Abstract
Background
Mucunain, a cysteine protease extracted from the spicules of cowhage, can induce intense itching as a pruritus mediator independent of histamine. It is well known that erythema, edema and vesicles on skin may occur immediately following contact with Mucunain. A recent study has demonstrated that Mucunain may trigger pruritus and inflammation on the left or right groin of each Maltese–Beagle atopic dog. The aim of this study was to investigate the cytotoxic effect of Mucunain on a cocultured skin epithelial cell model in vitro.
Methods
Functional recombination Mucunain (rtMucunain) were obtained following induction, purification, and renaturation. Human keratinocytes and dermal fibroblasts were cocultured to construct a cocultured cell model skin epithelium cell model. The growth inhibition was measured using an MTS test. Microscopy was used to observe morphological alterations in cell architecture. The DCFH-DA assay was used to evaluate reactive oxygen species production, the cells death type was identified using a Flow cytometry assay, and the pro-inflammatory cytokines IL-6 and −8 were detected using an ELISA assay. The ability to form an apoptosis network was investigated by a Western blot assay to assess apoptosis-related protein expression.
Results
Following purification and renaturation, rtMucunain was obtained with a yield of up to 50 mg/L. In the cocultured skin epithelial cells treated with rtMucunain, cell growth was inhibited in a dose- and time-dependent manner, with doses of 2.5 µM or higher resulting in significant cytotoxicity. Morphological analysis revealed increased cell dispersion and abnormal morphology, particularly following prolonged exposure. Levels of reactive oxygen species were elevated, indicating oxidative stress. Flow cytometry and Western-blot data demonstrated that the cells underwent apoptosis, with extrinsic apoptosis pathways (NF-κB and Caspase-8) being activated by rtMucunain. Furthermore, treatment with rtMucunain markedly enhanced levels of IL-6 and IL-8, suggesting a robust pro-inflammatory response.
Conclusion
rtMucunain showed a strong inflammation and apoptosis effect in a cocultured skin epithelium cell model. This effect may be due to the activated Caspase 8 signaling pathway, on which NF-κB might play an important role. It was the first time to clarify the cytotoxic effect of rtMucunain on the skin epithelial cell model, and the comprehensive understanding the damaged mechanisms of rtMucunain on normal skin cells can provide a data basis for the skin irritated experiment of rtMucunain and its analogues.
Introduction
Itching is a subjective sensation that can immediately lead to scratching behavior. A variety of systemic disorders and dermatological diseases are usually accompanied by itching symptoms. Long-term chronic itching has a significant impact on patients’ physical, mental, and quality of life. It has been confirmed that multiple signaling pathways are involved in the transduction process of itching, including histamine-dependent and histamine-independent itch signaling pathways, 1 and the efficacy of therapies remains largely limited. As so far, the prevalent mediators of itching include histamine, biogenic amines, neuropeptides, eicosanoids, proteases, cytokines and others.
Mucunain, as a cysteine protease, was firstly extracted from the spicules of cowhage, and has been proven to evoke more intense itch on histamine-independent itch signaling pathway than histamine-dependent itch signaling pathway. 2 Mucunain is an exogenous PAR-2 ligand and PAR4, 3 and induces an itching characterized by burning and pricking sensations. This process is mediated by a population of capsaicin-sensitive afferent nerve fibers, all mechano-heat-sensitive C fibers (CMHs) are activated, which are distinct from mechanically-insensitive C fibers (CMi) mediating histamine-induced itching.4,5 The elevated levels of PAR-2 were also expressed in the epidermis and cutaneous nerve fibers of chronic pruritic patients, including those with atopic dermatitis (AD). It has been identified that Mucunain can be used as an ideal pruritogens for the research of chronic pruritus disease such as AD. 6 In addition to pruritus, Mucunain can also trigger inflammation responses. 7 Plant proteases, such as bromelain, ficin and papain, are signaling molecules and can activate human PAR2 and PAR4 via cleaving the N-terminus ‘tethered ligand’ of the receptors themselves, and PARs activation is associated with pain and inflammation.8,9 A variety of plant proteases, such as ficin and papain can usually be used for anthelmintics by the capacity of enzymatic attack and digestion of the worms. 10 However, there have been little studies on the cytotoxic effects induced by plant proteases.
The skin is composed of the epidermis and dermis, with approximately 90% consisting of keratinocytes. Keratinocytes can stimulate fibroblasts to produce growth factors, in turn, dermal fibroblasts are capable of producing pro-inflammatory cytokines and chemokines.11,12 With the rise of alternative toxicology and the extensive application of tissue engineering technology in toxicology, tissue-engineered skin models for skin irritation testing have become a focal point of research. The coculture model of keratinocytes and fibroblasts was extensively developed in the 1990s,13–15 and possess the advantage of the naturally occurring crosstalk between cell types, 16 which makes it an ideal model for assessing skin irritation. Even though the pain, edema, erythema, and even vesicles are also occurred after primate and rodent skin contact with Mucunain,2,3,10,17 the cytotoxicity of Mucunain is still unknown. 9 In our study, we constructed a cocultured skin epithelial cell model to assess the cytotoxic effect of exposure to truncated recombinant Mucunain, which exhibits bioactivity, and indues continuous Ca2+ change in Hela cells stably expressed PAR2 and PAR4 receptors. 18 Our findings would provide foundational data on the potential application of Mucunain as a histamine-independent pruritus mediator.
Material and Methods
Strains and Reagents
Standard chemically transformation competent strains of E. coli: DH5α competent cells and Rosetta (DE3) competent cells were from Tiangen (Beijing, China). The expression plasmids pET-28a-Sumo was purchased from Novagen (Darmstadt, Hesse). The SUMO protease was from Solarbio (Beijing, China). DMEM/F12 medium, K-SFM medium, 1% (v/v) penicillin-streptomycin, 10% (v/v) fetal bovine serum (FBS), serum extracts, epidermal growth factor (EGF), T4 DNA ligase, restriction endonuclease Kpn Ⅰ and Xho Ⅰ, 1 kb DNA Ladder, and pre-stained protein MW marker 26616 were purchased from Thermo Fisher Scientific (Waltham, MA). Human IL-6 and IL-8 ELISA kits were purchased from Ray Biotech (Norcross, GA). Cell Titer® 96 AQueous One Solution Reagent was purchased from Promega (Madison, WI). The reactive oxygen species (ROS) assay kit was obtained from ABP Biosciences (Rockville, MD). Polyvinylidene fluoride membrane was purchased from Millipore (Billerica, MA). Anti-Caspase 3 (ab13847), anti-Caspase 8 (ab25901), anti-Caspase 9 (ab286147), anti-Bax (ab32503), anti-Bad (ab32445), anti-Bcl-2 (ab32124), anti-NF-κB-p65 (ab16502), anti-Bid (ab32060), anti-CYLD (ab137524) and anti-β-actin (ab8227) were purchased from Abcam (Cambridge, UK). PE Annexin V Apoptosis Detection Kit I was purchased from BD Biosciences (San Diego, CA). IRDye 800 conjugated with affinity-purified anti-Rabbit IgG antibody was purchased from Rockland (Philadelphia, PA).
Cells Culture
Human primary keratinocytes HSK and dermal fibroblasts HSF were purchased from NICR (National Infrastructure of Cell Line Resource, Beijing, China). Human primary dermal fibroblasts were grown in monolayer and cultured in DMEM/F12 medium, supplemented with 1% (v/v) penicillin-streptomycin (100 μg/mL penicillin, 100 μg/mL streptomycin) and 10% (v/v) FBS. Human primary keratinocytes were cultured in K-SFM medium, supplemented with 1% (v/v) penicillin-streptomycin, 500 μg/mL serum extracts, and 5 ng/mL EGF. Cells were grown in 25 cm2 tissue culture flasks at 37 °C under an atmosphere of 5% CO2 in a humidified incubator and passaged every 5–7 days.
Construction of Human Skin Epithelial Cell Model
The human skin epithelial cell model was constructed as follows. Briefly, human keratinocytes were inoculated at a density of 5 × 104 cells/well in 6-well plate and cultured at 37 °C under an atmosphere of 5% CO2 in a humidified incubator for 24 h, and then the dermal fibroblasts were inoculated in 6-well plate at a density of 1.25 ×104 cells/well. Human keratinocytes and dermal fibroblasts were inoculated as a ratio of 2 × 103:0.5 × 103 cells/well in 96-well plate. K-SFM and DMEM/F12 medium were used at a ratio of 3:1.
Expression, Renaturation and Purification of the Recombinant rtMucunain
The active Mucunain gene (93-309) was synthesized and cloned into the pUC57 vector by Nanjing Genscript Biologic Technology Limited Company (Nanjing, China), and the plasmid rMucunain-pUC57 was obtained. The vectors pET-28a (+)-Sumo and rMucunain-pUC57 were digested with Kpn Ⅰ and Xho Ⅰ for linearization. The linearized vector and the active Mucunain gene fragment (93-309) were ligated by T4 DNA ligase and then transformed into strain DH5α of E.coli to obtain the recombinant plasmid rtMucunain-pET-28a-Sumo.
The fusion protein Sumo-
Cell Proliferation Assay
The evaluation of the influence of rtMucunain on the proliferation of skin epithelial cells was conducted using 3-(4, 5-dimethylthiazol-2-yl)-5-(3-carboxy-methoxyphenyl)-2-(4-sulphophenyl)
-2H-tetrazolium (MTS) assay. After coculture in 96-well plate for 24 h, the cells were treated with 2.5 μM, 5 μM, 10 μM, 20 μM and 40 μM rtMucunain for 5 h, 24 h and 48 h. A volume of 20 L of Cell Titer® 96 Aqueous One Solution Reagent was dispensed into each well of the 96-well plate at intervals of 0 h, 5 h, 24 h, and 48 h. Following incubation for 60 min, the absorbance at an optical density of 490 nM was determined using the multi-detection microplate reader (BIO-TEK Instruments Inc., Winooski, VT). Calculated the mean and standard error of mean (SEM) for each concentration, and the cell proliferation and inhibition rate were calculated according to the formula “Cell proliferation=
Fluorescent Detection of Intracellular ROS Production
The dichlorofluorescein (DCF) method was employed to determine the intracellular ROS level on skin epithelial cells that were stimulated by rtMucunain. After coculture in a 6-well plate for 24 h, the cells were treated with rtMucunain in a dose of 5 μM for 5 h and 24 h. Followed by washing twice with 0.01 M PBS (pH7.4), the treated cells were incubated with 15 μM DCFH-DA for 30 min at 37 °C. Subsequently, cells were thoroughly rinsed three times with HBSS and incubated with DMFM/F12 medium at 37 °C for 10 min. The fluorescent signal was detected with a Cytation cell imaging multi-mode reader (BIO-TEK Instruments Inc., Winooski, Vermont) with an excitation wavelength of 485 nm and an emission wavelength of 590 nm.
Flow Cytometry
The type of cell death was investigated by flow cytometry. The skin epithelial cells were cocultured in a 6-well plate and treated with rtMucunain for 5 h and 24 h, respectively. After collection and rinse with PBS (0.01 M, pH 7.4), the treated cells were resuspended in 100 μL binding buffer (10 mM HEPES pH 7.4, 140 mM NaCl, and 2.5 mM CaCl2) containing 5 μL 7-ADD and 5 μL Annexin V-FITC, and incubated in the dark for 15 min at room temperature. And then the 400 μL binding buffer was added to the sample tubes and analyzed by FACS Calibur (Becton Dickinson, Franklin Lakes, NJ). At least 10,000 events were recorded for each sample in the FL1 channel (green fluorescence: 530/30 nm) and in the FL2 channel (orange-red fluorescence: 585/42 nm). Data analysis was performed with FlowJoTM software (version 10.0.6, Tree Star, Ashland, OR).
Western Blot Analysis
Apoptosis-related proteins were assayed by Western blot following the protocol. In brief, cocultured cells treated with 5 μM rtMucunain for 5 h and 24 h were collected. Proteins were separated by 15% SDS-PAGE and transferred to polyvinylidene fluoride membranes. The membranes were probed with polyclonal rabbit antibodies against anti-Caspase 3, anti-Caspase 8, anti-Caspase 9, anti-Bid, anti-CYLD, anti-NF-κB-p65, anti-Bax, anti-Bad, anti-Bcl-2 and anti-β-actin (Abcam, Cambridge, UK) overnight at 4 °C and incubated with IRDye 800 conjugated with affinity purified anti-Rabbit IgG antibody for 1.5 h at room temperature. The membranes were scanned by the Odyssey Infrared Imaging System (LI-COR Bioscience, Lincoln, NE).
ELISA Assay of Cytokine IL-6 and IL-8
An enzyme-linked immunosorbent assay (ELISA) was used to detect the expression levels of intracellular interleukins on skin epithelial cells. Following the standard operating procedures of Human IL-6 and IL-8 ELISA kit, the supernatants of 4∼5 × 105 cocultured epithelial cells treated with 5 μM rtMucunain for 5 h and 24 h were collected to analyze the secretion of IL-6 and IL-8 for ELISA. The absorbance was measured using the multi-detection microplate reader (BioTek Instruments Inc., Winooski, VT, USA) at an optical density of 450 nm.
Data Statistics and Analysis
All in vitro experiments were done in triplicate and repeated three times, and the values were recorded as the Mean ± SEM. Statistical difference between Mean values was calculated by T-test, and statistical significance was taken as *p<0.05. Statistical analysis of the data was performed by GraphPad Prism 8.0.1.
Results
Cloning, Expression and Purification of Recombinant Mucunain in E. coli Rosetta (DE3)
The full length of Mucunain gene encodes the entire precursor protein (propeptide and mature peptide). Mucunain consists of three parts: I29 inhibitor domain, C1A cysteine proteases domain, and GRAN domain (Figure 1). 20

The composition of Mucunain. Mucunain includes 422 amino acids, which can be divided into three domains according to their functions: I29 (4-59) inhibitory domain, C1 (93-309) cysteine proteases domain, and GRAN domain (333-389).
Based on the predicted results of structural activity and literature research, it is speculated that the active site is located at position 93–309 of the amino acid sequence. Therefore, the truncated tMucunain-pUC-57 encoding the active domain 93–309 of Mucunain was constructed by gene synthesis, and was subcloned into pET-28a (+)-Sumo, the recombination plasmid rtMucunain-pET-28a-Sumo was obtained (Figure 2 A). After a series of optimizations, the fusion protein His6-Sumo-rtMucunain was produced with an approximate molecular weight of 42 kD, and predominately presented as inclusion bodies following induction with 0.05 mM IPTG at 18 °C for 48 h (Figure 2 B). Subsequently, the inclusion bodies were sonicated, dissolved in 8 M urea, and cleaved using SUMO Protease. The His6-Sumo-rtMucunain was then isolated through an agent-resistant Ni-NTA affinity chromatography column. Refolding produced recombinant rtMucunain with a yield of up to 50 mg/L (Figure 2 C). rtMucunain displyed superior bioactivity than natural Mucunain in our previous study 18 and was used to investigate the cytotoxic effects of Mucunain on skin epithelial cells.
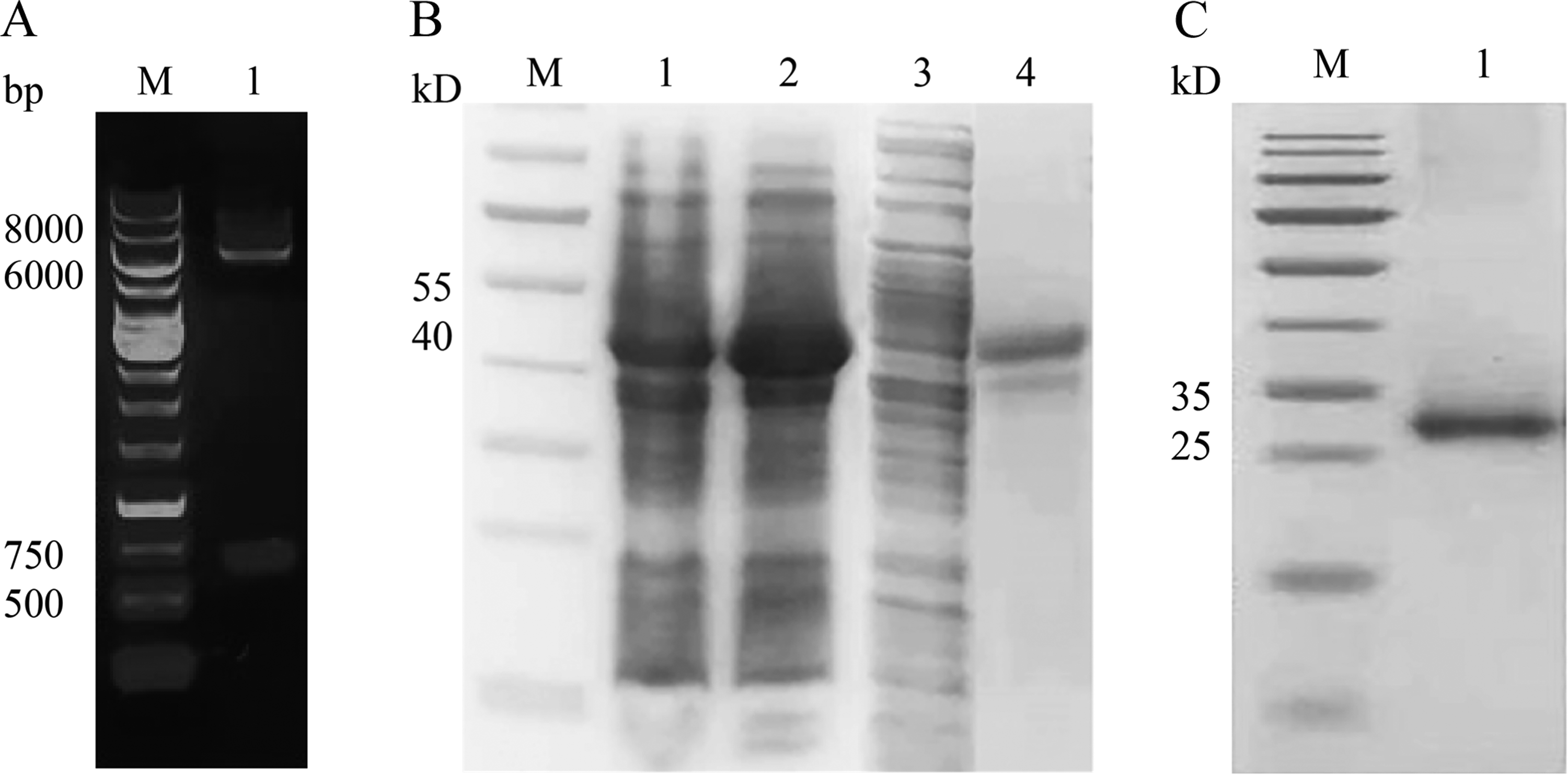
Cloning, expression, and purification of recombinant truncated rtMucunain. A. The gel image of plasmid rtMucunain-pET-28a-Sumo digested by Kpn Ⅰ and Xho Ⅰ, M, 1 kb DNA Ladder; B. The 15% SDS-PAGE analysis of rtMucunain-pET-28a-Sumo expressed in E. coli Rosetta (DE3), lanes 1 and 2 were the uninduced and induced fusion protein, respectively; lanes 3 and 4 were the fusion protein in the supernatant and precipitation, respectively, M, Pre-stained protein MW marker 26616; C. The 15% SDS-PAGE analysis of purified and refolded rtMucunain, M, Pre-stained protein MW marker 26616.
Inhibition of Proliferation
To determine whether rtMucunain has any effect on cell proliferation, the MTS assay was performed to detect the proliferation of cells treated with rtMucunain in different doses at different time points, which is a colorimetric method for determining the number of viable cells in proliferation or cytotoxicity assay. As Figure 3 A, B shown, the viability of cells treated with 2.5 μM rtMucunain for 5 h had been obviously inhibited, and totally abolished for 48 h in dose of 40 μM rtMucunain. Along with the exposure time, the cell growth treated with rtMucunain was inhibited in a time- and dose- dependent manner. The inhibited percentages of cells by rtMucunain in dose of 2.5 μM, 5 μM, 10 μM and 40 μM were respectively up to almost 11%, 16%, 27%, 40%, and 67% at 24 h, and ultimately reached to 42%, 43%, 56%, 60%, and 83% at 48 h. In addition, curve fitting was performed on the inhibition rate of cell proliferation, and the IC50 of rtMucunain was determinately 32.19 μM at 24 h, as calculated using GraphPad Prism 8.0.1 (Figure 3 C). The effect of cell proliferation treated with 2.5 μM and 5 μM rtMucunain was not significant difference at 24 h, therefore, in subsequent experiments, 5 μM rtMucunain was chosen as an exposure concentration.

Proliferation of skin epithelial cells was inhibited by rtMucunain. The proliferation of cells treated with rtMucunain was analyzed by MTS assay at 0 h, 5 h, 24 h and 48 h. A, B. Effect of rtMucunain on the proliferation of skin epithelial cells in dose of 2.5 μM, 5 μM, 10 μM, 20 μM and 40 μM; Cell proliferation was represented as the average OD490 ± SEM, **p<0.01; C. Cytotoxicity was represented as the concentration of rtMucunain inhibiting cell growth at 24 h by 50% (IC50).
Morphological Effects of rtMucunain on Skin Epithelial Cells
To investigate the cytotoxic effect of rtMucunain on skin epithelial cells in a dose of 5 μΜ, the morphological alterations in cell architecture were observed by microscope. As shown in Figure 4, by contrast, after 5 h of exposure to rtMucunain, phenotypic alterations of cells were observed, the boundaries of treated cells became indistinct, and cellular swelling was evident. Following an extended exposure time of 24 h, morphological alterations included the emergence of apoptosis bodies, cytoplasmic blebbing, and slight detachment of the cells. At 48 h, a significant number of cells had detached from the dishes. These findings suggested that the cytotoxicity of human skin epithelial cells might be triggered by rtMucunain in a time-dependent manner.
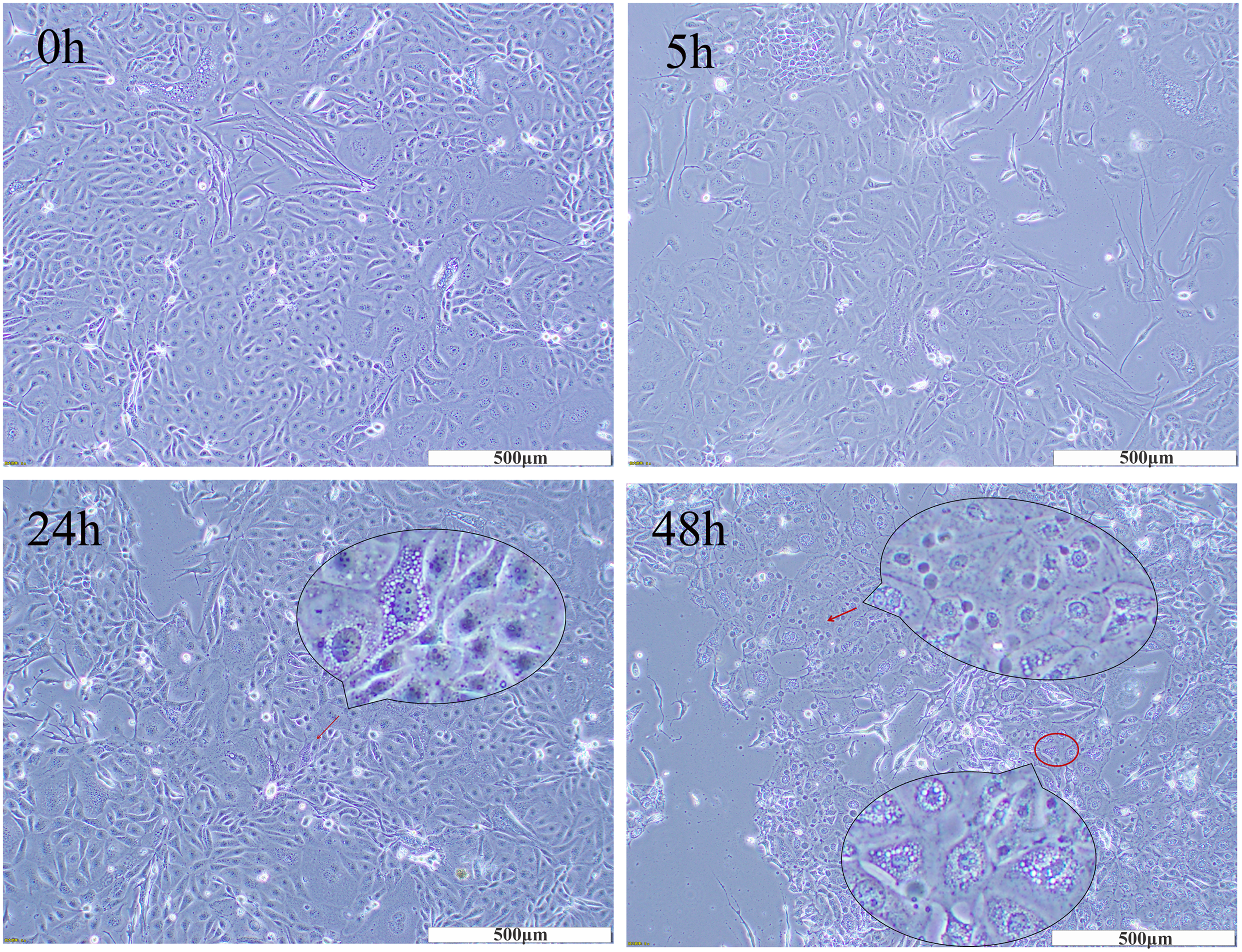
Morphological alterations of skin epithelial cells in response to rtMucunain. Cocultured cells treated with rtMucunain for 5 h, 24 h and 48 h were photographed by microscopy, in × 100 magnifications. Red circle: blebbing of cytoplasm; Red arrow: apoptotic bodies.
rMucunian Increased the ROS Level of Cocultured Skin Epithelium Cells
In view of the cell proliferation inhibition and the morphological alterations of skin epithelial cells treated with rtMucunain, we hypothesized that the elevated intracellular ROS might occur after exposure to rtMucunain. The endogenous production of ROS was measured using the DCF method. Cells were treated with rtMucunain in a dose of 5 μM for durations 5 h and 24 h, 100 μM TBHP served as a positive control, beknown as an effective ROS inductor after one hour of exposure to cells. 21 Notably, the intensity of intracellular fluorescence in cells treated with rtMucunain was significantly enhanced compared to the control group, indicating that rtMucunain could indeed induce intracellular ROS formation in a time- dependent manner (Figure 5). The level of ROS may act as one potential switch that modulates between apoptotic and nonapoptotic roles of caspase-8 within target cells. 22 Therefore, we proposed that the intracellular formation of ROS could be a plausible mechanism underlying cell damage contributing to apoptosis.

Effect of rtMucunain on the accumulation of ROS in cocultured skin epithelial cells. Fluorescence imaging system was employed to detect the production of ROS in skin epithelial cells pretreated with 5 μM rtMucunain for 5 h and 24 h using DCFH-DA. Cellular ROS (green color), Control (black and white color). In × 200 magnifications. Scale bar was 100 μm. The histogram was represented as the relative fluorescence intensity in treated skin epithelial cells.
rtMucunain Induced Apoptosis via the Caspase 8 Activation
Morphological alteration, inhibition of cell proliferation, and the release of cellular ROS within the skin epithelial cells suggested the apoptosis might be triggered by rtMucunain. To investigate this further, a flow cytometry assay was first conducted to determine the type of cell death in skin epithelial cells exposure to rtMucunain for 5 h and 24 h. The cells were stained with a combination of Annexin V-FITC and 7-Amino-Actinomycin (7-ADD) to identify early apoptotic cells as well as in late apoptosis or already dead. Surprisingly, apoptosis was observed in cells treated with rtMucunain at 5 h and 24 h, moreover, extending the treatment duration resulted in a transition from a viable to early/late stages apoptosis. In contrast to untreated cells, the rate of late apoptosis was separately increased by 2.3% at 5 h and 12.27% at 24 h (Figure 6 A). Subsequently, we aimed to identify which apoptotic signaling pathways were activated by rtMucunain by assessing the expression levels of Caspase 3 (effector), Bad, Bax (Pro-apoptotic) and Bcl-2 (apoptosis inhibitor). The results showed that, after exposure to 5 μM rtMucunain for both 5 h and 24 h, there was a significant reduction in pro-Caspase 3, Bad and Bax compared to untreated cells. Conversely, active-Caspase 3 were elevated, while, Bcl-2 was undetectable (Figure 6 B, C). In line with our above noted findings, these results confirm that cell apoptosis was indeed induced by rtMucunain. To further elucidate the mechanism underlying the effects on cocultures keratinocytes and dermal fibroblasts induced by rtMucunain, we examined whether Caspase 8 and Caspase 9 (initiators) were activated following treated with rtMucunain. As shown in Figure 6 D and E, the pretreatment with 5 μM rtMucunain for 5 h and 24 h led to a marked reduction in the expression of pro-Caspase 8, in contrary, Caspase 9 were not detected (Data not shown). Finally, downstream genes associated with Caspase 8- such as Bid and CYLD were also found to be obviously downregulated in treated cells, indicating that the apoptotic signaling pathway within skin epithelial cells induced by rtMucunain might operate through Caspase 8 rather than Caspase 9 or Bad.
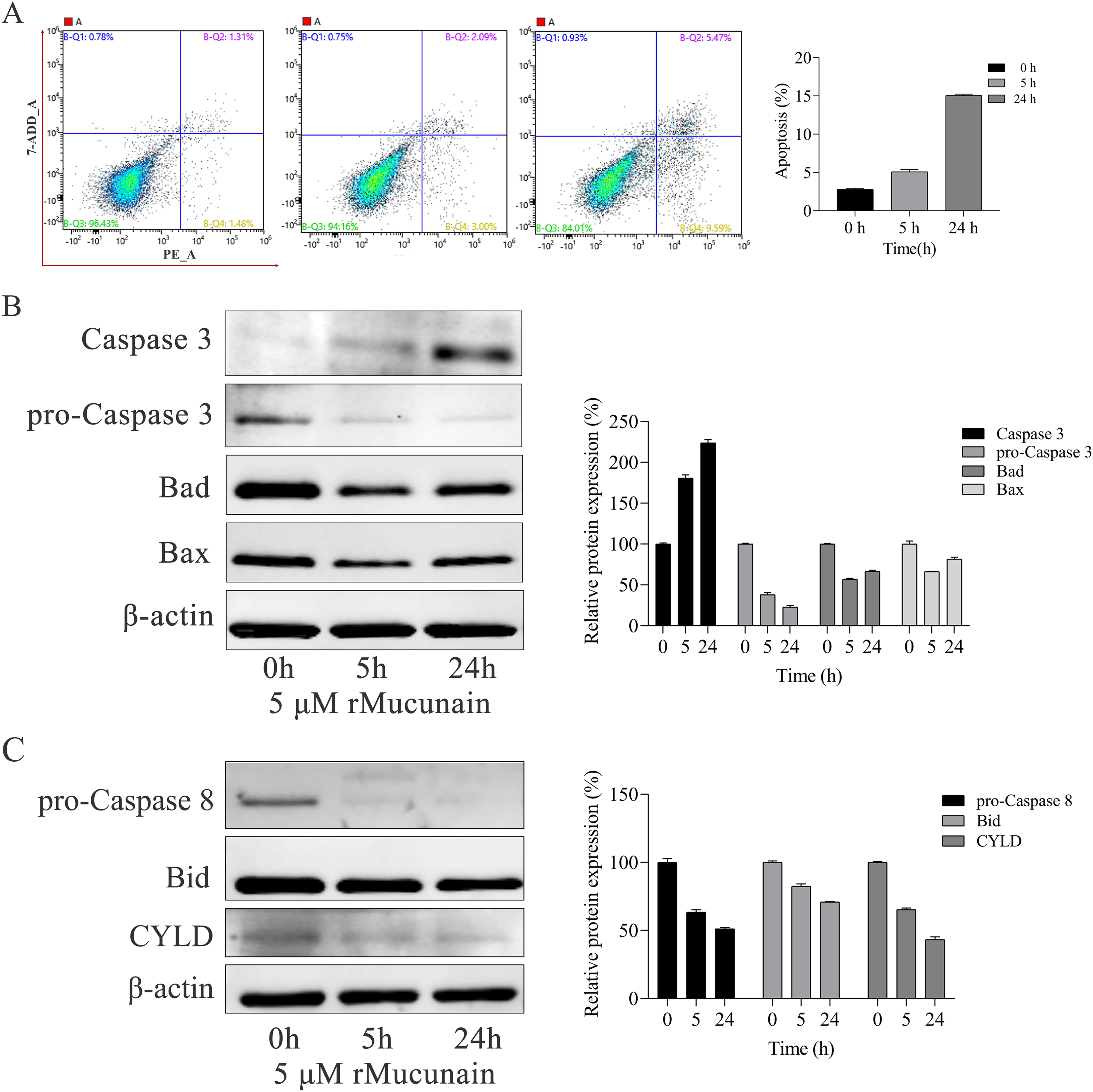
Apoptotic activation of skin epithelial cells treated with rtMucunain. A. Flow cytometric assay was performed by binding of Annexin V-FITC and uptake of 7-ADD to evaluate the early apoptosis and late apoptosis. Q1, dead cells (7-ADD-positive, annexin V-FITC-negative), Q2, late apoptotic cells (7-ADD-positive, annexin V-FITC-positive); Q3, living cells (7-ADD-negative, annexin V-FITC-negative); Q4, early apoptotic cells (7-ADD-negative, annexin V-FITC-positive); Values in the Q4 and Q2 quadrants represent percentage of cells showing early apoptotic and late apoptotic, respectively. The histogram was represented as the Mean ± SEM of three independent experiments. *P < 0.05, **P < 0.01 versus control. B, C. Protein levels of Caspase-3/8, Bad, Bax, and Bcl-2 in cocultured skin epithelium cells were assessed after pretreatment with 5 μΜ rtMucunain for 5 h and 24 h.β-actin was used as a loading control. Results are representative of one experiment out of three. The histogram was represented as the relative protein expression of above noted proteins, Data represent Mean ± SEM of three independent experiments. **p<0.01 versus control.
rMucunain Increased the Lever of IL-6 and IL-8 in Cocultured Skin Epithelium Cells
NF-κB pathway is widely recognized as a prototypical pro-inflammatory signaling pathway, closely related to ROS and Caspase 8.23,24 Additionally, activated Caspase 8 has the capacity to suppress NF-κB activity, thereby mediating apoptosis.25,26 We hypothesized that activated Caspase-8 by rtMucunain might lead to the inactivation of NF-κB. As anticipated, we observed that the inactivation of NF-κB as early as 5 h post-stimulation with rtMucunain, reaching statistical significance at 24 h (Figure 7 A, B). Overexpression of NF-κB-p65 can significantly upregulate the mRNA expression levels of inflammatory cytokines such as TNF-α, IL-6, IL-8, and IL-1β. Subsequently, we assessed that the levels of IL-6 and IL-8 genes through an ELISA assay, which are NF-κB-targeted genes, 25 and are also closely associated with ROS 27 Our findings revealed that the increasing secretion of both IL-6 and IL-8 was induced by rtMucunain in a time-dependent manner (Figure 7 C). In accordance with our results, it has been reported that the influence on IL-6 production is more pronounced than that on IL-8 in human keratinocytes. 28 This finding indicated that inflammation and apoptosis induced by rtMucunain might be attributed to an increase in the production of IL-6 and IL-8 via inactivated NF-κB.
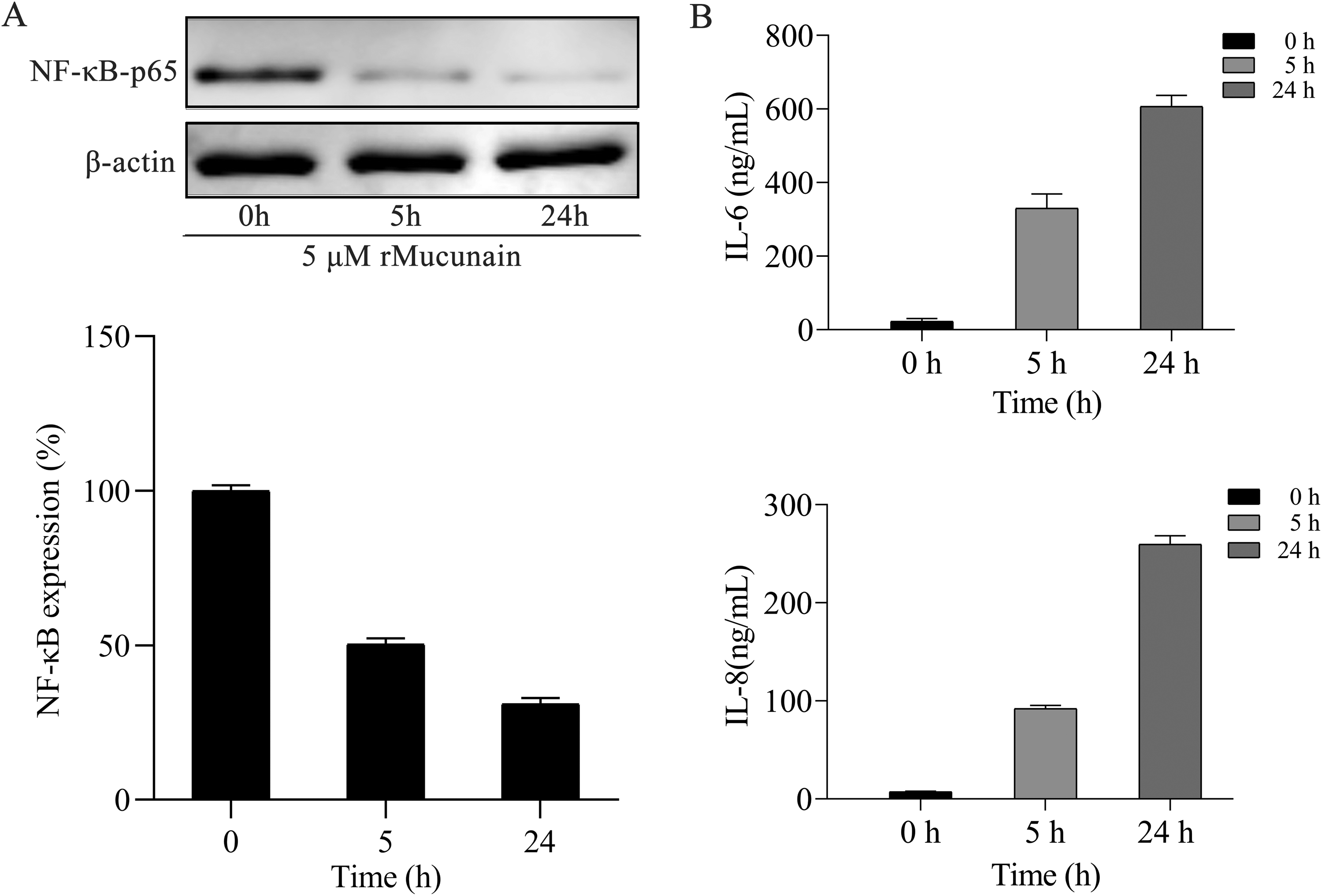
Rtmucunain could induce the release of IL-6 and IL-8 through inactivated NF-κB. A. Western blot analysis compared the protein level of
Discussion
Mucunain exists in small spicules that cover the pods of cowhages, however, extracting native Mucunain in large quantities from cowhage spicules is extremely challenging. As so far, only one study has been published on recombinant expression of Mucunain. 3 In our study, the synthetic gene encoding the active domain 93–309 of Mucunain predicted was cloned into pET-28a-Sumo to express rtMucunain in E.coli. Following a series of selections in induction time and temperature, as well as varying IPTG concentrations in expression strains of E.coli, such as BL21 (DE3) competent cells, BL21 (DE3) PlysS competent cells, Rosetta (DE3) competent cells, and Origami (DE3) competent cells, fusion protein was consistently present as inclusion bodies. Hence, the inclusion bodies were subsequently subjected to purification by a Ni-NTA affinity chromatography column and renaturation by dialysis. Finally, rMucunain was obtained, with a yield of up to 50 mg/L in flasks, which exerts better bioactivity than natural Mucunain in our previous study. 18
Mucunain-induced itching and inflammation through PAR2 activation are frequently seen in AD, a typical inflammatory paradigm of severe pruritus brought on by aberrant skin inflammation and epidermal barrier collapse. It has been demonstrated that applying cowhage spicules and Mucunain to the left or right groin of each Maltese–Beagle atopic dog, like in other species causes pruritus and inflammation. 7 Mucunain-induced itch is an appropriate model for studying itch in AD and other chronic pruritic diseases. 6 However, antihistamines are ineffective at relieving chronic itching. It is crucial to investigate the mechanisms and treatments of pruritus using animal models with Mucunain. Given the variability of irritation between species and the growing awareness of animal welfare, preclinical safety evaluations utilizing a skin epithelial cell model are required before human exposure to Mucunain. Plant proteases, such as bromelain, ficin and papain, have inhibited the growth of endothelial cells and fibroblasts. The viability of fibroblast cell decreases following papain treatment after 12 h and 24 h. 29 Bromelain can moderately attenuate the proliferation of endothelial cells and fibroblasts in dose of 36 × 10−3 IU/ml. At 72 × 10−3 IU/ml, almost all cells are detached from the surface. 30 Bromelain and ficin can also inhibit cell proliferation in human umbilical vein endothelial cells. 31 Whereas, little is known about the cytotoxicity of Mucunain. Currently, there are hardly any reports that intestinal worms are dead with injection of Mucunain. 10 We presumed that skin epithelial cells exposure to rtMucunain were susceptible to immune responses, even to apoptosis. As anticipate, rtMucunain inhibited the proliferation of cocultured skin epithelial cells in a time- and dose-dependent manner, and the inhibition rate of proliferation was nearly up to 45% in doses of 20 μM and 40 μM rtMucunain at 5 h, indicating that a higher concentration of rtMucunain could trigger the growth inhibition of cocultured skin epithelial cells in a short period of time. Moreover, rtMucunian also caused morphological alterations of the cells in time- dependent manner, including cytoplasmic blebbing and slight detachment. These results suggested that rtMucunain might trigger the cytotoxicity in human skin epithelial cells in a time- and dose-dependent manner. It is widely recognized that IL-6 and IL-8 can induce abnormal inflammation and proliferation of epithelial cells.32,33 Our findings revealed that rtMucunain also induced the increasing secretions of IL-6 and IL-8 in a time- dependent manner. In line with our results, the effect on the production of IL-6 is greater than that on IL-8 in human keratinocytes. 28 The maintenance of high concentrations of pro-inflammatory cytokines, including IL-6 and IL-8, can also cause apoptosis of epithelial cells and gingival fibroblasts. 34 Taken together, our results suggested that the inhibited effect on skin epithelial cells following exposure to rMucunain might be related with the increasing secretion of IL-6 and IL-8.
Notably, the emergence of the apoptotic bodies suggested that the apoptosis might have occurred in the cocultured skin epithelial cells after exposure to rtMucunain in some time, and FACS analysis further revealed the event. It is well known that ROS are essential by-products of cellular metabolism, and elevate levels of ROS can cause both apoptotic and necrotic cell death, depending on the severity of the oxidative stress. 35 Exposure to environmental pollutants and chemotherapeutic drugs can also trigger apoptosis, which is frequently mediated by ROS. For instance, NP exposure has been shown to induce apoptosis in GC-1 spg cells through mitochondrial ROS-mediated death signaling. 36 Our findings demonstrated that rtMucunain induced an increase intracellular ROS in skin epithelial cells in a time-dependent manner, suggesting that the intracellular formation of ROS might be one contributing factor to cell damage leading to apoptosis.
During apoptosis, three major downstream pathways of the death signal are involved: the mitochondria pathway, endoplasmic reticulum pathway, and death receptor pathway. 37 A complete implementation of apoptosis involves the interaction of a diverse of proteins and signal transducers as well as cascades of signaling pathways. The generation of ROS is consistently associated with Caspase 8 activation, Bid cleavage, and NF-κB activation. 23 In this study, apoptosis was induced by exposure to rtMucunain in cocultured skin epithelial cells that were involved in death receptor-mediated Caspase 8 signaling. Active Caspase 3 was induced by rtMucunain exposure in the cocultured skin epithelial cells, which was accompanied by a downregulation of pro-Caspase 3 and pro-Caspase 8. It was imperative that the Bid be downregulated, which can be cleaved into tBid by Caspase 8, and subsequently translocate to the outer membrane of mitochondria where it inhibits the anti-apoptotic functions of Bcl-2 and Bcl-XL, while activating Bax and Bak. 38 Nevertheless, our finding revealed a significant decrease in Bad and Bax, indicating that tBid did not translocated into mitochondria to promote mitochondrial apoptosis throughmodulation of the balance between pro-apoptotic and anti-apoptotic members of the Bcl-2 family. Meanwhile, Caspase 8 cleaved CYLD to generate a survival signal, conversely, loss of Caspase 8 can impete CYLD degradation leading to necrosis. 39 By reason of the foregoing, we speculate that rtMucunain induces apoptosis primarily via the Caspase 8 signaling pathway rather than through mitochondrial pathway.
NF-κB is closely related with ROS and Caspase 8, and exhibits distinct effects in various cell types and circumstances. Numerous studies have been reported that activated NF-κB can perform a pro-apoptotic role, inducing apoptosis in islet β cells as well as HK-2 human proximal tubular epithelial cells.40,41 In other instances, a reduction in the levels of NF-κB-p65 has been shown to exacerbate inflammation and apoptosis in epithelial cells.42–45 In accordance with our findings, the apoptosis observed in human skin epithelial cells treated with rtMucunain was accompanied by activated Caspase 8 and inhibition of NF-κB-p65. The NF-κB pathway is widely recognized as a prototypical pro-inflammatory pathway. Notably, overexpression of NF-κB-p65 significantly up-regulates the mRNA expressions of inflammatory cytokines such as TNF-α, IL-6, and IL-1β, leading to inflammation in bovine rumen epithelial cells. 46 The IL-8 gene, one of the NF-κB-targeted genes, which is constitutively expressed in the HL-60 cell line, can also be suppressed at the mRNA and protein levels due to considerable inactivation of NF-κB triggered by tmTNF-α. 47 Given the relationship between NF-κB and these cytokines, along with our finding that rMucunain could induce an increase in secretion levels of these cytokines, we hypothesize that rMucunain might lead to the inactivation of NF-κB, thereby resulting in elevated levels of IL-6 and IL-8, and requires our further investigations. Summing up along with the analysis, our results meant that the inflammation and apoptosis of the cocultured skin epithelium cells induced by rtMucunain might be mediate through Caspase 8 signaling pathway, in which NF-κB-p65 was also involved (Figure 8).

Schematic diagram showed the apoptosis signaling pathway might be triggered by rtMucunain. rtMucunain induced the apoptosis in cocultured skin epithelia cells via Caspase 8 pathway, in which NF-κB-p65, IL-6, and IL-8 were also involved.
Conclusion
In conclusion, this study provides a comprehensive analysis of the mechanisms by which rtMucunian induces damage in a cocultured skin epithelial cells model. Higher concentrations of rtMucunian have the potential to inhibit cell proliferation, alter cell morphology, induce oxidative stress, promote apoptosis, and affect key signaling pathways. Taken together, all of the experimental results indicate that the effects of rtMucunian treatment on cocultured skin epithelial cells are dose- and time- dependent. The study for the first time demonstrates that the Caspase 8 and NF-κB signaling pathway played a crucial role in the inhibition of proliferation and the occurrence of apoptosis on cocultured skin epithelial cells exposure to rtMucunain. These findings hold significant implications for the utilization of Mucunain in skin treatments and underscore the necessity for further investigation to explore its therapeutic potential and safety.
Footnotes
Acknowledgments
The authors thank the anonymous reviewers for their insightful comments.
Author Contributions
Y.L., Y.Y. and Y.S. conceived and designed the experiments; Y.L. and Z.W. performed the experiments; F.W. and Y.Y. analyzed the data; Y.L. and Z.W. wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
