Abstract
The current study aimed at investigating the therapeutic effects of rubusoside on type 2 diabetes mellitus (T2DM) mice models as an alternative hypoglycemic candidate drug. T2DM mice models were established with a combination of streptozotocin (STZ) intraperitoneal injection and high-fat diet. After 10 weeks of rubusoside intragastric administration (100, 200 mg/kg/day) to the mice, the body weight, fasting blood glucose, glucose tolerance, and blood lipids were measured. The liver protein expression levels of p-AMPK, GLUT2, GLUT4 and total antioxidant capacity were also investigated.
After 10 weeks of rubusoside administration, the levels of blood glucose and lipids were decreased in T2DM mice. Compared with the model group, rubusoside administration significantly decreased the liver mass-to-body weight ratio, upregulated p-AMPK and GLUT4, and downregulated GLUT2 expression levels in the liver. Activities of superoxide dismutase (SOD), catalase (CAT), and gluathione peroxidase (GSH-Px) were increased, and the concentration of malondialdehyde (MDA) was decreased to reduce oxidative stress in the liver. Liver hematoxylin and eosin (H&E) pathological analysis also showed that rubusoside had a protective effect on T2DM mice liver.
These results demonstrate that rubusoside could be used as an anti-diabetic candidate drug, and that its hypoglycemic mechanism might be related to the activation of adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK) to modulate the expression of GLUT2 and GLUT4. Finally, rubusoside could also increase total antioxidant capacity to protect the liver from oxidative stress.
Introduction
Diabetes mellitus (DM) morbidity is in constant increase, and its rate in China is as high as 10%. It is predicted that more than 600 million people will develop type 2 diabetes mellitus (T2DM) by 2045, worldwide1,2. DM primary clinical manifestation is an increase in the blood glucose range, which can be divided into type 1, type 2, and other diabetes. It can cause a wide range of chronic diseases and metabolic syndromes, such as cardiovascular disease 3 , hypertension 4 , obesity 5 , and nonalcoholic fatty liver disease 6 . T2DM accounts for more than 90% of the total cases and has become a global public health problem7,8.
As an important organ for regulating blood glucose, the liver has attracted considerable attention in the study of DM. Adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK) is one of the critical targets for T2DM treatment. Studies have shown that AMPK activation plays a vital role in the regulation and control of glucose transport (GLUT) in the liver, and therefore, also exerts a regulatory role in blood glucose of T2DM patients9-11. Previous studies have found that activation of the AMPK signaling pathway increases the stimulation of GLUT4 translocation, and blood glucose influx that is mediated by GLUT. Glucose enters β cells through a facilitated diffusion via the GLUT and is retained inside the cell through its phosphorylation by glucokinase. Moreover, GLUT4 transmits intracellular glucose delivery from the extracellular space12-14. GLUT2 is primarily found in the hepatocyte plasma membrane, and during the fasting state, it increases intracellular glucose production, and therefore, would favor glucose efflux15,16. Due to the continuous hyperglycemia, it will induce reactive oxygen species in the body. The study of Chis et al and others have shown that in streptozotocin (STZ)-induced diabetic models, there is a high oxidative stress that is due to a persistent and chronic hyperglycemia, which reduces the activity of the antioxidant defense system, thus promoting the production of free radicals. 17 These later lead to glycosylation and lipid oxidative modification, protein modification, and DNA damage.
Recently, Chinese medicines have received extensive attention in the treatment of DM, and shown to play a vital role18,19. Rubusoside is one of the main components of Rubuosideus corchorifolius (Rosaceae and Rubus). This functions in the heat clearing and moisturizing of lungs, provides cough and phlegm relief, swelling reduction, and muscle function enhancement.
In our previous study, we found that rubusoside effectively reduces blood glucose levels. In this study, we investigate whether the hypoglycemic mechanism of rubusoside is related to the activities of AMPK and if this hypoglycemic mechanism is related to antioxidant stress and organ protection. For this, we investigated the anti-diabetic effect of rubusoside in the STZ-induced T2DM mice model and detected the expression of p-AMPK in liver to clarify the hypoglycemic mechanism of rubusoside and verify its antioxidant capacity in preventing oxidative stress.
Results
High Performance Liquid Chromatography (HPLC) Analysis of Rubusoside
The extraction rate of rubusoside was 72%, and from the two peaks that were detected by liquid chromatography, one of them corresponded to rubusoside
Rubusoside Effect on Blood Glucose and Lipids in T2DM Mice
To evaluate the hypoglycemic effect of rubusoside on blood glucose in T2DM mice, we tested fasting blood glucose and oral glucose tolerance in each group of mice: control (Group 1), model (Group 2), rubusoside treated with 100 mg/kg/day (Group 3), rubusoside treated with 200 mg/kg/day (Group 4), and metformin treated (Group 5). The results showed that the fasting blood glucose concentrations of the model group were significantly different from those of the control group (Figure 1A). After 10 weeks of rubusoside administration, the groups that received different concentrations of rubusoside had a decline in fasting blood glucose, but the levels of glucose in the high-dose intervention group recovered to the normal range compared to the control group, indicating that rubusoside reduces fasting blood glucose in T2DM mice. Oral glucose tolerance test (OGTT) was also operated to confirm the hypoglycemic effect of rubusoside (Figure 1B-C).
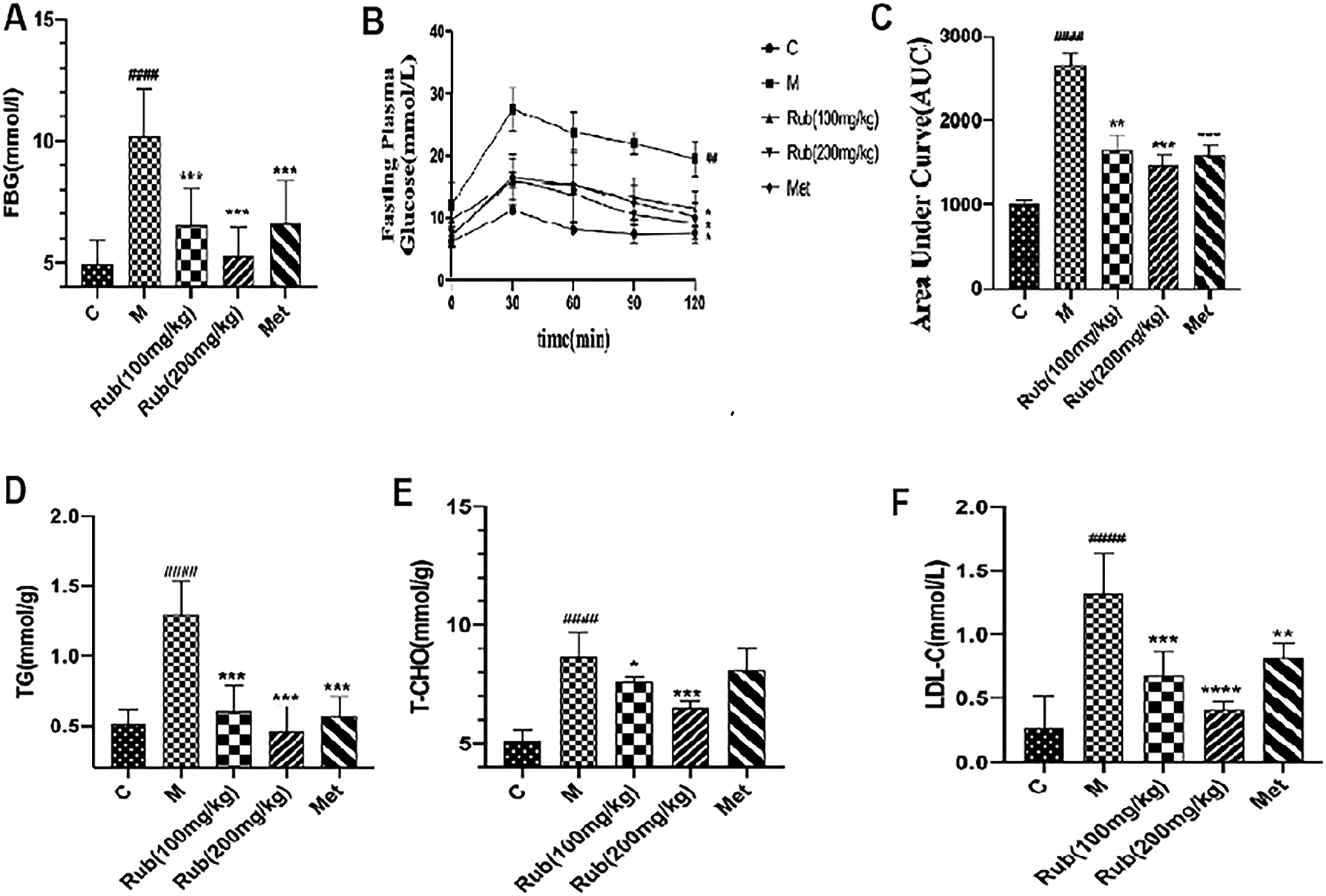
Effect of rubusoside on blood glucose and lipids.
To evaluate further the hypoglycemic effect of rubusoside on T2DM mice, we measured the blood glucose concentrations of triglyceride (TC), total cholesterol (TG), and low-density lipoprotein cholesterol (LDL-C). The blood glucose concentrations of TC, TG, and LDL-C in the model group were significantly different from those of the control group. After 10 weeks of rubusoside administration, a decrease in the concentrations of TC, TG, and LDL-C in the blood glucose of mice that were treated with different concentrations of rubusoside was observed (Figure 1D-F). The results showed that rubusoside also improves the levels of blood lipids in T2DM mice.
Rubusoside Effect on Liver Mass-to-Body Weight Ratio in T2DM Mice
The liver is one of the critical organs that are involved in the regulation of blood glucose. To evaluate the effect of rubusoside on T2DM mice liver, we measured the liver mass-to-body weight ratio in each group of mice. After 10 weeks of rubusoside administration, the results showed that the liver mass-to-body weight ratio of the mice that were administered with rubusoside was significantly lower than that of the model group, indicating that rubusoside decreases the liver mass-to-body weight ratio of T2DM mice. (Figure 2A-C)

Rubusoside effect on the liver weight-to-body weight ratio.
Rubusoside Effects on p-AMPK, GLUT2, and GLUT4 Protein Expression in the Liver
To determine the hypoglycemic mechanism of rubusoside, we investigated the protein expression levels of p-AMPK, GLUT2, and GLUT4 in the liver. After 10 weeks of rubusoside administration, the results showed an upregulation of p-AMPK and GLUT4 protein expression levels, but a downregulation in that of GLUT2. These findings suggest that the mechanism of glucose reduction by rubusoside might be related to modulating p-AMPK, GLUT2, and GLUT4 expression levels. (Figure 3A-C)

Expression of p-AMPK, GLUT2, and GLUT4 in the liver after rubusoside administration.
Rubusoside Effect on Liver Oxidative Stress in T2DM Mice
Oxidative stress plays an important role in the progression of T2DM. To evaluate the effect of rubusoside on oxidative stress in T2DM mice liver, we investigated the levels of superoxide dismustase (SOD)

Evaluation of the improvement in oxidative stress ability after 10 weeks of rubusoside administration. After treatment of each dose group with rubusoside, its influence on the biological activity of (A) CAT, (B) SOD, (C) GSH-Px, and (D) MDA were measured.
Hyphology Analysis on Rubusoside Effect in the Liver
To explore the liver morphological changes following rubusoside administration, a hyphology analysis was performed. The liver pathological damage in the model group was obvious, and after rubusoside administration, the liver damage was significantly reversed. This confirms that rubusoside improves the liver damage of T2DM mice (Figure 5A-E). This conclusion was consistent with the observation that rubusoside improves the liver mass-to-body weight ratio and that rubusoside has an anti-oxidative protective effect in the liver.

Histological analysis of T2DM mice liver treated with rubusoside for 10 weeks (× 400).
Discussion
In recent years, identifying effective hypoglycemic drugs in traditional Chinese medicines or natural products has become a research hotspot. Particularly, studies have shown that glycoside extracts from a large number of Chinese medicine source plants have anti-diabetic effects19,20. Xu et al and others showed that total saponins from Stauntonia chinensis have significant hypoglycemic and hypolipidemic activities in diabetic db/db mice 21 . Zhang et al and others showed that hollyhock flowers (Althaea rosea (L.) Cav.) have significant reduction effects on serum glucose and lipid levels in KK-Ay mice after 8 weeks of administration 22 . Chen et al and others showed that iridoid glycosides from Cornus officinalis have an anti-diabetic effect, which alleviates the increase in blood glucose and serum insulin in male Wistar rats that were induced by a high-fat diet and STZ 23 . These studies proved that some glycoside-containing Chinese herbal medicinal extracts might have a hypoglycemic effect.
After 10 weeks of rubusoside administration, the concentration of fasting blood glucose and OGTT results in T2DM mice confirmed that rubusoside had an ideal hypoglycemic and hypolipidemic (TC, TG, and LDL-C) effect in T2DM mice.
AMPK plays a major role in the liver glucose uptake and is one of the important targets for T2DM treatment24,25. Several studies have proved that AMPK improves the body’s levels of blood glucose and lipids after their activation26-28. GLUT2 and GLUT4 play critical roles in the regulation of liver intracellular GLUT. GLUT2 mainly promotes the elimination of glucose in hepatocytes, while the GLUT4 effect is opposite to that of GLUT2. Reyhaneh’s study showed that S. bachtiarica (Lamiaceae) extract exerts its effects by decreasing oxidative stress markers and by modulating the expression of GLUT genes. The study also reported that S bachtiarica extract upregulates GLUT2 expression and downregulates GLUT4 expression in the liver25,29. Moreover, another study on p-AMPK in the liver of T2DM showed that GLUT2 expression was downregulated, while the expression of GLUT4 was upregulated 16 . It also showed that rubusoside activates the AMPK pathway in the liver of T2DM mice and improves the expression of GLUT2 and GLUT4 in T2DM mice. This might be an essential mechanism that is mediated by rubusoside in lowing blood glucose.
STZ is currently commonly used to generate T2DM animal models and most scholars believe that oxidative stress plays an important role in the development of STZ-induced diabetes16,30. Furthermore, glucose uptake by the liver is significant to the body’s glucose homeostasis. Studies have shown that oxidative stress had a significant effect on liver function 31 . The study of Molehin et al showed that lipid peroxidation was an important event in oxidative stress. The increase in MDA level indicates the enhancement of lipid peroxidation that leads to liver injury and failure of the antioxidant defense mechanisms 32 . Therefore, improving the oxidative stress level in T2DM mice is necessary to prevent diabetes occurrence and development33,34. Hu et al showed that after its activation, AMPK reduces active oxygen in the body and plays a role in anti-oxidative stress 35 . In this study, previous data related to the level of oxidative stress were tested and we found that rubusoside increases the levels of SOD, CAT, and GSH-Px, and decreases the levels of MDA in T2DM mice, therefore, reducing oxidative stress. Meanwhile, liver hyphology analysis and liver mass-to-body weight ratio also proved that the liver function in T2DM mice was significantly improved after rubusoside administration. It also indicated that rubusoside reduces oxidative stress and protects the liver, which might be an important mechanism for its hypoglycemic effect.
Conclusion
This study confirmed that rubusoside could reduce blood glucose and lipids in T2DM mice, and that the mechanism is associated with AMPK activation. The results showed that after its activation by rubusoside, AMPK reduces GLUT2, increases GLUT4 expression, and inhibits oxidative stress in the liver of T2DM mice, while lowering blood glucose, protecting the liver and preventing further development of the disease. These results demonstrated that rubusoside prevents T2DM through multi-target efforts.
Materials and Methods
Drugs and Reagents
Serum glucose, SOD, GSH-Px, CAT, MDA, TG, TC, and the LDL-C assay kits were purchased from Nanjing Jiancheng (Jiangsu, China), STZ from Sigma-Aldrich (St Louis, USA), the high-fat and high-sugar feed from Experimental Animal Technology (Changchun China), D-glucose from Sangon Biotech (Shanghai China), and GLUT2 and GLUT4 enzyme linked immunosorbent assay (ELISA) assay kits from Huacheng (Changchun, China).
Experimental Animals
Eight-week-old male C57BL/6J mice, weighing 20 to 22 g, were purchased from Liaoning Changsheng (Shenyang China). The animals were kept in a standard animal laboratory (temperature 21-23°C, humidity 50%). The mice were adaptively fed for 7 days before the start of the experiment. All laboratory and animal care procedures were performed in accordance with the regulations that were approved by the Animal Care and Use Committee of Jilin Medical College (Approval number: 2019-LW003).
HPLC Conditions
The dried leaves of R corchorifolius were crushed, and 40 mg sifted powder was put into a 20 mL conical bottle to which 10mL distilled water was added. The mixture was stirred with a magnetic agitator (100 r/min) at 65°C. After stirring and extracting for 30 min, repeated twice, the water extract was collected and stored at 4°C, ready to use.
Chromatographic conditions: Column: Agela Diamonsil-C18 (150 mm × 4.6 mm, 5 μm); Mobile phase methanol: trifluoroacetic acid (0.05% v/v) = 75:25; Detection wavelength: 208 nm; Flow rate: 1.0 mL/min; Column temperature: 30°C; Injection volume: 5 μL.
Experimental Design
The mice were fed for 7 days with a common diet to promote their environmental adaptation. They were fed with a high-sugar and high-fat diet for 4 weeks, and then intraperitoneally injected with a prepared STZ solution (130 mg/kg). After 7 days, fasting blood glucose levels were measured using the serum glucose kit (fasting for 12 h and free drinking water). Mice with fasting blood glucose ≥ 7.8 mmol/mL were used for the experiments. The mice were randomly divided into 5 groups according to blood glucose level, and followed these intervention regimens (n = 10): Group 1, Control group (C), Group 2 Model group (M), Group 3 rubusoside (Rub) (100 mg/kg), Group 4 rubusoside (Rub) (200 mg/kg), and Group 5 metformin (Met).
Groups 1 and 2 had daily intragastric administration of normal saline. Groups 3 and 4 were given 100 mg/kg/day and 200 mg/kg/day of rubusoside, respectively, and Group 5 was given 200 mg/kg/day of metformin for 10 weeks. At the end of the experiment, blood was taken from the back of the eyeball and the mice were sacrificed by cervical dislocation. For the detection of fast blood glucose and lipids, serum was obtained from blood samples after centrifugation at 3000 rpm for 10 min.
Oral Glucose Tolerance Test
The mice were fasted for 12 h and then given glucose (2 g/kg) by intragastric administration. The blood glucose level was measured at 0, 30, 60, 90 and 120 min. The blood samples were centrifuged at 3000 rpm for 10 min to obtain serum and detect blood glucose.
GLUT2, p-AMPK, and GLUT4
After 10 weeks of rubusoside administration, livers from each group of mice were collected. Liver suspensions were obtained by centrifugation at 2000 rpm for 30 min. The levels of GLUT2, p-AMPK, and GLUT4 in the livers were detected using the corresponding ELISA kits, and according to the manufacturers’ instructions.
Oxidative Stress and Anti-Oxidation
After 10 weeks of rubusoside administration, the livers from each group of mice were collected. Liver suspensions were obtained by centrifugation at 2000 rpm for 30 min. SOD, GSH-Px, MDA, and CAT levels were determined using commercially available assay kits, and according to the manufacturers’ instructions.
Histological Examination
The livers were removed and fixed in 4% paraformaldehyde for 24 h, then embedded in paraffin. Slices of 3 mm were obtained and used for hematoxylin and eosin (H&E) staining. A Nikon microscope (Nikon Eclipse 80i; Tokyo, Japan) was used to examine the slices, and the data were collected by randomly scanning the slices using a computer with imaging software (ACT-2U version 1.52; Nikon).
Statistical Analysis
GraphPad Prism 8 software (San Diego, California, USA) was used for data analysis, and the experimental results were expressed as an average of ± SD. Statistical analysis to determine statistical differences was conducted using either the Student’s t-test or one-way analysis of variance.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project Agreement for Science & Technology Development, Jilin Province (grant numbers 20180201033YY and 20200404135YY).
Data Availability
The data used to support the findings of this study are included within the article.
Ethical Approval
This study was approved by the Administration Committee of Experimental Animals, Jilin Province, China.
Statement of Human and Animal Rights
All experimental procedures involving the animals were conducted in accordance with the Committee of Jilin Medical College (Approval number: 2019-LW003), China and approved by the Administration Committee of Experimental Animals, Jilin Province, China.
Statement of Informed Consent
There are no human subjects in this article, and therefore, informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
