Abstract
Renal ischemia reperfusion injury (RIRI) is one of the main causes of acute kidney injury. This study aimed to explore whether tubeimoside-1 (TBMS1) could protect against RIRI. RIRI mice model and hypoxia/reoxygenation (H/R)-induced NRK-52E cells were used in this study. The renal pathology was observed by hematoxylin and eosin staining to calculate the tubular injury score. The levels of serum creatinine and blood urine nitrogen were analyzed by a Hitachi model 7180 automatic analyzer. The expressions of tumor necrosis factor alpha (TNF-α), interleukin-1 beta (IL-1β), interleukin 6 (IL-6), Bax, cleaved caspase-3, cleaved caspase-9, total caspase-3, and total caspase-9 in renal tissues and NRK-52E cells were detected by western blot analysis. The levels of TNF-α, IL-1β, and IL-6 in serum and NRK-52E cells were measured by a commercial enzyme-linked immunosorbent assay kit. The renal cell apoptosis in renal tissues was analyzed by TUNEL assay, and NRK-52E cell apoptosis was detected by flow cytometry analysis. CCK-8 assay was used to analyze the viability of NRK-52E cells after the indicated treatment. As a result, the renal tissues that were seriously damaged in mice with RIRI could be alleviated by TBMS1. Therefore, 50 mg/kg TBMS1 was chosen for the animal experiment. Renal cell apoptosis was increased in renal tissues of mice with RIRI. These changes could be partially reversed by TBMS1 treatment. TBMS1 improved the viability, and reduced the inflammation and apoptosis of H/R-induced NRK-52E cells. In conclusion, TBMS1 ameliorates RIRI by promoting viability and suppressing apoptosis and inflammation of renal cells.
Ischemia reperfusion injury (IRI) refers to the damage caused by the restoration of blood supply following a long time of ischemia in tissues or organs. 1 Renal ischemia reperfusion injury (RIRI) is a common cause of acute renal failure (ARF) and the main factor affecting the early recovery of renal function after kidney transplantation. It not only causes delayed graft function, but also affects the immune mechanism to promote the occurrence of acute rejection. 2 RIR can lead to apoptosis of renal tubular epithelial cells, renal tubular-interstitial inflammatory response, and ARF. 3 -5 During the development of RIR, the occurrence of inflammatory cell infiltration and the production of inflammatory factors plays important roles in the early stage of RIRI. 6 Therefore, inhibiting renal tubular epithelial cell apoptosis and inflammatory response, and promoting proliferation of renal tubular epithelial cells induced by RIR may be useful for RIRI treatment.
Materials and Methods
Animals
All animal experiments were approved by the Animal Ethics Committee of Beijing Tongren Hospital, Capital Medical University. Eighty wild-type C57BL/6 male mice (10 weeks old, 25‐30 g) were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). All mice were raised and had free access to food and water in a controlled room with moderate temperature (22 ± 2 °C) and humidity (55%-60%) with a 12:12 hours light–dark cycle.
RIRI Model
Mice adaptively fed for 7 days were randomly divided into 8 groups (
Hematoxylin-Eosin Staining
Renal tissues were fixed in 4% (v/v) paraformaldehyde for 48 hours at room temperature, embedded into paraffin, and sliced into 4-µm-thick sections. Then, the sections were stained with hematoxylin and eosin. The tubular injury score was calculated according to the percentage of damaged tubules in the outer medulla. The magnitude of cell loss and necrosis was scored based on 5 levels. Scores ranged from 0 to 4 based on the percentage of tubules affected (0: <10%; 1: 10%-25%; 2: 25%-50%; 3: 50%-75%; 4: >75%). 16 A renal pathologist blinded to the mice group assignments graded the tubular injury under the high-power field of a light microscope.
Cell Culture
NRK-52E cells were provided from American Type Culture Collection (Rockville, MD, USA). All the cells were cultured in Dulbecco’s modified Eagle medium containing 10% fetal bovine serum, 100 U/mL penicillin and 100 µg/mL streptomycin in an incubator with 5% CO2 at 37 °C. The medium was exchanged every 24 hours.
H/R Induction and TBMS1 Treatment
The H/R procedure was conducted as previously described with modifications. 17 Briefly, confluent NRK-52E cells were incubated for 24 hours in an anaerobic chamber equilibrated with 5% CO2, 5% H2, and 90% N2 to form hypoxia cells. After the hypoxia treatment, the cells were moved to a regular incubator with 21% oxygen and treated with TBMS1 in different concentrations (1, 5, 10, 15, and 20 µM) and 50 µM RVT for 4 hours. NRK-52E cells in dimethyl sulfoxide (DMSO) group were conventionally cultured in medium containing DMSO. After H/R induction and TBMS1 treatment, a hemocytometer was used to count cell numbers.
CCK-8 Assay
After the indicated treatment of NRK-52E cells, they were seeded in 96-well plates to which was added 10 µL of CCK-8 reagent per well. When the plates were incubated in the dark for 4 hours, the absorbance value of each well was measured at 450 nm with a PerkinElmer Microplate reader.
Serum Biochemistry and Enzyme-Linked Immunosorbent Assay
The levels of serum creatinine (Scr) and blood urine nitrogen (BUN) were analyzed by a Hitachi model 7180 automatic analyzer. Tumor necrosis factor alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-6 (IL-6) in serum and NRK-52E cells were measured by a commercial ELISA kit referring to the manufacturer’s instructions.
TUNEL Assay
Briefly, renal tissue sections were washed with phosphate-buffered saline (PBS) and treated with protease K at room temperature. Next, sections were sealed with 3% H2O2 for 5 minutes and then incubated completely with TUNEL reaction solution at 37 °C for 1 hour. After PBS washing, sections were treated with 50 mL converter-POD at 37 °C for 30 minutes. Diaminobenzidine was added for full color development at room temperature for 10 minutes. Finally, fluorescence microscopy was used to analyze the stained sections.
Western Blot Analysis
RIPA lysis buffer was used to lyse renal tissues and NRK-52E cells. After lysis, total proteins were extracted from both and quantified by a protein assay kit. Then, proteins (40 µg/lane) were separated with 10% sodium dodecyl sulfate-polyacrylamide gels and transferred to polyvinylidene difluoride membranes, which were blocked with 5% fat-free milk for 2 hours at room temperature. The membranes were incubated with primary antibodies against TNF-α, IL-1β, IL-6, Bax, cleaved caspase-3, cleaved caspase-9, total caspase-3, and total caspase-9 at 4 °C overnight, followed by buffer washing. Next, horseradish peroxidase-conjugated antibody was added to the membranes for incubation at room temperature for 1 hours. Finally, the protein bands were observed using an enhanced chemiluminescence system.
Flow Cytometry Analysis
Apoptosis of NRK-52E cells was detected by fluorescein isothiocyanate (FITC) Annexin V and propidium iodide (PI) dual staining in accordance with the manufacturer’s protocol. Annexin V-FITC/PI binding was analyzed by fluorescence-activated cell sorting, and FlowJo software was used to analyze the data.
Statistical Analysis
Data are presented as mean ± standard deviation. One-way analysis of variance was used coupled with Tukey’s post hoc test to evaluate intergroup differences. SPSS 23.0 statistical software was performed for statistical analysis.
Results
TBMS1 Protects Against RIRI
In mice with RIRI, renal tissues were seriously damaged. RIRI mice were, respectively, treated with different concentrations of TBMS1. After TBMS1 treatment with concentrations from 1 to 50 mg/kg, the renal tissue injury was gradually improved. After 50-100 mg/kg TBMS1 treatment, the renal tissue showed little, but not significant injury as TBMS1 concentration increased, which was consistent with the alleviative effect of RVT treatment on RIRI (Figure 1(A)). The tubular injury score was increased in mice with RIRI, while decreased when mice were treated with 1-50 mg/kg TBMS1. The tubular injury scores were not statistically changed after 50-100 mg/kg TBMS1 treatment on mice with RIRI. The tubular injury score after 50 mg/kg TBMS1 treatment was not statistically different from RVT treatment, either. Therefore, 50 mg/kg TBMS1 was determined for the next experiment (Figure 1(B)). The levels of Scr (Figure 1(C)) and BUN (Figure 1(D)) in serum were upregulated in mice with RIRI, and TBMS1 treatment downregulated their levels.
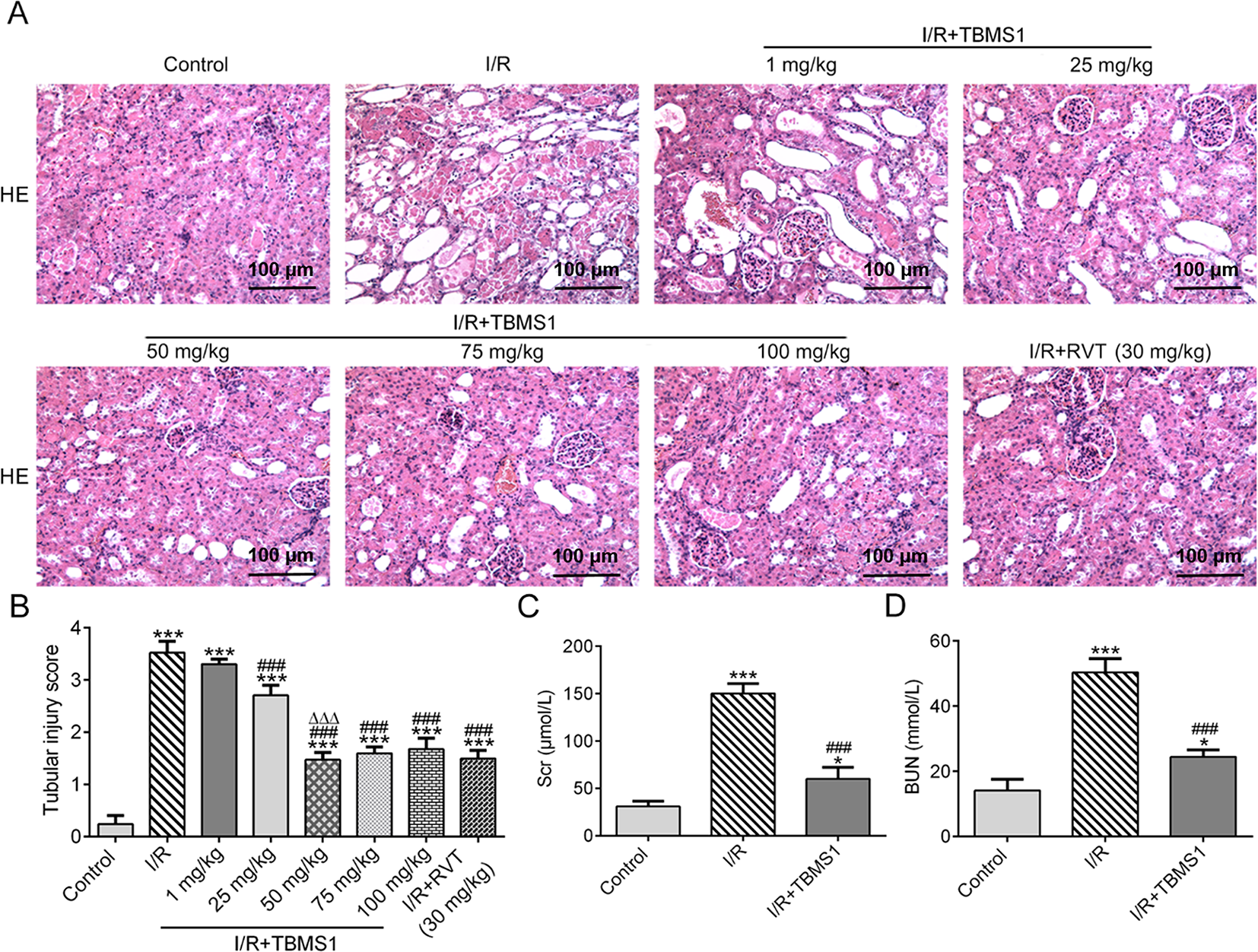
TBMS1 protects against RIRI. (A) The renal pathology of mice with RIRI after TBMS1 treatment was observed by H&E staining. (B) The tubular injury score of mice with RIRI after TBMS1 treatment was calculated according to the percentage of damaged tubules in the outer medulla. ***
TBMS1 Alleviates Inflammation in Renal Tissues of Mice With RIRI
The expression of TNF-α, IL-1β, and IL-6 in renal tissues of mice with RIRI was increased, but this was inhibited by TBMS1 treatment (Figure 2(A)). Also, the levels of TNF-α, IL-1β and IL-6 in serum of mice with RIRI were increased, and TBMS1 treatment decreased their levels (Figure 2(B)-(D)).
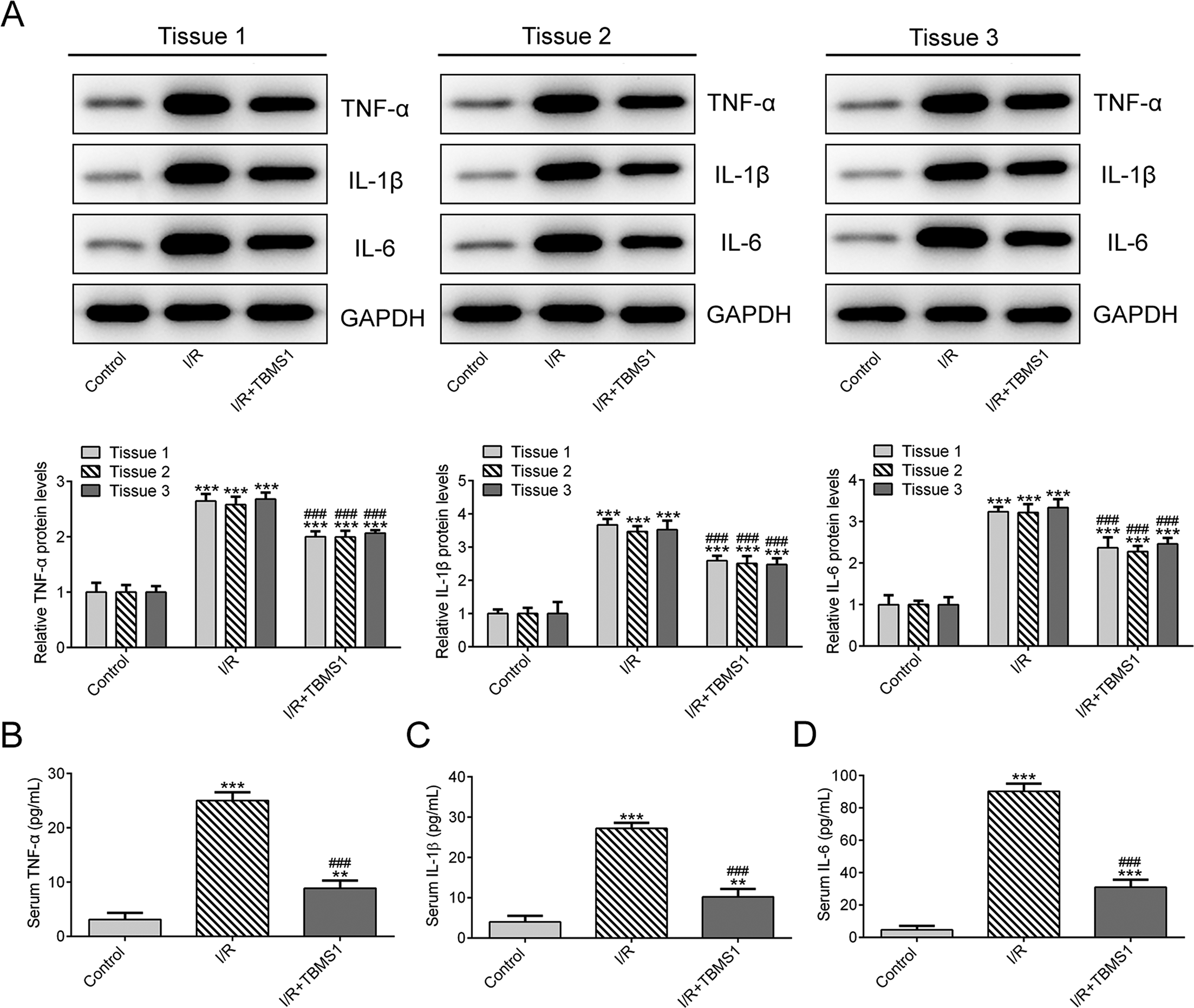
TBMS1 alleviates inflammation in renal tissues of mice with RIRI. (A) The protein expressions of TNF-α, IL-1β, and IL-6 in renal tissues of mice with RIRI after TBMS1 treatment were detected by western blot analysis. ***
TBMS1 Inhibits Apoptosis of Renal Cells in Mice With RIRI
The apoptosis of renal cells was enhanced in mice with RIRI, but TBMS1 treatment suppressed this effect (Figure 3(A)), which was consistent with the changes in apoptosis index (Figure 3(B)).
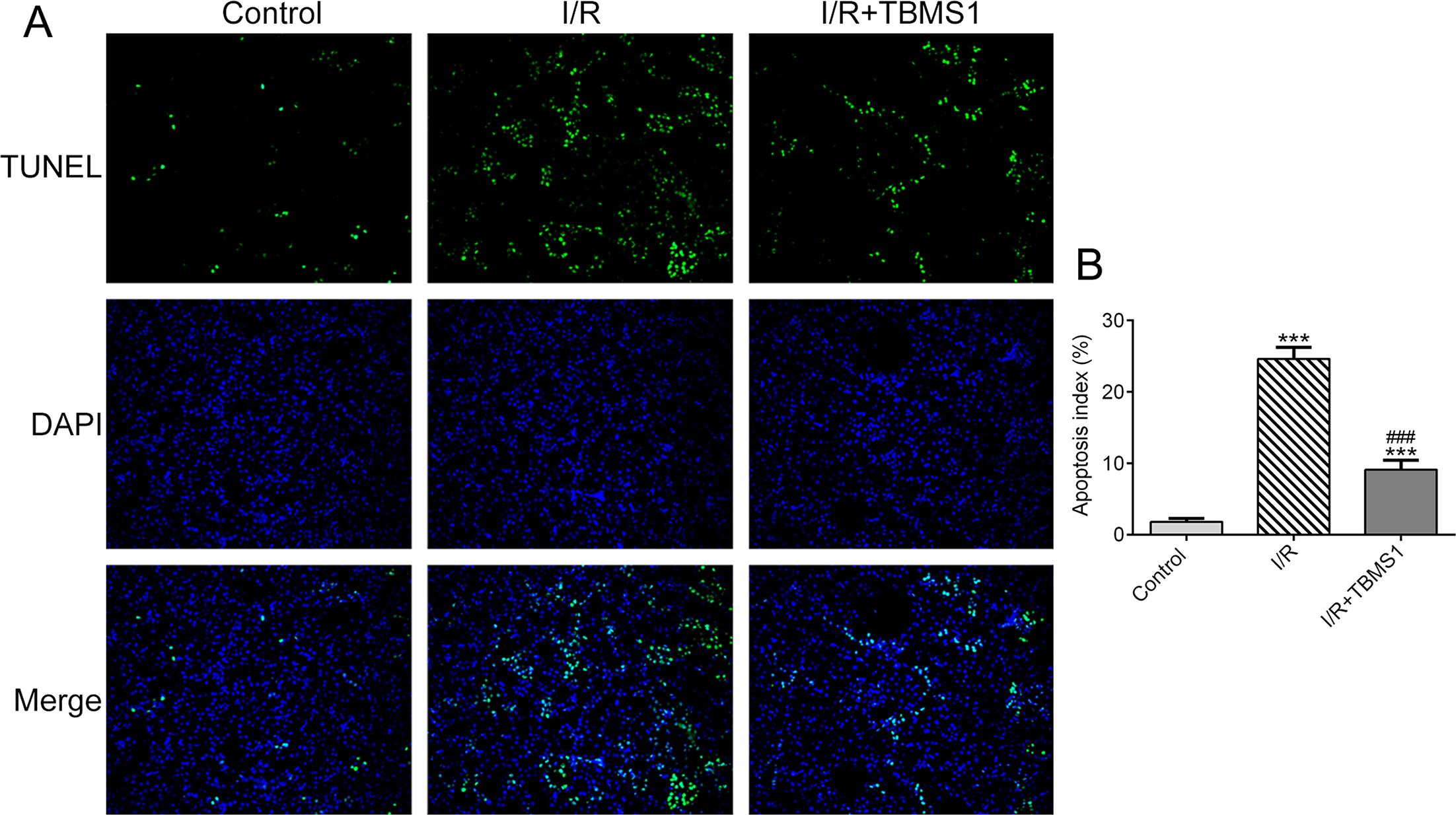
TBMS1 inhibits apoptosis of renal cells in mice with RIRI. (A) The apoptosis of renal cells in mice with RIRI after TBMS1 treatment was analyzed by TUNEL assay. (B) The apoptosis index was obtained from the data of TUNEL assay. ***
TBMS1 Affects the Expression of Apoptosis-Related Proteins in Renal Tissues of Mice With RIRI
As shown in Figure 4, the expressions of Bax, cleaved caspase-3, and cleaved caspase-9 were increased in renal tissues of mice with RIRI, which could be reversed by TBMS1 treatment. The expression of total caspase-3 and total caspase-9 was not changed in these 3 groups.

TBMS1 affects the expression of apoptosis-related proteins in renal tissues of mice with RIRI. The protein expressions of Bax, cleaved caspase-3, cleaved caspase-9, total caspase-3, and total caspase-9 in renal tissues of mice with RIRI after TBMS1 treatment were analyzed by western blot analysis. ***
TBMS1 Promotes Proliferation of H/R-Induced NRK-52E Cells
Observed by microscopy, the cell number in the H/R group was significantly decreased compared with the DMSO group. When H/R-induced NRK-52E cells were treated with TBMS1, the numbers of H/R-induced NRK-52E cells were increased after treatment with 1 and 5 µM TBMS1, while there was no significant difference in cell numbers after 1 and 5 µM TBMS1 treatment of the cells. The number of H/R-induced NRK-52E cells was further increased after 10 µM TBMS1 treatment, and there was no significant difference in cell numbers after TBMS1 treatment at doses of 10, 15, and 20 µM (Figure 5(A)). CCK-8 assay showed that NRK-52E cells induced by H/R showed sharp declines in their growth curve, while the growth curve of H/R-induced NRK-52E cells treated with 1, 5, 10, 15, and 20 µM TBMS1 rose. The improvement produced by 10, 15, and 20 µM TBMS1 on cell growth was better than that of 1 and 5 µM TBMS1. There was no significant difference in cell growth after 1 and 5 µM TBMS1 treatment, and there was no significant difference in cell growth after TBMS1 treatment at doses of 10, 15, and 20 µM (Figure 5(B)). Therefore, 10 µM TBMS1 was chosen for the cell experiments.
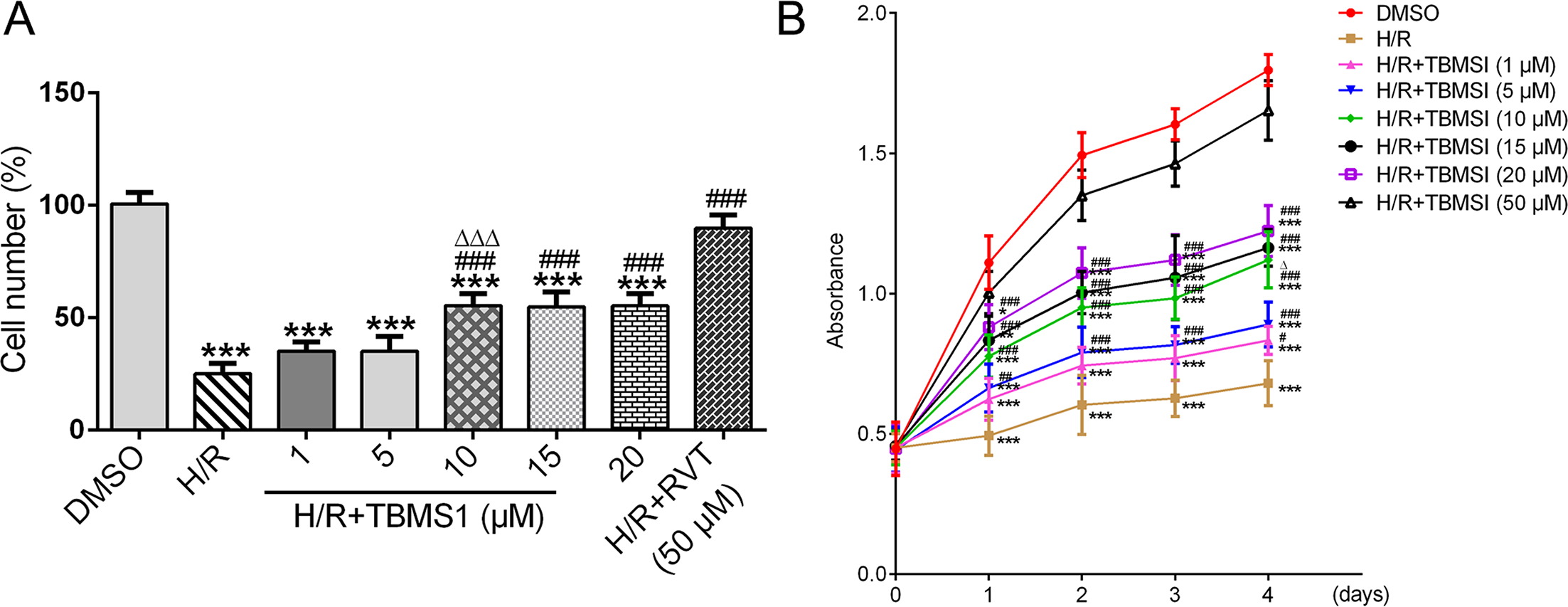
TBMS1 promotes proliferation of H/R-induced NRK-52E cells. (A) The cell number of H/R-induced NRK-52E cells after TBMS1 treatment was counted by a hemocytometer. ***
TBMS1 Alleviates Inflammation in H/R-Induced NRK-52E Cells
The protein expression of TNF-α, IL-1β, and IL-6 in H/R-induced NRK-52E cells was increased, but this effect was decreased by treatment with TBMS1 (Figure 6(A)). In the culture supernatant of NRK-52E cells, ELISA indicated that levels of TNF-α, IL-1β, and IL-6 were increased in the H/R group, but decreased in the H/R + TBMS1 group (Figure 6(B-D)).
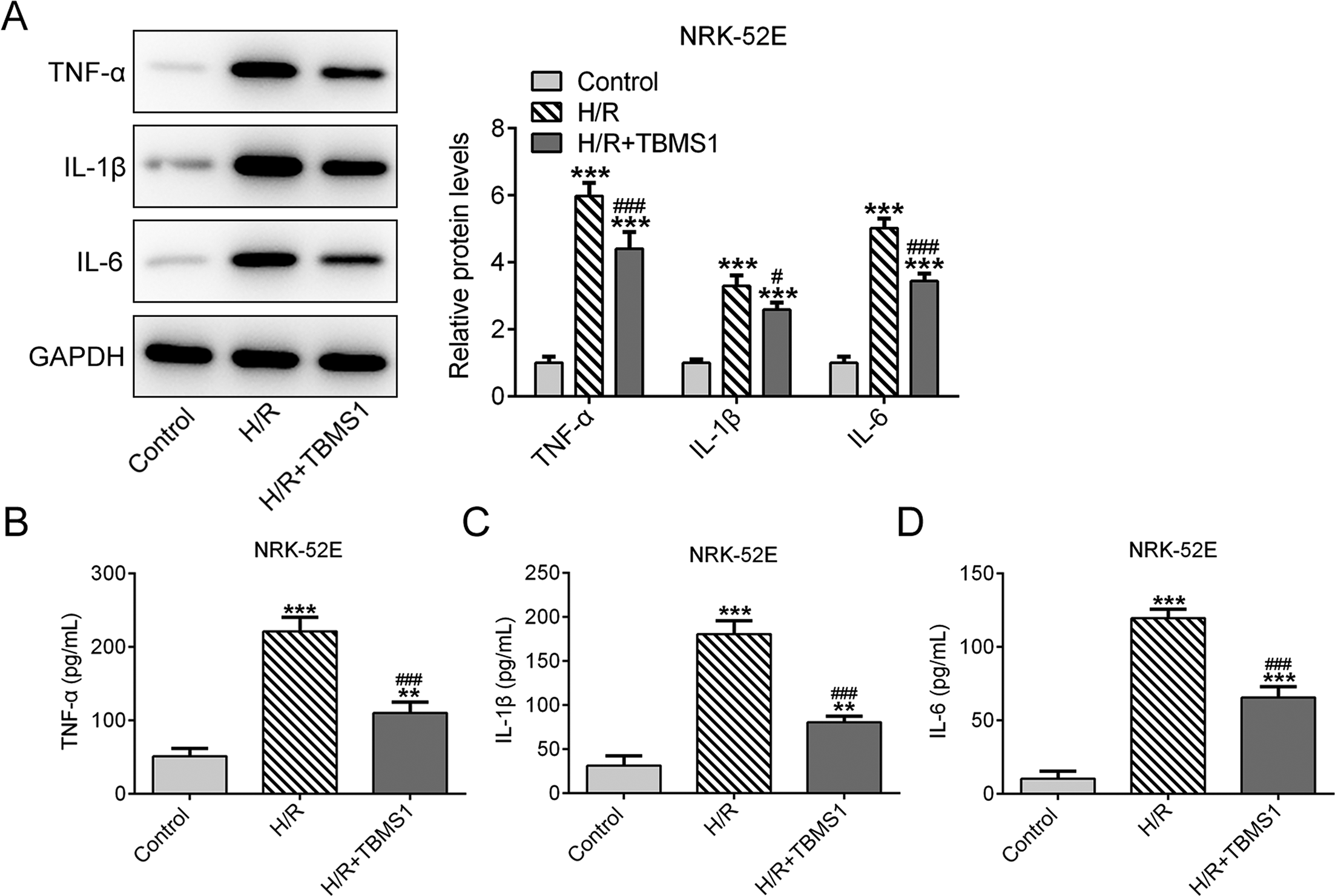
TBMS1 inhibits apoptosis of H/R-induced NRK-52E cells. (A) The protein expressions of TNF-α, IL-1β, and IL-6 in H/R-induced NRK-52E cells after TBMS1 treatment were detected by western blot analysis. ***
TBMS1 Inhibits Apoptosis of H/R-Induced NRK-52E Cells
The apoptosis of NRK-52E cells after H/R induction was increased, but TBMS1 treatment suppressed this (Figure 7(A)). Likewise, the expressions of Bax, cleaved caspase-3, and cleaved caspase-9 were increased, but TBMS1 treatment decreased these. The expressions of total caspase-3 and total caspase-9 were not statistically different in these 3 groups (Figure 7(B)).

TBMS1 inhibits apoptosis of H/R-induced NRK-52E cells. (A) The apoptosis of H/R-induced NRK-52E cells after TBMS1 treatment was detected by flow cytometry analysis. **
Discussion
After renal ischemia, reperfusion can aggravate the injury of tissues or organs, and even cause irreversible injury. 18,19 RIRI has insidious clinical manifestations, rapid progression, and high mortality, and its incidence has been increasing in recent years. 20 Although the diagnosis of RIRI has been greatly improved, the incidence and mortality of the disease are still high. 21 Therefore, reducing new RIRI cases is a hot topic in clinical research and discussion.
The use of traditional Chinese medicine in the treatment of diseases has a long history. Wang et al
22
indicated that RSV alleviated oxidative stress and inflammation to relieve I/R-induced renal injury in rats. Han et al
23
demonstrated that cordycepin suppressed the development of RIRI by inhibiting inflammation, apoptosis, and oxidative stress. Long et al
24
reported that macelignan increased the expressions of Bax and caspase-3 while decreasing the expression of Bcl-2, and suppressed inflammation in renal epithelial cells rats with RIRI. TBMS1 extracted from the tubers of
In conclusion, TBMS1 ameliorated RIRI by inhibiting apoptosis of renal cells and inflammation in mice with RIRI. Also, TBMS1 improved viability, and inhibited apoptosis and inflammation of H/R-induced NRK-52E cells. The above experimental results demonstrated that TBMS1 has a potential therapeutic effect on RIRI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
