Abstract
This is a visual representation of the abstract.
Introduction
Acute kidney injury (AKI) is a common renal disease characterized by a rapid decline in renal function, ultimately leading to acute renal failure and even causing other types of organ failure. AKI causes 2 million deaths worldwide every year and is a serious global disease. 1 Currently, there are no effective treatments or preventive measures against AKI. Numerous research reports indicate that drugs, especially antitumor drugs, and antibiotics are some of the important causes of AKI due to obvious side effects of these agents on the renal tissue. The results of clinical diagnostics indicate that cisplatin (CDDP) causes drug-induced renal injury. Clinical studies have shown that cisplatin has dose-dependent nephrotoxicity, and the incidence rate of renal damage induced by cisplatin in the cases of clinical hemotherapy ranges from 25% to 35%, thus greatly limiting clinical applications of the drug. 2 Oxidative stress caused by mitochondrial dysfunction, the accumulation of intracellular reactive oxygen species (ROS), and inflammation was linked to pathophysiological changes common in cisplatin-induced AKI (CIAKI). Entry of cisplatin into renal tubular cells provokes oxidative stress followed by inflammation, leading to cisplatin-induced renal dysfunction. 3 In addition, the characteristics of cisplatin-induced inflammation and oxidative stress are not conducive to normal kidney morphology and function, resulting in AKI. 2
In addition to common oxidative stress and inflammation, the regulatory role of macrophages in immune regulation in the mechanism of AKI cannot be ignored. Macrophages are divided into proinflammatory (M1) and anti-inflammatory (M2) macrophages 4 and have complex effects on the manifestations and progression of AKI due to functional plasticity of these cells. 5 Previous studies have shown that macrophages may be effectors in animal models of AKI induced by ischemia/reperfusion. 6 Renal tubular cell death occurs in AKI. M1 macrophages primarily release proinflammatory mediators and damage the proximal tubule of the outer layer of the renal medulla during the early stages of AKI. Macrophages amass and consume apoptotic cells or their released components as the disease advances. 7
Mango is a very popular tropical fruit. The ancient Chinese medicine book “Compendium of Materia Medica” recorded the medicinal effect of mango, including benefit the stomach, stop vomiting and nausea, so it is a widely recognized natural product for both medicine and food. Recent studies have found that mangiferin(MF; chemical formula C19H18O11), a representative compound of mango, has anti-inflammatory, antioxidant, and immunoregulatory effects. 8
MF belongs to the group of organic xanthones and antioxidant effects on oxidative stress-related nephropathy. 9 MF can alleviate renal inflammation and oxidative stress, alleviate renal interstitial fibrosis and functional impairment. 10 Previous studies have shown that MF alleviates AKI, including CIAKI, through antioxidant and anti-inflammatory effects.11–13 However, only a few studies have explored the potential therapeutic mechanisms of the effects of MF in addition to suppression of oxidative stress and inflammation, such as regulatory mechanisms of macrophages in AKI mentioned above. The study of Akter S et al 14 suggested that MF may be able to prevent inflammatory response and cell apoptosis in a rat spinal cord injury model. Therefore, we speculated that the protective mechanism of action of MF on CIAKI is not limited to the antioxidant and anti-inflammatory effects.
The present study is different from the previous studies, which have been focused on the antioxidant and anti-inflammatory effects of MF. The aim of this study was to explore additional potential mechanisms of action of MF in the treatment of CIAKI by combining the transcriptome and network pharmacology methods and to explore whether this protective mechanism is related to immunomodulatory characteristics of macrophages.
Materials and Methods
Experimental Animals
As shown in Figure 1, 8-weeks male C57BL/6 mice weighing 21–24 g (n = 24; SLAC Lab Animal Co., Ltd, Shanghai, China) were divided into four groups and housed in a pathogen-free environment with a 12-h light/12-h dark cycle. Mice had unrestricted access to food and water. All animal studies followed the National Institutes of Health's “Guidelines for the Care and Use of Laboratory Animals”. The animal study was reviewed and approved by the Committee on Ethics of Medical Research, Naval Medical University (registration number: NMUMREC-2021-006). Mice were divided into four groups of six animals each: the control (CON), CIAKI (CDDP), low-dose MF therapy (LMF), and high-dose MF treatment (HMF) groups. According to the protocol of Holditch SJ et al, 2 mice in the CDDP, LMF, and HMF groups were treated with an intraperitoneal (i.p.) injection of cisplatin (15 mg/kg; HPLC ≥ 98%; Ye Yuan Biotechnology Co., Ltd, Shanghai, China), and in the CON group mice were treated with equal volume of physiological saline solution. According to the protocol of Hu QH et al, 15 immediately after i.p. injection of cisplatin, mice in the LMF and HMF groups were treated with MF (75 and 150 mg/kg, respectively; HPLC ≥ 98%; Ye Yuan Biotechnology Co. Ltd, as shown in Figure 2(A)). I.p. injection of MF solution in saline at the concentrations of 18.75 mg/mL and 37.5 mg/mL were administered to mice. After 24 h, the right kidney was promptly taken out and store in the −80 °C refrigerator for further biochemical analysis; the blood was also collected and centrifuged. The left kidney was harvested for pathological evaluation and extensively fixed in 10% buffered formalin.


Biochemical Analysis of Renal Function
Serum creatinine (Scr) and blood urea nitrogen (BUN) levels were assayed by an automatic biochemical analyzer (Chemray 240, Shenzhen, China).
Real-Time Polymerase Chain Reaction (PCR)
Cryopreserved tissue samples were ground, and total RNA was extracted from the kidney tissue using TRIzol reagent. Then use reverse transcription reagents to reverse transcribe RNA into cDNA (Yeasen Bio, Shanghai, China). SYBR Green assay kit (Yeasen Bio) and ABI Prism 7500 system (Applied Biosystems, Foster City, CA, USA) were used for quantitative real-time PCR (qPCR). qPCR was carried out with the forward and reverse primers, cDNA, H2O, and SYBR Green PCR Master Mix. The amplification was performed at 50°C for 2 min and 95°C for 10 min, followed by 45 cycles at 95°C for 15 s and 60°C for 1 min.
Kidney injury molecule 1 (KIM-1; gene ID 171283) is a biomarker of AKI. The KIM-1 primer sequences were as follows: (forward) TCAGGAAGCTGAGCAAACATTA and (reverse) AGTGCCATTCCAGTCTGGTT. Relative gene expression was quantified using the 2-ΔΔCt technique. Actin was used as a housekeeping gene.
Hematoxylin and Eosin (HE) and Immunohistochemical (IHC) Staining
After deparaffinization of the paraffin-embedded sections in water, the tissue sections were placed in a pressure cooker containing citric acid antigen retrieval buffer (pH 6.0) for antigen retrieval, followed by immersion in 3% hydrogen peroxide solution to destroy endogenous peroxides. Enzymes, BSA, or serum were used to block the sections, and the corresponding blocking solutions were discarded. The slides were incubated overnight at 4°C with primary antibodies (all from Servicebio, China) against interleukin-2 (IL-2, 1:200), interleukin-9 (IL-9, 1:200), monocyte chemoattractant protein–1 (MCP-1, 1:200), colony stimulating factor 1 (CSF1, 1:200), matrix metalloproteinase 13 (MMP13, 1:200), nuclear factor kappa-B (NF-κB, 1:200), and macrophage inflammatory protein 1α (MIP-1α, 1:200).
Immunofluorescent Staining (IF)
Water was used to deparaffinize the paraffin-embedded sections, and the tissue sections were heated in the presence of citric acid antigen retrieval solution. BSA was applied dropwise to the dry sections and incubated for 30 min. After gently shaking off the blocking solution, the sections were incubated overnight at 4°C with rat anti-mouse F4/80 antibodies (Servicebio, 1:500) and subsequently with a secondary antibody (Servicebio, 1:400). The slides were washed three times in PBS (pH 7.4). The slides were dried, and 4′,6-diamidino-2-phenylindole (DAPI) staining solution was used to stain the sections for 10 min at room temperature in dark.
Cytokines Assay
The levels of inflammatory factors in the renal tissue were measured by Cytokine Immunoassay. Renal tissue was homogenized in PBS containing protease and phosphatase inhibitors (Shanghai Ya Mei Biological Medicine Science and Technology Co., Ltd, Shanghai, China) and centrifuged to obtain the supernatant. Bio-Plex Pro Mouse Cytokine 23-plex Assay (M60009RDPD, Bio–Rad) was used to measure the levels of 23 mouse cytokines, among them, the cytokines with significant changes are IL-2, IL-10, IL-13, MCP1, MIP-1α, and keratinocyte-derived chemokine (KC).
Gene Expression Microarray Analysis16–21
TRIzol reagent (Invitrogen), a RNeasy Mini kit (Qiagen), and other kits were used to extract total RNA from all samples. An Agilent 2100 bioanalyzer (Agilent Technologies, Palo Alto, CA, USA), NanoDrop (Thermo Fisher Scientific, Inc.), and 1% agarose gels were used to quantify and determine the quality of the total RNA preparations. Subsequently, a library was generated using total RNA with a 1 µ g RNA integrity value higher than 6.5. The libraries for next-generation sequencing were prepared according to the manufacturer's protocol. A poly(A) mRNA magnetic isolation module or an rRNA removal kit were used for isolation of poly(A) mRNA. The first chain synthesis reaction buffer and random primers are used for mRNA fragmentation and priming.
The synthesis of the first strand cDNA was accomplished using ProtoScript II reverse transcriptase, while the synthesis of the second strand cDNA was accomplished using a mixture of second strand synthases. After purification on the beads, double-stranded cDNA was treated with End Prep enzyme mix to modify both ends and to add dA-tailing in a single reaction; subsequent the T-A ligation procedure was used to attach the adaptors to both ends. The beads were used for size selection of adaptor-ligated DNA, and the fragments of ∼420 bp (with an insert size of approximately 300 bp) were retained. The P5 and P7 primers were used to amplify all samples by PCR; bridging PCR used the two primers to anneal with the flow cell, and the P7 primer with a six-base indicator was used for multiplexing. The PCR products were purified using the beads, validated using a Qsep100 analyzer (Bioptic, Taiwan, China), and was quantified by a Qubit3.0 fluorometer (Invitrogen, Carlsbad, CA, USA). After multiplexing, load the libraries with various indexes onto the Illumina HiSeq instrument according to its instructions (Illumina, San Diego, CA, USA). A 2 × 150 bp paired-end (PE) configuration was used for sequencing; HiSeq Control software (HCS), OLB, and GAPipeline-1.6 (Illumina) running on a HiSeq instrument were used for imaging and base calling. GENEWIZ was used to process and analyze the sequences.
Network Pharmacology
The process of Network Pharmacology followed the methods in our previous published studies.
Statistical Analysis
Gene Expression Microarray Data Analysis
Quality was controlled to eliminate the technical sequences, such as adapters, PCR primers, their fragments, and the base reads lower than 20, and the pass filtering of the data in the fastq format was performed by Cutadapt (V1.9.1) to yield high-quality clean data.
Genome websites, including UCSC, NCBI, and ENSEMBL, with reference genome sequences and gene model annotation files of related species were used for mapping. Then, the reference genome sequence was assessed with Hisat2 (v2.0.1). Finally, the reference genome was assigned based on the clean data by Hisat2 software (v2.0.1).
The transcripts in the fasta format were converted using the known gff annotation archives and subsequent indexing for expression analysis. Then the generated file was used as a reference, HTSeq (v0.6.1) was used to forecast the levels of the gene and isoform expression according to the clean paired-end data.
Differential expression analysis is achieved through a negative binomial distribution model based on the DESeq2 Bioconductor software package. The evaluations of the dispersion and logarithmic fold-variations included the data-boosted prior distributions, and the Padj values of the genes were set to <0.05 to test differentially expressed genes.
Other Data Analysis
The outcomes were analyzed by one-way ANOVA and Tukey's post hoc test; the data are shown as the mean and standard error of the mean (SPSS version 23.0; Inc., Chicago, IL, USA). The graphs were generated using GraphPad Prism (version 9.0, USA). A statistically significant difference corresponded to a P value less than 0.05.
Results
Initially, we evaluated the protective effect of MF on renal injury. Next, we combined the results of DNA microarray analysis and network pharmacology to explore other potential protective mechanisms of MF other than anti-inflammatory effects. Finally, this hypothesis was verified by pathological immunohistochemical and immunofluorescence staining.
Mangiferin Ameliorates Cisplatin-Induced AKI in Mice
To determine whether MF treatment improves renal function in CIAKI mice, the levels of Scr, BUN and KIM-1 were measured as indicators of renal function (Supplementary table 1). As shown in Figure 2(B), Scr and BUN increased in mice of the CDDP group compared with that of the CON group, and significantly elevated in the LMF and HMF group. The expression level of KIM-1 mRNA in the LMF and HMF groups was considerably lower than that in the CDDP group. In addition, HE stained renal tissue sections are imaged by an optical microscope (Figure 2(C)). Interstitial cell infiltration was significantly more severe in the CDDP group than that in the CON group, and it decreased after MF intervention. The improvement was more pronounced in mice of the HMF group than that of the LMF group. Therefore, MF had a protective effect on CIAKI mice.
Mangiferin Reverses the Changes in the Levels of Inflammatory Factors in the Renal Tissue
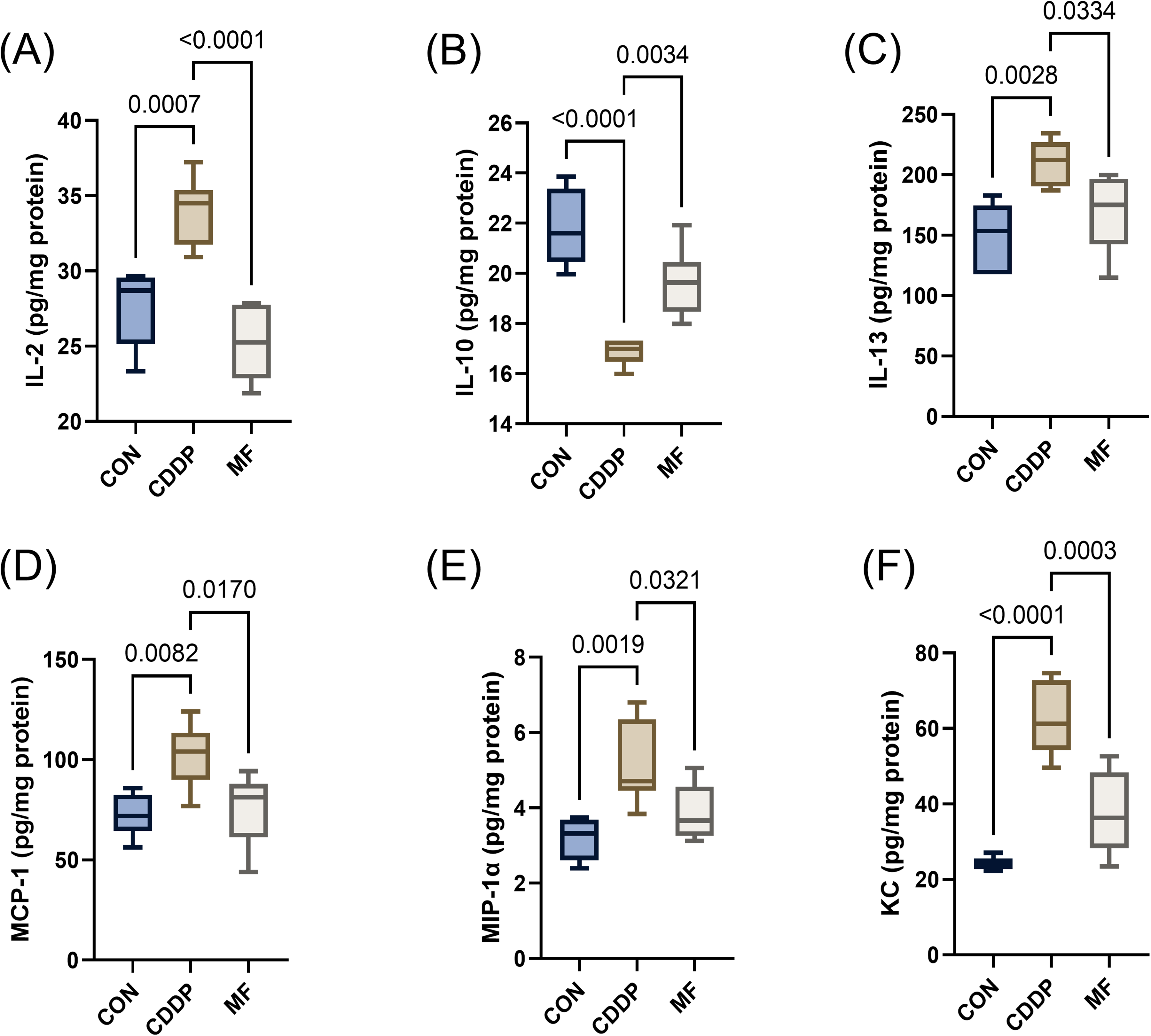
To assess the treatment of MF on inflammatory factors in renal tissue, the levels of inflammatory factors in the renal tissues were determined by a Cytokines Assay kit (Supplementary table 1). The results showed that CDDP group had higher levels of IL-2, IL-13, MCP-1, MIP-1α, and KC in the renal tissues and decreased levels of IL-10 compared with CON group. As an anti-inflammatory cytokine, IL-10 has been suggested to directly relieve CIAKI. 22 After treatment with MF, the changes in the levels of inflammatory factors in the renal tissue were reversed (Figure 3).
Related Targets and Pathways of Mangiferin in Cisplatin-Induced Acute Kidney Injury
The network pharmacology method was used to identify the relevant targets and pathways of MF in the treatment of CIAKI. The STITCH and PharmMapper databases were used to predict the targets of MF, yielding 475 targets after integration and duplicate removal (Supplementary table 2). The top 10 GO terms (Figure 4(A)) and the top 20 KEGG pathways (Figure 4(B)) were identified, and the results are listed in Supplementary table 3. Three GO keywords were associated with the collagen-containing extracellular matrix, leukocyte migration, and signaling receptor activator activity. The results of KEGG analysis showed that the targets of MF were related to cytokine–cytokine receptor interactions and chemokine signaling pathways.
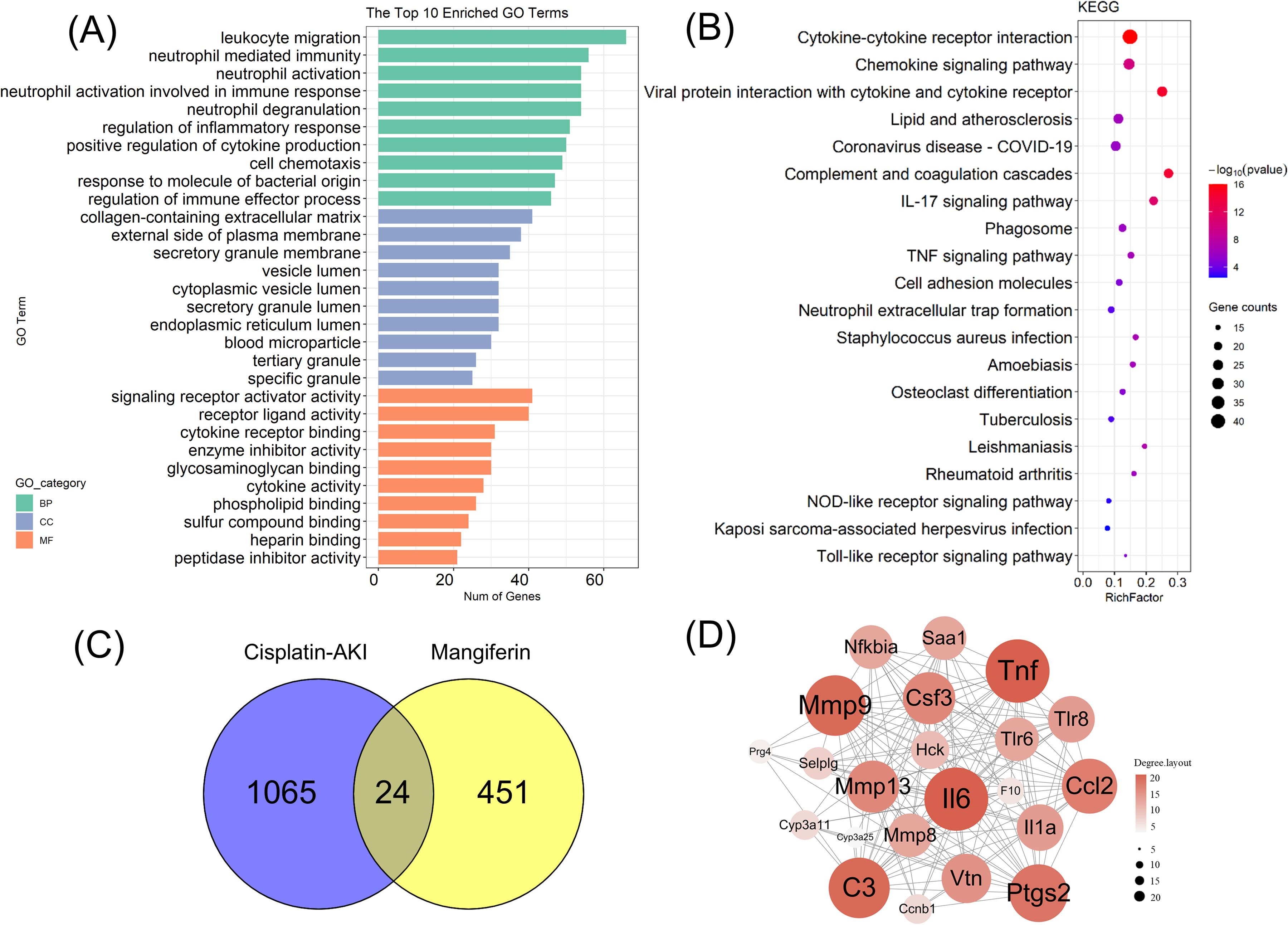
Next, the intersection of CDDP-AKI (Supplementary table 4) and MF targets were examined, and 24 MF-CDDP targets were obtained (Figure 4(C)). In the degree method of Cytoscape software, TNF, IL-6, C3, MMP9, and PTGS2 were the top 5 hub nodes identified by the PPI network as illustrated in Figure 4(D) (Supplementary table 5). CIAKI is directly associated with these nodes.23–27
Mangiferin Influences the Expression of Inflammation- and Macrophage-Related Proteins in Cisplatin-Induced Acute Kidney Injury
The expression of related proteins was detected to clarify the mechanism by which MF influenced CIAKI. Figure 5(A) indicated that the expression of IL-2, IL-9, MMP13, MCP-1, CSF-1, NF-κB, and MIP-1α was considerably higher in the CDDP group than that in the CON group. After MF treatment, the expression of these markers was decreased.

The results of immunofluorescence analysis showed that F4/80-positive cells emitted the green fluorescence signal. The number of F4/80-positive cells in the CDDP group were increased compared to the CON group, and it decreased in the MF group (Figure 5(B)).
Discussion
CIAKI is characterized by high levels of numerous cytokines and immune cell infiltration. These factors promote the progression of AKI mediated by balance of pro- and anti-inflammatory factors. In addition, immune regulation plays an important role in AKI. 28 Therefore, identification of anti-inflammatory and immunomodulatory drugs is very important for prevention of AKI.
In our network pharmacology work, TNF, IL-6, C3, MMP9, and PTGS2 were the top 5 hub nodes in MF treatment of CIAKI. TNF 29 and IL-6 30 are pleiotropic pro-inflammatory cytokines and were upregulated in the kidneys of mice following cisplatin treatment. And Pharmacological inhibition of TNF production, neutralization of TNF, or TNF deficiency attenuated cisplatin-induced cytokine production and kidney damage in mice, suggesting a critical role of TNF in CIAKI. 31 Recent studies 32 have shown that the family of MMPs plays a role in AKI and that MMP9 can activate the PI3 K/AKT pathway to resist apoptosis. Genetic deficiency of C3 prevented immunothrombosis, glomerular filtration rate drop, and ischemic necrosis at 24 h of cholesterol crystal embolism-induced AKI. 33 And syndrome-related AKI mice in the GSE44925 cohort highly expressed the ferroptosis markers ACSL4 and PTGS2. 34
Present studies indicated that mangiferin confers its renoprotective effects against AKI and chronic kidney disease predominantly through protection against inflammation (reducing the inflammatory cytokines like TNF-α, IL-6, IL-18, IL-8), scavenging of ROS in oxidative stress, anti-apoptotic and anti-fibrotic effects in renal fibrosis. 10 MF treatment can also alleviate CIAKI in mice by upregulating the levels of NF-κB, IL2, MCP-1, and MIP-1α. In CIAKI mice, renal neutrophil infiltration is promoted by caspase 1-dependent proinflammatory cytokines, such as NF-κB-dependent cytokines, MCP-1, and MIP-2.22,31,35 The activation of MCP-1 coupled with additional secretion by injured renal parenchymal cells promotes the chemotaxis of acute inflammatory cell populations, such as natural killer cells, neutrophils, monocytes, and T lymphocytes.2,36 This infiltration further increases the concentrations of inflammatory mediators released by neutrophils and mediates additional renal dysfunction. 2 Moreover, an increase in the proinflammatory factor IL2 in AKI mice was consistent with the data of a previous study. 37 Therefore, we speculated that MF may protect the kidney by blocking these proinflammatory mediators, thereby limiting the infiltration of neutrophils during CIAKI. However, the blockade of these proinflammatory mediators can limit neutrophil infiltration during CIAKI, which is not sufficient to completely abolish nephrotoxic AKI. Hernando Gomez et al 38 noted that neutrophils migrate into the peritubular capillaries of the renal cortex and medulla, increasing inflammatory mediators in the peritubular capillaries and aggravating inflammation in the renal tubules. Therefore, we suggest that the protective effect of MF on the kidney is not limited to the suppression of proinflammatory mediators.
According to the data of the literature, monocytes/macrophages exert critical influence on renal injury and renal repair. Monocytes can differentiate into various macrophage subsets, such as M1 or M2 macrophages, in vitro. 39 M1 macrophages release chemokines, proinflammatory cytokines (KC, TNF-α, IL-6, IL-12, and NF-κB-dependent cytokines) and express inducible nitric oxide synthase to mediate renal tubular injury in AKI caused by ischemia/reperfusion (I/R)-induced injury.6,40,41 The experimental results indicated that MF treatment alleviated the upregulation of the levels of KC and NF-κB in CIAKI mice, and we speculate that the protective effect of MF may reduce the proinflammatory effect by downregulating the level of M1 macrophages. In addition, M2 macrophages have anti-inflammatory effects. The STAT3/STAT6 signaling pathway is activated by M2 macrophages, resulting in the production of anti-inflammatory molecules, including IL-10 and TGF-β.42–44 Present study indicated that MF treatment alleviated the CIAKI-induced downregulation of IL-10 levels, and we speculate that the protective effect of MF may achieve an anti-inflammatory effect by upregulating the level of M2 macrophages.
On the other hand, M2 macrophages create extracellular matrix, which is critical in the expansion of renal fibrosis after damage. The results of the present study showed that the levels of CSF-1 and MMP13 were upregulated in the model group and were downregulated after MF treatment. Therefore, we suggest that MF upregulated the level of CSF-1 and stimulated an increase in M2 macrophages, promoting the repair of the renal tubules; continuous expression of these nutritional factors and chemokines by macrophages may promote the retention, survival, and continuous recruitment of macrophages in continuously damaged kidneys.45,46 Additionally, the secretion of nutritional factors activates and supports myofibroblast survival and changes the balance of the extracellular matrix mediated by MMP to form obvious proinflammatory scars. 47
Overall, results of the present study indicated that MF played the anti-inflammatory, antioxidant, and renal protective roles in CIAKI by regulating macrophages. The major limitation of the present study is that we did not elucidate the relevant mechanism how MF played the protective roles in CIAKI by regulating macrophages, which need further experiments to explore.
Conclusion
Mangiferin can relieve cisplatin-induced AKI by regulating inflammatory factors and reducing inflammation. The results of comprehensive network pharmacological analysis and immunopathological verification showed that MF played the anti-inflammatory, antioxidant, and renal protective roles in CIAKI by regulating macrophages. Further exploration is needed to determine the mechanism by which mangiferin exerts its biological activities.
Supplemental Material
sj-xlsx-1-npx-10.1177_1934578X251325035 - Supplemental material for Protective Effect of Mangiferin on Cisplatin Induced Acute Kidney Injury in Vivo
Supplemental material, sj-xlsx-1-npx-10.1177_1934578X251325035 for Protective Effect of Mangiferin on Cisplatin Induced Acute Kidney Injury in Vivo by Jin Cheng, Maoting Li, Chenqi Li, Xiangtong Li, Rui Li, Yinyin Zhang, Hongtao Lu, Zhenzhen Zhao and WenRui Liu in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
We thank Prof. Hui Shen, Xuejun Sun and Zhiyong Guo (Naval Medical University, Shanghai, China) for their technical assistance.
Authors’ Contributions
Zhenzhen Zhao, Jin Cheng, and Hongtao Lu: Study design. Maoting Li, Chenqi Li, and Yinyin Zhan: Methodology. Jin Cheng, Maoting Li, Xiangtong Li, Rui Li, Wenrui Liu, and Hongtao Lu: acquisition, analysis, and interpretation of the data. Maoting Li and Hongtao Lu: Writing - original draft. Jin Cheng, Zhenzhen Zhao, and Hongtao Lu: Writing - review & editing. Jin Cheng, Hongtao Lu, and Wenrui Liu: Funding acquisition.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statements
The animal study has been approved by the Committee on Ethics of Medical Research, Naval Medical University (registration number: NMUMREC-2021-006).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Clinical Research Special General Project of Shanghai Municipal Health Commission (NO.202340097), the Health Service Key Project of Naval Medical Center of PLA(22M3204), the Foundation of Naval Medical University (2022MS003), the Science and Technology Innovation Plan Of Shanghai Science and Technology Commission (23Y11920500), The Young Medical Talents Training Program of Shanghai PudongNew Area Health Commission (PWRq2021-38), the Scientific Research Program of Shanghai Pudong New Area Health Commission (the Youth Program, PW2023B-13)the Talents Training Program of the Seventh People's Hospital, Shanghai University of Traditional Chinese Medicine (QMX2022-01), the National Natural Science Foundation of China (81801955), and the Construction Project of National Comprehensive Reform Pilot Zone for Traditional Chinese Medicine Development in Pudong New Area, Training of Advanced Talents in Integrated Traditional Chinese and Western Medicine (PDZY-2024-0711).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
