Abstract
Background
Xiaoqinglong Decoction (XQLD) is a traditional Chinese medicine formula used for the treatment of allergic rhinitis and asthma, including combined allergic rhinitis and asthma syndrome (CARAS), due to its anti-inflammatory and anti-seizure properties. However, the pharmacological activities and the underlying molecular mechanisms of XQLD remain to be elucidated. Hence, we investigated the effects of XQLD against inflammation in an ovalbumin (OVA)-induced CARAS model in BALB/c mice.
Methods
BALB/c mice were sensitized by intraperitoneal injection and aerosol inhalation of OVA. XQLD or dexamethasone was administered by oral gavage prior to OVA challenge for 7 consecutive days. Specific airway resistance (sRAW) was evaluated 24 h after the final challenge with OVA. Enzyme-linked immunosorbent assay was used to measure the levels of serum inflammatory factors. The nasal mucosa and lung tissue were examined for general morphology and goblet hyperplasia using hematoxylin and eosin or periodic acid Schiff staining. Flow cytometry was employed to analyze the frequencies of helper T lymphocytes in peripheral blood. Immunohistochemistry was performed to determine the expression of transcription factors in helper T lymphocytes and γδ TCR.
Results
XQLD significantly ameliorated the symptoms of CARAS model mice, suppressed serum levels of interferon-γ, interleukin (IL)-2, IL-4, IL-5, IL-17, while increasing levels of IL-10, transforming growth factor-β. Epithelium impairment, cilia loss, eosinophil infiltration, and goblet cell metaplasia were significantly reduced in the nasal mucosa and lung tissue sections. XQLD restored the balance of Th1/Th2 and Th17/Treg cell frequency in peripheral blood. Furthermore, XQLD down-modulated the expression of GATA-binding protein 3 (GATA3), retinoic acid receptor-related orphan receptor γt, and γδ T cell receptor (TCR) in the nasal mucosa, along with GATA3 and γδ TCR in the lung tissue.
Conclusions
XQLD alleviated OVA-induced CARAS in BALB/c mice through its anti-allergic effects and modulation of the immune system.
Introduction
Combined allergic rhinitis and asthma (CARAS) is a respiratory disorder characterized by simultaneous allergic reactions in the upper and lower airways, manifested with the coexistence and mutual influence of allergic rhinitis (AR) and bronchial asthma (BA). 1 CARAS is presented by recurrent nasal congestion, runny nose, sneezing, nasal itching, cough, shortness of breath, chest tightness, and wheezing, exacerbated by exposure to cold air, irritating substances, allergens, or physical exertion. CARAS poses a growing global health challenge with a rising prevalence worldwide. Research indicates a close link between AR and BA, with inflammation playing a crucial role in the communication between the upper and lower airways. 2 AR triggers asthma attacks by increasing inflammation and hyperresponsiveness in the lower airways upon exposure to allergens, while bronchial challenges worsen nasal mucosa inflammation through eosinophil infiltration. 3 The concept of ‘one airway, one disease' 4 underscores that AR and BA represent distinct manifestations at different sites of the same underlying pathology.
CARAS pathophysiology is characterized by immediate airway hypersensitivity following aeroallergen exposure. 1 Despite differing symptoms, the physiological and pathological changes in AR and BA share similarities due to the respiratory tract's continuity. Both conditions are type I hypersensitivities mediated by serum immunoglobulin E (IgE), characterized by eosinophil infiltration in respiratory pathology. Dendritic cells play a crucial role in antigen presentation following initial exposure to allergens. They stimulate helper T cells (Th) to differentiate into helper T lymphocyte 2 (Th2), leading to the secretion of various interleukins, triggering the differentiation, maturation, and migration of eosinophils, as well as IgE antibody responses. Upon reexposure to allergens, the sensitized body experiences symptoms including sneezing, runny nose, nasal itching, and wheezing, triggered by mast cell release of active mediators such as leukotrienes and histamine, leading to increased vascular permeability, gland secretion, and smooth muscle contraction. Advances in research have highlighted the role of γδ T cells as pro-inflammatory mediators in allergic airway inflammation. 5 Notably, the regulation of γδ T cells by IgE responses suggests that allergic reactions transcend adaptive immunity. 6 Acting as a bridge between innate and adaptive immunity, γδ T cells can activate and degranulate mast cells independently of IgE, challenging the notion that IgE is the sole initiator of mast cell activation. This discovery opens new possibilities for immunomodulatory treatments for CARAS and offers a fresh perspective on preventive and therapeutic approaches in traditional Chinese medicine (TCM). 7
CARAS treatments have evolved with the widespread adoption of nasal and inhaled corticosteroids, particularly following the introduction of drugs such as omalizumab, marking the onset of CARAS-targeted therapies. However, while these seemingly targeted approaches effectively suppress airway eosinophilic inflammation, they can cause ‘nontargeted’ side effects by interfering with the physiological functions of other organs, given the widespread distribution of various interleukins and their receptors throughout the body. Prolonged corticosteroid use can lead to side effects such as epistaxis, immune suppression, increased susceptibility to secondary infections, nocturnal drowsiness, and diminished sleep quality.8–10 Current utilization of allergen-specific immunotherapy in less than 10% of patients with AR or BA highlights significant room for enhancement in CARAS treatment strategies. Within TCM frameworks, CARAS is categorized under “Biqiu” (鼻鼽) and “Xiaobing” (哮病), aligning with the holistic principles espoused by TCM. The TCM approach to CARAS management emphasizes a comprehensive treatment strategy guided by the concept of ‘together treatment of the lung and nose’, offering a distinctive therapeutic profile.
A notable traditional Chinese remedy for AR and BA is Xiaoqinglong Decoction (XQLD), tracing its origins back to Zhang Zhongjing's “Treatise on Febrile and Miscellaneous Diseases” from 205 AD. XQLD is known for its efficacy in alleviating respiratory conditions by dispelling exterior cold and warming the lungs, comprising ingredients including Ephedra sinica, Neolitsea cassia, Paeonia lactiflora, Asarum heterotropoides, Glycyrrhizae Radix rhizoma, Pinellia ternata, Schisandra chinensis, and Zingiber officinale. XQLD has been reported to be effective and safe in the treatment of respiratory diseases such as AR, BA, and bronchitis.11–13 Pharmacological studies indicated that XQLD exerted antiallergic effects by antagonizing the histamine H1 receptor, significantly reducing levels of nasal mucosal lymphopoietin and specific IgE (sIgE), and inhibiting the release of Th2 cytokines and mast cell degranulation triggered by IgE.14–16 XQLD has been demonstrated to suppress airway inflammation by decreasing serum levels of interleukin (IL)-4 and IL-5.17,18 Clinical and research studies support the efficacy of XQLD in the treatment of allergic rhinitis and asthma, yet further investigations are warranted to clarify its impact on CARAS.
The study aimed to investigate the anti-allergic and anti-inflammatory effects of XQLD and understand its underlying cellular mechanism using an ovalbumin-induced murine CARAS model. Evaluations were carried out on nasal symptoms, airway resistance, serum IgE levels, T cell cytokines, and histological analysis of nasal mucosa and lung tissue. The results demonstrated that treatment with XQLD alleviated allergic symptoms, improved airway resistance, reduced inflammation in respiratory tissues, and modulated ovalbumin (OVA)-specific IgE and cytokine levels. These findings suggest that XQLD holds promise as a therapeutic intervention for CARAS.
Materials and Methods
Plant Materials and Main Reagents
The herbs in the XQLD were evaluated as follows: E. sinica (Inner Mongolia, China), N. cassia (Guangxi, China), P. lactiflora (Anhui, China), A. heterotropoides (Liaoning, China), G. rhizoma (Inner Mongolia, China), P. ternata (Sichuan, China), S. chinensis (Liaoning, China) and Z. officinale (Yunnan, China). All herbs were authenticated by the Beijing Institute for Drug control. Reference standards ephedrine hydrochloride, cinnamic acid, paeoniflorin, asarinin, 6-gingerol, liquiritin, and glycyrrhizic acid were purchased from Biorbyt (Wuhan, China), schisandrol A was purchased from Yuanye Bio-Technology Co., Ltd (Shanghai, China).
Acetonitrile (orb1296807, purity >99%) was purchased from Biorbyt (Wuhan, China), HPLC-grade phosphoric acid (49685), Ovalbumin (A2512, OVA) and analytical-grade methanol (1.06018) were purchased from Sigma (St. Louis, Missouri, USA). Dexamethasone (S1322) was provided by Selleck (Shanghai, China). ELISA kits for the detection of IL-2 (BMS601), IL-4 (BMS613), IL-5 (BMS610), IL-10 (BMS614), IL-17 (BMS6001), IFN-γ (BMS606-2), TGF-β (BMS608-4), and IgE (EMIGHE); antibodies of T-bet (PA5-28881), GATA3 (MA1-028), RORγt (PA5-23148), Foxp3 (700914), and γδ TCR (MA5-44050) were obtained from Invitrogen (Carlsbad, CA, USA). The Mouse T Lymphocyte Subset Antibody Cocktail (558391) for flow cytometry was purchased from BD Biosciences (Franklin Lakes, NJ, USA).
Preparation of XQLD
The XQLD formulation consists of eight herbs and the doses were as follows. E. sinica, 9 g; N. cassia, 9 g; P. lactiflora, 9 g; A. heterotropoides, 3 g; G. rhizoma, 9 g; P. ternata, 9 g; S. chinensis, 9 g; Z. officinale, 9 g.
The herbs were soaked in 500 mL of distilled water for 30 min and then subsequently boiled for 20 min. The resulting decoction was collected, and the residue underwent a second round of boiling with another 500 mL of distilled water. The combined decoction was then filtered through sterile gauze and concentrated using a rotavapor at a final concentration of 1.43 g/mL, equivalent to 1.0 g of dried herbal material per milliliter. The extracted XQLD was stored at −20°C and diluted with distilled water prior to intragastric administration.
Preparation of Standard Solutions
Seven reference compounds, namely ephedrine hydrochloride, cinnamic acid, paeoniflorin, asarinin, 6-gingerol, schisandrol A, liquiritin and glycyrrhizic acid, were accruately weighed and dissolved in methanol to prepare the stock solutions with concentrations ranging from 0.204 to 0.576 mg /mL. The stock solution was then diluted to the required concentration to establish a standard curve. All solutions were stored at 4°C until analysis.
Chromatographic Conditions
Chromatographic separation was carried out following the previous study, 19 using the Agilent ZORBAX Eclipse XDB-C18 column (Analytical, 250 mm × 4.6 mm, 5 μm), equipped with Shimadzu SPD-20A High-Performance Liquid Chromatography (Shimadzu, Japan). The mobile phase consisted of 0.1% phosphoric acid water (A) and acetonitrile (B) using a gradient elution of 5% B at 0–10 min, 10% B at 10–30 min, 12% B at 30–50 min, 15% B at 50–54 min, 17% B at 54–70 min, 22% B at 70–78 min, 26% B at 78–90 min, 28% B at 90–97 min, 35% B at 97–108 min, 49% B at 108–125 min, 57.9% B at 125–135 min and 95% B at 135–140 min. The flow rate was 0.8 mL/min with an injection volume of 10 μL, and the detection wavelength was set at 254 nm.
Animals
Forty female BALB/c mice, aged 6-8 weeks (weighing approximately 18 to 22 g each), were purchased from Charles River Laboratories Animal Technology Co., Ltd (Beijing, China), and utilized as experimental animals. The mice were acclimatized to the new environment for 1 week. The adopted diet was provided ad libitum, with free access to water and standard chow, comprising 54% mixed carbohydrate, 19% protein and 3% lipid by weight, and was maintained under controlled conditions with a temperature of 25 °C, humidity of 40–60% and a light/dark cycle of 12 h. All procedures strictly adhered to the use and care of laboratory animals outlined by the People's Republic of China's legislation and the National Research Council's Guide, and approved by the Institutional Animal Care and Use Committee of the Beijing Hospital of Traditional Chinese Medicine (No. SQ-2021-02-62, granted in February 2021). All efforts were made to ameliorate the welfare and to minimize animal suffering.
The mice were randomly assigned to four groups (8 per group) using a computer-generated random sequence as follows: (1) naive group (Control); (2) CARAS group; (3) XQLD group, CARAS mice treated with XQLD, 6.0 g/kg according to the conversion by body surface area; (4) CARAS mice treated with dexamethasone as a positive control (DEX).
Induction of CARAS by OVA
Animal experiments were carried out according to Tang's design 20 with minor modifications. Briefly, CARAS mice were sensitized with 500 μL of OVA solution by intraperitoneal injection on days 1, 7, and 14, and initially challenged with 50 μL of 5% OVA by nasal drip for three consecutive days over three weeks, followed by a second challenge of 5 mL of 5% OVA through aerosol inhalation for another three successive days over three weeks. Mice in the naive group were sensitized and challenged with phosphate-buffered saline (PBS) instead of OVA. CARAS mice were gavaged with distilled water, XQLD water extraction (6.0 g/kg, equivalent to 8.58 g / kg of dried herbal material for a 70 kg adult), or dexamethasone (1.0 mg/kg) as a positive control one hour after each challenge. On day 60, retroorbital blood, spleen, nasal mucosa, and lung tissue samples were collected from all mice for laboratory analysis following the euthanasia with pentobarbital sodium euthanasia (0.1 mL/20 g, i.p.). (Figure 1)

Experiment protocol. CARAS mice were sensitized with 500 μL of OVA solution via intraperitoneal injection on days 1, 7, and 14 and challenged with 50 μL of 5% OVA in PBS by nasal drip for three consecutive days over three weeks, and with 5 mL of 5% OVA through aerosol inhalation for three successive days over three weeks. The Control group was sensitized and challenged with PBS. The Control and CARAS groups were administered distilled water orally, the XQLD group and the DEX group received XQLD and dexamethasone, respectively. Control, the naive group. CARAS, combined allergic rhinitis and asthma. XQLD, CARAS mice treated with Xiaoqinglong Decoction, 6.0 g/kg. DEX, CARAS mice treated with dexamethasone as a positive control.
Measurement of Nasal Symptoms
Local allergic symptoms were monitored for 30 min following the airway challenge (day 59). The frequency of sneezing and nasal rubbing, and the severity of runny nose were recorded using the scoring system used by Narita et al 21 The sneezing was classified on a scale from 1 (1-3 sneezes) to 3 (≥11 sneezes). The severity of runny nose was scored as: 1 (flowing into the anterior nostril); 2 (flowing through the anterior nostril); and 3 (severe rhinorrhea). The itching of the nose was recorded as 1 (occasional scratching) or 2 (continuous scratching).
Determination of Specific Airway Resistance
24 h following the final OVA challenge, specific airway resistance (sRaw) in response to CARAS was evaluated by measuring the Penh value of the mice using 4-chamber whole-body plethysmography (Buxco Research Systems, Troy, NY), which provides non-invasive and free conditions, slightly modified from Wang's research. 22 Briefly, mice were placed in unrestrained whole-body plethysmograph chambers and acclimated for 5 min, then exposed to PBS aerosolization for 30 s and recorded for 3 min. The mice were then exposed to 6.25, 12.5, and 25 mg/mL methacholine (Sigma-Aldrich) for 30 s, followed by a 3-min recording period. Each measurement was conducted following a timed procedure of 1 min nebulization, 4 min monitoring and a 1 min break. Respiratory flow was measured with a pneumotachograph and transmitted to Buxco FinePointe software for functional analysis to calculate sRaw.
Determination of serum OVA-Specific IgE and Cytokines
12 h after anesthesia, retroorbital blood was collected and centrifuged until sedimentation at 4 °C. The samples were diluted using the ELISA kit 10 times and added with different concentration standards, and the sample was diluted into a 96-well plate which was coated with an enzyme label. Concentrations of OVA-specific IgE, IL-2, IL-4, IL-5, IL-10, IL-17, interferon-γ (IFN-γ), and transforming growth factor-β (TGF-β) were detected using ELISA kits, following the manufacturer's instructions.
Histochemical Analysis
The nasal mucosa and lung tissue were harvested following euthanasia of the mice. The tissues were fixed in 10% paraformaldehyde, dehydrated, and embedded in paraffin. Paraffin-embedded tissue samples were sectioned into 4 μm slices, then stained with hematoxylin and eosin (H&E) for general morphology and Periodic Acid-Schiff (PAS) for goblet hyperplasia. Histopathological changes were assessed and photographed using an optical microscope (Eclipse Ci, Nikon, Japan).
Flow Cytometric Analysis
Single-cell peripheral blood suspensions were prepared, and cells were evenly distributed in tubes and washed with PBS. Cells were then collected, centrifuged to remove the supernatant, and subsequently incubated in darkness at 20 °C for 30 min before gradually freezing at a rate of 1 °C per minute, and finally stored in liquid nitrogen. The thawed cells were prepared for flow cytometry staining by gently swirling the vial in a 37 °C water bath for 1 min. Subsequently, the thawed cells were transferred to a centrifuge tube containing a pre-warmed complete growth medium, centrifuged at 200 × g for 5 min, the supernatant was decanted and the cells were resuspended. To identify Th1, Th2, Th17 and regulatory T cells (Treg), the samples were incubated in darkness at 25 °C for 30 min with anti-mouse CD4 PerCP-Cy5.5 and anti-mouse CD8 PE-Cy7. Anti-IL-4 and anti-IFN-γ antigen antibodies were used to distinguish between Th1 (CD4 + IFN-γ+) and Th2 (CD4 + IL-4+) cells. Th17 (CD4 + IL-17+) and Treg (CD25 + Foxp3+) cells were detected and stained with phycoerythrin (PE) anti-mouse IL-17 and anti-mouse Foxp3-PE-Cy5. For the detection of γδ T cells, mouse monoclonal antibody CD3 FITC and γδ T cell receptor (TCR) antibody staining were added after stimulation with phorbol ester and monensin, respectively. Cells were counted by flow cytometry using a BD FACS versus flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA), and data were analyzed with FlowJo software (BD Biosciences, Franklin Lakes, NJ, USA).
Immunohistochemistry
Immunohistochemistry was used to detect the expression of transcription factors T-box expressed in T cells (T-bet), GATA-binding protein 3 (GATA3), retinoic acid receptor-related orphan receptor γt (RORγt), forkhead box protein P3 (Foxp3), and γδ TCR γδ in the nasal mucosa and lung tissue, following the manufacturer's instructions. The tissue was sliced into 4μm sections, paraffin-embedded, and baked at 60 °C for 2 h. After dewaxing with xylene for 10 min, twice, the sections were treated with absolute ethanol for 3 min, twice, followed by 95% ethanol for 2 min, twice, and 80% ethanol for 2 min. The sections were then subjected to high temperature and pressure for antigen retrieval and serum was added to block for 20 min. The corresponding primary antibodies were added and incubated overnight at 4 °C. The secondary antibody was then applied and incubated for 1 h at room temperature. Diaminobenzidine was used for 1 min to develop the staining, followed by counterstaining with hematoxylin for 1 min. The sections were dehydrated with absolute ethanol, cleared with xylene, and mounted with neutral gum. The average optical density (AOD) of the positive cells expressed per unit was analyzed using ImageJ software (National Institutes of Health, USA).
Statistical Analysis
Data were expressed as means ± SD, and non-parametric values were expressed as median and interquartile range. One-way analysis of variance (ANOVA) was used to compare parametric data, and the Student-Newman-Keuls post hoc test was used when there were statistical differences between the groups. Kruskal-Wallis analysis, followed by Dunn's multiple comparison test, was used to evaluate the nonparametric data. P values < 0.05 were considered statistically significant. IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, N.Y., USA) and GraphPad Prism for Windows, version 9.5.1 (GraphPad Software, Massachusetts, Boston, USA) were used for statistical analysis.
Results
HPLC Chromatogram of XQLD
A total of eight peaks were identified as ephedrine hydrochloride (peak 1), paeoniflorin (peak 2), liquiritin (peak 3), cinnamic acid (peak 4), glycyrrhizic acid (peak 5), 6-gingerol (peak 6), schisandrol A (peak 7), and asarinin (peak 8) and compared to retention time-based standards (Figure 2). The standard curve was established to quantify the eight index components in the XQLD. The concentrations of ephedrine hydrochloride, paeoniflorin, liquiritin, cinnamic acid, glycyrrhizic acid, 6-gingerol, schisandrol A and asarinin in the XQLD were 0.78, 4.30, 2.09, 1.57, 3.39, 0.52, 0.91, and 1.43 mg/g in the XQLD sample, respectively.
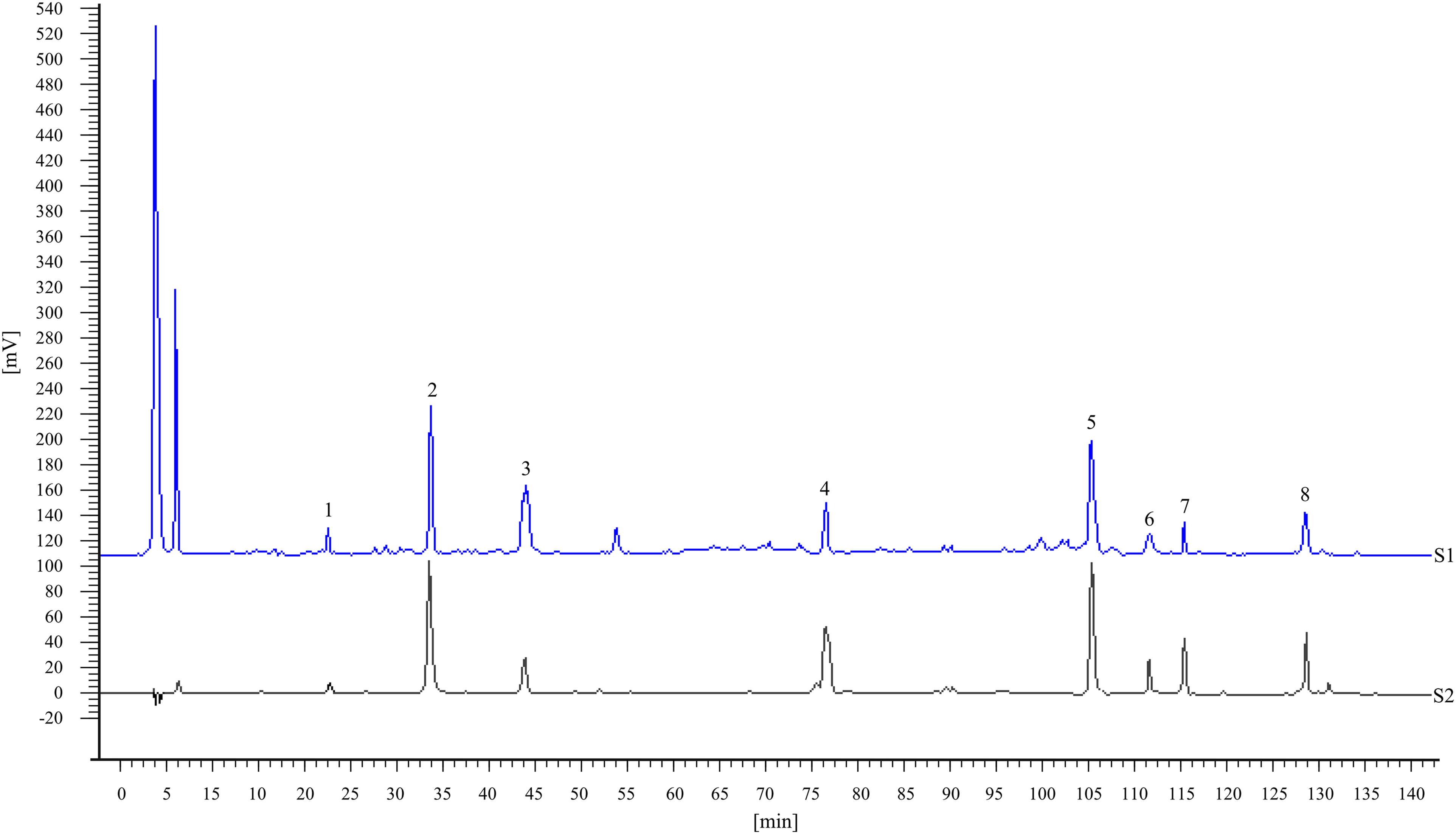
Representative high-performance liquid chromatography fingerprint of (S1) XQLD sample and (S2) mixed standard solutions. Ephedrine hydrochloride (1), paeoniflorin (2), liquiritin (3), cinnamic acid (4), glycyrrhizic acid (5), 6-gingerol (6), schisandrol A (7), and asarinin (8).
XQLD Ameliorated Allergic Symptoms and sRAW in CARAS Model Mice
Sneezing, nasal rubbing, and increased sRAW are the primary symptoms of CARAS. To investigate the effects of XQLD in OVA-induced CARAS model mice, we evaluated the severity of the allergic symptoms and sRAW in different groups. Within 15 min following the last OVA challenge, intense sneezing and nose-rubbing were observed in CARAS mice, compared to those of the Control group (Figure 3, P < 0.01). Notably, both sneezing and rubbing were significantly alleviated in the XQLD group (Figure 3, P < 0.05). Similarly, the DEX group exhibited markedly reduced nasal rubbing and sneezing compared to the CARAS group. The whole-body plethysmography revealed a significantly increased sRaw in CARAS model mice compared to the Control group, with a distinct decrease in sRaw observed in the XQLD and DEX groups, respectively (Figure 3). These findings indicate the successful establishment of an OVA-induced CARAS model in mice, with XQLD demonstrating notable alleviation of allergic symptoms and reduction in sRAW.

Establishment of ovalbumin (OVA)-induced mouse model of combined allergic rhinitis and asthma syndrome. The frequencies of sneezing (A) and nasal rubbing (B) were evaluated on day 60. Following aerosolized 1× phosphate-buffered saline and various doses of methacholine, and specific airway resistance (C, D) was measured using a Buxco system 24 h after the last OVA challenge. Control, the naive group. CARAS, combined allergic rhinitis and asthma. XQLD, CARAS mice treated with Xiaoqinglong Decoction, 6.0 g/kg. DEX, CARAS mice treated with dexamethasone as a positive control. sRAW, specific airway resistance. Data are presented as mean ± SD, n = 8. *P < 0.05, **P < 0.01.
XQLD Reduced Inflammatory Changes in the Nasal mucosa and Lung Tissue in the CARAS Mouse Model
Inflammation in the nasal cavity and lung plays a critical role in the progression of CARAS. We assessed the histopathological changes, including reduced cilia and eosinophil infiltration determined by H&E staining, and goblet cell metaplasia by PAS staining in sectioned nasal mucosa or lung tissue. Compared to the control group, the CARAS mice exhibited significantly elevated levels of eosinophils and goblet cells in the sections, accompanied by distinct epithelial damage and loss of cilia. Relatively, no apparent lesions were observed in the Control. In contrast, mice in the XQLD group and DEX group showed significantly reduced injuries in these sections, characterized by decreased eosinophil infiltration, reduced metaplasia of goblet cells, improved epithelial integrity, and cilia conditions Figure 4.

Representative photographs of stained sections of the nasal mucosa and lung tissues. (A) Nasal mucosa staining, hematoxylin and eosin (H&E) staining, scale bars = 50 µm. (B) Lung tissue, H&E staining, scale bars = 50 µm. (C) Nasal mucosa, Periodic acid-Schiff (PAS) staining, scale bars = 50 µm. (D) Lung tissue, PAS staining, scale bars = 50 µm. Control, the naive group. CARAS, combined allergic rhinitis and asthma. XQLD, CARAS mice treated with Xiaoqinglong Decoction, 6.0 g/kg. DEX, CARAS mice treated with dexamethasone as a positive control. Data represent one of three independent experiments.
XQLD Modulated serum OVA-Specific IgE and Cytokine Levels of CARAS Model Mice
IgE serves as the primary mediator of allergic reactions, triggering rapid activation of mast cells and basophils, leading to the release of inflammatory mediators. Serum OVA-specific IgE levels were significantly higher in CARAS mice compared to the Control (P < 0.01, Figure 5). Dexamethasone reduced serum OVA-specific IgE in OVA-induced CARAS mice (P < 0.05), while no significant changes were observed in the XQLD group (P > 0.05). The concentrations of IFN-γ, IL-2, IL-4, IL-5, IL-17 in the XQLD and dexamethasone groups exhibited a significant decrease compared to the CARAS group (P < 0.05 or 0.01), and TGF-β were significantly increased (P < 0.01). IL-10 was notably upregulated in the DEX group, while it decreased in the XQLD group compared to the CARAS mice. Furthermore, we analyzed the correlation between XQLD-mediated alleviation of allergic symptoms, serum OVA-specific IgE and inflammatory cytokines. The results indicated significant positive correlations between OVA-specific IgE, IFN-γ, IL-2, IL-4, IL-5, IL-17, and sneezing and nasal rubbing in XQLD-treated CARAS mice (P < 0.05 or 0.01, Figure 6).
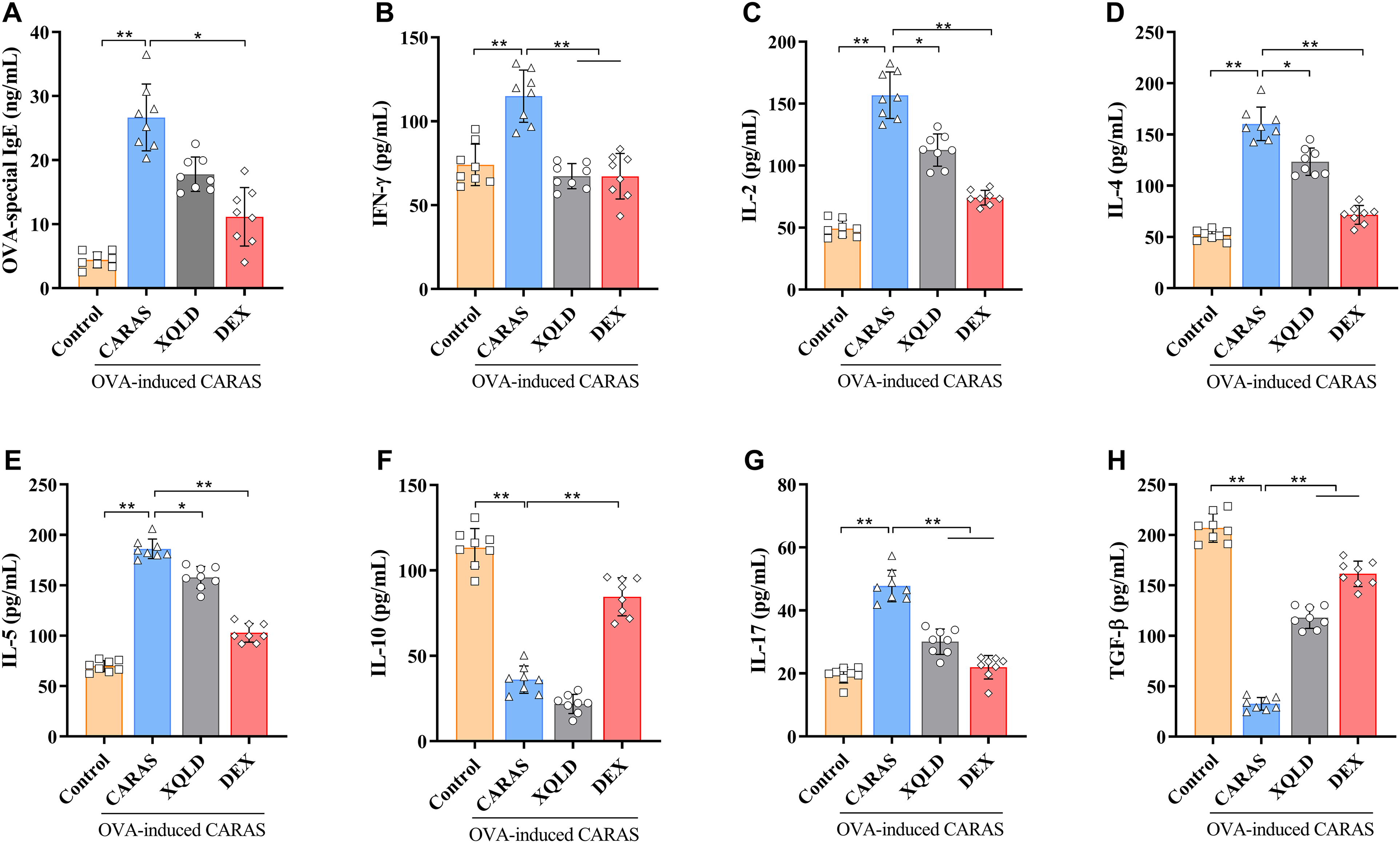
ELSIA analysis of OVA-specific IgE (A) and IFN-γ (B), IL-2 (C), IL-4 (D), IL-5 (E), IL-10 (F), IL-17 (G), TGF-β (H) in serum. Control, the naive group. CARAS, combined allergic rhinitis and asthma. XQLD, CARAS mice treated with Xiaoqinglong Decoction, 6.0 g/kg. DEX, CARAS mice treated with dexamethasone as a positive control. Data are presented as mean ± SD, n = 8. *P < 0.05, **P < 0.01.

Correlation between allergic symptoms and inflammatory mediators in Xiaoqinglong Decoction-treated combined allergic and rhinitis syndrome mice. (A-G) Correlation between the count of sneezing and OVA-specific IgE (A) and IFN-γ (B), IL-2 (C), IL-4 (D), IL-5 (E), IL-10 (F), IL-17 (G), TGF-β (H) in serum. (I-O) Correlation between OVA-specific IgE and IFN-γ (I), IL-2 (J), IL-4 (K), IL-5 (L), IL-10 (M), IL-17 (N), TGF-β (O) in serum.
XQLD Modulated the Frequencies of Th1, Th2, Th17, Treg, and γδ T Cells in Peripheral Blood of CARAS Mice
Th are believed to be involved in the development and progression of CARAS in patients. Specifically, these cells are responsible for releasing cytokines that induce inflammation in the airways and nasal passages. Understanding the involvement of Th in CARAS is essential for developing effective treatment approaches that target the underlying immune dysfunction. As shown in Figure 7, the percentages of Th2, Th17 and γδ T cells were significantly higher in CARAS mice compared to the Control group (P < 0.05). In the XQLD group, higher proportions of Th1 and lower proportions of Th2 and Th17 cells were observed compared to the CARAS group (P < 0.05). While the average percentages of Th1, Th2, Th17 and γδ T cells in the XQLD group were lower than those in the DEX group, the differences were not statistically significant (P > 0.05). Thus, XQLD led to an increase in the percentages of Th1 cells and a decrease in Th2 and Th17 cells in the OVA-induced CARAS murine model.
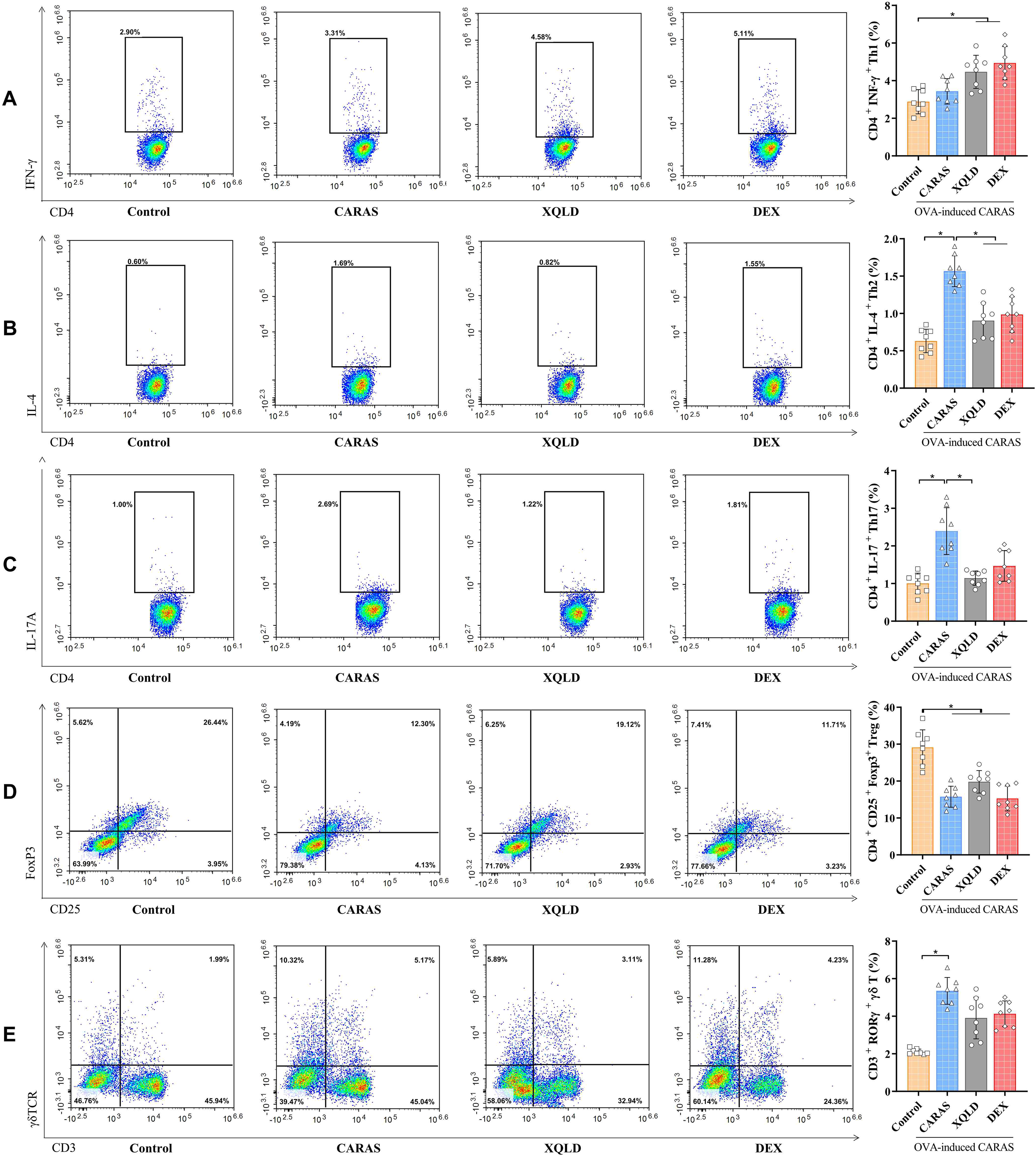
Flow cytometry analysis of Th1 (A), Th2 (B), Th17 (C), treg (D), and γδ T cells (E) in the peripheral blood of each group. Representative flow cytometry graphs of three independent experiments are displayed. Control, the naive group. CARAS, combined allergic rhinitis and asthma. XQLD, CARAS mice treated with Xiaoqinglong Decoction, 6.0 g/kg. DEX, CARAS mice treated with dexamethasone as a positive control. Data are presented as mean ± SD, n = 8. *P < 0.05.
XQLD Regulated the Expression of T-Cell Subset Molecules in the Nasal mucosa and Lung Tissues of CARAS Mice
Transcription factors within T helper cells play a critical role in determining the cell's fate and functionality. The impact of XQLD on the expression of T-bet, GATA3, Foxp3, RORγt and γδ TCR in the nasal mucosa and lung tissues of mice was assessed through immunohistochemistry, as outlined in Figure 8. Expression levels of T-bet, GATA3, RORγt, and γδ TCR were significantly elevated (P < 0.05) in CARAS mice in comparison to the Control group, both in nasal mucosa and lung tissue. Treatment with XQLD led to a decrease in the expression of GATA3, RORγt (except for the lung) and γδ TCR mRNA (P < 0.05). While there were no significant differences in Foxp3 expression among the groups in the nasal mucosa, a notable increase was observed in the lung sections of mice treated with XQLD compared to those of the Control group.
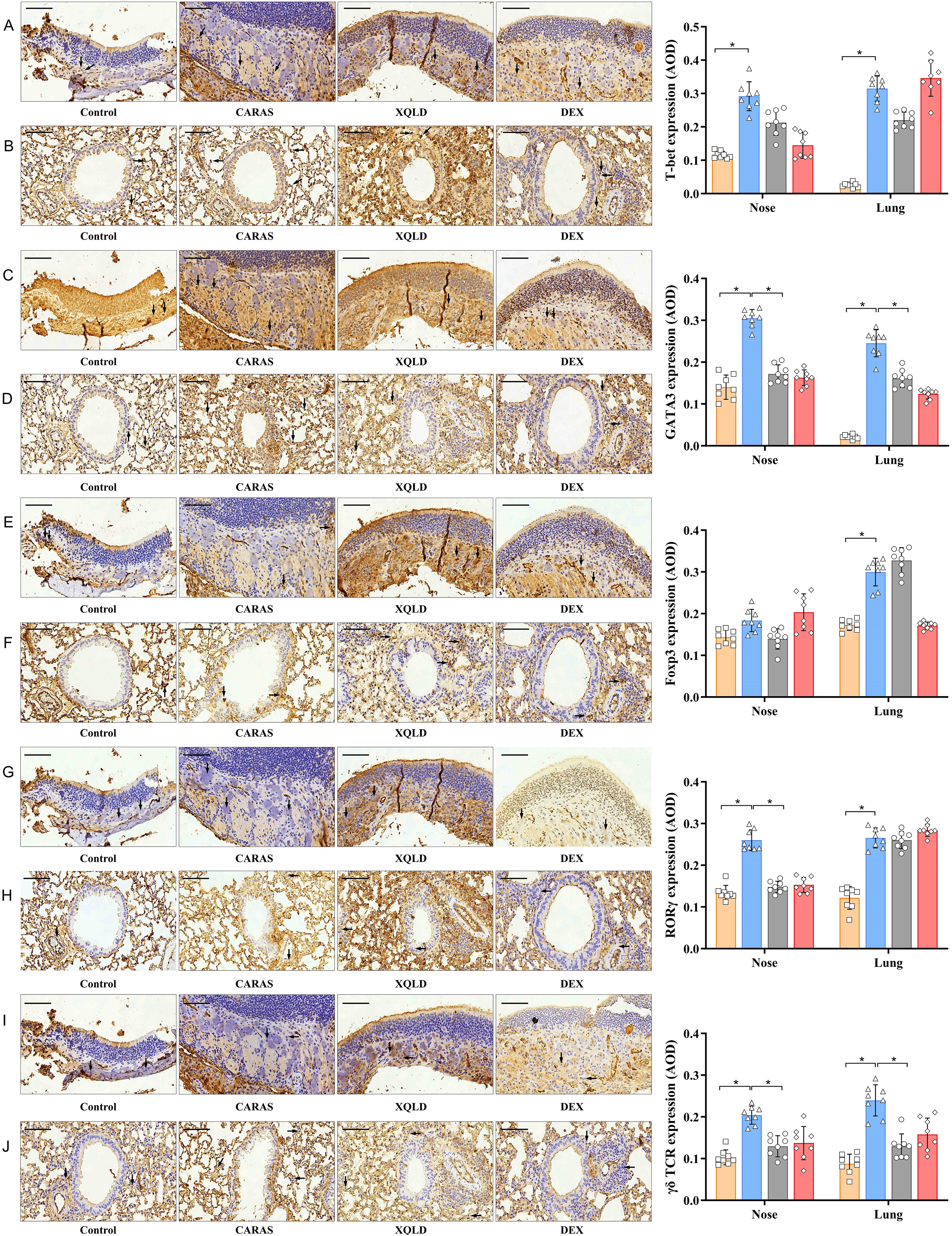
Effects of XQLD on the expression of the T lymphocyte-related proteins T-bet, GATA3, Foxp3, RORγt and γδ TCR in the nasal mucosa (A, C, E, G, I) and lung tissue (B, D, F, H, J) of mice in each group. Immunohistochemistry, scale bars = 50 µm. The photomicrographs exhibit the representative morphology of the nasal mucosa of mice in each group. Control, the naive group. CARAS, combined allergic rhinitis and asthma. XQLD, CARAS mice treated with Xiaoqinglong Decoction, 6.0 g/kg. DEX, CARAS mice treated with dexamethasone as a positive control. T-bet, T-box expressed in T cells. GATA3, GATA-binding protein 3. RORγt, retinoic acid receptor-related orphan receptor γt. Foxp3, forkhead box protein P3. TCR, T cell receptor. Data represent one of three independent experiments. AOD, average optical density. *P < 0.05.
Discussion
The research demonstrated that XQLD has anti-inflammatory and immunoregulatory effects in a murine model of CARAS. XQLD treatment reduced inflammatory changes in the nasal mucosa and lung tissue, decreased the levels of OVA-specific IgE and cytokines (IFN-γ, IL-2, IL-4, IL-5, and IL-17), and modulated the frequencies of Th1, Th2, Th17, Treg, and γδ T cells in the peripheral blood of CARAS mice. These findings suggest that XQLD may be a potential therapeutic option for the treatment of CARAS.
In this study, the HPLC analysis of XQLD revealed that ephedrine hydrochloride, paeoniflorin, liquiritin, cinnamic acid, glycyrrhizic acid, 6-gingerol, schisandrol A, and asarinin are the primary components. Ephedrine hydrochloride, paeoniflorin, cinnamic acid and schisandrol A exhibit various pharmacological activities, including anti-allergic, anti-inflammatory, antioxidant and immunoregulatory effects, as documented in previous studies.23–26 These components contribute to the relaxation of bronchial smooth muscles and the alleviation of nasal congestion. Liquiritin and glycyrrhizic acid demonstrate anti-allergic and immunomodulatory properties through the inhibition of mast cell degranulation.27,28 6-Gingerol and asarinin exhibit anti-asthmatic effects by suppressing the Th2 cell-mediated allergic response.29,30
Elevated levels of antigen-specific IgE have been demonstrated to be associated with allergic inflammation and airway hyperresponsiveness in the murine model of CARAS. 31 Allergen-induced IgE increased cytokine secretion, involving IL-4, IL-5, and IL-10 by eosinophils, basophils, and mast cells. 32 Our investigation revealed that XQLD significantly reduced OVA-specific IgE levels, suggesting its efficacy in suppressing allergic inflammation.
The development of CARAS is mainly attributed to the predominant differentiation of Th2 cells and the overexpression of related cytokines such as IL-4 and IL-5. 33 IL-4, as a key regulator of IgE secretion, effectively promotes B cell differentiation into plasma cells. 34 IL-5 plays a wide role in eosinophil cell differentiation, maturation, proliferation and chemotaxis, while also inhibiting apoptosis and promoting IgE secretion in coordination with IL-4.35,36 IL-17 contributes to the Th2-mediated immune response, enhancing eosinophil infiltration into the airways, and independently increasing airway hyperresponsiveness in the absence of Th2 cells. 37 The levels detected by ELISA in our study were consistent with changes in OVA-specific IgE. While the CARAS group exhibited a significant increase in levels of IL-4, IL-5, and IL-17, both the XQLD group and the DEX group demonstrated marked reductions.
IL-2 and IFN-γ enhance Th1 cell bioactivity and immune function, inhibiting the intensity of the humoral immune response by suppressing Th2 cell proliferation. 38 Consequently, they also hinder the production of Th2-related cytokines such as IL-4 and IL-5, exerting positive regulatory effects on allergic diseases. IL-10 exhibits robust anti-inflammatory activity in inhibiting the production of IL-2 and IFN-γ in mRNA reverse transcription. It inhibits Th2 cell differentiation, subsequently reducing type-2 cytokines. 39 IL-10 can inhibit the expression of Eotaxin mRNA in eosinophils, exerting anti-allergic effects and promoting accelerated eosinophil apoptosis. 40 TGF-β functions as a crucial regulator of the immune balance between Th17 and Treg. Low levels of TGF-β induce Th17 cell differentiation, promoting inflammatory effects, whereas high levels of TGF-β promote Treg cell production, playing an anti-inflammatory role. 41 Currently, IL-10 + and IFN-γ + γδ T cells have been found to increase during the treatment of atopic dermatitis, which is the most common co-occurence of asthma and allergic rhinitis. 42 The current study demonstrated a significant elevation in serum IL-10 and TGF-β levels in the XQLD group compared to CARAS mice, aligning with the DEX group. The levels of IL-2 and IFN-γ in the XQLD mice were significantly lower than those in CARAS mice, consistent with the findings of Tanaka. 43
The cellular mechanism underlying CARAS involves a series of allergic reactions primarily induced by the immune imbalance of Th1/Th2 and Th17/Treg cells. The immune system response to internal and external stimuli and the development of CARAS are closely associated with cytokines released by polarized Th cells, with some variability in the populations of these polarized cells. IL-17, secreted by Th17 cells, exerts a potent influence on granulocyte recruitment and the promotion of differentiation and maturation of various types of inflammatory cells. Treg cells act as a negative feedback regulator of T cell activation, exhibiting immunosuppressive effects to maintain immune tolerance and homeostasis through extensive inhibition of T effector cells.44,45 XQLD demonstrated a significantly capability to reduce Th2 and Th17 cell differentiation in peripheral blood, as well as the expression of GATA3 and RORγt in the nasal mucosa, indicating its anti-allergic effects on T-cell polarization.
As research on allergic rhinitis progresses, γδ T cells are emerging as a new focus in the pathogenesis and treatment of CARAS. Research has substantiated extensive infiltration of γδ T cells in the nasal mucosa of CARAS patients, with significantly higher cell frequencies than those of healthy individuals, positively correlated with peripheral blood eosinophils, macrophages, mast cells, and IL-17+ T cells of the patients.33,46 γδ T cells enhance IgE secretion by B cells, thereby stimulating cascade reactions. 47 In this study, both the peripheral blood γδ T ratio and the expression of γδ TCR in the nasal mucosa of mice in the high XQLD group were lower than those of the CARAS group, suggesting that XQLD inhibited T-cell differentiation of γδ T cells and the inflammatory response.
This study may have certain limitations. In clinical practice, the doses for various herbal drugs of XQLD are established. However, this study focused solely on determining the most appropriate dose to observe the pharmacological effects of XQLD based on the human-to-mouse dose conversion factor, while neglecting experimental observations in low and high-dose groups. This oversight hinders the elucidation of the dose-dependent effects and drug toxicity of XQLD in anti-inflammatory and anti-allergic pathways. To improve the understanding of its pharmacological properties, future research should incorporate various dose groups. Additionally, potential variations in the chemical composition of XQLD result from small deviations in the extraction processes, despite implemented quality control measures. Therefore, future research and applications could benefit from the development of a more stable dosage form. Furthermore, clinical studies on CARAS should validate the anti-allergic effects of XQLD, which should be considered in the future.
The results of this study provide information on the potential therapeutic role of XQLD in CARAS. These results contribute to the understanding of XQLD as a treatment option in the field of allergic respiratory diseases.
Conclusions
In conclusion, XQLD significantly inhibited allergic reactions by reducing serum OVA-specific IgE and various cytokines and restoring the balance of Th1 / Th2, Th17 / Treg and γδ T cells. Furthermore, XQLD modulated the expression of the T lymphocyte transcription factors mRNA and γδ TCR in the nasal mucosa and lung tissue of mice with allergic rhinitis. These results suggested that XQLD could attenuate allergic reactions and proved its effectiveness as an alternative medicine in the treatment of CARAS.
Footnotes
Abbreviations
Author Contributions
Data Availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All procedures were approved by the Institutional Animal Care and Use Committee of the Beijing Hospital of Traditional Chinese Medicine (No. SQ-2021-02-62, granted in February 2021), Beijing, China.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China [grant number 82174448, 82205203, 82374529].
