Abstract
Introduction
This study examines the α-glucosidase inhibition and antioxidant impact of Moringa oleifera (MO) leaves, which have long been adopted for their medicinal features.
Methods
GC/MS, or gas chromatography/mass spectrometry, was utilized to evaluate the chemical content of MO extracts. The DPPH as well as ABTS assays were utilized to assess antioxidant activity, while the Folin-Ciocalteu reagent was utilized to clarify the total phenolic amount. Its potential for managing diabetes was confirmed by evaluating α-glucosidase inhibition in vitro.
Results
Twenty-five compounds were shared between the 160 compounds in the EtOH extract and the 140 in the EtAc extract. Both Palmitic acid (3.40% in EtAc and 3.57% in EtOH) and α-Linolenic acid (2.95% in EtOH and 2.85% in EtAc) were found at significant levels. The total phenolic amount was 157.14 ± 14.46 mg GAE/g. Antioxidant activity was demonstrated through the DPPH test, which showed an IC50 of 43.01 µg/mL, as well as the ABTS assay, with an IC50 of 21.20 µg/mL. Strong α-glucosidase inhibition was observed with the EtOH-MO extract, resulting in an IC50 of 15.01 µg/mL.
Conclusion
Moringa oleifera extracts demonstrate significant antioxidant and α-glucosidase inhibition activities, attributed to their high phenolic content and beneficial fatty acids. These findings support MO's potential as a natural therapeutic compound for diabetes management as well as prevention.
Introduction
Moringa oleifera (MO), often listed as the drumstick tree or the “miracle plant,” is a tropical perennial deciduous plant that originates to the Moringaceae family and has generated a lot of curiosity due to its many health advantages. 1 Understanding the biological properties of both natural and synthetic molecules is essential for recognizing the potential of natural products. By integrating perspectives from other fields, researchers can acquire insights into the relationships between structure and activity, as well as effective therapeutic applications. 2
This rapidly increasing crop exhibits a variety of pharmacological impacts, like anti-diabetic, anti-cancer, anti-inflammatory, and antioxidant benefits, as well as containing a range of bioactive chemicals. 3 The discovery of new therapeutic targets and intervention strategies will lead to more individualized treatment plans, helping to determine which medications are best for each patient in terms of both risks and benefits. 4
MO seeds are an essential ingredient of Thai food, and throughout Western Asia, other parts of the plant are valued for their therapeutic qualities, which include regulating blood pressure, controlling thyroid function, improving digestive health, and exhibiting antibacterial effects.5-7 The plant is useful for food, cosmetic, pharmaceutical, as well as water treatment applications due to its many components, including leaves, pods, seeds, roots, as well as flowers. These parts are affluent in proteins, vitamins C as well as A, omega-3 fatty acids, antioxidants, as well as vital minerals like calcium, potassium, iron, as well as phosphorous.8,9
MO leaves have been shown to prevent blood glucose and urine glucose levels while improving glucose tolerance in studies involving both healthy as well as diabetic mice. Researchers consider that the fiber in MO leaves contributes to these blood sugar-lowering effects by reducing the rate at which glucose is absorbed in the intestines and postponing the emptying of the stomach.10-12
In prospective randomized placebo-controlled study, participants with type 2 diabetes mellitus (T2DM) who had not yet received treatment were given either 8 g of MO leaf capsules daily or a placebo for four weeks. There were no discernible differences in refraining plasma glucose (FPG) or HbA1C among the treatment and control groups in this trial, which involved 32 T2DM patients with a mean age of 55 as well as a mean HbA1C of 7.0%. The systolic blood pressure of the MO leaf group was 5 mmHg lower than baseline; however, this difference was not statistically significant. Interestingly, supplementing MO leaves did not cause any negative effects. 13
MO's potential for environmentally beneficial uses, such as wastewater treatment and nanoparticle manufacturing, has been the subject of recent advancements. 14 Because of their high protein content as well as nutritional value, the plant's leaves are especially noteworthy and can be used to treat malnutrition and as an nutritional supplement for athletes. 15 The many medicinal benefits of MO are attributed to its high levels of phytochemicals, including tannins, phenolics, flavonoids, sterols, saponins, carotenoids, tocopherols, as well as chlorophyll. Its anti-obesity, anti-cancer, anti-diabetic, anti-hypertensive, anti-arthritic, anti-inflammatory, as well as wound-healing qualities make it useful for the development of functional foods, cosmetic products, biodiesel, as well as pharmaceutical applications. 16 This work investigates the antioxidant as well as α-glucosidase inhibitory characteristics of MO leaves, which have long been utilized for their therapeutic benefits.
Materials and Methods
Plant-based Material Collection Along with Extraction
Fresh MO leaves were purchased from the Hafr Al Batin store in the Eastern Province of Saudi Arabia and were allowed to air dry in the shade. Extracts were prepared as described by Kumaran & Joel. 17 Ten grams of the crushed plant samples were taken out in 100 mL of 99% ethanol for 48 h with constant shaking. The extracts were then filtered by Whatman No. 4 paper as well as concentrated under vacuum at 40 °C via a rotary evaporator from Buchi R-215-Switzerland. Furthermore, the ethanol extracts were stored at 4 °C in the dark till further use.
GC-MS Analysis
The extraction method began with 10 g of dried, pulverized plant material, which was extracted separately with 250 mL of Ethanol (EtOH) as well as 250 mL of Ethyl acetate (EtAc). Each extraction took place at room temperature for 24 h with continuous stirring at 250 rpm. The resultant mixes were filtered via Whatman filter paper before being condensed to dryness with a rotary evaporator from Buchi- Germany). For analysis, 10 mg of each dried extract was derivatized using 150 μL of MSTFA (N-Methyl-N-trimethylsilytrifluoroacetamide, containing 1% Trimethyl-chlorosilane) as well as 150 μL of hexane, afterward incubated at 60 °C for 30 min. After filtering via a 0.22 μm syringe filter, then the solutions were transferred to 1.5 mL HPLC vials. GC-MS analysis was performed by injecting 1 μL samples. The analysis was conducted using Lab-Solutions GC-MS software on a Shimadzu with GC-MS-QP 2010 Ultra System (Kyoto, Japan). A Restek Rtx®-5 ms column, 30.0 m, 0.25 mm, and 0.25 μm, was employed for the separation of metabolites. In addition, Helium (99.9% pure) was employed as the carrier gas at a steady flow rate of 1.0 mL per min. Furthermore, the temperature program ran for 51.43 min, starting at 60 °C, held for three minutes, then ramping up to 140 °C at 7 °C/min, as well as finally reaching 300 °C at 5 °C per min along with a final hold of five min. Moreover, the temperature of the ionization and interface was kept at 250 °C. The full-scan mode was used to detect metabolites within the mass range of 50-650 amu. Additionally, a total volume of 10 μL was injected via a Shimadzu AOC-20i auto-injector in splitless mode. Compounds were identified by matching GC total ion chromatograms (TIC) as well as fragmentation patterns with libraries like NIST/EPA/NIH Mass Spectral Library (NIST 17).18-21
The Measurement of Total Phenols
The total phenolic content of the extract was measured using the Folin-Ciocalteu reagent, with gallic acid as the standard reference. 22 A 0.2 mL aliquot of the 10 mg/mL extract was combined with 1.5 mL of the Folin-Ciocalteu phenol reagent. After allowing the mixture to react for 5 min, 1.5 mL of 6% sodium carbonate was added, and the solution was left to stand at room temperature for 90 min. The absorbance was measured at 725 nm, and the results were expressed as mm of gallic acid equivalent per g of extract (mg GAE/g). All assays were performed in triplicate.
Assessment of Antioxidant Activities
The antioxidant activities of MO extract, dissolved in ethanol, were evaluated based on their ability to scavenge 2,2-diphenyl-1-picrylhydrazyl (DPPH) as well as 3-ethyl-benzothiazoline-6-sulfonic acid (ABTS) radicals. Additionally, both DPPH as well as ABTS assays were conducted as described by Al-Mustafa and Al-Thunibat. 23
DPPH Assay
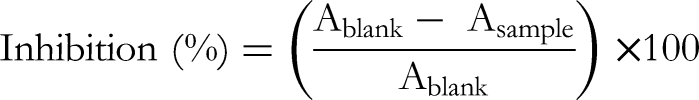
For the DPPH assay, 50 µL of each extract (final concentration of 10 to 50 µg/mL) was mixed with 950 µL of DPPH solution. After 30 min of incubation in the dark, the absorbance was observed at 517 nm through a Hitachi spectrophotometer. The DPPH radical scavenging properties were calculated via the following formula:
ABTS Assay
For the ABTS assay, an ABTS mixture was prepared by mixing 2.45 mM ABTS in 2.45 mM potassium persulfate in water. The prepared solution was incubated at room temperature for 12 h in the dark. After that, the absorbance of the mixture was adjusted to 0.7 ± 0.02 at 734 nm by diluting it in 5 mM phosphate-buffered saline (PBS) at pH 7.4. Then, 50 µL of each extract (final concentration of 10 to 50 µg/mL) was mixed with 950 µL of the ABTS mixture, followed by 10 min of incubation in the dark. The absorbance was then measured at 734 nm using a spectrophotometer. The radical scavenging impact was calculated using the same formula as for the DPPH assay. 25
Measurement of α-glucosidase Inhibition Effectiveness
The α-glucosidase Induction test was performed according to the method described by Indrianingsih et al (2015),
26
with some alterations. A 1500 μL solution containing 0.1 M phosphate buffer (pH 7), 5 mM p-nitrophenyl-α-D-glucopyranoside, and 1 U/mL α-glucosidase, with or without varying concentrations of MO ethanolic extract, was continuously measured at 405 nm for 10 min. Acarbose was utilized as the positive control. The percentage inhibition was identified using the following equation:
Statistical Analysis
The results of this study are presented as means ± standard deviation (SD). Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS, version 16), and statistical significance was assessed with a P-value of less than .05.
Results
GC-MS Detection of Phytochemicals
GC/MS examination of the extracts’ chemical composition revealed that the EtOH and EtAc extracts had different profiles. The EtOH extract contained 160 compounds, while the EtAc extract had 140, with 25 compounds shared between both extracts. These chemicals were identified by comparing the mass spectrum patterns with those in the Wiley 9 and NIST reference libraries. Table 1 displays the relative abundances of the common compounds as peak area percentages, and Figure 1(a and b) show the chromatographic patterns corresponding to these abundances. Figure 2 illustrates the clarified molecular structures of the main compounds that were identified.

GC–MS Profile of the Plant Extracts by a- EtOH and b- EtAc.

Chemical Compounds of the Major Identified Components in EtOH and EtAc Extracts by GC-MS.
GC-MS Assessment of EtOH and EtAc Extracts of MO Extract.

Total Phenolic Amount
The Folin-Ciocalteu reagent was employed to quantify the total phenolic components in the EtOH-MO extract, which revealed a total phenolic content of 157.14 ± 14.46 mg GAE/g.
Evaluation of Antioxidant Activity
MO's antioxidant capacity reflects the effectiveness of its oxygen radical-neutralizing components, particularly its phenolic constituents (Figures 3 and 4).

Antioxidant Activity of EtOH-MO Extracts Determined Based on DPPH % of Inhibition.

Antioxidant Activity of EtOH-MO Extracts Determined Based on ABTS% of Inhibition.
Assessment of α-Glucosidase Inhibitory Activity
The in vitro inhibition of α-glucosidase was evaluated to confirm the traditional use of MO extract in diabetes treatment. Patients with non-insulin-dependent diabetes mellitus use α-glucosidase inhibitors (Figure 5), which delay carbohydrate digestion as well as absorption, thereby reducing postprandial hyperglycemia.23,27,28

In Vitro Effect of EtOH-MO Extract on α-Glucosidase Activity (1 mg/mL at Absorbance 405 nm).
Discussion
This study identified several key chemicals in MO extracts, with Palmitic acid and α-Linolenic acid being the most prevalent. The GC/MS analysis revealed distinct profiles for the EtOH and EtAc extracts (Table 1). Both extracts exhibited high levels of Palmitic acid, a common saturated fatty acid found in various organisms, with concentrations of 3.40% in the EtAc extract and 3.57% in the EtOH extract. Additionally, α-Linolenic acid was significantly present at 2.95% in the EtOH extract and 2.85% in the EtAc extract, contributing 6.97% and 5.80% of the total extracts, respectively. Another important compound identified was N,N-Dimethyloctanamide, with concentrations of 0.14% in the EtOH extract and 0.40% in the EtAc extract. Notably, N,N-dimethyloctanamide may interact with the enzyme's active site, potentially inhibiting its activity and slowing the conversion of disaccharides to glucose.
These findings highlight the potential health benefits of MO extracts and underscore the importance of further investigation into their biochemical properties and therapeutic applications. The biological implications of these findings are significant, particularly concerning α-Linolenic acid. Prior studies indicate that α-Linolenic acid, along with other unsaturated fatty acids like Palmitoleate, protects renal proximal tubular cells from cell death and endoplasmic reticulum (ER) stress induced by Palmitic acid. This protective mechanism involves the reduction of critical ER stress markers, including phosphorylated eIF2α, CHOP, and GRP78. Furthermore, these protective effects extend to other ER stress inducers, such as salubrinal and tunicamycin, suggesting that unsaturated fatty acids play a vital role in maintaining cellular homeostasis by blocking ER stress pathways and preventing apoptotic cell death.27,29-34
The total phenolic content in the EtOH-MO extract was quantified using the Folin-Ciocalteu reagent, yielding a measurement of 157.14 ± 14.46 mg GAE/g. This finding highlights the significant concentration of phenolic compounds, which are well known for their antioxidant properties and potential health benefits. The improved extraction efficiency of the EtOH extract can be attributed to the use of a water-ethanol mixture, 20 which enhances both polarity and the solubility of the phenolic compounds. This combination allows for better solubilization, resulting in a more effective extraction process. Supporting our results, Dehshahri et al28 reported a phenolic content of 88.05 ± 1.08 mg/g in the methanolic leaf extract of Moringa peregrina, underscoring the vital role of phenols from medicinal herbs as significant antioxidants and free radical terminators. Furthermore, The average phenolic content in the ethanolic extracts of MO seeds was 11.85 mg GAE/g, 35 while the flowers contained 19.31 mg/g of gallic acid equivalent total phenolics in dry extract, although these extracts exhibited only moderate antioxidant activity. 36
The antioxidant properties of these phenolic compounds, due to their redox capabilities, enable them to act as reducing agents and singlet oxygen inhibitors. 37 The DPPH method is a reliable technique for evaluating the antioxidant properties of plant extracts. 38 Our findings confirmed that MO exhibited potent antioxidant activity, as demonstrated by the DPPH percentage inhibition and IC50 measurements. The antioxidant capacity of the extract was assessed at 25.33 µg/mL, yielding an IC50 value of 43.01 µg/mL. Additionally, the EtOH extract of MO exhibited substantial antioxidant capacity, as indicated by the ABTS percentage inhibition values and IC50 levels, shown in Figure 4. Further antioxidant analysis demonstrated strong capabilities through two assays. The ABTS test revealed an antioxidant ability of 41.80 μg/mL, with an IC50 value of 21.20 μg/mL, while the DPPH assay yielded an IC50 of 70.05 μg/mL. Although both extracts had higher IC50 values compared to BHT, a widely used synthetic phenolic antioxidant, this finding suggests that the extracts may provide a safer alternative, minimizing the risks associated with enzymatic or lipid modifications, as well as potential carcinogenic and mutagenic effects. 39
MO is acclaimed for its antioxidant properties, with its leaves, seeds, pods, and flowers containing various bioactive compounds that enhance its efficacy. Rich in flavonoids, phenolic acids, and tannins, MO acts as a potent free radical scavenger. Compounds like quercetin and kaempferol are particularly effective in neutralizing reactive oxygen species (ROS). Additionally, high concentrations of vitamins C and E further bolster antioxidant activity by preventing lipid peroxidation and protecting cellular structures from oxidative stress. Bioactive compounds in MO, including Tetratetracontane, promote the production of endogenous antioxidants like glutathione and catalase, thereby reducing oxidative damage to DNA, proteins, and lipids. In vitro studies have shown that MO leaf extracts exhibit significant DPPH radical scavenging effects.6,37-41 In vivo studies involving animal models treated with MO leaf extract reported reduced levels of oxidative markers, such as malondialdehyde (MDA), alongside increased activity of antioxidant enzymes.42,43 Furthermore, supplementation with MO powder or extracts has been linked to reduced oxidative stress markers and improved antioxidant status in individuals experiencing oxidative stress due to chronic conditions or environmental factors. 44
The EtOH-MO extract displayed the highest α-glucosidase inhibition (IC50 = 15.01 µg/mL), demonstrating a clear, concentration-dependent effect on α-glucosidase activity, as depicted in Figure 5. Previous investigations have highlighted the hypoglycemic effects of compounds like β-sitosterol and stigmasterol found in Dillenia indica L. 34 Furthermore, phenolic compounds with flavonoid structures have been shown to regulate diabetes by improving glucose and lipid levels. 43 Notably, compounds such as quercetin, epicatechin, kaempferol, and naringenin effectively inhibit the α-glucosidase enzyme, a crucial target in antidiabetic treatments.45-47 Research by Jiménez et al (2016) 48 confirmed that high-polarity extracts of Hamelia patens were more effective inhibitors compared to low-polarity extracts. The pathogenesis of diabetes mellitus often results in the production of ROS, leading to glucose oxidation, increased lipid peroxidation, and heightened insulin resistance. The antioxidant activity of phenolic compounds may therefore play a role in managing and preventing the pathogenesis of diabetes mellitus. 49
Our study highlighted the presence of N,N-dimethyloctanamide in MO leaves, a compound with potential α-glucosidase inhibitory properties. Such inhibitors are essential for managing postprandial hyperglycemia in Type 2 Diabetes Mellitus (T2DM) by delaying carbohydrate breakdown and glucose absorption. The interaction of N,N-Dimethyloctanamide with the enzyme's active site could decrease its activity, slowing the conversion of disaccharides into glucose. Additionally, our study identified significant constituents in MO, including α-Linolenic acid, Stearic acid, 3-octenoic acid, and Tetradecanoic acid. Previous studies have noted that Tetradecanoic acid can inhibit α-glucosidase by up to 78%. 50 ,51 Both Palmitic and Tetradecanoic acids have been recognized as significant fatty acids contributing to α-glucosidase inhibition.50,51
While this study on the leaves of MO provides valuable insights into its strong antioxidant and α-glucosidase inhibitory properties, it has certain limitations. Firstly, there is a need for future molecular docking studies to elucidate the exact mechanisms of α-glucosidase inhibition by the fatty acids present in the extracts. These studies could help clarify interactions involving hydrogen bonding, hydrophobic interactions, or salt bridging. Furthermore, although mechanistic insights into these compounds are currently limited, similar derivatives have shown promise in enhancing glycemic control and mitigating diabetic complications through improved metabolic regulation.51,52,53 This study is among the first to conduct a comprehensive GC-MS assessment of the EtOH and EtAc extracts of MO, which sheds light on their potential health benefits. Despite these limitations, the findings contribute valuable insights into the therapeutic applications of MO, particularly regarding its antioxidant properties and α-glucosidase inhibition. Further research is essential to explore these mechanisms and validate the therapeutic potential of MO in clinical settings.
Conclusion
The leaves of Moringa oleifera are a potent natural treatment that has strong antioxidant and α-glucosidase inhibitory properties. According to the study, ethanol and ethyl acetate extracts have unique chemical profiles with high levels of palmitic acid and α-linolenic acid, which support their potential as medicines. With a total phenolic concentration of 157.14 ± 14.46 mg GAE/g, MO's remarkable antioxidant ability is highlighted. Robust DPPH and ABTS tests confirmed this capability, demonstrating efficient free radical neutralization. For instance, the EtOH extract showed excellent α-glucosidase inhibition (IC50 = 15.01 µg/mL), confirming its long-standing use in diabetes treatment by postponing the absorption of carbohydrates and lowering postprandial hyperglycemia. With its abundance of phenolic compounds and beneficial fatty acids, MO is a great option for natural diabetes preventive and treatment methods. MO is a useful addition to the toolkit of natural therapeutic substances because of its many health benefits as it could be a highly effective natural antioxidant source, contributing to health promotion and disease prevention. Its diverse bioactive compounds offer promising applications in nutrition, medicine, and industry, which demand more research. The effectiveness of MO in clinical settings and its potential for incorporation into dietary supplements should be investigated in future research.
Footnotes
Acknowledgements
For their unwavering support during the research investigation, the researchers would like to thank the Deanship of Scientific Research at their linked universities.
Author Contributions
Conceptualization, writing the original draft, reviewing and editing: L.H., A.A., I.A., R.A., F.A., H.A., G.M., M.H. Formal analysis, investigations, funding acquisition, reviewing, and editing: M.S., M.M., G.M., A.A., H.M. Resources, data validation, data curation, and supervision: L.H., A.A., G.M., F.A.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This article contains all of the data created or examined during this investigation.
Statement of Informed Consent
This article does not use human subjects, and informed consent is not relevant.