytes. MNC were collected according to institutional guidelines, including written informed consent from each donor. Collection procedures were approved by Ethics and Scientific Committee of the Mexican Institute of Social Security (IMSS, ethics approval number CNIC R-2021-785-025). The MNC were purified with FicollPaque Plus (Pharmacia Biotech, Uppsala, Sweden) by centrifugation at 400 × g at room temperature for 30 min according to the manufacturer's protocol. Once the cells were obtained, they were resuspended in RPMI medium) with 10% FBS, and they were counted using a hemocytometer, previously stained with a trypan blue solution, verifying the viability of 95%. 34
A total of 1 × 105 cells/well were cultured into 96-well plates and treated with different concentrations of compounds (12.5, 25, 50, and 100 µg/mL) for 48 h. RPMI medium with 10% FBS was used as culture medium for cell growth. At the end of this time, the cells were collected, washed with phosphate buffer saline (PBS), and stained with DAPI at 500 ng/mL for 15 min in darkness, according to manufacturer instructions. The samples were analyzed by flow cytometry with a FACSVerse flow cytometer (BD Bioscience, San Jose, CA, USA). The IC50 obtained in this analysis (µg/mL) was changed to µM.
Apoptosis Assay
Cells (1 × 105 cells/well) were cultured in 96-well plates and treated with compounds at two different concentrations (½ and 1 × IC50), to be later incubated for 48 h. After this time, cells were washed with PBS and stained with annexin V-FITC (BD Bioscience) and incubated in the dark for 15 min; subsequently, the cells were washed with PBS and stained with DAPI at 500 ng/mL for 15 min in darkness. Then they were analyzed by flow cytometry with a FACSVerse flow cytometer (BD Bioscience, San Jose, CA, USA).
In Silico Studies
The ADME (absorption, distribution, metabolism and excretion) properties of compounds, such as molecular weight (MW), hydrogen bond acceptor (nHBA), hydrogen bond donors (nHBD), lipophilicity (cLogP), rotatable bonds (nROTB), and topological polar surface area (TPSA), and pharmacokinetic parameters were predicted using SwissADME. 35
Protein target structures for molecular docking studies were retrieved from RCSB Protein Data Bank, 36 selecting BCL-2 in complex with a small molecule inhibitor targeting BCL-2 BH3 domain (N-(6-{4-[(4’-chlorobiphenyl-2-yl)methyl]piperazin-1-yl}-1,1-dioxide-1,2-benzothiazol-3-yl)-4-{[(2R)-4-dimethylamino)-1-(phenylsulfanyl) butan-2-yl]amino}-3-nitrobenzenesulfonamide) (PDB-ID: 4IEH) and Epidermal Growth Factor Receptor tyrosine kinase domain with erlotinib as an inhibitor (PDB-ID: 1M17). Molecular docking studies were performed using software Autodock4 (v.4.2). 37 Protein and ligands were prepared using Autodock Tools (ADT, v.1.5.7) to obtain proper inputs for Autodock4. The polar hydrogen charges of Gasteiger-type were assigned, and the nonpolar hydrogens were merged with the carbon atoms. All the protein was considered as a rigid body and the ligands being flexible. All the torsions and rotable bonds in the ligands were defined. The grid box for ligands were centered at the co-crystalized inhibitor, with dimensions of 70 × 70 × 70 with a spacing of 0.375 Å. The search was performed with the Lamarckian Genetic Algorithm as is implemented in Autodock4 code. The population of 150 individuals was mutated with a mutation rate of 0.02 and evolved for 10 generations. The number of the docking runs was 100. The best binding mode was selected based on the lowest energy binding and more populated cluster. Molecular graphics and analyses were performed with UCSF ChimeraX v1.8.38,39
Analysis of Results
All cytometric data were analyzed using FlowJoTM v. 10.9 software. Results of viability were calculated by GraphPad Prism software version 9. IC50 values (the concentration of the compound that causes 50% of cell proliferation inhibition) were obtained from dose-response curves using GraphPad Prism software version 9. The experiments were performed in triplicate. The data were expressed as means ± SEM. Statistical significance was calculated with a one-way analysis of variance (ANOVA) followed by Dunnett's post-hoc test, applying a p < 0.05 significance level.
Results
In the present study and based on previous studies about cytotoxic effects of betulin and
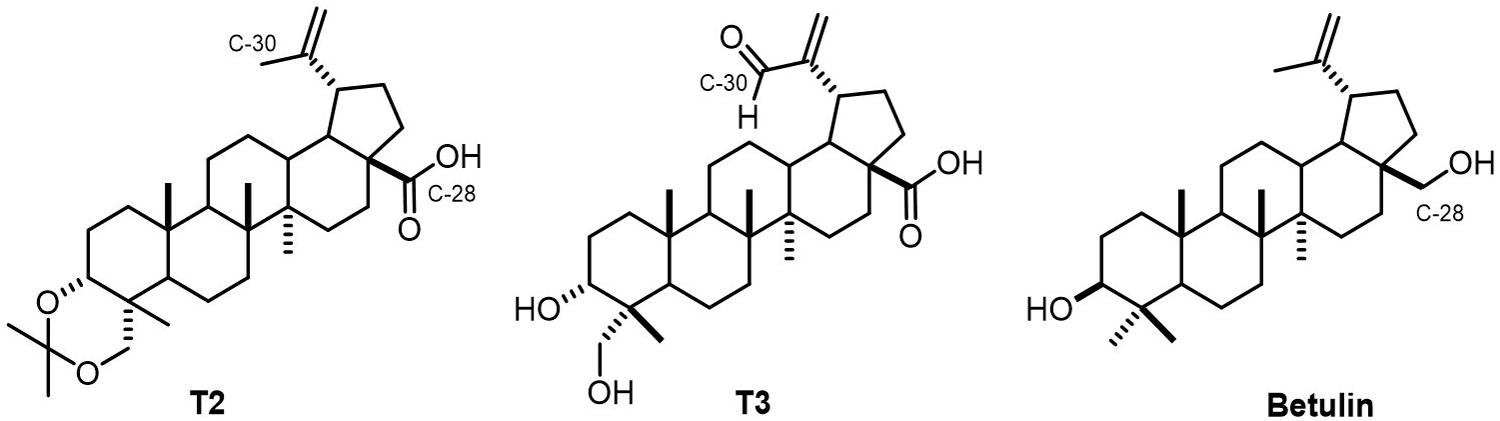
Chemical structures of
The structural modification of

Semi-synthesis of 3α,23-O-isopropylidenyl-3α,23-dihydroxylup-20(29)-en-28-oic derivatives (
Subsequently, from
The allylic oxidation reaction on C-30 of
The effect on the viability of leukemic cells was evaluated using different concentrations of
The Mean Inhibitory Concentrations of Triterpenes and Derivatives Evaluated Against Leukemia Cell Lines and Peripheral Blood Mononuclear Cells.

MNC: normal mononuclear cells; SI: selectivity index (ratio of IC50 in the MNC line over IC50 in the leukemia cell line); N/A: no active.
On the other hand,
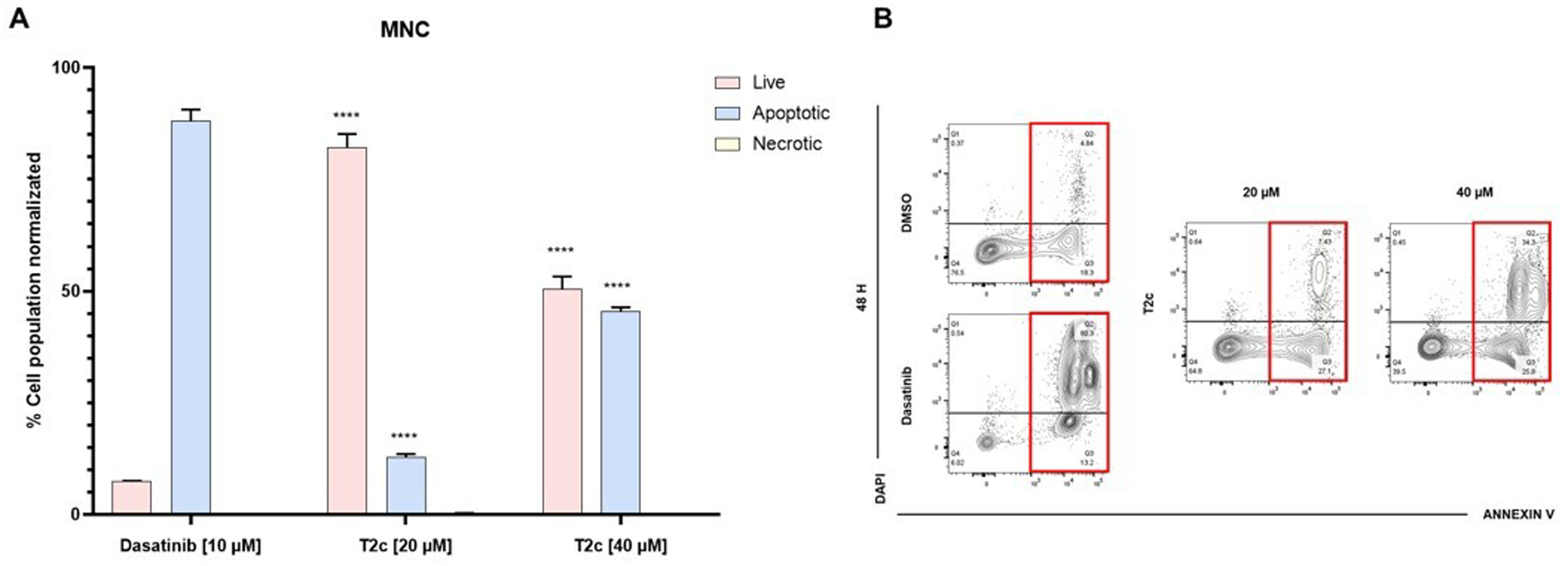
The safety of the compounds was measured by evaluating the effect on normal mononuclear cells (MNC). When compounds
Because
For the REH cell line (Figure 1), death by apoptosis was 93.29 ± 1.29% in the positive control, while for the active compounds at ½ IC50 were: 95.81 ± 2.17% (

Under the same conditions, the apoptotic rate for the CCRF-CEM cell line (Figure 2) was 94.84 ± 1.07% in the positive control. Apoptotic rates at ½ IC50 of the most active compounds were 82.50 ± 0.90% (

When evaluated on THP-1 cell line (Figure 3) the positive control showed 32.14 ± 3.15% of apoptosis. Of the evaluated compounds, only

For the MOLT-4 cell line (Figure 4), the apoptotic rate was 96.13 ± 0.71% for the positive control, while for

(A)
When tested against the JURKAT cell line (Figure 5) the positive control showed 97.10 ± 0.11% apoptosis. Of the evaluated compounds at ½ IC50,

Under the same conditions, only the effect of
Based on the above results, it was decided to perform a molecular docking analysis using key molecular targets involved in apoptotic processes. Among these are members of the BCL-2 family, which are related to the control and regulation of apoptotic mitochondrial events (eg, the anti-apoptotic protein BCL-2); these proteins block apoptosis through the intrinsic pathway and are found at elevated levels in cancers, especially blood cancers, thus representing an important molecular target.40–42 On the other hand, the vascular endothelial growth factor receptor epidermal (EGFR) is also a relevant target. In leukemia, EGFR is deregulated, which plays an important role as it is involved in key signaling pathways responsible for cell survival and proliferation. In addition, it has downstream signaling pathways associated with the BCL-2 family. As a result, EGFR kinase activity is a promising therapeutic target in the treatment of leukemia.43–45
For molecular docking analyses, the crystal structure of BCL-2 was used in complex with a small molecule inhibitor targeting the interaction of the BH3 domain of BCL-2 (Figure 7) and the EGFR tyrosine kinase domain with erlotinib as inhibitor (Figure 8). The derivatives (

Binding modes of

Binding modes of
Docking Score for Compounds

As a complementary study, we submitted the molecular structures of the six compounds to in silico analysis via the SwissADME platform to predict their physicochemical and pharmacokinetic properties (Table 3). 35
In Silico Predicted Physicochemical and Pharmacokinetic Parameters of Compounds

nHBA: num. H-bond acceptor; nHBD: num. H-bond donors; cLogP: lipophilicity (consensus Log Po/w); nROTB: num. rotatablebonds; TPSA: topological polar surface area; GI absorption: gastrointestinal absorption; CYP1A2: cytochrome P450 1A2; CYP2C19: cytochrome P450 2C19; CYP2C9: cytochrome P450 2C9; CYP2D6: cytochrome P450 2D6; CYP3A4: cytochrome P450 3A4.
Lipinski's rule of six for compound selection was applied as an initial approach. Only compounds
Discussion
Natural products and molecules based on their structures have aroused great interest in cancer research and therapy. 7 Triterpenes, compounds isolated from natural sources, present diverse biological activities, being of great interest in cancer research due to their antineoplastic properties. 13 In addition, they represent a key scaffold to generate new derivatives with greater biological activity, achieving a wide structural diversity through chemical modifications in their C-3, C-17, C-20, and C-28 positions, which can enhance their effects on cancer cells.14,47
In this context, to improve the biological activity of the lupane-type triterpene
During the process of structural modification of compound
Importantly, this molecule is similar to the compound 3α,23-dihydroxy-30-oxolup-20(29)-en-28-oic acid (
The α,β-unsaturated carbonyl groups, such as aldehydes or ketones, induce a cytotoxic effect through the formation of Michael-type adducts, interacting with thiols in proteins and peptides, which impacts cancer progression and drug resistance.4,48 In addition, these compounds can induce apoptosis through the mitochondrial pathway, a key cell death pathway and a promising therapeutic target in cancer treatment. 48
There are numerous reports on the anticancer activity of lupane-type triterpenes.49–51 Triterpenes, such as betulinic acid, betulin, lupeol and 23-hydroxybetulinic acid, show anticancer activity by inhibiting the proliferation of several cell lines, including leukemic ones.24,52 Their main mechanism of action is the induction of apoptosis, through loss of mitochondrial membrane potential, 53 DNA fragmentation, 53 release of proapoptotic proteins such as Bax, cytochrome c and Smac,53,54 activation of caspases, production of reactive oxygen species and glutathione depletion.55,56 In addition, these compounds have been found to arrest the cell cycle in the G1/S phase. 30
The selective inhibitory activity of betulinic acid is of interest for the study of other lupane-skeleton triterpenes, as well as to determine their mode of action. In a previous study, structural modification of the triterpene 3α,24-dihydroxylup-20(29)-en-28-oic acid by the introduction of a methyl group at C-3 generated the derivative 3α-methoxy-24-hydroxylup-20(29)-en-28-oic acid with enhanced antileukemic activity in K562 and HL60 cell lines. 31 In addition, this derivative was found to promote apoptosis by generating ROS and altering mitochondrial membrane potential. Molecular docking analyses revealed that compound 3α-methoxy-24-hydroxylup-20(29)-en-28-oic acid has an affinity against BCL-2 and EGFR proteins. 31
Due to the reports described on lupane-type triterpenes and the possible antileukemic potential of
In the present results, it was observed that compounds
On the other hand, betulin at IC50 has a mild apoptotic effect on REH (16.48 ± 1.52%) and THP-1 (39.66 ± 2.46%) cells. However, it is documented to have a low bioavailability, which prevents it from producing the desired therapeutic effect under in vivo conditions. Therefore, the introduction of a phosphate group can improve various properties such as bioavailability and solubility, as well as enhanced biological activity. 55
Moreover, pentacyclic triterpenes, such as betulin and betulinic acid, have been shown to inhibit proliferation in leukemic cells and to have effects on the BCL-2 family of proteins, which are involved in the control and regulation of mitochondrial apoptotic events, suggests the usefulness of molecular docking analyses on the BCL-2 protein using
In addition, EGFR was selected because it is considered a promising therapeutic target. It plays an essential role in cell survival and proliferation and has a downstream signaling pathway associated with the BCL-2 family, and it has been shown that it is dysregulated in leukemia.43–45 In the present results, the
Another derivative that appears interesting for further studies is
As can be seen from the results, compounds
high gastrointestinal absorption, a key goal in drug discovery.35,46 The low solubility of
These results indicate that
Limitations of Study
This research allowed us to determine which structural modification enhances the biological activity, the effect on apoptosis, and the possible target observed in the molecular docking analysis. In this study we chose two proteins overexpressed in leukemia cell lines and involved in apoptosis, EFGR and BCL-2. Nevertheless, it is possible that in the future a larger quantity of the active compound (
On the other hand, in the molecular docking analysis, the ligand as well as the protein were prepared following standard AutoDock Vina conditions, without an explicit assignment of protonation states at pH 7.4. However, the visualization in ChimeraX may not accurately reflect the actual protonation used in docking.
Conclusion
The triterpenes
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251328289 - Supplemental material for Semi-Synthetic Derivatives from a Lupane-Type Triterpene Isolated from Phoradendron wattii with Anti-Leukemia Activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X251328289 for Semi-Synthetic Derivatives from a Lupane-Type Triterpene Isolated from Phoradendron wattii with Anti-Leukemia Activity by Mario J. Noh-Burgos, Sergio García-Sánchez, Fernando J. Tun-Rosado, Antonieta Chávez-González, Rosa E. Moo-Puc, Sergio R. Peraza-Sánchez and Carlos J. Quintal-Novelo in Natural Product Communications
Footnotes
Acknowledgments
R.E.M.-P. thanks the IMSS Foundation, A.C., for the facilities used at Centro de Investigación en Salud Jesús Kumate Rodríguez. Additional thanks to María Antonieta Chávez González for the donation of leukemia cells (CCRF-CEM, JURKAT, MOLT-4, REH, and THP-1) and for the facilities used at Laboratorio de Células Troncales Tumorales, Unidad de Investigación Medica en Enfermedades Oncológicas, UMAE Hosptital de Oncología, CMN Siglo XXI, IMSS. Thanks also to M.Sc. Luis W. Torres-Tapia for his guidance in the Natural Products Chemistry Laboratory of the Biotechnology Unit at CICY.
Author Contributions
Conceptualization, R.E.M.-P. and A.C.-G.; methodology, M.J.N.-B., S.G.-S.; formal analysis, M.J.N.-B. and R.E.M.-P.; investigation, M.J.N.-B., C.Q-N., S.G.-S., F.J.T.-R., A.C.-G., S.R.P.-S., and R.E.M.-P.; resources, R.E.M.-P.; data curation, M.J.N.-B., S.G.-S., and F.J.T.-R.; writing: original draft preparation, M.J.N.-B.; writing: review and editing, M.J.N.-B., C.Q-N., S.G.-S., F.J.T.-R., A.C.-G., S.R.P.-S., and R.E.M.-P.; visualization, M.J.N.-B. and R.E.M.-P.; supervision, C.Q-N., S.G.-S., and A.C.-G.; project administration, R.E.M.-P.; funding acquisition, R.E.M.-P. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Materials
The data used to support the findings of this study are included within the article and are available from the first author upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This article does not contain any studies with human or animal subjects. In the case of MNC, these were collected according to institutional guidelines, including written informed consent from each donor. All procedures were approved by Ethics and Scientific Committee of the Mexican Institute of Social Security (IMSS), ethics approval number CNIC R-2021-785-025.
Funding
This work was supported by the Consejo Nacional de Humanidades Ciencias y Tecnologías de Mexico (CONAHCYT) under grant A1-S-10616 CB 2017–2018; M.J.N.-B. received a scholarship from CONAHCYT (application number 774184); R.E.M.-P. thanks Fundación IMSS, A.C., for providing facilities at the Centro de Investigación en Salud Dr. Jesús Kumate Rodríguez.
ORCID iDs
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
This article does not contain any studies with human or animal subjects. In the case of MNC, these were collected according to institutional guidelines, including written informed consent from each donor. All procedures were approved by Ethics and Scientific Committee of the Mexican Institute of Social Security (IMSS), ethics approval number CNIC R-2021-785-025.
Supplemental Material
Figures S1-S2. Nuclear magnetic resonance spectra of compound T2a [Figure S1. 1H-NMR (400 MHz, CDCl3), Figure S2. 13C-NMR (100 MHz, CDCl3)]. Figures S3-S4. Nuclear magnetic resonance spectra of compound T2b [Figure S3. 1H-NMR (400 MHz, CDCl3), Figure S4. 13C-NMR (100 MHz, CDCl3)]. Figures S5-S6. Nuclear magnetic resonance spectra of compound T2c [Figure S5. 1H-NMR (400 MHz, CDCl3), Figure S6. 13C-NMR (100 MHz, CDCl3)]. Figures S7-S8. Nuclear magnetic resonance spectra of compound T2d [Figure S7. 1H-NMR (400 MHz, CDCl3), Figure S8. 13C-NMR (100 MHz, CDCl3)]. Figures S9-S10. Nuclear magnetic resonance spectra of compound T2e [Figure S9. 1H-NMR (400 MHz, CDCl3), Figure S10.13C-NMR (100 MHz, CDCl3)]. Figures S11-S12. Nuclear magnetic resonance spectra of compound T2f [Figure S11. 1H-NMR (400 MHz, CDCl3), Figure S12. 13C-NMR (100 MHz, CDCl3)]. Supplemental material for this article is available online.
Trial Registration Number Data
Not applicable because this article does not contain any clinical trials.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
