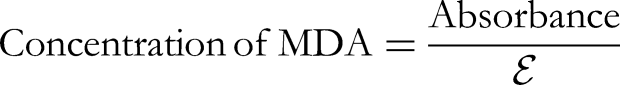Abstract
Objective
Insulin resistance is a key feature of, type 2 diabetes mellitus (T2DM), and it has been linked to an environmental pollutant bisphenol-A (BPA). Gastrodin, a derivative of Chinese medicinal herb, has antioxidant and anti-inflammatory properties that can potentially ameliorate BPA-induced insulin resistance in albino rats.
Method
The study involved five groups of six rats each, including normal control, BPA-treated, metformin-treated, and gastrodin plus BPA-treated groups Insulin sensitivity index, homeostasis model assessment- index of insulin resistance (HOMA IR), and fasting insulin levels were assessed. Using ELISA, the study measured protein levels, including High-Sensitivity C-reactive Protein (hs-CRP,) Glucose Transporter 1 (GLUT1), Interleukin-6 (IL-6), Tumor Necrosis Factor-alpha (TNF-α), AMP-Activated Protein Kinase (AMPK), Inducible Nitric Oxide Synthase (iNOS), caspase-3, and Nuclear factor erythroid 2–related factor 2 (Nrf2), and analyzed factors like thiobarbituric acid reactive substances-malondialdehyde (TBARS-MDA), reduced glutathione, and catalase activity.
Results
BPA administration causes insulin resistance in albino rats, reducing insulin sensitivity. Gastrodin treatment attenuates resistance, improves glucose levels, and increased insulin sensitivity. Gastrodin also mitigates oxidative stress.by reducing reactive oxygen species (ROS) production and restoring antioxidant enzyme activities, while TBARS-MDA was reduced. Studied inflammatory indicators included; C-reactive protein, Tumor necrosis factor (TNF-α), and Interleukin 6. Notably, the administration of bisphenol-A caused a 2–3 fold increase in the serum levels of hs-CRP, TNF-α, and IL-6. All treatments by gastrodin (high and low doses) decreased these observed effects by bisphenol-A to values similar to the standard (metformin) and normal control rats. Gastrodin exerted its protective benefits via activating the AMPK/Nrf2 signaling pathway, increasing AMPK phosphorylation and Nrf2 nuclear translocation, resulting in improved cellular defense against oxidative stress. Indicators of possible inflammations on bisphenol-A treatment were also investigated.
Conclusion
This shows that gastrodin might be a promising therapeutic drug for the prevention and treatment of insulin resistance-related problems.
Introduction
Bisphenol A (BPA) is a chemical compound with the formula C15H16O2 commonly found in plastic products, 1 it is used to strengthen polycarbonate plastics and thus has garnered widespread applications in food storage and packaging. 1 It leaches into food or water once the plastic has been exposed to overheating, acidic or basic conditions or constant use. 2 As a result of this, its potential health hazards have raised concerns worldwide. BPA is widely recognized as an endocrine disruptor with a weak estrogenic effect, but studies have also linked it to cardiovascular diseases, obesity and type 2 diabetes.3–5 Animal studies have linked long-term BPA exposure to glucose intolerance, insulin resistance ultimately leading to type 2 diabetes. 6
Insulin resistance is the inability of insulin-targeting tissues to respond to the actions of insulin, resulting in high glucose levels, which is a prominent sign of type 2 diabetes mellitus (T2DM) 7 ; Insulin regulates key metabolic processes, including glucose production, glycolysis, glycogenolysis, and lipogenesis. These processes produce glucose, pyruvic acid, glycogen, and fatty acids, leading to triglyceride production and lipolysis.8–10 When the above roles of insulin in metabolism are altered due to decreased insulin production or sensitivity, insulin resistance is said to have occurred
There may however, be hope in the form of gastrodin, a natural compound found in traditional Chinese herb called Gastrodia elata Blume. 11 Gastrodin has long been used for the treatment of illnesses such as headache, epilepsy, stroke and convulsions in china. 12 Recently, however, more researchers have shown more interest in its anti-diabetic activity. In a recent study, it was found to significantly lower fasting blood glucose levels while increasing insulin sensitivity index values. 13
The AMP-Activated Protein Kinase / Nuclear factor erythroid 2–related factor 2 (AMPK/Nrf2) signaling pathway participates in several physiological functions, including stress response, antioxidant defense, and metabolic regulation. Nrf2 has a role in antioxidant defenses and is associated with adipocyte differentiation, obesity, and insulin resistance. 14 AMPK, an energy sensor, enhances autophagy under stress circumstances. 15 The AMPK and Nrf2 pathways are interrelated, as both are stimulated by identical stressors and may collaboratively safeguard cells. 16 Extended oxidative stress may lead Nrf2 to inhibit AMPK, thereby regulating autophagy to prevent cellular damage. 15 This regulatory connection may be pertinent in chronic illnesses linked to oxidative stress, including neurological disorders, certain cancer types, and metabolic diseases. 15 The AMPK-Nrf2 signaling pathway regulates metabolic and endocrine systems and may diminish the risk of lung infections. 17
We investigated the effects of gastrodin on Bisphenol-A (BPA)-induced insulin resistance in albino rats in this study. Our main points of discussion was on the mechanism of activation of AMPK/NrF2 pathways via their increased concentrations in serum of BPA-induced rats after gastrodin administration, the potential therapeutic benefits of gastrodin, and the implications of these findings for human health. More so, the impact of gastrodin on serum oxidative stress indices, and inflammation markers were also investigated
Materials and Procedures
Reagents
Bisphenol-A, insulin, and Gastrodin were acquired from Sigma (St. Louis, MO, USA); Trichloroacetic acid, Ellman's reagent Dithiobis(2-nitrobenzoic acid (DTNB), and thiobarbituric acid were obtained from CUSABIO, as were Randox and Elisa kits.
Purchasing Animals for Research
Albino rats of mature age were acquired from the Animal House of the Faculty of Veterinary Medicine at the University of Nigeria. These rats were then placed in well-ventilated cages located in the Biochemistry Department at Ebonyi State University Abakaliki. During a seven-day acclimatisation period in a sterile laboratory environment, they were given free access to water and the standard mouse chow.
Insulin Resistance Induction, Experimental Design, and Procedures
The experiment segregated albino rats (6-8 weeks) into five groups, each consisting of six rats, and designated them as groups A-E. Group B was administered bisphenol A, while Groups C, D, and E were given both bisphenol A and metformin. Group A was given distilled water. Each treatment was orally provided for a duration of 21 consecutive days, commencing from the beginning date. Bisphenol A dose was as earlier used by Pinafa et al, 18 while the doses of gastrodin and metformin were used with modifications from Zhang et al. 19 Table 1 shows the animal grouping and treatments.
Collection and Homogenization of Sample
The albino mice were rendered unconscious with Ketamine (80 mgkg−1, ip) and then dissected after a 12 h overnight fast at the last day of the experiment (6 PM to 6 AM). Subsequently, blood samples were obtained and placed in unadorned test tubes, while the livers were extracted and put to a sterile container without any additives. The container was then preserved in a refrigerated environment. The substance was cleansed in a frigid 1.5% potassium chloride (KCl) solution, dried using a blotting paper, and measured in terms of weight. The tissues were disrupted using 0.1 M phosphate buffer at a pH of 7.2, followed by centrifugation at 2500 g for 15 min. The liquid portion, known as the supernatant, was used for examination. The experiment adhered to the guidelines set out by the National Institute of Health for the proper care and use of laboratory animals. The experiment was authorised by the Ethical Review Committee of the Department of Biochemistry at Ebonyi State University, with the issued identification number EBSU/BCH/ET/22/031.
Fasting Blood Glucose Levels Measurement
The study was conducted using the methodologies outlined by Carper et al 20 The rats had a 12-h period of fasting, after which blood was carefully obtained from their tails. The blood glucose levels was measured using a glucometer (ACCU-CHECK® GO, Hoffmann-La Roche Ltd).
Measurement of Insulin Sensitivity Index and Fasting Serum Insulin Levels
The insulin sensitivity index (ISI) was determined by using the enzyme-linked immunosorbent assay (ELISA) after the administration of the last dosage of gastrodin. The ISI is obtained by multiplying the blood glucose levels (measured in mg/dL) by the blood insulin levels (measured in μ/L). 13 The Homeostatic Model Assessment of Insulin Resistance (HOMA-IR) is calculated by multiplying the fasting glucose levels (measured in millimoles per litre) by the fasting insulin levels (measured in micromoles per litre), and then dividing the result by 22.5. or the Homeostatic Model Assessment of an Insulin Resistance (HOMA-IR) maybe determined by multiplying the fasting glucose levels (measured in mg/dL) by the fasting insulin levels (measured in millimoles per litre), and then dividing the product by 405. 21
Biochemical Analysis
The research used enzyme-linked immunosorbent assay (ELISA) to quantify the levels of many proteins, such as fasting serum insulin, high-sensitivity C-reactive protein (hs-CRP), Glucose Transporter 1 (GLUT1), Interleukin-6 (IL-6), Tumor Necrosis Factor-alpha (TNF-α), AMP-activated protein kinase (AMPK), Inducible Nitric Oxide Synthase (iNOS), caspase-3, Nuclear Factor Erythroid 2–Related Factor 2 (Nrf2), and leptin. The test used a quantitative sandwich enzyme immunoassay method, using pre-coated microplates with antibodies that were specific to the target protein. The intensity of the colour produced was directly proportional to the amount of protein binding. The intensity was evaluated after the cessation of development. Glucose oxidase method was used to quantify serum glucose concentrations, as indicated in the Randox assay kit received from Randox R Laboratories Ltd, Ardmore, United Kingdom. While hemoglobin A1C (HBA1c), was measured by method of. 22 The technique entails the measurement of haemoglobin glycosylation by quantifying the formaldehyde generated during the oxidation of glycated haemoglobin using periodate and detecting it using fluorometric methods.
Evaluation of Oxidative Markers
Assay for Lipid Peroxidation
The quantity of thiobarbituric acid reactive substances (TBARS) 20 was calculated using the Buege and Aust technique.
23
The experiment consisted of introducing 50 µL of the sample and 1 mL of TBARS reagent into sterile Eppendorf tubes, heating the mixture until it became yellow, subjecting it to centrifugation, and measuring the absorbance at 535 nm. The extinction coefficient was used to compute the TBARS-Malondialdehyde (MDA), in accordance with the established technique.
Determination of Antioxidant Activity
Catalase activity was measured using the Aebi method. 24 Super oxide dismutase (SOD) was measured using the method reported by Li. 25 The glutathione test (GSH) method is based on an idea first proposed by Ellman in 1959 and revised by Hu in 1994. 26 The glutathione peroxidase activity was measured using the method modified by Hu and described by Ellman in the determination of sulhydryl groups. 27 The concentration of nitric oxide was determined using the Bryan and Grisham 2007 technique. 28 The Gries reaction is a biphasic diazotization process that generates dinitrogen trioxide (N2O3) by the production of nitrous acid. The compound undergoes a chemical reaction with sulfanilamide, resulting in the formation of diazonium ions. These ions are then combined with N-(1-napthyl)ethylenediamine to form a chromophoric azo product that exhibits a high level of absorption at a wavelength of 540 nm. The sample is combined with Griess Reagent, which consists of Sodium hydroxide (NaOH) and deionized water. When a secondary or tertiary amine is present, iodine in the Griess Reagent is converted into iodide ions (I-) by reduction. These iodide ions then react with excess iodine to produce a brownish complex.
Data Analysis
The data was analyzed using Analysis of Variance (ANOVA), Turkey Post Hoc test, and Graph Pad Prism 7 software, with statistical significance determined at P values below .05, presenting mean and standard deviation.
Results
Impact of Gastrodin on Blood Glucose and its Regulatory Proteins

Impact of gastrodin on blood glucose and indicators of insulin resistance. The results are presented as mean ± standard deviation (n = 6). Bars with different labels and cross lines are significantly different at p < .05 (NC- Normal control, BPA-Biphenol A control group, MET-Metformin treatment, GSTD1, and GSTD2-Gastrodin treatments; low and high dosesS).

A: impact of gastrodin on blood glucose and its regulatory proteins. The results are presented as mean ± standard deviation (n = 6). Bars with different labels and cross lines are significantly different at p < .05. B: Impact of gastrodin on energy regulatory proteins. The results are presented as mean ± standard deviation (n = 6). Bars with different labels and cross lines are significantly different at p < .05.
Impact of Gastrodin on Indicators of Lipid Peroxidation and Oxidative Stress
Figure 3 (a and b) shows the effect of gastrodin on lipid peroxidation and oxidative stress markers. Malondialdehyde (MDA), an indication of lipid peroxidation, rose by nearly 1000% after bisphenol-A injection compared to normal control rats (Figure 3aA). This was decreased by over 700% in metformin and lower dose gastrodin treatments and by over 900% in gastrodin higher dose treatment compared to bisphenol-A control group. A similar trend was observed in nitric oxide levels (concentration) in bisphenol-A control group compared to the standard (metformin) and gastrodin treatments (Figure 3aB). In Figure 3aC and aD, reduced glutathione (GSH), and glutathione peroxidase (GPx) activity decreased significantly (p < .05) on bisphenol-A administration compared to the normal control group. These observed decreased in GSH and GPx due to bisphenol-A, were significantly increase (p < .05) on administration of gastrodin, and this was better than the effect of metformin especially at the higher dose. Decreased activity of catalase, superoxide dismutase (SOD), and nuclear factor erythroid 2–related factor 2 (Nrf2), which decreased on bisphenol-A administration were all increased (p < .05) in gastrodin administration, and these observed increase were more significant in the higher dose of gastrodin treatment (20 mg/kg.b.wt) (Figure 3bA–bC). A reciprocal effect was observed on the tissue concentration of inducible nitric oxide synthse (iNOS). On bisphenol-A administration, iNOS increased significantly compared to the normal control group. This increase in the concentration of iNOS in bisphenol-A administration was significantly, decreased on gastrodin treatment in a manner similar to the normal, and controls.
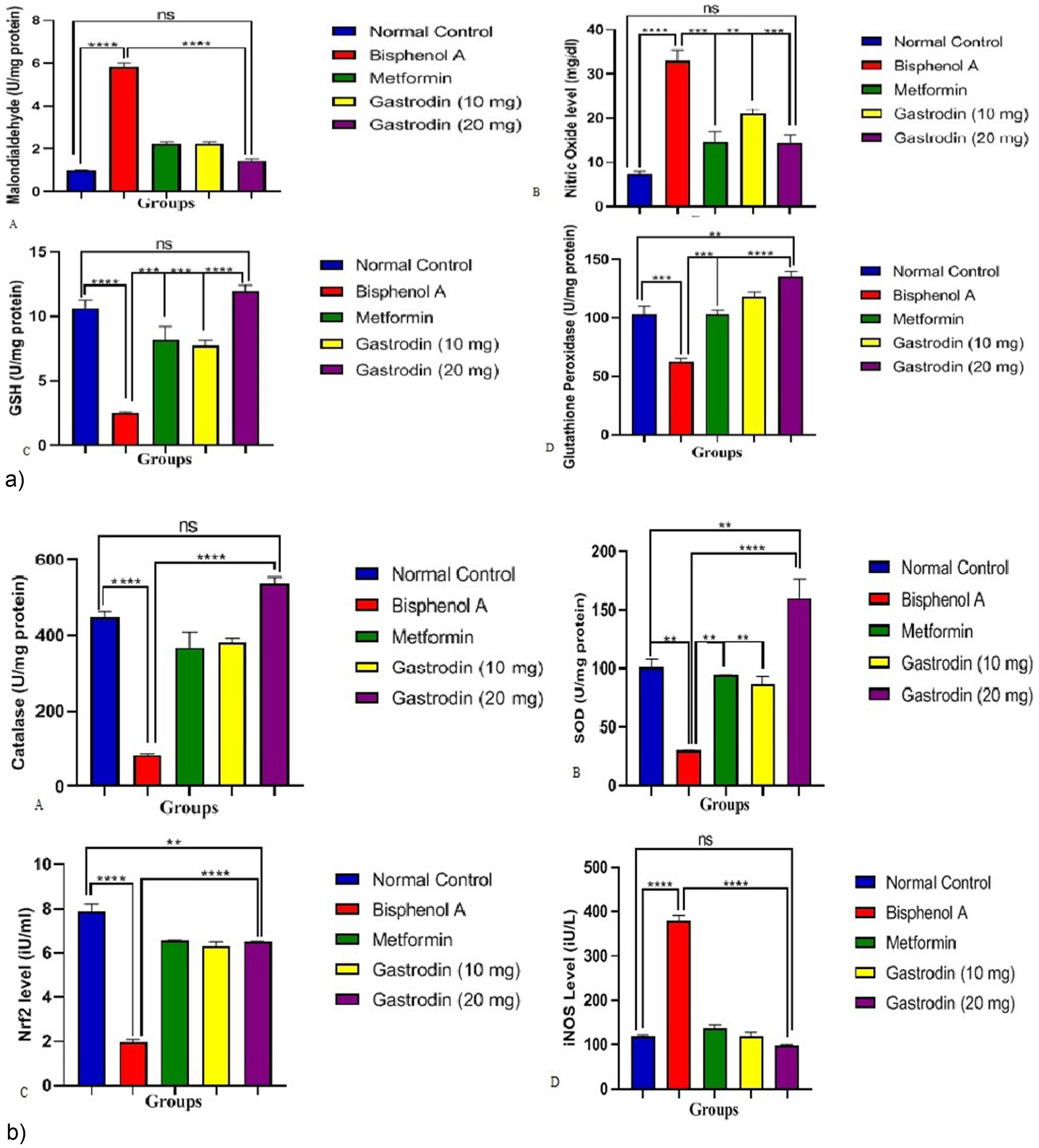
A: impact of gastrodin on indicators of lipid peroxidation and oxidative stress. The results are presented as mean ± standard deviation (n = 6). Bars with different labels and cross lines are significantly different at p < .05. B: Impact of gastrodin on indicators of lipid peroxidation and oxidative stress. The results are presented as mean ± standard deviation (n = 6). Bars with different labels and cross lines are significantly different at p < .05.
Impact of Gastrodin on Inflammation Indicators
Indicators of possible inflammations on bisphenol-A treatment were also investigated. Studied inflammatory indicators included; C-reactive protein, Tumor necrosis factor (TNF-α), and Interleukin 6. (Figure 4A–C) Notably, the administration of bisphenol-A caused a 2–3 fold increase in the serum levels of high sensitive C-reactive protein (hs-C-RP), TNF-α, and interleukin-6 (IL-6). All treatments by gastrodin (high and low doses) decreased these observed effects by bisphenol-A to values similar to the standard (metformin) and normal control rats.

Impact of gastrodin on inflammation indicators: the results are presented as mean ± standard deviation (n = 6). Bars with different labels and cross lines are significantly different at p < .05.
Discussion
The research discovered that Bisphenol-A has the ability to decreased insulin resistance in Albino Rats by reducing oxidative stress and stimulating the activation of proteins involved in energy utilisation and oxidative stress regulation. Several reports have examined the impact of bisphenol A (BPA) on the AMP-activated protein kinase (AMPK)/ Erythroid 2–Related Factor 2 (Nrf2), pathway. BPA was shown to trigger apoptosis in human retinal pigment epithelial cells by inhibiting the Nrf2/HO-1 pathway and diminishing AMPK phosphorylation. 29 In contrast, AMPK activation facilitates the nuclear accumulation of Nrf2 by phosphorylating it at Ser550, therefore increasing its antioxidant response. 30 BPA promotes autophagy in ovarian granulosa cells via the AMPK/mTOR/ULK1 signaling pathway, potentially influencing folliculogenesis. 31 High doses of BPA (>10 μM) were demonstrated to activate the Nrf1/2-ARE pathway in HEK 293 cells, enhancing the expression of antioxidant genes. 32 These findings indicate that BPA can influence the AMPK/Nrf2 pathway, however its effects may differ based on cell type and concentration.
The administration of gastrodin in our study at concentrations of 10 and 20 mg/kg.bwt resulted in a considerable reduction in blood glucose levels, indicating a concentration-dependent impact on blood glucose concentration. Gastrodin exhibited better modulation of blood glucose levels in comparison to metformin, indicating its advantageous impact on blood glucose management, 33 and phenolics show anti-diabetic properties by improving insulin action, decreasing fatty acid levels, and increasing glucose absorption in skeletal muscle and liver cells. 34
More so, the activity of fasting insulin (FI) was investigated in this study, it was observed that FI significantly increased (p < .05) in bisphenol-A group versus normal control an indication of induction of insulin resistance in bisphenol-A administration. 35 ISI significantly decreased in bisphenol-A, a major cause of insulin resistance (Figure 1C) compared to normal control group, 36 the HOMA-IR was also assessed (Figure 1D), and the value for HOMA-IR increased in bisphenol-A in comparison to the normal control, another indicator of insulin resistance. 21 HbA1C blood test which measures average blood sugar levels over time was also measured (Figure 2B). Similarly, HbA1C which increased in bisphenol A administration by over 125% compared to control was decreased (p < .05) on gastrodin administration at both lower and higher doses. Implying gastrodin as a phenolic glycoside has a strong modulation effect on blood glucose regulation, a result that was in agreement with the report by Deka et al 37
In another phase of the study, we measured the impact of gastrodin on the tissue levels of GLUT-2 glucose transporter, and AMP-activated protein kinase (AMPK), one of the focal points of this study, understanding the role of GLUT-2 and AMPK in glucose and energy utilization. The result was very striking. The tissue levels of GLUT-2 and AMPK were significantly decreased (p < .05), in bisphenol-A administration, and these observed decreased were significantly increased in gastrodin treatment. This is could be the possible mechanism of glucose regulation by phenolic glycosides in glucose, and energy pathways modulation as earlier reported. 37 Overall, gastrodin showed strong therapeutic potential by its reciprocal actions on its administration to the treated animals on these measured indices of glucose metabolism compared to bisphenol-A administration, and in some instances better than the standard drug, metformin.
The effect of gastrodin on indicators of lipid peroxidation and oxidative stress were also measured in this study. Results indicated that malondialdehyde (MDA) increased in bisphenol-A administration, and a similar trend was observed in nitric oxide, Inducible nitric oxide synthase (iNOS), concentration in bisphenol-A control group. More so, reduced glutathione (GSH) concentration decreased, with both catalase, glutathione peroxidase (GPx) activity, superoxide dismutase, and nuclear factor erythroid 2–related factor 2 (Nrf2), following the same trend on bisphenol-A administration compared to the normal control group, indicating a state of lipid peroxidation and oxidative stress. 38 However, gastrodin administration attenuated the observed indicators of oxidative stresses in biphemol A administration.
Recent studies have shown the efficacy of gastrodin in addressing multiple health issues. Gastrodin has demonstrated efficacy in mitigating insulin resistance and boosting type 2 diabetes mellitus by increasing insulin receptor expression and activating the Phosphoinositide 3-kinase (PI3K)/ Protein Kinase B (AKT) signaling pathway. 39 Gastrodin alleviates inflammation, oxidative stress, and apoptosis generated by excessive glucose in podocytes through the activation of the AMPK/Nrf2 signaling pathway. 40 Gastrodin similarly mitigates kidney inflammation and fibrosis via the AMPK/Nrf2/HMGB1 pathway. 41 Moreover, gastrodin has demonstrated anti-fatigue properties in mice by maintaining glycogen levels, enhancing antioxidant activity, and upregulating the AMPK and Nrf2/HO-1 pathways. 42 Although these studies do not explicitly examine gastrodin's impact on Bisphenol-A-induced insulin resistance, they together imply that gastrodin's stimulation of the AMPK/Nrf2 pathway may be advantageous in this regard.
The precise mechanism through which gastrodin reduces BPA-induced oxidative stress in Wistar rats has not been thoroughly understood. 43 However, based on its antioxidant and anti-inflammatory properties, numerous pathways can be hypothesized. Gastrodin has antioxidant characteristics, which means it can scavenge free radicals and lower oxidative stress. 43 Exposure to BPA may impair the body's ability to defend against harmful substances by generating reactive oxygen species (ROS). However, this effect can be counteracted by Gastrodin, which enhances the activity of antioxidant enzymes such as SOD, CAT, and GPx, while reducing the levels of MDA, nitric oxide, and iNOS. 12
Gastrodin also has anti-inflammatory properties, suppressing the production of pro-inflammatory cytokines and mediators. 44 It indirectly relieves oxidative stress caused by BPA by lowering inflammation. Gastrodin protects mitochondria by retaining mitochondrial membrane potential, lowering ROS generation, and preserving mitochondrial DNA integrity. 45 It has the potential to affect several signaling pathways involved in oxidative stress and cellular damage, such as activating the Nrf2 pathway, which is a key regulator of cellular antioxidant defenses. 46 This could be the mechanism of gastrodin reported in this investigation, as its antioxidant, anti-inflammatory, mitochondrial protective, and signaling modulatory capabilities contribute to its potential benefits.
Indicators of potential inflammation in response to bisphenol-A therapy were also studied. C-reactive protein, tumor necrosis factor (TNF-α), and interleukin-6 were among the inflammatory markers studied. (Figure 4A–C) Notably, bisphenol-A treatment elevated serum concentrations of C-reactive protein, tumor necrosis factor (TNF-α), and interleukin-6 by 2–3 fold. All gastrodin treatments (high and low doses) reduced the observed bisphenol-A effects to levels comparable to the standard (metformin) and normal control rats.
Gastrodin as a naturally occurring chemical has been researched for its possible medicinal effects in a variety of illnesses, including inflammation.3,47,48 In the context of the parameters obtained in Wistar rats, gastrodin was discovered to have biochemical consequences in relation to inflammation caused by bisphenol-A (BPA) administration. C-reactive protein (CRP), tumor necrosis factor (TNF-α), and interleukin-6 (IL-6) were the inflammatory markers evaluated in this study. These indicators are routinely used to detect and measure the severity of inflammation in the body. The results showed that BPA treatment increased serum concentrations of CRP, TNF-α, and IL-6 by 2–3 folds. This shows that BPA caused the rats to develop an inflammatory reaction. The reported effects of BPA on inflammatory markers were minimized when the rats were given gastrodin (at both high and low dosages). CRP, TNF-α, and IL-6 levels in gastrodin-treated rats were equivalent to those in standard (metformin) and normal control rats. These data imply that gastrodin may be able to reduce the inflammatory response caused by BPA therapy. Gastrodin's effects may be mediated through altering the activity of inflammatory pathways or by lowering the synthesis of inflammatory mediators.
Study Limitation
The possible shortcoming of this study was that only albino rats were utilized in the study, which perhaps cannot emphasize the variety of the effects caused by Bisphenol-A (BPA) and the treatment of gastrodin as it would in humans. More comprehensive, more extensive and more complex animal models as well as human studies will be required to ascertain the efficacy of gastrodin in the treatment of BPA-induced insulin resistance.
Summary and Conclusion
In conclusion, Gastrodin has considerable therapeutic efficacy in alleviating insulin resistance, oxidative stress, and inflammation. Its administration led to a significant decreased in glucose level, lipid peroxidation, and pro-inflammatory indicators, surpassing metformin, particularly at elevated doses. Gastrodin's augmentation of antioxidant enzyme activities and stimulation of the AMPK/Nrf2 further emphasizes its significance in the management of metabolic illnesses, such as type 2 diabetes and BPA-induced oxidative stress. These data indicate that Gastrodin is a good option for future therapeutic use.
Grouping and Animal Treatments.

Footnotes
Acknowledgement
NA
Author's Contributions
OUO, NJA, and DEU conceived the study, ODO, PMA, ENN, and GUU Laboratory procedure, PMA, DEU, and OUO, conducted data analysis, UNO, PNO,, PUM, wrote the original draft, UAI, DEU and EUA read and approved the final draft before submission. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
Human and Animal Rights
The experiment adhered to the guidelines set out by the National Institute of Health for the proper care and use of laboratory animals. The experiment was authorised by the Ethical Review Committee of the Department of Biochemistry at Ebonyi State University, with the issued identification number EBSU/BCH/ET/22/031.
Ethical Approval
This work was authorized by the Department of Biochemistry Ethical Review Committee at Ebonyi State University in Abakaliki, Nigeria. The approval number is EBSU/BCH/ET/22/031
Informed Consent Statement
Not applicable
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.