Abstract
Background
Diabetes mellitus is marked with hyperglycemia due to insulin impairment and could be managed with α-glucosidase inhibitors, known to have side effects. Therefore, this study profiled the in vitro anti-diabetic activity of Xylopia aethiopica seeds, isolated and characterized its compounds, and in silico profiled these ligands against α-amylase and α-glucosidase to ascertain their benefit-to-risk ratio.
Methods
The extraction, isolation, characterization, evaluation of anti-diabetic activity, investigation of the extract's anti-proliferative activity against the breast cancer cell line (MCF7), retrieval of α-amylase and α-glucosidase, ADMET, and docking studies were done according to standard protocols; eluents were isolated and characterized using thin layer and column chromatography, and GC-MS; pharmacokinetic properties were evaluated with the help of PKCSM and ADMET Lab 2 tools; docking analysis was performed using PyRx; Biovia Discovery Studio for 2D visualization; and PyMol software for 3D visualization of the ligand-protein interactions.
Results
After 72 h, the extract's cytotoxic value against the MCF-7 cell line was 57.03 μg/mL. Additionally, the extract (0.15) and (1.13) had the best inhibitory activity (IC50) (µg/ml) of α-amylase and α-glucosidase in a dose-dependent manner when compared favorably with acarbose (0.12) and (1.09), respectively. The chromatographic analyses yielded bulked fractions (A-D); fraction C (2135 mg) was further semi-purified to sub-fractions C2 (AS3) (17 mg), C4 (AS8) (21 mg), and C7 (AS11) (26 mg), and yielded xanthumin, metolachlor, Z-10-tetradecen-1-ol acetate, 13-docosenoic acid methyl ester, and alpha-bisabolol with reported anti-diabetic activities after GC-MS analysis. Docking with 7K9N and 3BC9 revealed the binding affinities (kcal/mol) of 1-Chlorotetradecane (−5.8) as well as 1-Chlorotetradecane (−5.2); 1-(4-Bromobutyl)-2-piperidinone (5.0); and 11,12-Dibromo-tetradecan-1-ol acetate (5.0) as the best when compared with metformin (−4.4, −3.7), respectively. All the compounds obeyed Lipinski's criteria, showed inactivity to hepatotoxicity, carcinogenicity, and immunotoxicity, and were non-inhibitors of mono-oxygenases.
Conclusion
These ligands’ better antineoplastic potency, pharmacokinetic, binding affinities, physicochemical, and drug-like properties as compared to metformin and acarbose drugs imply they could control hyperglycemia with a higher benefit-to-risk ratio; hence, their potential as lead compounds for the production of anti-diabetic drugs.
This is a visual representation of the abstract.
Background
Hyperglycemia and accompanying symptoms like polyuria, weight loss, and blurred eyesight are hallmarks of diabetes mellitus, a metabolic disorder.1,2 It comes in two varieties: Type 1 (driven by an autoimmune response that destroys the β-islets of Langerhans) and Type 2 (driven by insulin resistance and hyper-insulinemia).3,4 In 2021, out of 500 million adults living with diabetes mellitus worldwide, 5 about 5.8% lived in Nigeria 6 whereas the prevalence rate in Cross Rivers State, Nigeria, was 4.1%. 7 The majority of oral anti-diabetic medication classes, including biguanides (metformin) and α-glucosidase inhibitors (acarbose), have a high benefit-to-risk ratio, but they still have a lot of side effects. These medications improve the effective management of diabetes mellitus by inhibiting the enzymes that metabolize carbohydrates and controlling hyperglycemia. α-glucosidase and α-amylase inhibitors are known to reduce and stop the breakdown of carbohydrates, which slows the absorption of glucose and aids in the control of postprandial blood glucose levels.8-11
However, to avert the numerous side effects that emanate from the conventional oral anti-diabetic drug administration, there is a need to patronize traditional medicinal approaches that entail the use of medicinal plants such as Xylopia aethiopica seeds. The plant (Annonaceae), otherwise called guinea pepper and African pepper in English, is commonly known as Uda in Igbo as well as Ata in Ibibio and Efik languages. 12 Its ethnopharmacological and scientific uses include the treatment of diabetes mellitus, stomach ache, bronchitis, and dysentery. 13 It is worthy of note that the numerous phytochemicals in the plant's seed are believed to either mediate their anti-diabetic activity through the inhibition of carbohydrate-metabolizing enzymes, enhancement of insulin sensitivity, or reduction of oxidative stress. 14
Nevertheless, some of the challenges in the patronage of ethnobotanicals are the limited information on the plants’ chemical constituents as well as the technical know-how in the isolation and characterization of its bioactive compounds through chromatographic and spectroscopic techniques. In addition, evidence abounds on the prediction of the various mechanisms of actions of compounds through both in vivo and in silico studies, thereby enhancing the understanding of the pathogenesis of diseases and their management.15,16 One of the computational models that has enhanced the prediction of the mechanism of action of compounds and ligand-protein interactions, as well as the discovery and development of new chemotherapies, is molecular docking.
Since the aim of ligand-protein docking is to anticipate the predominant binding mode of a ligand with a protein of known three-dimensional structure, molecular docking is a computational method utilized in structural molecular biology and computer-assisted drug design.13,16 More so, it is used to predict how small molecules bind to target proteins.16,17 Therefore, in order to treat diabetes mellitus, it is essential to comprehend the molecular interactions between possible medications and target proteins. Hence, this study was designed to extract, isolate, and characterize bioactive compounds from X. aethiopica seeds, validate the extract's in vitro anti-diabetic activity, evaluate the anti-proliferative activity of the extract against MCF-7 human breast cancer cell lines for cytotoxicity assays, as well as dock these ligands against some anti-diabetic target proteins implicated in diabetes mellitus pathogenesis.
Materials and Methods
Collection and Preparation of Xylopia aethiopica Seeds
A taxonomist named Dr Michael Eteng of the Department of Botany at the University of Calabar in Cross River State, Nigeria, identified and verified the X. aethiopica seeds that were acquired from the botanical farm of Arthur Jarvis University. The voucher specimen UCB/S/0269 was then placed in their herbarium.
Sample Processing, Extraction, and Isolation Protocol
To lower the moisture content, X. aethiopica seeds were rinsed under running water and allowed to dry in the shade for two weeks. An electric blender was then used to grind the dried materials into a coarse consistency. After macerating the coarse material (703.24 g) in 70% ethanol (12 L) for 72 h while shaking occasionally, sieve cloth and filter paper were used for filtration. After disposing of the waste, the filtrate was dried out in a rotary evaporator set at 40 °C to produce 138.8 g of ethanol extract, which was then employed in an in vitro anti-diabetic test. After the validation of the anti-diabetic activity, the extract (40 g) was subjected to isolation process through column chromatography (CC), and the appropriate solvent systems: n-hexane (300 ml), dichloromethane (700 ml), ethyl acetate (2.4 L), and methanol (200 ml) were used to elute the various fractions. This procedure produced 208 fractions, and each of these fractions passed through two distinct TLC analysis phases (the pooling process and then the bulking process). Subsequently, nineteen (19) pooled and four (4) bulked fractions (A–D) were produced based on similarity in their Rf values. Following CC using silica gel, bulked fraction C (2135 mg) was eluted using the appropriate solvent mixtures to yield 38 pooled sub-fractions and three (3) bulked sub-fractions C2 (AS3) (17 mg), C4 (AS8) (21 mg), and C7 (AS11) (26 mg) after their subjection to TLC analysis. Additionally, thin-layer chromatography was performed using analytically pre-coated TLC silica gel 60 from Sigma-Aldrich. The TLC plates were then examined under UV light with both long (366 nm) and short (254 nm) wavelengths. Furthermore, Sephadex LH-20 from Sigma-Aldrich and the silica gel Kiesel gel 60 (200-400 mesh, Merck) enhanced column chromatography (CC) analysis.
The Evaluation of the Cytotoxic Activities of X. aethiopica Seeds on Cancer Cell Lines
The American Type Culture Collection (ATCC, Manassas, VA, USA) provided the human breast cancer (MCF-7) cell lines used for cytotoxic activity. In a humidified environment at 37 °C and 5% CO2, they were kept in Dulbecco's Modified Eagle Medium (DMEM) F12 (Life Technologies GIBCO, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (FBS) (Sigma-Aldrich St. Louis, MO), glutamine, phenol red, and 50 μg/mL gentamycin. The extract's impact on the growth of MCF-7 human breast cancer cells was assessed using the MTT technique for the cytotoxic investigation. 18 These cells were treated with the medication after being seeded at a density of 5 × 104 cells in 96-well plates. The cells were rinsed with PBS after 24, 48, and 72 h. Each well was then filled with 0.2 mL of growth media and 50 μL of MTT reagent (2 mg/mL). After four more hours of growth, when a purple precipitate was evident, the medium was taken out and 150 μL of dimethyl sulfoxide was added. To completely liberate the crystals, the wells were shaken for ten minutes. A microplate reader was used to detect cell viability at 570 nm, and each concentration was examined in triplicate. The assay found that mitochondrial dehydrogenase reduced MTT [3-(4,5-dimethylthiazolyl)-2,5-diphenyl-tetrazolium bromide] to a blue formazan result, which represented proper mitochondrial and cell viability function. 19 The value of cell cytotoxicity at 50% (IC50) in the MTT experiment was defined as the quantity of the extract that resulted in a 50% reduction of absorbance compared with untreated cells. The percent inhibition of MCF-7 cells was computed using optical density.20,21
Enzyme Inhibition Assay
The extract's ability to inhibit the enzymes that break down carbohydrates was evaluated against α-amylase and α-glucosidase. The amylase assay's principle was a direct method that measured the amount of starch that was converted to glucose; the substrate was soluble starch; the dinitrosalicyclic acid (DNSA) method was used to improve detection; the experiments were conducted in triplicate on 96-well polystyrene plates; absorbance was measured at 540 nm using an ELISA plate reader; ideal conditions for the assay were pH 6–7 at 37 °C; and the percentage inhibitions were computed as follows: inhibition (%) = (Ac - At)×100/Ac, where Ac is the absorbance of the control and At is the absorbance in the presence of the tested extract. 22 Additionally, the glucosidase assay's principle was to detect the amount of glucose released from specific substrates—the substrate was p-nitrophenyl β-D-glucopyranoside (pNPG), which produced p-nitrophenol; detection was colorimetrically enhanced by measuring nitrophenol at 405 nm wavelength using an ELISA plate reader; the experiments were conducted in triplicate on 96-well polystyrene plates; the assay's ideal conditions were pH 4–7 at 37 °C; the percentage inhibitions were computed as follows: inhibition (%) = (Ac×100/Ac. 23
Alpha-Amylase Activity
The extract's ability to inhibit α-amylase activity was assessed using a modified version of the approach developed by certain researchers. 22 0.2 ml of 2 units/ml α-amylase solution, extract (20-100 μg/ml), and 10 min of incubation at 30 °C made up the reaction mixture. A 0.2 ml solution of 1% starch in 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M NaCl) was then added, and the mixture was incubated for five minutes. 1.0 ml of 3.5-dinitrosalicylic acid (DNSA) was used as a colorimetric indicator to stop the reaction (1 g of DNSA in 50 ml of reagent-grade water, 30 g of sodium potassium tartrate tetrahydrate in 20 ml of 2 M NaOH). The reaction was then heated for 10 min at 90 °C in a water bath. After cooling and diluting the mixture with five milliliters of distilled water, the absorbance at 540 nm was measured. The control sample was considered to have 100% enzyme activity since it contained saline buffer rather than X. aethiopica seed extract.
Alpha-Glucosidase Activity
The approach was used to determine the extract's inhibitory effect on α-glucosidase activity. 23 In particular, for 15 min, Eppendorf tubes with 50 μl of 10 mM phosphate buffer (pH 6.8), 10 μl of α-glucosidase solution (0.5 units/ml) extracted from rat intestinal acetone powder (Sigma-Aldrich), and 20 μl of X. aethiopica seed extract at varying concentrations (20-100 μg/ml) were incubated at 37 °C. Following the addition of 20 μl of 2.5 mM 4-nitrophenyl-β-d glucopyranoside, the reaction mixture was incubated for an additional 20 min at 37 °C. 50 μl of 0.1 M sodium carbonate was added to the reaction mixture following incubation. In the control tests, an equivalent volume of buffer was used in place of the ethanol extract. The absorbance at 405 nm was used to determine the extract's % inhibitory action on α-glucosidase activity.
Ethical Approval
The approval for this study was duly given by Arthur Jarvis University's Research Committee (approval number: AJU/RC/0147), and compliance was judiciously monitored.
Gas Chromatography-Mass Spectrometry Analysis of the Sub-Fractions
The Agilent 7890, a gas chromatograph coupled to an Agilent MS model 5975C MSD detector (Agilent Technologies, USA), was used to record the gas chromatography-mass spectrometry (GC-MS) data of the sub-fractions C2 (AS3), C4 (AS8), and C7 (AS11) (each 10 mg). A 30 m × 0.25 mm × 0.25 µm HP5-MS column containing 5% phenyl-methylpolysiloxane was employed with a helium gas flow at 10 psi. The injector temperature was set at 280 °C, and the oven temperature was raised to 300 °C at a rate of 10 °C per minute for five minutes after beginning at 150 °C for three minutes. The sub-fractions were immediately injected in n-hexane to create TMSi derivatives using N,O-bis(trimethylsilyl) trifluoroacetamide (BSTFA) as explained, and the mass spectrometer was run in the electron ionization mode at 70 eV.24,25 The compounds were identified by comparing the retention durations and mass spectra fragmentation patterns of the isolates using the ChemStation data system and the National Institute of Standards and Technology (NIST) database. Retention times, retention indices, area sum percentage, peak area percentage, and compound names were all noted.
The Assessment of the Physicochemical and Pharmacokinetic Characteristics of the Bioactive Compounds
Prediction of Target and ADMET Properties of the Ligands
The absorption, distribution, metabolism, excretion, and toxicity (ADMET) characteristics of the reference drug and the ligands were predicted using their various canonical strings or Simplified Molecular-Input Line-Entry System (SMILES) obtained from the PubChem web platform (https://www.ncbi.nlm.nih.gov/pccompound) in 3D conformation using the online programs ADMET Lab 2.0 and PKCSM. 26 The Ghose profiles of the five chosen ligands and Lipinski's rule were among the pertinent metrics that were noted. Additionally, the SWISS target prediction tool improved the identification of the various ligands’ targets. 27
Target Proteins’ Properties, Selection, and Molecular Docking
The study used the protein data bank (PDB) https://doi.org/10.2210/pdb7K9N/pdb and https://doi.org/10.2210/pdb3BC9/pdb to obtain the target proteins, α-glucosidase (7K9N) and α-amylase (3BC9), respectively. 28 The protein-ligand interactions were determined through the use of Auto Dock Vina 4.2, while Biovia Discovery Studio software was used for the visualization of their interactions. The proteins were prepared for interaction by the elimination of water molecules, heteroatoms, and ligands. Following their production, they were docked against the different ligands, with metformin serving as a reference.
Statistical Analysis
Data collected were analyzed using one-way analysis of variance (ANOVA) followed by Tukey's multiple comparison post-test by using GraphPad Prism software, Inc., La Jolla, CA, USA.
Results
In vitro α-Amylase and α-Glucosidase Activities of X. aethiopica Seed Extract
The results (Figure 1) showed the α-amylase and α-glucosidase activities following assay with X. aethiopica seed extract. Moreover, the inhibitory activity IC50 (inhibitory concentration 50%) of α-amylase and α-glucosidase by the extract (0.15) and (1.13) in a dose-dependent manner compared favorably with acarbose (0.12) and (1.09), respectively.
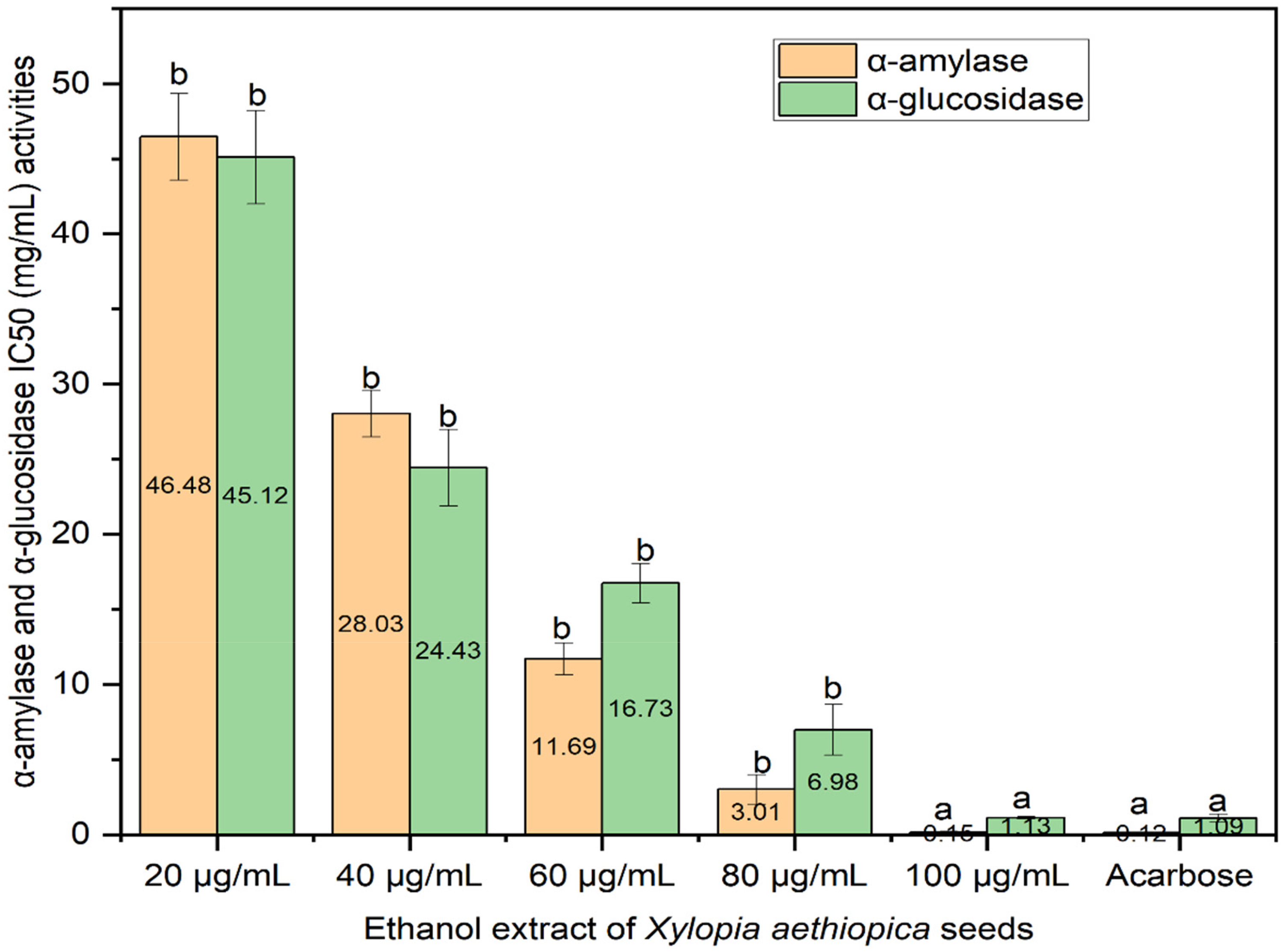
The inhibitory effect of X. aethiopica seeds on α-amylase and α-glucosidase in comparison with acarbose.
Cytotoxicity Assay of X. aethiopica Seeds
The results demonstrated that the proliferation of MCF-7 human breast cancer cells was significantly inhibited in a concentration-dependent manner. Moreover, as the treatment time increased, the cell viability gradually decreased, indicating a time-dependent effect (Figure 2). The IC50 values of ethanol extract for both 48 and 72 h were 75.42 ± 0.7 μg/mL and 57.03 ± 0.7 μg/mL. In addition, the experimental results showed that the proliferative activity of MCF-7 human breast cancer cells was not significantly affected by the extract in the first 24 h in the concentration range of 10–100 μg/mL (Figure 2). After 72 h of treatment, the viability of the cells could have decreased due to the senescence stage, continuous depletion of nutrients, and accumulation of metabolic wastes.
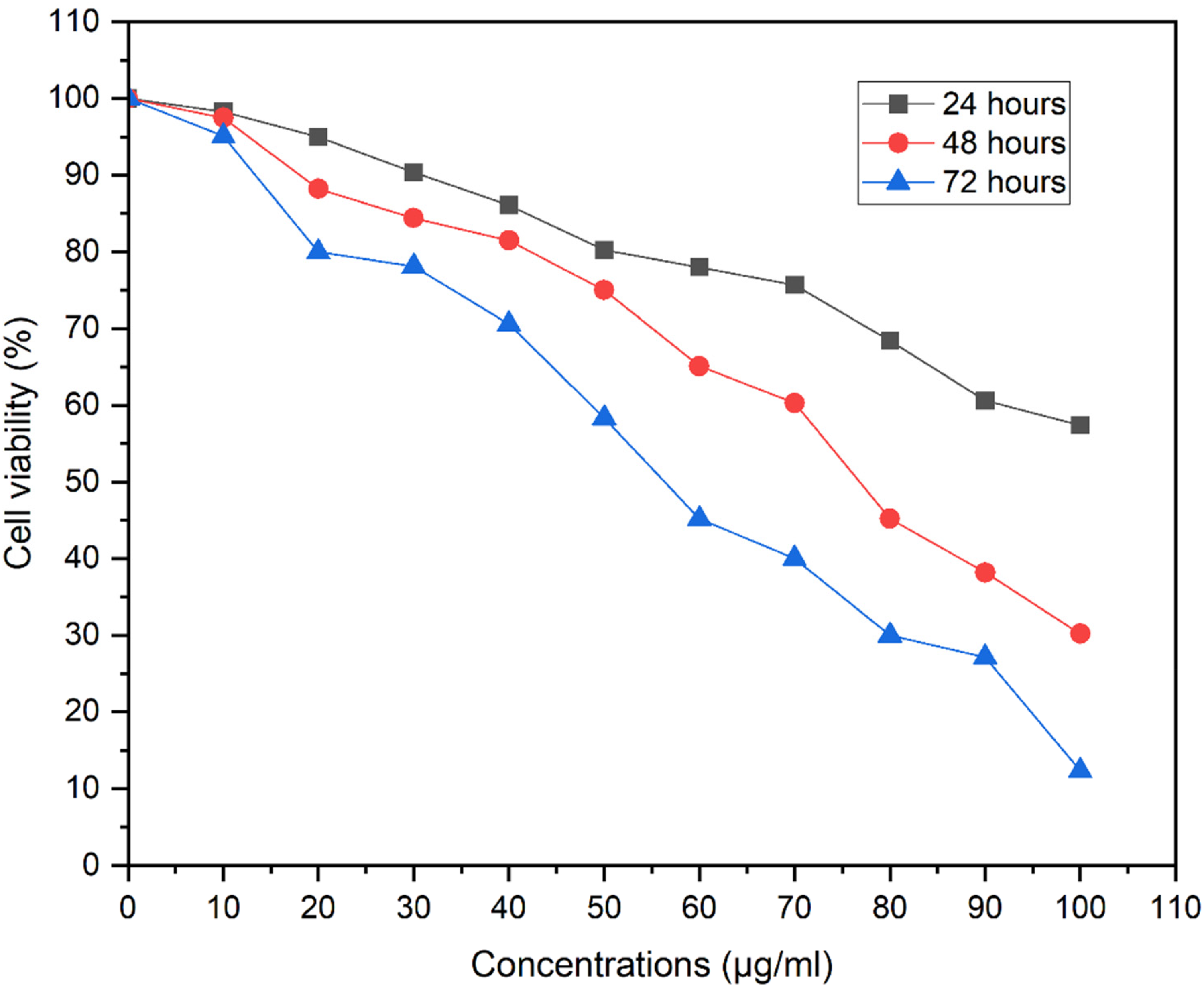
Cytotoxicity MTT assay of X. aethiopica seeds.
GC-MS Analysis of X. aethiopica Seeds
Gas chromatography-mass spectrometry analysis showed that twenty-five, fourteen, and ten chemical compounds (Tables 1–3 and Figures 3–5) were isolated and characterized from sub-fractions C2 (AS3), C4 (AS8), and C7 (AS11), respectively. The presence of the following bioactive compounds justified the ethnopharmacological usage of this plant seed for the treatment of different ailments. They include: chloroacetic acid, propyl ester, xanthumin, 9-oxabicyclo[6.1.0]nonane, and N-[4-bromo-n-buty]-N-[3-[N-2-piperidinone, metolachlor, Z-10-tetradecen-1-ol acetate, oxalic acid, allyl hexadecyl ester, 13-docosenoic acid methyl ester, alpha-bisabolol, 11,12-dibromo-tetradecan-1-ol acetate, etc On the basis of their peak area percentages, the compounds were categorized into four groups, namely, the highly abundant (>8%), moderately abundant (3 to 7.99%), less abundant (1 to 2.99%), and the least abundant or minor compounds (<1%).

Chromatogram for AS3.

Chromatogram for AS8.

Chromatogram for AS11.
GC-MS Analysis of Sub-Fraction C2 (AS3).

GC-MS Analysis of Sub-Fraction C4 (AS8).

GC-MS Analysis of Sub-Fraction C7 (AS11).

Molecular Docking of Ligands
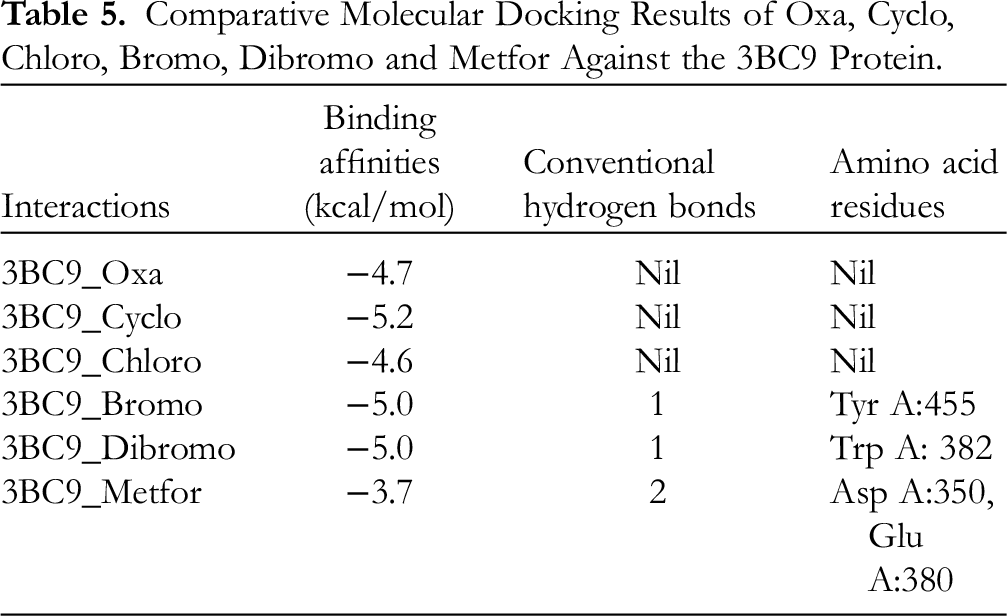
The results (Tables 4 and 5) showed the binding affinities from the docking analysis of oxalic acid, allyl hexadecyl ester (Oxa); 1-(4-bromobutyl)-2-piperidinone (Bromo); 11,12-dibromo-tetradecan-1ol acetate (Dibromo); cyclopentane, (4-octyldodecyl)- (Cyclo); and 1-chlorotetradecane (Chloro) ligands with 7K9N and 3BC9 proteins, respectively. For the docking of the above ligands with 7K9N (Figure 6), 4-octyldodecyl-cyclopentane (−5.8) revealed the best binding affinity interaction (kcal/mol) that was better than standard drug, metformin (4.4) through interactions with the following amino acids (Thr 407 and Arg 632) sequences at the α-glucosidase active site. Also, the binding affinities (kcal/mol), conventional hydrogen bonds, and the interacting amino acids of the five ligands with the 7K9N receptor are shown in Table 4. Moreover, all the ligands’ interactions (Figure 3 and Table 5) at the active site of the α-amylase were enhanced through the formation of hydrogen bonds, hydrophobic interactions, and electrostatic interactions with Thr 470, Arg 632, and Thr 407 at the active sites of the protein. In addition, 4-octyldodecyl-cyclopentane (−5.2) still revealed the best interaction with a binding affinity (kcal/mol) that was better than standard drug, metformin (−3.7).
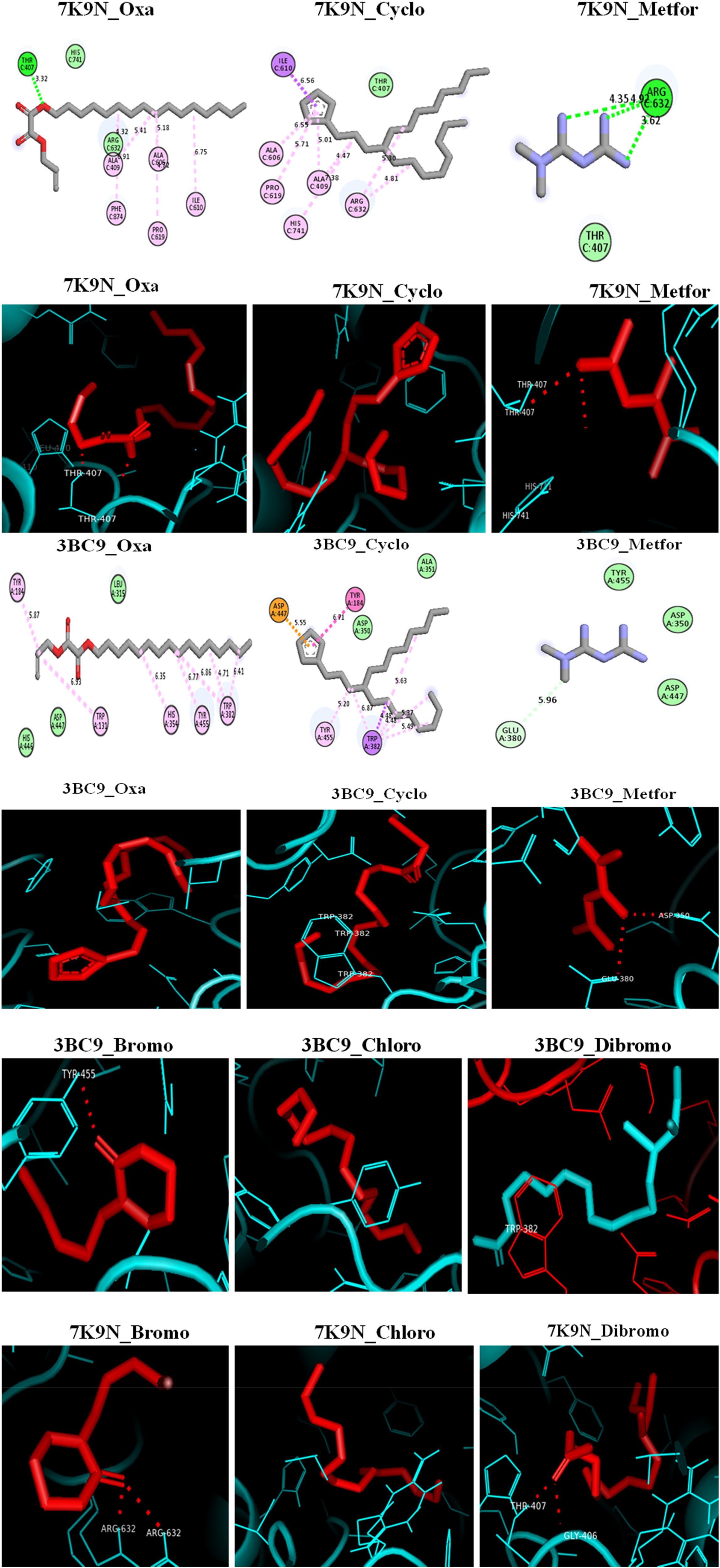
2D and 3D visualization of ligands’ interactions with 7K9N and 3BC9 receptors.
Comparative Molecular Docking Results of Oxa, Cyclo, Chloro, Bromo, Dibromo, and Metfor Against the 7K9N Protein.

Comparative Molecular Docking Results of Oxa, Cyclo, Chloro, Bromo, Dibromo and Metfor Against the 3BC9 Protein.
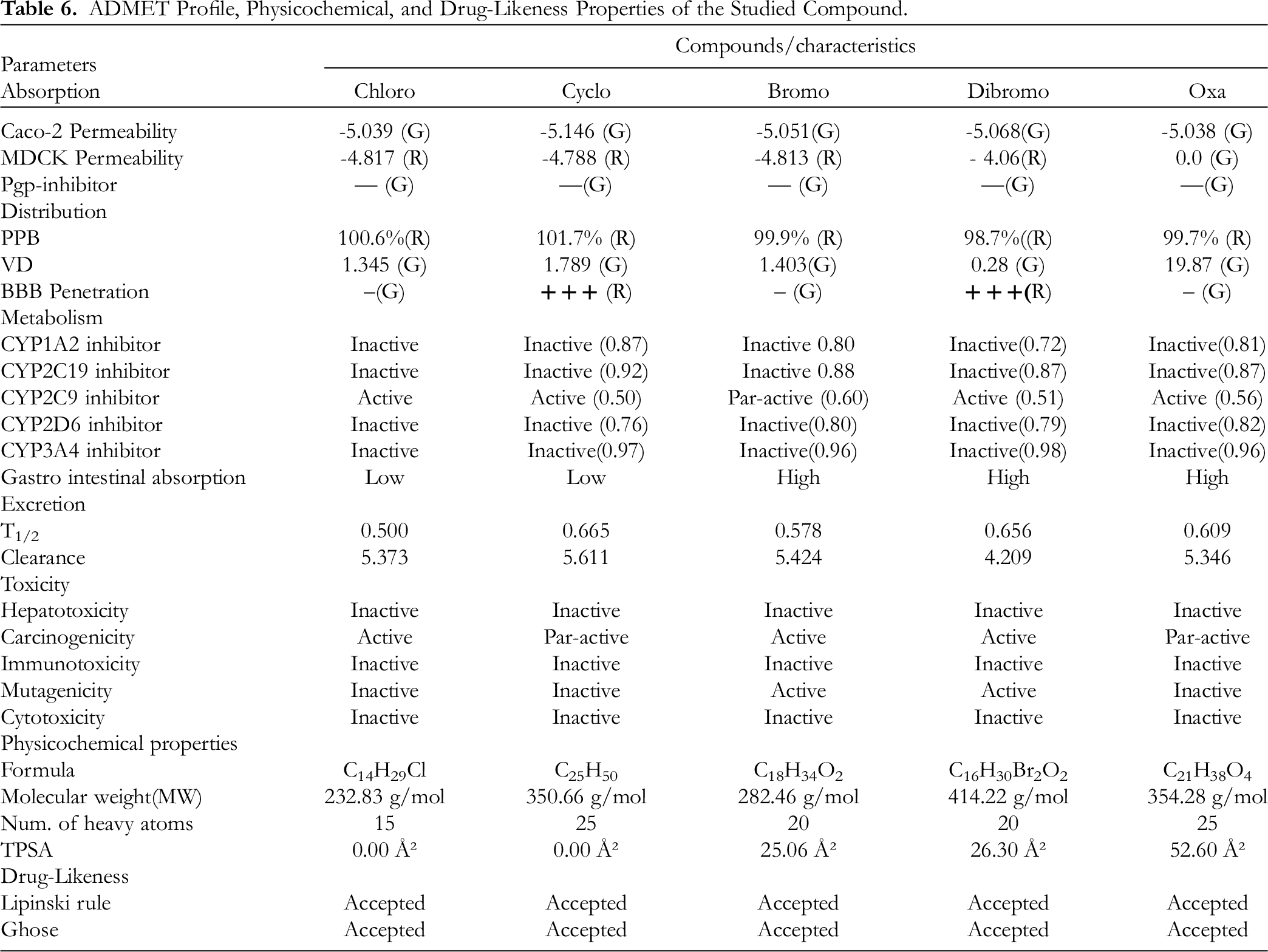
The Investigated Ligands’ ADMET Profile, Physicochemical Characteristics, and Drug-Likeness
This essentially separates compounds according to their drug-like and non-drug-like properties. Lipinski's rule of five qualifies the drug-like characteristics of compounds based on molecular mass (<500 Dalton), molar refractive index (40-130), partition coefficient (Log P ≤ 5), hydrogen bond donors (≤5), and hydrogen bond acceptors (≤10). All five of the ligands that were chosen for this study met Lipinski's Ro5 requirements for being potential therapeutic candidates (Table 6). The selected ligands were subsequently examined by PKCSM for their ADMET (absorption, distribution, metabolism, excretion, and toxicity) profile. The result (Table 6) revealed that 1-(4-bromobutyl)-2-piperidinone, 11,12-dibromo-tetradecan-1ol acetate, and 1-chlorotetradecane were possible carcinogens, while 4-octyldodecyl-cyclopentane and oxalic acid allyl hexadecyl ester were partial carcinogens. It also suggested that oxalic acid, allyl hexadecyl ester, 4-octyldodecyl-cyclopentane, and 1-chlorotetradecane were non-mutagens, whereas 1-(4-bromobutyl)-2-piperidinone and 11,12-dibromo-tetradecan-1ol acetate revealed their capacities in the induction mutagenicity.
ADMET Profile, Physicochemical, and Drug-Likeness Properties of the Studied Compound.
Discussion
Diabetes mellitus as a metabolic disorder is life-threatening, characterized with hyperglycemia, and attendant generation of free radicals that could possibly induce neoplasm. 29 The result of the cytotoxicity assay revealed that X. aethiopica seeds possess anti-proliferative activity against human breast cancer MCF-7 cell lines. Therefore, the compounds in the extract could have activated the metabolizing enzymes (cytochrome P-450) that enhance biotransformation of carcinogens, induction of apoptosis, and subsequent mitigation of neoplasm through the prevention of non-interaction of carcinogens with DNA and cellular proteins. The observed result showed consistency with the reports20,21 on the X. aethiopica's anti-proliferative property.
Moreover, the control of the resultant postprandial hyperglycemia being a criteria in diabetes mellitus management necessitated this study on the discovery of anti-diabetic agents with a higher benefit-to-risk ratio because bioactive constituents from medicinal plants are very critical in the discovery and development of new therapeutic agents. 30 Despite the numerous folkloric histories on the use of ethnobotanicals in disease management, this ethnopharmacological claim still lacks reproducibility and scientific validations; therefore, this study has validated the anti-diabetic potentials of X. aethiopica seeds and revealed that the isolated and characterized compounds could inhibit the various carbohydrate hydrolyzing enzymes in a dose-dependent manner. The inhibition of α-amylase and α-glucosidase has been emphasized as a therapeutic and management approach for the control of hyperglycemia because it delays the absorption of glucose from the small intestinal tract's enterocytes.8,9,31 The hydrolysis of the linear α-(1.4)-glycosidic bonds of large insoluble carbohydrates into oligosaccharides and disaccharides is catalyzed by salivary α-amylase, while α-glucosidase further catabolizes starch and oligosaccharides. These are subsequently absorbed through the small intestines into the hepatic portal vein, raising the postprandial glucose level. 32 Also, acarbose, a common oral antidiabetic medication compared well with all concentrations of the ethanol extract of X. aethiopica seeds,and showed remarkable inhibitory potentials against α-amylase and α-glucosidase. Therefore, the inhibition of α-amylase and α-glucosidase helped and regulated hyperglycemia by delaying the breakdown of carbohydrates thereby, causing the lowering of postprandial plasma glucose level seen in diabetic circumstances. In terms of the inhibitory activity of cinnamon bark species and their combined effect with acarbose against intestinal α-glucosidase and α-amylase activities, this finding was consistent with report. 23
More so, the GC-MS analysis revealed that the following compounds were highly abundant: capsaicin N1-tetrahydrofuran-2-ylmethyl-2-(4-chlorobenzylidene) hydrazine-1-carbothioamide (13.42%); 13-docosenoic acid, methyl ester (12.67%); Alpha-bisabolol (12.21%); N-[4-bromo-n-butyl]-2-piperidinone (9.45%); and 4-octyldodecyl-cyclopentane (8.22%). 2-piperidinone ester had earlier been reported to possess antidiabetic activity 33 ; methyl esters have antidiabetic, anti-tumor, and analgesic phytocompounds 34 ; and octadecadienoic acid and its methyl esters possess anticancer, hepatoprotective, and anti-arthritic activities. 35 Also, the only moderately abundant compound, N-[4-bromo-n-butyl]-2-piperidinone (3.47%) had been reported to possess antimicrobial activities. 36 More so, the less abundant compounds were carboxylic acid, 3-methylbut-2-enyl ester, (2.85%); metolachlor (2.43%); 13-docosenoic acid, methyl ester (2.02%); oxalic acid, allyl hexadecyl ester (1.78%); N-[4-bromo-n-butyl]-2-piperidinone (1.65%); 1,2-dibromododecane (1.52%); N-[4-bromo-n-butyl]-2-piperidinone (1.05%). However, oxalic acid, allyl hexadecyl ester possesses acaricide properties and can be used to control ticks and mites. 37 Moreso, compounds such as chloroacetic acid (0.90%); xanthumin S-(Propoxythiocarbonyl) thiohydroxylamine (0.64%); isobutylamine-2-nonenoic acid (0.38%); ethyl ester xanthumin (0.34%); chloroacetic acid (0.24); and S-(Propoxythiocarbonyl) thiohydroxylamine xanthumin (0.13%) were ranked as the least abundant. Therefore, the chemical constituents in X. aethiopica seed reported in this study had a similar chemical profile with few quantitative variations 33 ; hence, the basis of its numerous biological activities.
Furthermore, in contrast to costly and time-consuming in vivo laboratory techniques, in silico screening of natural products during drug discovery and development has become essential since it reduces the probability of a therapeutic molecule failing at a later stage. 38 The chosen drugs in this investigation demonstrated minimal binding affinities that were superior to those of metformin, according to the docking analysis. According to Asanga et al, 39 a ligand's stability and accuracy of engagement with the target protein are indicated by a lower docking score or binding affinity. Thus, by inhibiting α-amylase and α-glucosidase activities, all of the chosen compounds actively docked the target proteins; nevertheless, when compared to metformin, 4-octyldodecyl-cyclopentane was the best ligand with anti-diabetic potential. This result was similar to the report 40 on oleanolic acid isolated from the fruit of X. aethiopica that bonded the α-amylase and α-glucosidase with minimum binding energy values.
Consequently, the subjection of the selected ligands through ADMET profiling revealed variations in their activities. On the basis of hepatotoxicity, immunotoxicity, and cytotoxicity induction, all the selected ligands revealed inactivity, implying that they could not induce toxicity in the liver, immune system, and cells, respectively. However, 1-(4-bromobutyl)-2-piperidinone; 11,12-dibromo-tetradecan-1-ol acetate; and 1-chlorotetradecane showed that they could induce carcinogenicity as they revealed their potentials as active carcinogens. On the possibility of mutagenicity induction, oxalic acid, allyl hexadecyl ester, 4-octyldodecyl-cyclopentane, and 1-chlorotetradecane showed that they are non-mutagens and therefore could not cause mutations, whereas 1-(4-bromobutyl)-2-piperidinone and 11,12-dibromo-tetradecan-1-ol acetate revealed potential mutagenic properties.
Conversely, CYP450 as a superfamily of heme-containing enzymes is known to enhance the biotransformation of drugs to fast-track their therapeutic indices and elimination to avert multi-organ toxicity. All the selected ligands in this study acted as non-inhibitors of all the monooxygenases with the exception of 4-octyldodecyl-cyclopentane that revealed itself to be partially active against CYP2C9; therefore, the ligands could easily be bio-transformed to active metabolites and eliminated in order to avert toxicity. More so, drug efficacy and safety are highly critical in the quest for lead compound identification and optimization during drug discovery and development.26,41 This validation is often done through the evaluation of the numerous pharmacokinetic properties during in silico analysis. 42 In this study, Lipinski's rule of five was obeyed by all the selected ligands, implying that they have drug-like properties and could further be subjected to lead optimization. Consequently, further evaluation on drug absorption and distribution revealed that the best route for the administration of the selected ligands, with the exception of 4-octyldodecyl-cyclopentane and 1-chlorotetradecane, was through GIT, implying that these compounds’ oral administration could enhance good bioavailability and volume distribution in the various interstitial tissues with limited first-pass effect. Therefore, the observed physicochemical, drug-like, binding affinities, and pharmacokinetic properties of these ligands showed that they are the lead compounds for lead optimization to unravel new antidiabetic drugs.
Conclusion
X. aethiopica seed ethanol extract significantly inhibited the primary carbohydrate hydrolyzing enzymes (α-amylase and α-glucosidase) in a dose-dependent manner more than acarbose, and it also had a strong cytotoxic effect on MCF-7 cell lines. Numerous chemicals, their retention times and indices, as well as the area total percentage, were discovered when this extract was further separated using CC and TLC and characterized using GC-MS. The isolated compounds had already been reported to possess anti-diabetic, antioxidant, anti-inflammatory, anti-microbial, and anticancer activities. Moreover, the ligand-receptor docking and simulation approach that was adopted in this study accelerated the discovery of the following anti-diabetic agents (lead compounds) in the plant seeds; they include: 1-(4-bromobutyl)-2-piperidinone; 11,12-dibromo-tetradecan-1-ol acetate; 1-chlorotetradecane; oxalic acid; allyl hexadecyl ester; and 4-octyldodecyl-cyclopentane. These lead compounds possessed remarkable anti-diabetic potentials based on the evaluation of their binding interactions and affinities, docking potentials, bond distances, amino acid residues, ADMET, and physicochemical profiling with α-amylase and α-glucosidase as compared with metformin. Hence, they are good candidates for lead optimization and further rational drug design processes for anti-diabetic drugs.
Strengths and Limitations
The strengths of this study include:
The ethanol extract of X. aethiopica seed was used to isolate and characterize 49 bioactive components. By inhibiting α-amylase and α-glucosidase, these compounds’ anti-diabetic properties were confirmed. Based on their pharmacokinetic and docking potentials, the following anti-diabetic drug candidates were identified: oxalic acid allyl hexadecyl ester (Oxa), 1-(4-bromobutyl)-2-piperidinone (Bromo), 11,12-dibromo-tetradecan-1-ol acetate (Dibromo), Cyclopentane 4-octyldodecyl (Cyclo), and 1-chlorotetradecane(Chloro). In silico nature: Molecular docking is a potent tool, but its predictions require experimental validation through in vitro and in vivo research. Limited financial resources: The availability of cutting-edge technologies and instrumentation that may have improved the scope and depth of the sudy was a serious challenge.
Among the limitations were:
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251325717 - Supplemental material for Exploring Anti-diabetic Activity of Xylopia aethiopica Compounds as Potential Inhibitors of α-Amylase and α-Glucosidase: Experimental and Computational Approach
Supplemental material, sj-docx-1-npx-10.1177_1934578X251325717 for Exploring Anti-diabetic Activity of Xylopia aethiopica Compounds as Potential Inhibitors of α-Amylase and α-Glucosidase: Experimental and Computational Approach by Edet Effiong Asanga, Chinedum Martins Ekeleme, Imaobong Etima Udoh, Aniekeme Ndisa Inyang, Paul Sunday Thomas, Akaninyene Paul Joseph, Deborah Kenneth Ani, Favour Nseabasi Anwana, Onyekachi Emmanuel Kanu and Jude Efiom Okokon in Natural Product Communications
Footnotes
List of Abbreviations
Acknowledgements
The authors are grateful to members of staff in NIPRD, Abuja, Nigeria, for carrying out GC-MS analysis and other technical assistance, as well as the editor and reviewers for the review processes.
CRedit Authorship Contribution Statement
Conceptualization and design: Asanga EE.
Acquisition, analysis, and interpretation of data: Asanga EE, Ekeleme CM.
Drafting, revision, and resources: Asanga EE, Joseph AP, Okokon JE, Inyang AN, Thomas PS, Anwana FN, Ani DK, Kanu OE.
All the authors read the manuscript and the authors’ list and consented to the publication.
Data Availability
Data will be made available by the corresponding author on request.
Declaration of AI-Assisted Technologies
The authors used QuillBot software to edit and review the content of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The approval for this study was duly given by Arthur Jarvis University's Research Committee (approval number: AJU/RC/0147), and compliance was judiciously monitored.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
All the authors contributed their quota to the research; they have thoroughly read and have approved the manuscript to be published.
Statement of Human and Animal Rights
This was not applicable in this study.
Statement of Informed Consent
This was not applicable as human subjects were not part of the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
