Abstract
Background of the study
The global increase in the number of diabetes cases reported yearly is alarming, especially in developing countries. The study assessed the toxicological and antidiabetic effects of the leaf extract of Albizia zygia, examined the antihyperglycaemic activity of the partitioned fractions and identified the phytocompounds in the most active fraction.
Methodology
The acute and sub-chronic toxicity tests of extract of A. zygia were carried out and followed by histopathological, haematological and biochemical analysis of vital organs of the experimental animals. Antihyperglycaemic and streptozotocin-induced antidiabetic studies of the extract were carried out and followed by antihyperglycaemic activity of the partitioned fractions. The most active fraction was subjected to spectroscopic analysis using GC-MS to identify its constituents.
Results
A. zygia extract elicited no signs of acute toxicity in the animals up to the dose of 5000 mg/kg. In the heamatological and biochemical studies, the extract significantly improved the recruitment of white blood cells (WBC) at 1000 mg/kg extract with 11.4 (103/μL) WBC compared to control group with 5.2 (103/μL) WBC. The extract also demonstrated a pronounced biphasic effect on the expression and activity of aspartate transaminase enzyme (AST) at 250 and 500 mg/kg. Additionally, the A. zygia extract elicited a 60% reduction in the blood glucose level after four hours in the antihyperglycaemic studies and an 85% reduction in the streptozotocin-induced antidiabetic studies. Furthermore, the n-hexane fraction gave the highest percentage blood glucose level reduction among the partitioned fractions, while 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-isopropyl-1H-Indole-2-carboxylic acid was identified as the most abundant compound in the non-polar fraction.
Conclusion
The results of the antidiabetic activity of Albizia zygia leaf extract confirmed its potency and safety and therefore justified its ethnomedicinal use in the management of type II diabetes.
Introduction
Diabetes is one of the four non-communicable diseases (cancer, respiratory and cardiovascular diseases, and diabetes) attracting global attention. 1 It is an autoimmune disease once thought to be a disease of affluence, but is on the increase in the third-world countries of Asia and Africa. In many of these countries, the inaccessibility to effective therapy besides the endemic poverty, increased sendentary lifestyle and progression of various mutual diseases, is one of the driving forces of the disease.2,3
Diabetes is characterized by insufficient insulin release (type I) or the inability of the body to use the insulin produced (type II). Globally, the majority of diabetic patients suffer from type II diabetes.4,5 Diabetes of all types can lead to many complications such as heart attack, stroke, kidney failure, limb amputations due to non-healing of diabetic wounds, and loss of vision. 6 The risks are especially higher in pregnant women, with a possibility of fetal death. Lactating mothers are also at risk. Untreated cases of diabetes have resulted in life-threatening effects on the mother and child as evident in the increased risk of congenital malfunction, stillbirth, perinatal death, obstetric complications, and maternal mortality.7–9
Based on International Diabetes Federation report, more than 537 million adults between age 20–79 have been living with diabetes since 2021. 9 However, the incidence has been on the increase with mortality found to occur before the age of 70 years. This is particularly true in the low economic countries. 1 To mitigate avoidable diabetes-induced mortality, early detection and access to critical healthcare and drugs are very crucial. The inadequate access to scarce first-line antidiabetic drugs such as insulin, metformin, sulfonylureas, and other hypoglycaemic agents, coupled with perennial poverty in most of the low-income countries also introduced another challenge in the fight against the menace of the disease, thereby sparking a resurgent of interest in the use of alternative medicines.10–12 The use of herbal medicines in the treatment of diseases including diabetes predates the introduction of orthodox drugs into Africa. 13 The low cost and anecdotal effectiveness of herbs underline the renewed investigations of nature including medicinal plants for putative hypoglycaemic agents for the regulation of blood glucose level and hence diabetes.2,14 There is a dire need for scientific validation of the claims by traditional healers using medicinal plants in the management of diabetes.
Albizia zygia DC J.F. Macbr. (family Leguminosae) is a medium-sized, deciduous and gum-producing tree. It is widely found in Nigeria and other tropical African countries. Nigerian local names include Nyie avu” (Igbo) and “Ayunre” (Yoruba). 15 It is of economic importance because of its timber durability and immunity to termites’ attack. The leaves and the bark are used in Africa Traditional Medicine as remedies to treat fever, malaria, diarrhea, and edema, conjunctivitis. 16 Among the Yorubas in Southern Nigeria, it is used for the treatment of boils, piles, conjunctivitis, flu and scabies. 17 The bark of the plant has also been reported to be associated with the treatment of arthritis, sprain, cough, aphrodisiac, as a worm expeller and to counter female sterility. 17 The leaf and root of A. zygia are used in the management of feverish conditions and malaria,15,18–20 inflammation, diarrhoea, oedema, and as an antihelminthic. 21 The ethanol extract of the root and leaf are used as a cough suppressant, analgesic-antipyretic, aphrodisiac and antihyperglycemic agents.15,22 The decoction of the stem bark is used in the management of waist pain, and fever, a study conducted in 2023 showed that the root bark of A. zygia exhibited pronounced activity against venereal infections. 23
The antioxidant, antimicrobial, analgesic, anticancer, antiinflammatory properties of the methanol stem bark extract of Albizia zygia has been reported.24–26 Appiah-Opong et al 27 had reported the cytotoxic effects of extracts and root fractions of A. zygia. The hydroethanolic extract of the root exhibited apoptosis via induction of DNA fragmentation in Jurkat cells. The antibacterial activity of aqueous and hydro-ethanolic of the leaves and bark extracts of A. zygia. 28 In continuation of our investigation of medicinal plants of Nigerian flora as potent antidiabetic agents, toxicological and antidiabetic effect of Albizia zygia (DC.) J.F.Macbr. leaf extract was evaluated experimentally, and the identified chemical constituents of the extract were identified.
Materials and Methods
General Experimental
Rotary evaporator (RE301/601/801 model, Yamato Scientific America, Inc., U.S.A), Chiller (Churchill, Instrument Co. Ltd, U.K), Vacuum pump (MB 338618 model, Edwards High Vacuum Int., England), Oven (Hearson & Co. Ltd, London), Mettler electronic weighing balance (AB 54 model, Mettler Toledo, U.S.A), Ultra-violet (UV) lamp (254 and 366 nm) (Grant Instrument, U.K), Oral cannula, ACCU-CHEK Glucometer (model GB 11558973, Roche, Germany) with ACCU-CHECK test strips (Roche, Germany), UV spectrophotometer, Dutrao (Model SM 600, Shang Yhai Yong Chuang Medical Instrument Co. Ltd) spectrophotometric microplate reader, Automated haematology analyser, Centrifuge, Semi-automated biochemistry analyzer. Methanol, Tween 80, Normal Saline.
Preparation of Extract
Leaf of Albizia zygia was collected within the premises of Obafemi Awolowo University, Nigeria in December 2024. Identification and authentication of the plant was done by I. I. Ogunlowo of the herbarium section of the Faculty of Pharmacy, OAU, Ile-Ife. A voucher specimen was prepared, and deposited at the herbarium, with voucher number FP1 2672. The harvested leaf was oven-dried at 35 °C for four days. It was then milled, and about 2 kg powdered leaf of A. zygia was macerated in absolute MeOH (100%) for 72 h with intermittent shaken. The mixture was filtered, and the marc was re-extracted three times. The resultant filtrates were combined and concentrated in vacuo to obtain the A. zygia methanol leaf extract. The extract was then partitioned into n-hexane, dichloromethane, ethyl acetate to afford their corresponding and aqueous methanol fractions.
Animal Handling and Care
The experiment was carried out on healthy albino rats (120-150 g) of both sexes and bred under standard conditions. They were housed and allowed to acclimatized for 14 days on a natural cycle of light and dark at the animal care facility, Department of Pharmacology, College of Pharmacy, Afe Babalola University. The animals were allowed free access to food (standard pellets from Bendel Feeds, Nigeria) and water. The animal experimental protocols were approved by the College of Science Research Ethical Committee, Afe Babalola University, Nigeria with approval number CSREC/B-14/25. All procedures adhered to the international guidelines outlined in the NIH Guide for the care and use of laboratory animals (NIH Publication, No. 83-123, revised 1985).
Acute and Sub-acute Toxicity Tests
Normal rats weighing 120 to 150 g were deprived of food were administered methanol extract of A. zygia (ABE) in a range of 10 to 5000 mg/kg. The study was carried out in two distinct phases. In the first phase, a total of nine rats were used, divided into three groups of three. Each group received ABE at doses of 10, 100, and 1000 mg/kg, respectively, and the animals were observed over a 24-h period for signs of toxicity or death. Based on the results of Phase 1, twelve additional rats were used in Phase 2. These were assigned into four groups of three rats each and administered extract doses of 1000, 1600, 2900, and 5000 mg/kg, respectively. Following the administration of the extract, the animals were observed after one hour and monitored daily for 14 days for signs of severe toxicity, alterations in behaviour, and mortality. 29 The sub-acute toxicity test was conducted according to the modified OECD Test Guideline 407 (OECD, 2008). Four groups of 5 rats (n = 5) were administered graded doses (250, 500, and 1000 mg/kg) of A. zygia leaf extract and normal saline respectively every day for 28 days. On days 1, 7, 14, and 28, the blood glucose level of the rats were monitored. The animals were anaesthetized using chloroform and a 5 mL blood sample was collected by cardiac puncture after the 28th day and analysed. 30
Analysis of Haematological and Biochemical Markers in Sub-acute Toxicity Studies
After the study was completed, animals were anaesthetized with chloroform and blood sample of the animals were collected via cardiac puncture. The automated haematology analyser was used to test approximately 50 μL of blood on aspiration from a 2.5 mL blood sample for the haematological investigation. 31 In the analysis, parameters like packed cell volume (PCV), white blood corpuscles (WBC), Red blood corpuscles (RBC), Haemoglobin (HGB), Mean corpuscular volume (MCV), Mean corpuscular haemoglobin (MCH) and Mean corpuscular haemoglobin concentration (MCHC). Also, semi-automated biochemical analyser and analysing kits were used to perform a biochemical assay on a 3.5 mL blood sample. Biochemical markers such as Total cholesterol (TC), Aspartate transaminase (AST), Alanine transaminase (ALT), Lactate dehydrogenase (LDH), and Urea were analysed in the serum using commercial kits purchased from Randox Laboratories Ltd (Crumlin, UK) by the guidelines provided by the manufacturer. 31
Histopathological Examination
The histological analysis of vital organs such as the kidney, liver and pancreas was carried out according to the methods of Baker and Silverton (2014). Excised tissue samples of the kidney, liver and pancreas were separately fixed, dehydrated, cleared and impregnated in molten wax. The embedded tissues were then sectioned using a rotary microtome (Leica RM 2125 RTS), and the ribbons were floated out in a water bath at 40 °C. The sections were dried at 30 °C and stained for various histomorphological features followed by observation with a Leica DM 750 Microscope (Leica Microsystems Switzerland) enabled with a digital camera (Leica ICC50). 32
Antihyperglycaemic Activity of A. zygia Extract/Fraction
A glucose tolerance test was performed by administering glucose (10 g/kg, p. o) to 18 h fasted rats. Hyperglycaemic rats (blood glucose level of 7.0 mmol/L corresponding to 126 mg/dL after 0.5 h) were divided into six groups, each group of five rats (n = 5) was administered (p.o) with 1% Tween 80 in normal saline (negative control), A. zygia extract (50, 100, 200 and 400 mg/kg), and glibenclamide (5 mg/kg) as a positive control. Also, the partitioned fractions were administered with the most active dose of the extract. Blood glucose level lowering activity was determined over 4 h period from 0, 0.5, 1, 2 and 4 h by dropping blood collected from the tip of the tail of each rat onto a glucometer strip and the blood glucose level was recorded.33,34
Antidiabetic Effect of A. zygia Extract on Streptozotocin-Induced Diabetic Rats
Diabetes was induced in overnight-fasted healthy rats by intraperitoneal injection of freshly prepared streptozotocin at a dose of 65 mg/kg, dissolved in 0.1 M sodium citrate buffer (pH 4.5). Seventy-two hours post-induction, fasting blood glucose levels (FBGL) were measured, and rats with FBGL ≥ 12 mmol/L were classified as diabetic. These diabetic rats were then randomly divided into four groups, each containing five rats. Group A received 1% Tween 80 in normal saline (negative control), while Groups B was treated daily with A. zygia extract at doses of 50 mg/kg, respectively. Group C was administered glibenclamide at 5 mg/kg as the positive control. FBGL were monitored on days 1, 4, 7, 10, and 14 of treatment.2,35
GC-MS Analysis of the Most Active Fraction
The chemical constituents of n-hexane partitioned fraction of A. zygia was analysed with Gas Chromatography-Mass Spectrometry (GC-MS) on a Varian 3800/4000 system equipped with an Agilent CP-Sil 5 CB fused silica capillary column (30 m × 0.25 mm internal diameter). This setup was coupled to a Varian 4000 mass spectrometer operating in Electron Impact (EI) mode at 70 eV, with a mass scan range of m/z 30–1000 amu. The ion source and quadrupole were maintained at temperatures of 230 °C and 150 °C, respectively. The oven temperature programme began at 150 °C and was ramped up to a final temperature of 300 °C. A 1 µL aliquot of the sample was injected using a split ratio of 10:1. Nitrogen served as the carrier gas at a flow rate of 0.8 mL/min, with a total run time of 40 min.
Phytochemical identification was performed by comparing the obtained spectra to those in the NIST Mass Spectral Library (2009). Retention indices (RIs) were calculated by referencing a series of n-alkanes (C6–C28) under the same chromatographic conditions. Compound quantification was based on peak area normalization, without applying response factors.
To control for technical variability, all samples and their replicates were injected in a randomized sequence within a single analytical batch. Additionally, pooled quality control (QC) samples were injected periodically throughout the run to monitor instrument stability and support repeatability assessment
Statistical Analysis
The data are presented as the mean ± SEM, with the number of animals in each group indicated by (n). Analysis of variance (ANOVA) was done, followed by Bonferroni post hoc comparisons to identify the sources of significant differences. A p-value of less than 0.05 was considered statistically significant. Histomorphometric analysis of harvested tissues were analysed with Leica DM 750 Microscope (Leica Microsystems Switzerland) enabled with a digital camera (Leica ICC50) enabled with a ImageJ software.
Results
Extraction Yield
The cold extraction method of preparation of extract was used to preserve the active metabolites and prevent degradation, decomposition and isomerization of the active metabolites by heat. The extraction afforded a 14.59% yield of crude A. zygia methanol leaf extract.
Acute and Sub-chronic Toxicity
No acute signs of toxicity were observed in the treated animals following the administration of up to 5000 mg/kg dose of A. zygia extract. All animals in the experimental and control groups showed normal behavioural patterns. No treatment-related adverse reactions such as blisters, erythema, oedema and acute inflammation, and postural abnormalities such as distorted gait or impairment in food and water intake were observed, and no mortality was recorded.
Effects of A. zygia Extract on Haematological Parameters
The effect of A. zygia extract on key haematological indicators such as the PCV, blood cell count, MCV, and haemoglobin concentration of the treated animals against normal saline is presented in Table 1.
Effect of Extract on Haematological Parameters of Normal Rats.

Data showed the mean ± SEM haematological parameters at the different doses, n = 5. Values are expressed as mean ± SEM (n = 5). NS: Normal saline; AZ: A. zygia extract, PCV: Packed cell volume; WBC: White blood corpuscles; RBC: Red blood corpuscles; HGB: Haemoglobin; MCV: Mean corpuscular volume; MCH: Mean corpuscular haemoglobin; MCHC: Mean corpuscular haemoglobin concentration. Values with different superscripts within rows are significantly different (p < 0.05).
From the table, the A. zygia extract at the tested dose levels of 250, 500, and 1000 mg/kg exhibited no acute, chronic or sub-chronic toxic activities on the haematological parameters of all treated animals compared to the control group that received normal saline. The extract significantly improved the recruitment of white blood cells (WBC) at 1000 mg/kg extract with 11.4 (103/μL) WBC compared to the control group with 5.2 (103/μL) WBC. The extract had no acute or chronic toxic effects on the differential parameters of the blood such as the lymphocytes and granulocytes (Table 1). The effects of A. zygia extract were found to be biphasic from the control group to the treated groups (250, 500, and 1000 mg/kg).
Effects of A. zygia Extract on Biochemical Parameters
The toxic effects of A. zygia extract on the biochemical reactions in the treated animals were investigated by analysis of key biochemical toxicity indicators such as lactase dehydrogenase, aspartate transaminase, ALT and lipid profile and the results are presented in Table 2.
Effect of A. zygia Leaf Extract on Biochemical Parameters.

Data showed the mean ± SEM haematological parameters at the different doses, n = 5. Values are expressed as mean ± SEM (n = 5). NS: Control group; AZ: Extract of Albizia zygia; ALT: Alanine transaminase; AST: Aspartate transaminase; LDH: Lactate dehydrogenase. Values with different superscripts within columns are significantly different (p < 0.05).
The extract exhibited a pronounced biphasic effect on the expression and activity of aspartate transaminase enzyme (AST) at 250 and 500 mg/kg with 88.68 ± 6.14 and 90.53 ± 0 mmol/L compared with the effect of the extract at 1000 mg/kg with 286.58 ± 1.05 mmol/L as against the control group with 172.90 ± 2.89 mmol/L, while there was no significant difference in the ALT and LDH level of the experimental animals. Also, the extract exhibited a similar biphasic effect on the total cholesterol, triglyceride, high-density lipoprotein cholesterol and low-density lipoprotein cholesterol profile of the animals (Table 3).
Effect of A. zygia Leaf Extract on Lipid Profiles.
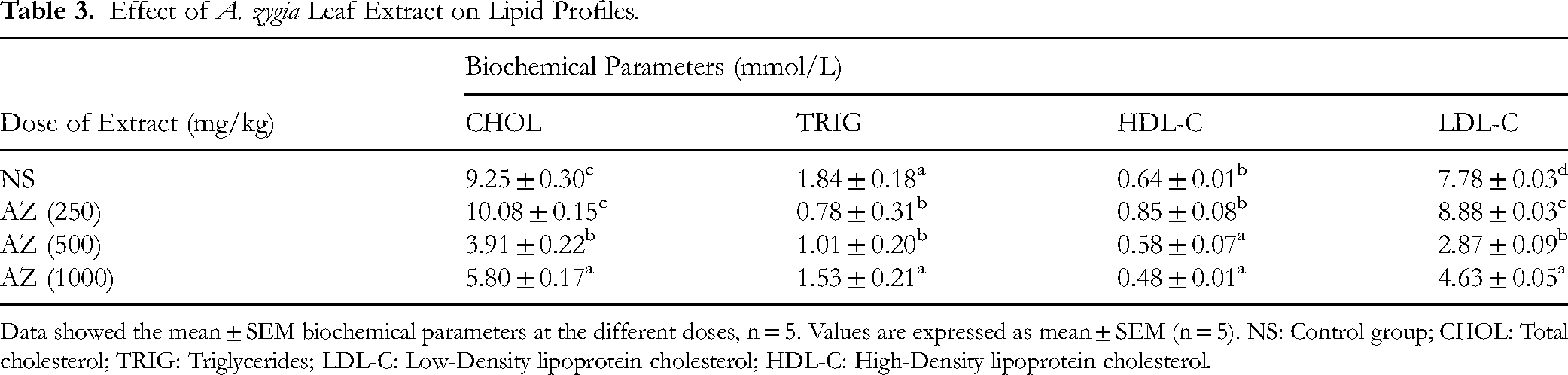
Data showed the mean ± SEM biochemical parameters at the different doses, n = 5. Values are expressed as mean ± SEM (n = 5). NS: Control group; CHOL: Total cholesterol; TRIG: Triglycerides; LDL-C: Low-Density lipoprotein cholesterol; HDL-C: High-Density lipoprotein cholesterol.
Histopathological Studies of the Effect of A. zygia Extract
The histological effect of A. zygia extract following the administration of the extract at 250, 500, and 1000 mg/kg, on vital organs were investigated. The photomicrograph of the kidney tissues (Figure 1), hepatic tissues (Figure 2) and the pancreas (Figure 3). In all tissues analysed, Plate A is the control, and plates B, C, and D tissues from treated animals respectively for the kidney, liver and pancreas tissues.
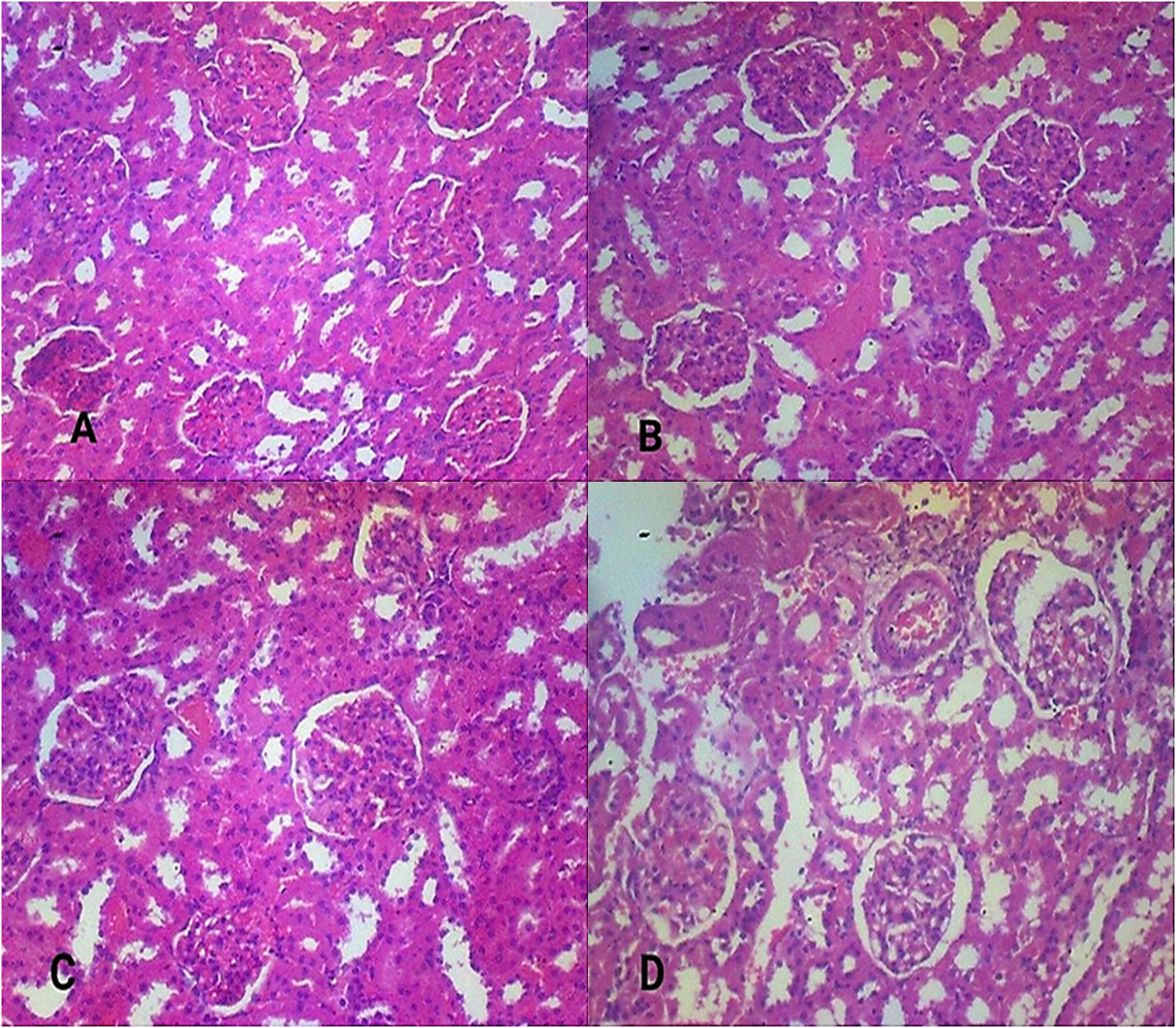
Photomicrographs of the Histology of Renal Tissue. A: Control Group, B-D: Treatment Groups with 250, 500 and 1000 mg/kg of A. zygia Extract, Respectively.

Photomicrographs of the Histology of Liver Tissue. A: Control Group, B-D: Treatment Groups with 250, 500 and 1000 mg/kg of A. zygia Extract, Respectively.

Photomicrographs of the Histology of Pancreatic Tissue. A: Control Group, B-D: Treatment Groups with 250, 500 and 1000 mg/kg of A. zygia Extract, Respectively.
Effects of A. zygia Extract on Blood Glucose Level of Normal Animals
The effects of the A. zygia extract at 250, 500, and 1000 mg/kg dose levels on the blood glucose level of normal rats were investigated. Normal rats were taken as animals which were not induced and not treated with standard drugs, rather with normal saline as control and the extract of A. zygia. This served to ascertain that the normal saline used as a negative control did not have a blood glucose-lowering effect on the animals and therefore it was free of any bioactive metabolite. The result as presented in Figure 4 showed a dose-dependent gradual lowering in the blood glucose level of the animals over 28 days. A significant lowering effect was observed from day 14 to day 28 at a dose of 1000 mg/kg as against distilled water-treated animals, with a gradual rise in blood glucose level from day 1 of the experiment to day 28.

Dose-Related Effect of A. zygia Leaf Extract on Normoglycemic Rats. NS: control; A. zygia Extract (AZ): 250, 500 and 1000 mg/kg Respectively. Values are Expressed as mean ± SEM (n = 5), (P < 0.05) Versus the Control Group Using Two-Way ANOVA Followed by Dunnett Multiple Comparison Test.
Blood-Glucose Lowering Effect of A. zygia Extract on Treated Animals
The hypoglycaemic activity of the A. zygia extract to lower blood glucose levels in diabetic animals is presented in Table 4. The extract was tested at 50, 100, 200, and 400 mg/kg doses against distilled water and glibenclamide at 5 mg/kg as negative and positive control respectively. A. zygia extract at 50 mg/kg exhibited an excellent dose-dependent monophasic hypoglycaemic activity comparable to that of glibenclamide from 0 through 4 h of the test, with a gradual reduction in the level of the glucose in the blood of treated animals compared with the animals treated with distilled water.
Blood-Glucose Lowering Effect of A. zygia Extract.

Data show the mean ± SEM blood glucose levels at the different time points expressed as percentages of levels at 0 h (T0), n = 6. Values in parentheses represent the percentage reductions in blood glucose levels relative to negative control for each time point. Values with different superscripts within columns are significantly different (p < 0.05). DW: negative control; GLI: Glibenclamide (positive control) and AZ: Albizia zygia extract.
Anti-Diabetic Activity of A. zygia Methanol Extract on Streptozotocin-Induced Diabetic Rats
The results of the effect of A. zygia extract on streptozotocin-induced diabetic rat are presented in Table 5.
Antidiabetic Activity of A. zygia in Streptozotocin-Induced Diabetic Rats.

Data show the mean ± SEM blood glucose levels at the different time points expressed as percentages of levels at 0 h (T0), n = 5. Values in parentheses represent the percentage reductions in blood glucose levels relative to negative control for each time point. Values with different superscripts within columns are significantly different (p < 0.05), one way ANOVA followed by the Student–Newman–Keuls’ test). NS: 1% of Tween 80 in normal saline (negative control).
The extract of A. zygia methanol (50 mg/kg) produced a significant, time-dependent blood glucose level reduction from day 1 to day 15. On day 15 of the experiment, the 50 mg/kg dose of the extract elicited 84% blood glucose level reduction and the result obtained at this time frame is comparable (p > 0.05) to the positive control.
Antihyperglycaemic Effect of A. zygia Partitioned Fractions
Table 6 summarizes the effect of partitioned fractions of Albizia zygia (n-Hexane, DCM, Ethyl acetate, and Aqueous) and standard drug (GLI) on blood glucose levels over time (0 to 4 h).
Effect of A. zygia Extracts and Standard Drug on Blood Glucose Levels.

Data show the mean ± SEM blood glucose levels at the different time points expressed as percentages of levels at 0 h (T0), n = 6. Values in parentheses represent the percentage reductions in blood glucose levels relative to negative control for each time point. Values with different superscripts within columns are significantly different (p < 0.05). NS: negative control; DCM: Dichloromethane, EtOAc: Ethyl acetate, GLI: Glibenclamide (positive control).
The dichloromethane and ethyl acetate fractions of A. zygia was devoid of activity while the aqueous fraction demonstrated a mild glucose lowering effect in glucose loaded rats after 4 h of administration. However, the n-hexane gave the highest antihyperglycaemic activity at 48% and the blood glucose level reduction was non-time dependent up to 4 h. Furthermore, the results obtained for the n-hexane fraction was comparable p > 0.05 at all-time interval.
Secondary Metabolite Profiling of n-Hexane Fraction
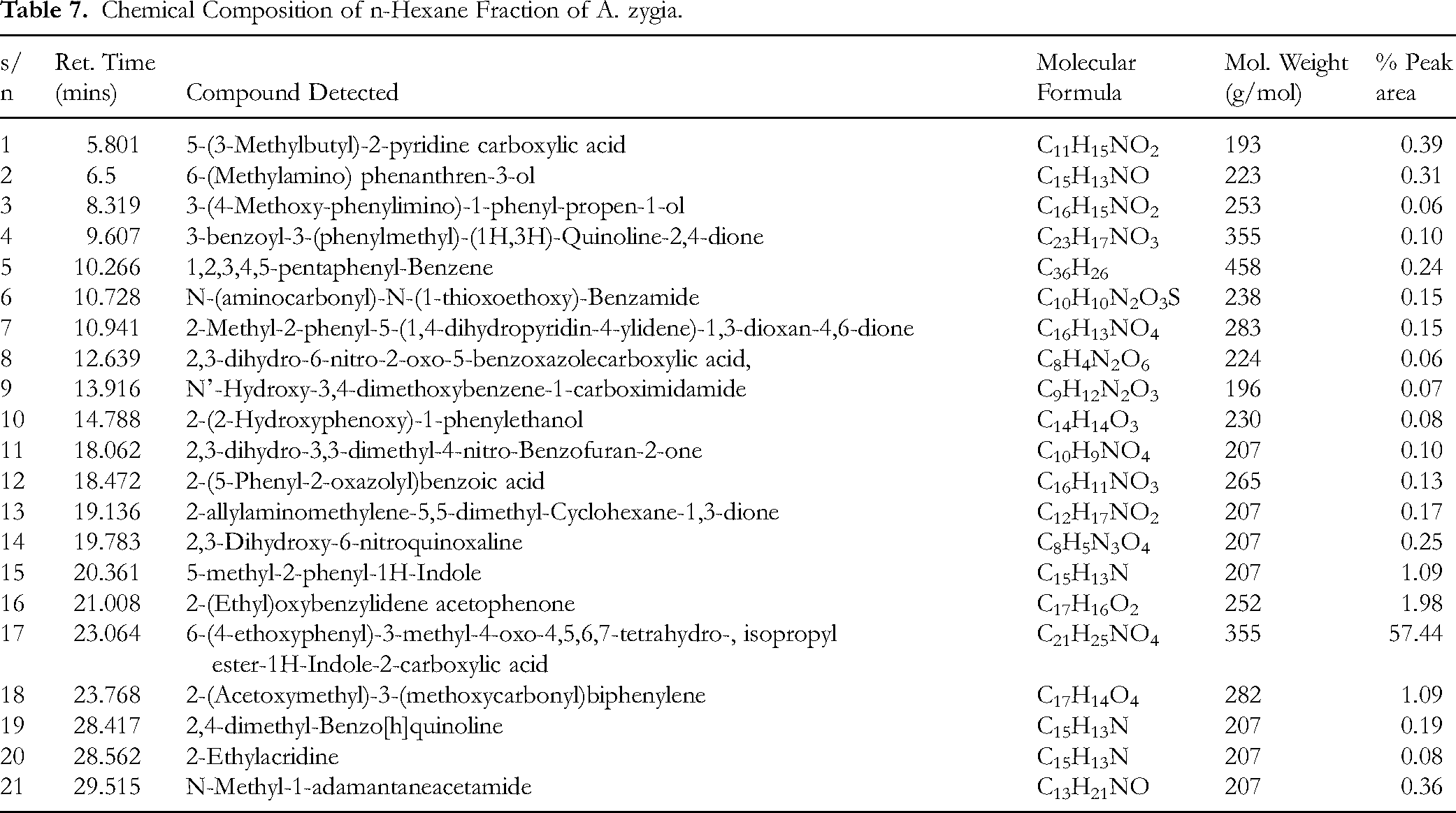
The GC-MS profiling of the most active fraction (n-hexane fraction) identified a total of 21 chemical constituents, as presented in Table 7, most of the chemical constituents detected were polycyclic aromatic nitrogenous heterocycles, comprising derivatives of indole and quinoxaline carboxylic acid esters. The % composition of the constituents was determined using the total % ion peak area and by comparison with relevant literatures and NIST library 2014 spectra matching. The structures of the identified constituents are shown in Figure 5.

Structures and Names of Chemical Constituents Identified in the n-Hexane Fraction.
Chemical Composition of n-Hexane Fraction of A. zygia.
Discussion
The study performed comprehensive safety investigation of A. zygia methanol leaf extract. In the acute toxicity study, no toxicity effect was observed in the experimental rats on administration of 5000 mg/kg A. zygia extract after daily monitoring period of 14 days. None of the rats died and there were no observable changes in the behaviour of the rats concerning breathing, cutaneous effect, sensory and nervous system responses, or gastrointestinal effect. The median lethal dose, LD50 of A. zygia leaf extract was, therefore, greater than 5000 mg/kg which indicated its safety and low risk of toxicity. The results are consistent with those obtained for Alternantera brasiliana and Acrocomia aculeata.2,36
Several studies has recommended repeated-dose administration to examine toxic effect of plant extract due to the fact that acute toxicity information does not give concise data for clinical application. 37 Therefore, this study further examined the toxic effect A. zygia extract through 28 days repeated dose administration study. Sunmonu and Oloyede 38 reported that haematological parameters are indicators of the toxic effects of plant extracts in living systems. Albizia zygia leaf extract at 250–1000 mg/kg dosage exhibited no toxic effect on the PCV, HGB and RBC levels in the normoglycemic rats. The haemopoietic system of experimental rats that were treated with the extract indicated a positive effect by the extract, evidenced by the maintenance of RBC, HGB, PCV, MCH, and MCHC levels by the extract throughout the experiment (Table 1). Administration of 500 and 1000 mg/kg doses of A. zygia extract resulted in a significant elevation of the WBC level, which is indicative of the possible immunomodulatory effect of the extract, particularly at higher doses.32,39
The AST and ALT are important enzymes and key indicators of liver dysfunction when they increase in serum. 40 From the results obtained, the effect of A. zygia leaf extract on the biochemical parameters of animals after treatment for 28 days, showed that there was no significant difference in the biochemical parameters when compared with the negative control. The extract exhibited no toxic effect on the ALT, LDH, and urea levels. This result suggested the safety of the extract on the heart, liver, and kidney of the rats. However, administration of the 250–500 mg/kg doses decreased the AST level, and in the 1000 mg/kg tested group of rats, the extract caused a significant increase in AST level. This may however, not indicate liver dysfunction or damage as a result of the administration of A. zygia extract or fractions. While ALT is predominantly found in the liver hepatocytes, and elevated levels of ALT are indicative of liver damage. (Table 3). High AST and ALT levels have been associated with liver diseases or hepatotoxicity (Brautbar and Williams, 2002; Desai et al, 2012). The extract gave a comparable effect with the control group on total cholesterol (TC), triglycerides (TRIG), low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein–cholesterol (HDL-C) at all its tested concentrations. This showed that the extract exhibited no adverse effect on the lipid profile of experimental rats (Table 4).
Vital organs like kidney, liver and pancreas are majorly responsible for metabolism and also purify and eliminate waste from the body. 41 The histomorphology of the kidney, liver and pancreas in all experimental and negative control groups were examined. The results showed well-outlined tubules with the cuboidal epithelium easily identifiable, the canals of some of the tubules are clear and unclogged. The glomeruli of the kidney are clearly outlined, the vascular and urinary poles are identifiable, and the filtration space between the glomerular podocytes and capsular boundary cells is normal. The histology of the pancreas showed pancreatic lobules with isolated pancreatic islets of Langerhans. The lobules are separated by interlobular septa. The pancreatic acini are numerous and healthy acinar cells are identifiable. The hepatic cells are aligned, and sinusoids are well-outlined, clear and unclogged. The central veins and portal triads of the vessels appear normal with no sign of vascular congestion or stenosis. The nuclei are seen with conspicuous nucleolus, some binucleate cells that imply active mitotic divisions are also recognizable. The extract therefore exhibited no toxic effects on the kidney, liver and pancreas. No alteration in the histoarchitectural pattern of the vital organs.
The regulation of blood glucose by plant extracts has become a trend in clinical applications, and more and more reports are focused on plant extracts.42,43 In this study, the blood glucose levels of the negative control normoglycemic rats that were given distilled water throughout the study did not change significantly, indicating the water was free of any bioactive constituents, while the blood glucose levels of the rats were affected by the extract at doses of 250 and 500 mg/kg, in a similar manner to the negative control on all days of the experiment (Figure 4). However, on days 14 and 21, the blood glucose levels of the animals decreased when the extract was given at a dose of 1000 mg/kg, suggesting a possible hypoglycaemic effect of the extract when given to non-diabetic people at greater doses, especially if taken over an extended period.
Consistently hyperglycaemia can be effectively managed with herbal medicines. 34 A similar activity profile to that of glibenclamide by plant extract and metabolites in the glucose-loaded rat model suggests an insulin-stimulating mechanism of action. From Table 5, there was a significant time-dependent reduction in blood glucose levels in the glibenclamide and extract-treated groups as against the negative control group, lasting up to 4 h. This indicates the animals’ homeostatic regulatory mechanisms and suggests a healthy state of their pancreas. The A. zygia leaf extract, at doses of 50, 100, 200, and 400 mg/kg, demonstrated a blood glucose level reduction from 0.5 to 2 h. However, the extract showed the most effective anti-hyperglycemic activity at 50 mg/kg dosage, which compares favourably with glibenclamide after 4 h (p > 0.05).
The permanent induction of diabetes in experimental rats using streptozotocin is an effective method in antidiabetic research due to its selective destruction of pancreatic beta cells, thereby causing reduction in insulin secretion. 2 In this study, healthy experimental rats were induced with streptozotocin, and the results showed that the diabetes induction was permanent in the negative control. The 50 mg/kg dose of the A. zygia extract gave time-dependent blood glucose level reduction which shows it exerted an early extrapancreatic and insulin-stimulating effect on prolong treatment in diabetic rats. The activity demonstrated by the A. zygia extract is similar to that of Alternantera brasiliana where the 50 mg/kg dose gave better antidiabetic effect that was comparable with the positive control and dietary formulation of experimental rat feeds with A. zygia leaf.2,22
To identify the most active fraction of the crude extract, the partitioned fractions were assayed for their antihyperglycaemic activity in glucose-loaded rats. The n-hexane fraction elicited the best antihyperglycaemic activity with a percentage blood glucose level reduction of 50%, while the aqueous fraction demonstrated a mild antihyperglycaemic effect. Also, the dichloromethane fraction exhibited no effect on the hyperglycaemic rats. This implies that the antidiabetic constituents in the A. zygia extract are largely resident in the n-hexane fraction. Profiling of the chemical constituents of the most active fraction (n-hexane fraction) identified 21 chemical constituents. The retention time, identified chemical components with names, molecular weight and formulae and total % peak area of constituents are presented in Table 7. The most abundant chemical constituent was 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-isopropylester-1H-Indole-2-carboxylic acid, which accounted for more than 50% of the total weight of the sample analysed. This is followed by 2-(Ethyl) oxybenzylidene acetophenone, 5-methyl-2-phenyl-1H-Indole, 2-(Acetoxymethyl)-3-(methoxycarbonyl)biphenylene. Most indole-based chemical constituents have been found to exhibit significant pharmacological importance as an antidiabetic, anti-inflammatory, anticancer and antimicrobial agents according to Ahmad et al, 44 Agus et al, 45 and Zhu et al. 46 Previous chemical investigations by Gunathilaka et al 47 identified 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-isopropylester-1H-Indole-2-carboxylic acid, as an antidiabetic agent from the ethyl acetate fraction of a marine seaweed (algae) Gracilaria eludis (Gracilariaceae). Therefore, existing literature is in agreement with the findings of this study. The antidiabetic activity of this compound has been linked to the insulin-sensitizing and activation of the glycogen synthase enzymes involved in the synthesis of glycogen. 47 QSAR studies have shown that the glucose-lowering activity of this compound is associated with 1H-tetrahydroindole-2-carboxylic acid moiety of the molecule. 47 Therefore, considering the critical importance of this compound as an antidiabetic agent, and from the GC-MS result obtained in this present study, it can safely be stated that 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-isopropylester-1H-Indole-2-carboxylic acid is the antidiabetic agent in the lipophilic fraction of A. zygia methanolic leaf extract. Further investigations into the biological role of 6-(4-ethoxyphenyl)-3-methyl-4-oxo-4,5,6,7-tetrahydro-isopropylester-1H-Indole-2-carboxylic acid, showed that it exhibited an antiobesity and antilipidemic activities. 48 Another indole-based chemical constituent identified in the lipophilic fraction of A. zygia is 5-methyl-2-phenyl-1H-Indole, which has been speculated to exhibit antiviral, antimicrobial and anti-inflammatory activities. 49
Limitation of the Study
The study assessed the toxicological and antidiabetic effect of the extract and fractions of A. zygia leaf. It also identified the phytochemicals in its active fractions. The molecular evidences of the effect of A. zygia extract on diabetic subjects can be exclusively studied. Also, the bioactivity guided isolation of the identified compounds and other phytocompounds that may be responsible for the antidiabetic activity of the n-hexane fraction can be performed.
Conclusion
This present study investigated the safety profile of A. zygia extract, evaluated its antidiabetic activity in both glucose loaded and streptozotocin-induced diabetic rats. The most active partitioned fraction was profiled to identify its chemical constituents. The acute toxicity study showed that the extract was safe up to 5000 mg/kg. Also, the methanol leaf extract elicited no significant toxic effects as indicated by the haematological and biochemical parameters. Furthermore, no acute and chronic toxic effects were observed on the kidney, liver and pancreas. The extract demonstrated good antihyperglycaemic activity in glucose loaded rat and excellent antidiabetic activity in streptozotocin-induced diabetic rats. The n-hexane fraction elicited the highest percentage blood glucose level reduction, thereby indicating that the non-polar phytocompounds are responsible for the antidiabetic activity. The study recommends extensive molecular investigation of the effect of extract of A. zygia in diabetic subjects as well as extensive phytochemical studies of the most active fraction.
Footnotes
Acknowledgment
The authors extend their appreciation to the Ongoing Research Funding program, (ORF-2025-620), King Saud University, Riyadh, Saudi Arabia.
Ethical Approval
This study was approved by the College of Science Research Ethical Committee (CSREC) of Afe Babalola University, Nigeria, with registration number: CSREC/B-14/25.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Authors Contributions
K.O.F. Investigation, supervision, data interpretation, writing-original draft, writing-review and editing, M.D.A. Supervision, investigation, data interpretation, writing-original draft, writing-review and editing. F.G.F. Investigation, supervision, software, manuscript writing and reviewing. S.B.O: Investigation, writing-original draft, writing-review and editing, resources. S.O.O: Investigation, data curation, formal analysis. S.A.A: Formal analysis, writing-original draft, writing-review and editing. A.A.A: Methodology, investigation. A.O.A: Methodology, investigation. T.I.A: Methodology, investigation. A.J.O: Investigation, methodology, data acquisition, writing-review and editing, funding acquisition. J.M.A: Investigation, methodology, formal analysis, funding acquisition. F.O.G: Data curation, formal analysis, writing-review and editing. S.O.F: Supervision, Investigation, methodology, writing- review and editing.
Funding
This work was supported by the Ongoing Research Funding program, (ORF-2025-620), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All the experiments were performed following the international ethical guidelines outlined in the NIH guide for the care, handling, and use of animals in research. A proposal, describing the handling and treatment of animals, was submitted, reviewed, monitored, and approved by the Afe Babalola University Ethical Review Board for the use of animals in research under the approval number CSREC/B-14/25.
