Abstract
Diallyl disulfide (DADS) is a garlic-derived organo-sulfur compound. This study was carried out to investigate the protective potential, antioxidant, and anti-inflammatory effects of this compound against cyclophosphamide (CP)-induced hepatotoxicity in rats. A single intraperitoneal dose of CP (200 mg/kg) resulted in a significant disturbance in hepatic function and oxidative stress, as well as inflammatory biomarkers. In addition, histopathological examination showed distinct changes and increased expression of proliferating cell nuclear antigen in hepatocytes. On the other hand, daily oral preadministration of DADS (200 mg/kg) for 10 days before the CP dose effectively attenuated the hepatotoxicity caused by CP administration as confirmed by significant amelioration of the aforementioned parameters in rat’s liver. It could be concluded that administration of DADS can diminish CP-induced hepatotoxicity through concurrent upregulation of antioxidant and anti-inflammatory responses that denote its possible potential clinical application against side effects of the CP drug.
Cyclophosphamide (CP) is a cytotoxic oxazaphosphorine nitrogen mustard alkylating drug used for the treatment of chronic and acute leukemia, lymphoma, myeloma, and cancers of the breast and ovary. It has been shown that CP treatment induces hemorrhagic cystitis, hepatic and lung damage, as well as cardiotoxicity and nephrotoxicity. 1,2 CP is metabolized by hepatic cytochrome P450 to form 4-hydroxycyclophosphamide that produces the chemically reactive metabolites phosphoramide mustard and acrolein that alkylate DNA and protein, producing cross-links. 3
During activation of CP by cytochrome P450, generation of reactive oxygen species (ROS) such as superoxide anions was demonstrated. 4 CP metabolites, and ROS generated in particular, can cause changes in cell redox balance, which leads to oxidative stress, resulting in cancer and healthy cell damage. 5
The clinical use of CP has been limited due to its ability to damage normal tissues, which usually resulted in multiple organ toxicity, mainly in the heart, testes, and urinary bladder. 6 Hepatotoxicity is a major side effect of CP as it is metabolized. Studies have suggested that oxidative stress is associated with its hepatotoxic effect. 7 Cyclophosphamide toxicity results from acrolein binding to cellular antioxidant nucleophiles such as glutathione (GSH) resulting in the depletion of the antioxidant defense system and initiation of lipid peroxidation (LPO). 8
CP administration results in the production of ROS, which diminish the activities of antioxidant enzymes and instigate peroxidative damage to the kidney, brain, and other vital organs. 9 In the same concern, CP administration modifies endogenous intensities of cytokines such as tumor necrosis factor alpha (TNF-α). 10
Plant-derived natural products are gaining wide attention in chemoprevention because of their little or no side effects against the chemically derived therapeutics. Diallyl disulfide (DADS) is a naturally occurring organo-sulfur compound, which intercedes in significant activities against urotoxicity, genotoxicity, nephrotoxicity, and cancer. 11
Experimental studies in rodents have shown that organo-sulfurs, such as diallyl sulfide (DAS) and DADS, as well as garlic powder, inhibit chemical-induced carcinogenesis in liver, lung, mammary gland, esophagus, and colon, when administered during the initiation or promotion stages. 12
DADS has been shown to reduce the hepatic DNA breaks induced by either N-nitrosodimethylamine or aflatoxin B1, as well as the mutagenicity of N-nitrosopiperidine and benzo[a]pyrene. These effects could be attributed to the modulation of drug-metabolizing enzymes, which play a key role in chemical activation and detoxication. 13
Therefore, this study intended to examine the possible protective effects of DADS on CP-induced hepatotoxicity and determine whether this effect was modulated through antioxidant and anti-inflammatory mechanisms in the liver.
Materials and Methods
Chemicals
DADS and CP were obtained from Sigma-Aldrich Co. (St Louis, MO, USA).
Experimental Animals
Thirty-two male adult albino rats weighing 120‐150 g used in the present studies were procured from the animal house of the National Cancer Institute, Cairo, Egypt. All the animals were kept under standard conditions at 23 ± 2 °C (12 hours light/12 hours dark cycle at room temperature) and were allowed free access to a standard diet and water ad libitum. Before starting the experiments, they were kept in an animal house for 1 week. All animal treatments were conducted according to the Ethics Committee of the National Center for Radiation Research and Technology in accordance with the recommendations for the proper care and use of laboratory animals (HIN publication No. 85‐23, revised 1985) in accordance with international ethical considerations.
Experimental Design
Animals were equally divided into 4 groups as follow: Group I: control that was injected with normal saline (0.5 mL/rat) for 10 days. Group II: were treated with DADS (200 mg/kg, oral) for 10 consecutive days. 14 Group III: rats were injected intraperitoneally (i.p.) with saline solution for 10 consecutive days; 1 hour later, they were injected with a single dose (200 mg/kg, i.p.) of CP 1 ; Group IV: rats were pretreated with DADS for 10 days; 1 hour after the last dose, rats were injected with CP. Twenty-four hours after the last doses of specific treatment, all animals were sacrificed, blood samples were collected by heart puncture, and serum samples were separated by centrifugation at 750×g at room temperature for 10 minutes for measurements of hepatotoxicity indices and lipid profile.
The liver was quickly excised, washed with saline, blotted with filter paper, and homogenized in ice-saline to yield a 10% (w/v) homogenate using a Branson sonifier (250, VWR Scientific, Danbury, CT, USA). The homogenate was then centrifuged at 10 000×g for 15 minutes at 4 °; the supernatant was taken to be used for measuring oxidative stress and inflammatory parameters.
Biochemical Assays
Determination of Liver Function Indices
The activities of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and gamma glutamyl transferase, and bilirubin contents (total and direct) were assayed in serum using commercial kits purchased from Biogamma (Italy) following the recommended methods. 15 -18
Determination of Lipid Profile
Total cholesterol, triglycerides, and high-density lipoprotein-cholesterol (HDL-C) were assayed in serum using commercial kits according to the published methods. 17,19,20
Determination of Hepatic Oxidative Stress Biomarkers
Malondialdehyde (MDA) was estimated as the end product of LPO, reduced GSH, according to published methods. 21,22 Protein carbonyl content (PCC) was determined using an abcam assay kit according to the manufacturer’s instructions. Superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px) activities in liver tissue homogenate were determined following the published methods. 23 -25 In the liver homogenate, the paraoxonase-1 (PON-1) level was determined using a commercial ELISA kit (Abcam, China), and NADPH oxidase 4 (NOX-4) level using a cusabio ELISA kit, according to the manufacturers’ instructions.
Determination of Hepatic Inflammatory Biomarkers
TNF-α, interleukin-6 (IL-6), and inducible nitric oxide (iNOS) levels were determined using an My BioSource ELISA kit, according to the manufacturer’s instructions. Nuclear factor-kappa B (NF-κB) was determined using a cusabio ELISA Kit, according to the manufacturer’s instructions.
Histopathological Study
Liver samples were washed with cold saline and then fixed in 10% neutral buffered formalin for at least 24 hours. The specimens were then dehydrated in an ascending series of ethanol, cleared in xylene, and then embedded in paraffin wax. Five-micrometer sections were processed using standard procedures and stained with hematoxylin–eosin (H&E) for the histological study. The stained slides were examined under a light microscope. 26
Immunohistochemical Expression of PCNA
All solutions were prepared according to the manufacturer’s recommendations, and serial unstained paraffin slides from histology were obtained and paraffin sections cleared and rehydrated according to the steps described by Campbell-Thompson et al. 27 Antigen retrieval using citrate buffer in a steamer at 95 oC for 30 minutes was performed and the slides were washed with a buffer for 5 minutes. The slides were loaded on mouse monoclonal anti-proliferating cell nuclear antigen (PCNA) antibody PC 10 Dako-autostainer (Dako, Santa Barbara, CA, USA) for 15 minutes and rinsed. After that, the slides were incubated with the first primary anti-rabbit immunoglobulin G for 30 minutes and rinsed twice, then incubated with the secondary antibody (Mach 2 HRP polymer) for 30 minutes and rinsed twice. Diaminobenzidine tetra-hydrochloride was used as a chromogen for 4 minutes and rinsed twice. Slides were washed with phosphate-buffered saline and counterstained in hematoxylin (1 minute), washed with water, and allowed to air dry for 2 hours. Finally, coverslips were placed over the slides using aqueous mounting media and scanned using an Aperio CS scanner (Aperio, Vista, CA, USA).
Statistical Analysis
Data are expressed as mean ± SEM (P < .05). Statistical analysis was carried out using one-way analysis of variance, followed by Tukey–Kramer multiple-comparisons test.
Results
DADS Treatment Improves Hepatotoxicity Biomarkers in CP-Administered Rats
To assess the severity of CP-induced liver injury, liver function tests were performed. The results revealed that CP administration caused noticeable liver injury, as indicated by significant elevation (P < .05) in the serum activity of AST, ALT, ALP, and γ-GT, as well as total and direct bilirubin levels, as shown in the rats in the CP-treated group (Table 1). On the other hand, DADS treatment of rats diminished the CP-induced liver damage and significantly improved (P < .05) the activity of AST, ALT, ALP, and γ-GT, as well as total and direct bilirubin levels.
Impact of DADS Pretreatment on the Liver Function Parameters of CP-Administrated Rats.

Abbreviations: ALP, Alkaline phosphatase, ALT, alanine aminotransferase, AST, aspartate aminotransferase, Bil. D, direct bilirubin; Bil. T, total bilirubin; CP, cyclophosphamide; DADS, diallyl disulfide; GGT, gamma glutamyl transferase.
Each value represents the mean ± SE.
*Significantly different from that of the control (P < .05).
#Significantly different from that of rats treated with the CP.
DADS Treatment Improves Lipid Profile Disturbance Induced With CP
In the same context, the data represented in Table 2 revealed that CP administration induced a significant increase in serum cholesterol, triglycerides, low-density lipoprotein cholesterol (LDL-C) and very-low-density lipoprotein cholesterol (VLDL-C) levels, accompanied by a significant reduction (P < .05) in the serum HDL-C level compared with the control group.
Impact of DADS Pretreatment on the Lipid Profile Against CP-Treated Rats.
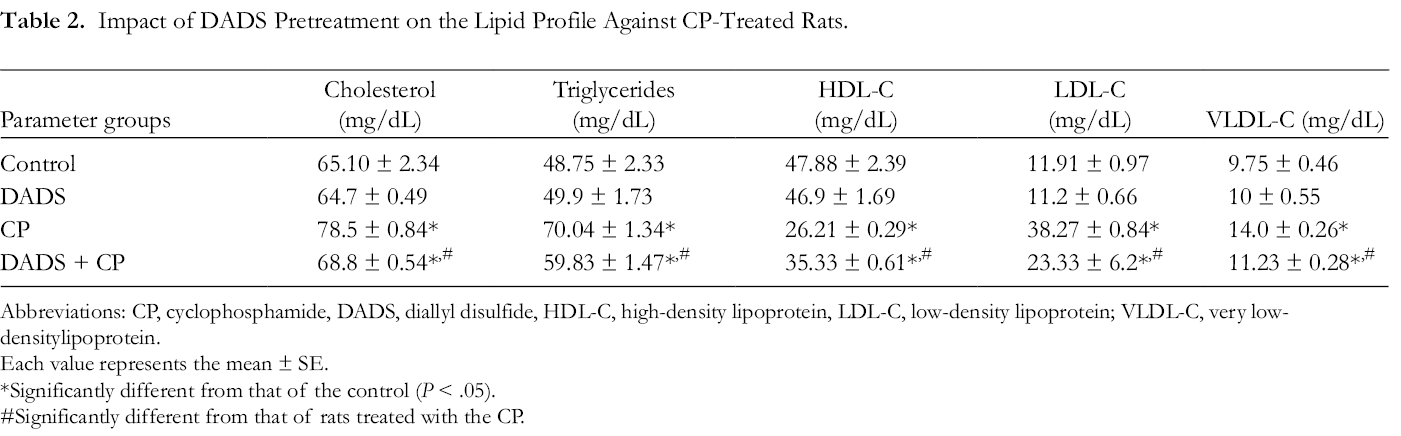
Abbreviations: CP, cyclophosphamide, DADS, diallyl disulfide, HDL-C, high-density lipoprotein, LDL-C, low-density lipoprotein; VLDL-C, very low-densitylipoprotein.
Each value represents the mean ± SE.
*Significantly different from that of the control (P < .05).
#Significantly different from that of rats treated with the CP.
Rats treated with DADS followed by CP administration resulted in a significant increase in HDL-C level. However, parameter levels that increased responding to CP treatment were only significantly improved in response to DADS pretreatment before CP administration compared with the CP group.
DADS Treatment Halts Oxidative Stress in the Liver of CP-Administrated Rats
CP administration to rats resulted in an imbalance in the oxidative status in hepatic tissue. As presented in Figure 1, a surge in MDA levels and PCC, paralleled by increases in NOX-4 activity, was observed in the CP group when compared with the control rats. On the contrary, CP treatment of rats diminished the beneficial PON-1, associated with a significant decrease (P < .05) in the hepatic activity of GPx and CAT, as well as decreasing GSH levels compared with the control group.

Effect of DADS treatment on the redox parameters in CP rats. Each value represents the mean ± SE. *Significantly different from that of the control (P < .05). #Significantly different from that of rats treated with CP. CAT, catalase; CP, cyclophosphamide; DADS, diallyl disulfide; GSH, reduced glutathione; GSH-Px: glutathione peroxidase; MDA, malondialdehyde; PCC, protein carbonyl content; SOD, superoxide dismutase.
On the other hand, the treatment of CP-administrated rats with DADS significantly reversed (P < .05) the redox imbalance exhibited by CP, as it achieved a reduction in MDA and PCC levels, as well as NOX-4 activity, as compared with the CP group. Moreover, DADS treatment managed to restore antioxidant defenses, as reflected by the significant increase (P < .05) in the activity of GPx and CAT, and the hepatic levels of GSH when compared with the CP group.
DADS Modulates Hepatic Inflammation Induced With CP
Hepatic injury is commonly associated with inflammation. Therefore, the possible role of inflammatory mediators and proinflammatory factors was surveyed in this study. As depicted in Figure 2, animals in the CP group showed a significant increase (P < .05) in the expression of TNF-α, NF-kB, iNOS, and IL-6 when compared with the control group. Meanwhile, concurrent pretreatment of CP rats with DADS for 10 days significantly reduced (P < .05) the expression of the proinflammatory factors TNF-α, NF-kB, iNOS, and IL-6 as compared with the CP group, and abrogated the inflammatory process, which plays a crucial role in liver toxicity.

Effect of DADS on inflammatory mediators in CP-induced liver toxicity. Each value represents the mean ± SE. *Significantly different from that of the control (P < .05). #Significantly different from that of rats treated with CP. CP, cyclophosphamide; DADS, diallyl disulfide; IL-6, interleukin-6; iNOS, inducible nitric oxide; NF-κB, nuclear factor-kappa B; TNF-α, tumor necrosis factor alpha.
Histopathological Examination
Histopathological examination of hepatic sections of the control group showed normal histological structure of hepatic lobules (a). DADS group showed no histopathological changes (b). On the other hand, the CP-treated rat group showed cytoplasmic vacuolization and apoptosis of hepatocytes (c). The DADS treatment prior to CP group showed slight vacuolization of hepatocytes with ameliorated hepatic structure (d) (H&E ×200) Figure 3.

Histological examination of the liver. (A) Control group showed normal histological structure. (B) DADS group showed no changes from normal histological structure. (C) CP group showed cytoplasmic vacuolization and apoptosis of hepatocytes. (D) DADS + CP group showed slight vacuolization of hepatocytes with ameliorated hepatic structure. CP, cyclophosphamide; DADS, diallyl disulfide.
Immunohistochemical Expression of PCNA
PCNA immunohistochemical examination of hepatic sections showed no expression of PCNA in the normal control group (a), as well as the DADS-treated group (b). The CP-treated group showed a strong positive expression of PCNA (c), while these strong positive expressions were ameliorated when DADS was administrated for 10 days before CP treatment, and showed a weak positive expression of PCNA (d) (Figure 4).

Immunohistochemical examination of hepatic sections. (A) Control group showed no expression of PCNA. (B) DADS group showed no expression of PCNA. (C) CP group showed strong positive expression of PCNA. (D) DADS + CP group showed weak positive expression of PCNA. CP, cyclophosphamide; DADS, diallyl disulfide.
Discussion
Some of the health challenges befalling humans around the world are chemically induced. Therefore, the present study inspected the protective potential of DADS in CP-induced hepatic damage in male rats. In the current study, administration of CP-induced hepatic toxicity was distinguished by increases in the activities of AST, ALT, ALP, and γ-GT, as well as total and direct bilirubin levels. These results agree with previous reports. 1,28,29 The rise in these markers might be a result of advanced injury in the hepatic cellularity as a subsidiary event subsequent to CP-induced ROS and more by its toxic metabolic by-product, acrolein. 30 In addition to the hepatic dysfunction, hepatic lipid accumulation was observed in CP-treated rats. DADS is one of the most abundant, oil-soluble organosulfur compounds derived from garlic. It has been demonstrated to have protective effects against several diseases. 31 In the present study, it was revealed that DADS treatment ameliorated hepatic dysfunction and reduced hepatic lipid accumulation in CP-treated rats probably by maintaining the hepatic cellular membrane integrity. 32,33
The current study focused on the role of DADS against the CP-induced oxidative stress in rats. CP is an inactive cytostatic, metabolized into active metabolites in the liver. In the present study, CP induced significant reduction in antioxidative enzymes GSH-Px, SOD, CAT, and PON-1 activities along with GSH contents, and a significant increase in MDA, PCC, and NOX-4 levels. This effect could be due to the capability of acrolein, to divest the cell of its natural defense-GSH system versus the ROS and promotes the free radical generation, initiation, and reproduction of LPO. 1 These free radicals are increased due to deterioration of the nitric oxide synthase system in the vasculature. 2 CP is a toxic chemical that produces various free radicals causing an increase in LPO, which converts polyunsaturated fatty acids into small and more reactive elements. It is also a measure of the damage caused to the membranes by free radicals. It is known that LPO is one of the principal causes of CP-induced toxicity; also, CP interferes with the tissue antioxidant defense system, produces highly reactive oxygen free radicals, and interacts with protein amino acids, thus causing structural and functional changes in enzymes. 34,35 Previous studies have suggested that DADS has potential as a treatment option for metabolic syndrome and acute liver injury due to its hepatoprotective and antioxidant effects. 36 Administration of DADS before CP fixed the oxidative stress marker levels and antioxidative enzyme activities in the liver tissues, demonstrating a defensive effect against ROS. This effect might be due to its capability to decrease oxidative stress or improving the antioxidant capacity in the cells, thus decreasing the susceptibility to oxidative damage. 37 -39 DADS ameliorated the level of antioxidant enzymes, shows that through its free radical scavenging activity could exert a beneficial action against pathophysiological alterations caused by free radicals, or might be due to the ability of DADS to protect against LPO. 40,41
Oxidative stress can activate inflammatory molecules. Previous observations recorded that chronic inflammation, oxidative stress, and cancer are closely linked. 42 In the present study, CP administration significantly augmented the levels of hepatic TNF-a, NF-kB, IL-6, as well as iNOS. These results are agreeing with earlier studies, which reported that TNF-α is a proinflammatory cytokine, initiates production and exudation of a cascade of inflammatory mediators such as cytokines, and has participated in aggravating acute inflammatory responses in a high dose of CP. 43,44 Administration of DADS before CP attenuated the alterations noticeably due to its antioxidant and anti-inflammatory properties, which is in agreement with previous reports. 41,45,46 NF-kB is a transcription factor, activates the transcription of many cytokines, including TNF-a, IL-1, and IL-6, which are vital in the production of acute inflammatory reactions. 47 DADS treatment before CP decreased the augmented levels of the inflammatory cytokines, IL-6, and TNF-α through blockade of NF-kB. 48 The protective effects of DAS may be due to its ability to reduce the metabolic activation of CP by the induction of CYP3A1 and the inhibition of CYP2C11. 40
Previous observations evidenced that NO is involved in acute and chronic inflammation. 49 It is reported that cytokines and NO produced by iNOS were involved in CP-induced hepatic injury. 50 The current study agrees with previous reports 51 that showed that CP caused a significant increase in NF-κB and iNOS expression levels, suggesting that activation of NF-κB and iNOS by CP might play a role in CP-induced inflammations. Agents that inhibit NF-kB may have beneficial therapeutic effects in the treatment of inflammatory diseases as a result of decreased iNOS and cyclooxygenase 2 expressions. Szabo and Billiar 52 recorded that DADS inhibits nuclear translocation of proinflammatory transcription factor NF-kB and the expression of tumorigenic enzyme COX2 via GSK-3 (α/β) inactivation. 53 This finding was consistent with our results. It has been reported that DADS was effective in reducing the levels of oxidized low-density lipoprotein, LPO, as well as NF-κB activity, revealing good anti-inflammatory and antioxidant properties. 32,54 In the current work, DADS led to a significant reduction in NF-kB and the subsequent regulation of inflammatory mediators caused by the treatment of CP. 31 Protective effects of DADS may be due to its ability to regulate proinflammatory cytokines production by inhibition of NF-κB and mitogen-activated protein kinase expressions, and its potent anti-oxidative capability through reduction of oxidative DNA damage in the liver. 40,41
In accordance with the previous biochemical profile, the histopathological and immunohistochemical examination of liver sections obtained from the CP group revealed severe pathological aberrations and cytoplasmic vacuolization and apoptosis of hepatocytes with strong positive expression of PCNA. This effect of CP was supported by the former findings. 55 However, liver sections from the DADS group showed preserved normal architecture of the hepatic lobule with no expression of PCNA. In the same context, the preadministration of DADS to the CP-treated rats resulted in the amelioration of changes elicited by CP with slight vacuolization of hepatocytes, and weak positive expression of PCNA revealed the hepatoprotective concept of DADS. Concerning the histological structure, this hepatoprotective effect of DADS is inconsistent with the observations of Somade et al. 56
Conclusion
It could be concluded that DADS remedy afforded hepatic protection against CP-induced oxidative and inflammatory responses, which was supported by the histopathological and immunohistochemical examination of PCNA.
Footnotes
Acknowledgment
The authors acknowledge Prof Dr Kawkab Abdel-Aziz, Pathology Department, Faculty of Veterinary Medicine, Cairo University for accomplishing the examination of liver sections in the current study.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Nantong University, China and approved by the Administration Committee of Experimental Animals, Jiangsu Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
