Abstract
Introduction
In recent years, with the development of the social economy and changes in diet, the incidence of gouty arthritis (GA), a disease closely related to a high-purine diet and defective purine metabolism, has significantly increased in China. 1 The pathology of GA mainly involves purine metabolism disorders, serum hyperuricemia (HUA), and monosodium urate (MSU) crystal precipitation in joints and synovium. 2 GA, characterized by tissue inflammation, pain, fever, redness, and stiffness, frequently affects the foot and ankle and seriously impairs the quality of life of patients with GA. 3 At present, GA therapies include nonsteroidal anti-inflammatory drugs, colchicine, and glucocorticoids. However, these drugs are associated with severe side effects, such as gastrointestinal disease, renal failure, and myopathy. 4 Therefore, alternative therapies for GA without adverse effects are urgently needed.
Berberine (BBR), an alkaloid isolated from Coptis chinensis and Phellodendri chinensis, has been widely used in China to treat infectious and inflammatory diseases including diarrhea and gastroenteritis.5,6 BBR is also effective in ameliorating metabolic diseases, such as HUA, hyperlipidemia, type 2 diabetes mellitus, and obesity.7–9 Recent studies have shown that BBR can inhibit the inflammatory response induced by MSU crystals and improve GA symptoms. Stimulation with MSU crystals significantly increased the levels of interleukin-1 beta (IL-1β), interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) in a GA mouse model and patients. 10 By activating the antioxidant pathway of nuclear factor erythroid-2-related factor and inhibiting the activation of nucleotide-binding oligomerization domain receptor 3 (NLRP3) inflammatory body, BBR reduced the expression of inflammatory factors. 11 Sanmiao pill (SMP), a traditional Chinese prescription, has been proven to be effective for treating GA, and its crucial anti-inflammatory ingredient BBR is concentrated in the knee and ankle joints of animals.12–14 Although both in vivo and in vitro studies have validated that BBR ameliorates GA, the systematic and multi-target mechanisms underlying the effects of BBR against GA have not been fully elucidated.
Network pharmacology is based on the interaction between several different disciplines, including bioinformatics, network biology, and polypharmacology. 15 Intercommunications between drug components and disease targets are scientifically analyzed in network pharmacology, guiding the prediction of potential pathways for intervention. Owing to the complex components and mechanisms of traditional Chinese medicine (TCM), network pharmacology analysis has been frequently used in TCM investigations. 16 Recent studies have utilized network pharmacology to explore possible genes, proteins, and functional pathways, and then validated the network pharmacology results using animal experiments, providing evidence for further applications of BBR as a novel therapeutic agent in the treatment of GA.
Materials and Methods
Prediction of BBR Targets
The targets of BBR were identified using the TCM Systems Pharmacology Database and Analysis Platform (TCMSP, https://old.tcmsp-e.com/tcmsp.php) and the Comparative Toxicogenomics Database (CTD, http://ctdbase.org/). In TCMSP, an oral bioavailability (OB) of ≥30% and drug likeness (DL) of ≥0.18 were considered screening criteria to select BBR targets. 17 The results obtained from the two databases were combined, and the targets of BBR were obtained by removing duplicates.
Acquisition of GA Targets
DisGeNET v7.0 (https://www.disgenet.org/) is a publicly available database that provides one of the largest collections of genes and mutations associated with human diseases. The current version includes 1 134 942 interactions between 21 671 genes and 30 170 illnesses. 18 GeneCards (https://www.genecards.org/) is a discovery platform that integrates gene-centric data from approximately 150 Web sources. “Gouty Arthritis,” “Podagra,” and “Chronic Gouty Arthritis” were searched as keywords in the above two databases to identify disease targets associated with GA, and the results obtained were then merged for de-duplication.
Construction of the Network of BBR-GA Common Targets
The common targets of BBR and GA were identified using the R platform, and the Venn diagram was obtained by using the DeepVenn platform (http://www.deepvenn.com/). The network of BBR-GA common targets was visualized by Cytoscape software 3.7.2 (https://cytoscape.org/). In the network, nodes represent the targets of BBR and GA while the edges indicate the correlations.
Construction of the PPI Network
Common targets for BBR and GA were uploaded to the STRING 11.5 platform (https://cn.string-db.org/) to generate the PPI network. In the function of “Multiple Proteins,” “Organism” was selected as “Homo sapiens,” and the “Required score” was set with the highest confidence (0.9) to construct the PPI network. The node degree value and information reflecting the relationship between target proteins were obtained. The core proteins in the network were mined based on the degree values of the network nodes.
Annotation of Biological Function and Signal Pathway
The common targets of BBR and GA were imported into the Database for Annotation, Visualization, and Integrated Discovery (DAVID, https://david.ncifcrf.gov/home.jsp) Bioinformatics Resources 6.8 to analyze the Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) signaling pathways. The biological function annotations of GO included biological processes, cellular components, and functional categories. A P-value of < .05 was used to select GO terms and KEGG pathways for visualization.
Animal Experiments and Sample Collection
Male specific-pathogen-free (SPF) Sprague-Dawley (SD) rats (average age: 8 weeks, weighing 290-310 g) were housed in ventilated cages at a regulated room temperature of 23 ± 2 °C and humidity of 55 ± 5% under a 12 h light/dark cycle. Normal chow and clean water were provided ad libitum. A yeast extract diet combined with potassium oxonate (750 mg/kg/d) (Yuanye Bio-technology Ltd, Shanghai, China) was used to induce HUA in rats. All animal experiments were approved by the Animal Experimental Ethics Committee of Zhejiang Chinese Medical University (2019012806) and complied with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
BBR (Yuanye Bio-technology Ltd), allopurinol (AL) (Yuanye Bio-technology Ltd), or control vehicle was daily administered to HUA rats via oral gavage for two weeks. The BBR dose was calculated using the following formula:
Cytokine and Biochemical Analyses
The concentrations of serum IL-6, IL-1β, and interleukin-8 (IL-8) were measured using an enzyme-linked immunosorbent assay (ELISA). ELISA kits (Meimian Industry) were maintained at room temperature for 30 min before usage. Standard solutions and test samples were added to the corresponding microwells and the plates were incubated at 37 °C for 30 min before quick washing (five times). Subsequently, the detection reagent and the substrate solution were added to the wells. Finally, the absorbance was measured at 450 nm immediately after the reaction was stopped with 50 µL stop solution. Serum uric acid levels were measured using appropriate standard kits according to the manufacturer's instructions.
Statistical Analysis
Data were analyzed using GraphPad Prism 8 (GraphPad Software Inc.) and are shown as the mean ± SEM. The Kruskal–Wallis test was used to compare differences among groups, followed by a false discovery rate (FDR) correction for multiple comparisons. Two-tailed P-values of < .05 were considered statistically significant.
Results
Screening BBR-GA Common Targets
In total, 272 BBR target genes were identified, with 17 genes from the TCMSP database and 255 genes from the CTD database. A total of 369 GA targets were screened after removing duplications by searching DisGeNET (208 genes) and GeneCards (201 genes). By using the R language and DeepVenn platform, 33 BBR-GA common targets were obtained (Figure 1a). As shown in Figure 1b, the network of BBR-GA common targets included nuclear factor-kappa B (NF-κB) subunit RelA, IL-6, IL-1β, mitogen-activated protein kinase 1 (MAPK1), and TNF.

Common targets of BBR and GA. (A) Venn diagram. (B) Network diagram of common targets. BBR, berberine; GA, gouty arthritis.
PPI Network Analysis of the BBR-GA Common Targets
To identify the key targets of BBR and GA, the STRING database was used to construct the PPI network. The network obtained was composed of 32 nodes and 63 edges, in which the nodes and edges represented targets and interactions, respectively. Nodes with more connecting edges are more crucial in the network (Figure 2a). The degree value of each node was calculated, with an average degree value of 3.94. As previously reported, 20 targets with degree values higher than the average were selected as key targets of BBR and GA. As shown in the bar chart (Figure 2b), targets such as RelA, toll-like receptor-4 (TLR-4), IL-6, TNF, MAPK1, and IL-1β had high degree values, suggesting that they are key targets in the PPI network.

PPI analysis of common targets of BBR against GA. (A) PPI network of BBR and GA. (B) Bar chart of PPI targets with degree values greater than average. BBR, berberine; GA, gouty arthritis; PPI, protein-protein interaction.
GO Annotations of the BBR-GA Common Targets
The common targets of BBR and GA were input into the DAVID platform to perform GO annotation, containing biological processes, cellular components, and function categories. The results were visualized using bar charts, and 229 GO terms were discovered, with a P-value of < .05. The most significant GO terms were inflammatory response, cellular response to lipopolysaccharide (LPS), immune response, LPS-mediated signaling pathway, activation of MAPK activity, cellular response to TNF, positive regulation of NF-κB transcription factor activity, positive regulation of IL-6 production, and cytokine activity (Figure 3). These results suggest that the common targets of BBR and GA mainly participate in the regulation of inflammation.

Go annotation of common targets of BBR and GA. (A) Biological processes. (B) Cellular components. (C) Function categories. GO, Gene Ontology; BBR, Berberine; GA, Gouty arthritis.
KEGG Pathway Analysis of the BBR-GA Common Targets
According to the results retrieved from the DAVID platform, 64 KEGG pathways were enriched in common targets of BBR and GA using a P-value of < .05 as the screening condition. The bubble plot visualizes the first 30 pathways, with the color of dots representing -log 10 (P-value) and the size representing the number of genes. The enriched pathways were mainly related to inflammatory regulation, such as the TNF, TLR, NF-κB, and MAPK signaling pathways, cytokine–cytokine receptor interaction, rheumatoid arthritis, and osteoclast differentiation (Figure 4).
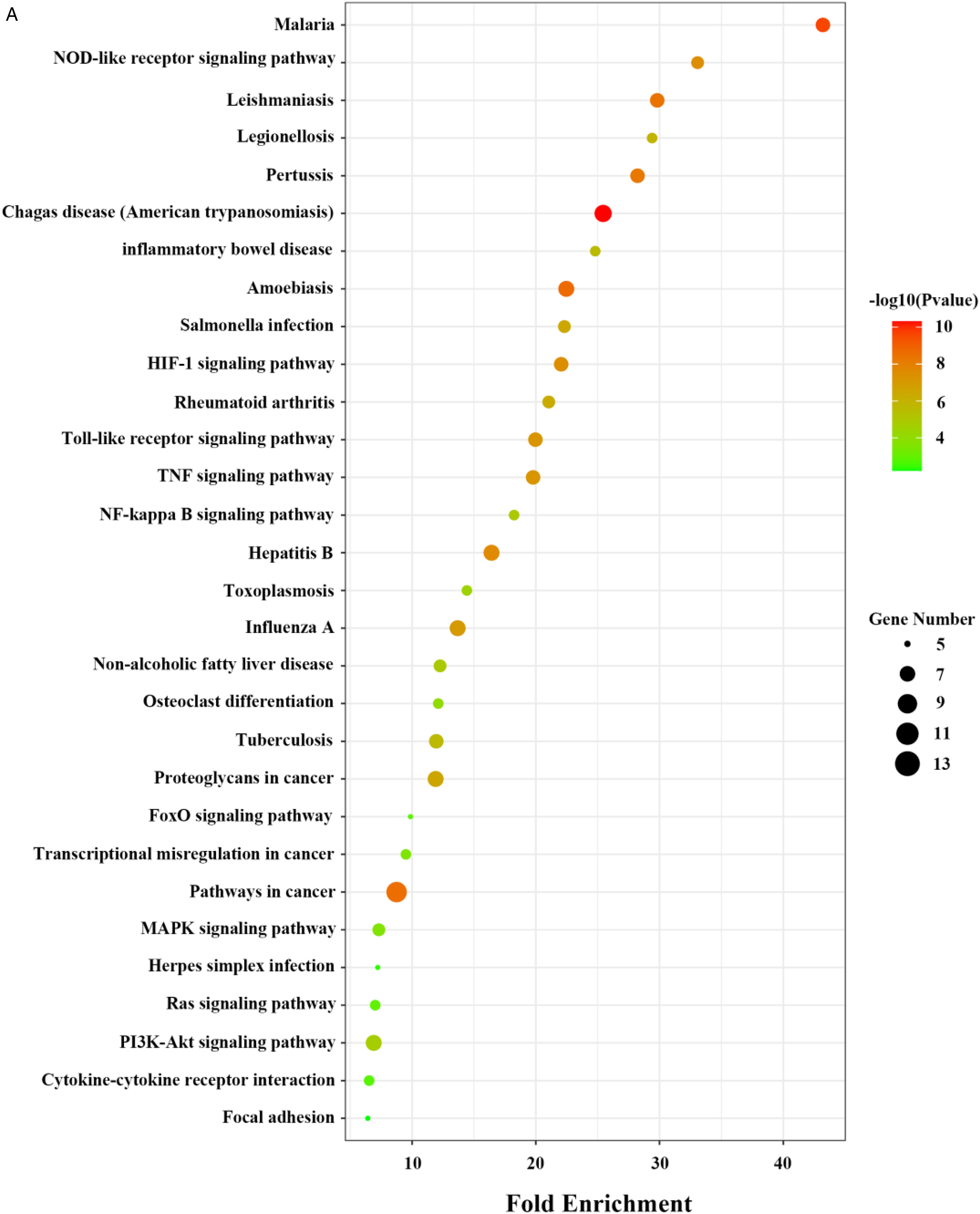
KEGG pathway analysis of common targets of BBR and GA. KEGG, Kyoto Encyclopedia of Genes and Genomes; BBR, berberine; GA, gouty arthritis.
BBR Decreased the serum Levels of Uric Acid and Cytokines
The serum uric acid level was significantly increased by the yeast extract chow combined with potassium oxonate, indicating that the rat model of HUA was successfully established. Moreover, the levels of the inflammatory factors IL-1β, IL-6, and IL-8 were elevated in the HUA group. As shown in Figure 5, compared to HUA rats, the BBR or AL intervention significantly decreased the serum levels of uric acid, IL-1β, IL-6, and IL-8 (P < .05).

Serum levels of uric acid and cytokines. (A) Uric acid, (B) IL-1β, (C) IL-6, and (D) IL-8. Data are expressed as the mean ± SEM (n = 6). *P < .05, **P < .01, ***P < .001. Con, control; HUA, hyperuricemia; BBR, berberine; AL, allopurinol.
Discussion
GA is a disease characterized by pain and inflammation in the foot and ankle joints caused by HUA. 21 BBR, which is extracted from C. chinensis, has been demonstrated as an effective ingredient for alleviating several types of inflammation, such as neuroinflammation, rheumatoid arthritis, and MSU crystal-induced inflammation.22,23 However, previous studies that have explored the mechanism by which BBR alleviates GA have focused mainly on a single target or pathway; thus, systematic mechanisms have not been fully elucidated. The current study exploited network pharmacology to elucidate the systematic mechanisms and targets underlying the protective effects of BBR against GA.
Our study used several databases to screen common targets of BBR and GA, which included the inflammatory factors IL-1β, IL-6, and TNF. IL-1β is a critical cytokine in the pathogenesis of GA and plays a central role in the initiation of acute inflammatory reactions. 24 MSU crystals have been reported to trigger the production and maturation of IL-1β through activation of NF-κB and recruitment of the NLRP3 inflammasome. 25 Mature IL-1β is released by immune cells, such as neutrophils and monocytes, binds to its receptors, and induces the production and secretion of other cytokines, thereby further aggravating the local inflammatory reaction. 26 Lin et al demonstrated that BBR inhibits NLRP3 expression and reduces the IL-1β levels in human monocytic THP-1 cells. 27 Previous in vitro experiments presented consistent results with our network pharmacology prediction (Figure 1) and in vivo results concerning IL-1β (Figure 5). One of the downstream targets of IL-1β is IL-6, which is a reliable marker that is positively correlated with inflammation and consistently elevated in the serum of patients with GA. 28 Ermiao pill, a TCM prescription used for thousands of years to treat GA, reduced the serum levels of IL-6 and alleviated ankle joint swelling in GA rats. 29 In our study, IL-6 was detected as a common target of BBR and GA (Figure 1), and the animal experiments demonstrated that BBR decreased the elevated IL-6 levels (Figure 5). MSU crystals have been shown to stimulate mononuclear macrophages to produce TNF on a large scale, which then binds to TNF receptor, enhancing the production of IL-1β. TNF deficiency or blockade of its receptor results in inhibition of neutrophil infiltration and IL-1β production.30,31 The cellular response to TNF and the TNF signaling pathway were identified using GO annotations and KEGG analyses (Figures 3-4). In our study, the IL-8 levels were increased in HUA rats and decreased by oral gavage with BBR (Figure 5). Although the IL-8 levels were not directly correlated with IL-1β and NLRP3 inflammasome expression, Kienhorst et al demonstrated that the IL-8 level was higher in GA patients compared with that in healthy controls, and that the circulating level of IL-8 was elevated during both the acute and intercritical phases of gout. 32
Our in vivo research showed that the expression levels of IL-1β, IL-6, and IL-8 could also be downregulated by AL (Figure 5), which is a specific inhibitor of xanthine oxidase. Xanthine oxidase is responsible for catalyzing the conversion of hypoxanthine to xanthine and xanthine to uric acid. AL is able to inhibit the formation of uric acid and precipitation of MSU crystals, which decreases tissue damage and inflammatory reactions. Reports have indicated that AL could reduce the expression of reactive oxygen species, normalize lipid peroxidation markers, decrease mitochondrial damage, and inhibit inflammatory signal pathways.33,34 Moreover, as a plant extract, BBR also presents an inhibitory effect on xanthine oxidase.35,36
The NF-κB signaling pathway plays a critical role in inflammatory reactions in numerous diseases, including GA. MSU crystals act on cell membrane TLRs (TLR2 and TLR4) as the initiating event and then trigger the activation of NF-κB. Activated NF-κB translocates to the nucleus and induces the transcription of inflammatory cytokines. 37 In the current study, both the TLR and NF-κB signaling pathways were identified as the combined targets of BBR and GA (Figure 4). A previous study reported that in MSU-induced GA, the expression levels of TLR4 and NF-κB were significantly increased. 38 Coptisine from C. chinensis has been shown to inactivate the NF-κB signaling pathway. 39 Zhang et al demonstrated that BBR inhibits the TLR4/NF-κB signaling pathway and reduces intestinal inflammation in diabetic mice. 8 RelA is a subunit of NF-κB and its phosphorylation regulates the transcription of NF-κB target genes. A previous study revealed that BBR attenuates adjuvant-induced arthritis in rats by downregulating the expression of phosphor-RelA and reducing the production of pro-inflammatory factors. 40 In the current study, RelA was detected as a common target of BBR and GA (Figures 1–2). The NF-κB signaling pathway is also activated by bacterial LPS. Numerous studies have shown that BBR attenuates LPS-induced inflammation. 41 In the current study, the LPS-mediated signaling pathway and cellular response to LPS were annotated in GO analysis as targets of GA and BBR (Figure 3).
In the present study, the MAPK signaling pathway was detected using KEGG analysis (Figure 4). There are three main MAPK types, namely, extracellular signal-regulated kinase-1/2 (ERK1/2), c-Jun N-terminal kinases (JNKs), and p38. 42 MAPKs activate the transcription factor NF-κB and regulate the expression of inflammatory factors, acting as the leading factors in inflammatory reactions during the pathogenesis of GA. Inhibiting the MAPK/ERK pathway significantly ameliorated inflammation of joints and articles, reduced the infiltration of leukocytes and microphages, and decreased damage to chondrocytes. 43 In GA model rats, the expression of p-ERK1/2, p-JNK, and p-MEK1/2 was increased, and the MAPK signaling pathway showed anti-inflammatory effects on GA rats. 44 A network pharmacology study concerning a TCM medicine, Porana sinensis Hemsl., indicated that it plays a role in the treatment of GA by regulating the MAPK pathway. 45
Further investigations are required to confirm the predicted inflammatory targets of BBR against GA, including more animal experiments and clinical trials. In our future experiments, RT-PCR and Western blot assays will be performed to confirm the level of TLRs, NLRP3, NF-κB, and MAPK, and related pathways.
Conclusions
In this study, a network pharmacology approach combined with experimental validation was used to clarify the underlying mechanism of the protective effects of BBR against GA. The network pharmacology analysis showed that BBR exerted its effect on GA partially by regulating the NF-κB, MAPK, and TLR signaling pathways. In addition, we showed that BBR improved GA by reducing the release of IL-1β and IL-6. The present study provides evidence supporting the clinical use of BBR for the treatment of GA.
Footnotes
Authors’ Contributions
Shan Fang, Yan Gao, and Yuan Fang designed and performed the experiments, analyzed the data, and wrote the manuscript. Zhijun Xie and Jing Sun conceived the study and revised the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable to this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the National Natural Science Foundation of China (grant number: 81803973) and New Faculty Launch Fund Project (grant number: 2022-JYB-XJSJJ058).
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Animal Experimental Ethics Committee of Zhejiang Chinese Medical University (2019012806) and complied with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.

