Abstract
Objective
Yougui pills (YGP) have been used clinically to treat diminished ovarian reserve (DOR), but, whether YGP could have a palliative effect in the DOR rat model and the underlying mechanisms have not been clearly elucidated. This study aims to investigate the therapeutic effect and potential mechanisms of YGP in rats with cortisol-induced DOR.
Methods
The cortisol-induced DOR model was used to investigate the protective effects and mechanism of YGP on DOR. The DOR model was established by intramuscular injection of 25 mg/kg cortisol for 28 consecutive days. Meanwhile, the rats were weighed and administered intragastrically YGP or progynova. To evaluate the mitigating effect of YGP on DOR, pharmacodynamic indicators were assessed including body weight, estrous cycle, H&E staining, and serum levels of AMH, FSH, LH, and cortisol. Oxidative stress (OS) and apoptosis pathways were further validated using Western blotting analysis.
Result
YGP significantly ameliorated the symptoms of DOR. Administration of YGP improved the ovarian index, estrous cycle, and body weight. After treatment with YGP, cortisol, FSH, and LH levels declined and AMH levels elevated. YGP increased the number of primary, secondary, and antral follicles, but decreased the number of atretic follicles. Mechanically, YGPtreated cortisol-induced DOR via the Keap1/Nrf2 signaling pathway which regulates OS. YGP also reversed the aberrant expression of apoptosis-related proteins Caspase-3, Bcl-2, and Bax.
Conclusion
YGP could reduce ovarian OS and apoptosis in cortisol-induced rats via regulating the Keap1/Nrf2 signaling pathway, thereby enhancing ovarian reserve function.
Introduction
Diminished ovarian reserve (DOR) is defined as a reduction in the number and quality of oocytes, with the main clinical manifestations including menstrual abnormalities, even amenorrhea and infertility. Increasing and younger incidence of DOR, the prevalence of DOR is reported to vary between 10% and 35%, 1 and may exceed 50% in women over 40 years of age. 2 It has a serious impact on the reproductive health and living quality of women at childbearing age and, if left untreated, it can progress to premature ovarian failure (POF), research has shown that the evolution from DOR to POF is a progressive process that takes approximately 1 to 6 years to complete. 3
The pathogenesis of DOR remains unclear. However, studies have indicated that it is associated with genetic factors, 4 oxidative stress (OS), 5 apoptosis, 6 and psychological stress. 7 OS is a consequence of the overproduction of reactive oxygen species (ROS) following a stimulus in vivo or in vitro, resulting in a significant imbalance between the oxidative and antioxidant systems. This leads to damage to the body. 8 An accumulation of excessive ROS could cause damage to proteins, lipids, and nucleic acids, which may result in mutations or deletions in nuclear and mitochondrial DNA. Nicotinamide adenine dinucleotide phosphate oxidase 4 (NOX4) represents an important source of ROS. It serves to catalyze the production of ROS via OS. 9 Superoxide dismutase (SOD) is a metalloenzyme that acts as an antioxidant in living organisms. 10 The oxidation of proteins results in the inactivation of SOD, which in turn leads to the induction of apoptosis and tissue structural damage. 11 Recent studies have demonstrated that OS triggers apoptosis in most germ cells and even in ovulated oocytes in the ovary, which represents a significant contributor to the fertility loss observed in patients with DOR.7,12 The Kelch-like ECH-associated protein 1-NF-E2-related factor 2 (Keap1-Nrf2) system is an important pathway for the resistance to OS. Keap1 is a redox damage sensor, and Nrf2 is a key transcription factor regulating a range of cytoprotective genes, such as NQO1 13 and SOD. It plays an essential role in the antioxidant response. Keap1 represses the activity of Nrf2 under homeostatic conditions, while Nrf2 is released from this suppression when it is exposed to cellular stress, including electrophilic and OS. 14
Currently, modern medical treatments of DOR involve hormone replacement therapy (HRT), assisted reproductive technology (ART),
15
dehydroepiandrosterone (DHEA),16,17 ovulation induction therapy, and other treatments. Although these treatments are available for the majority of t patients with DOR, there are numerous limitations. For instance, HRT has a considerable number of side effects and contraindications, which restrict its clinical application.
18
The efficacy of DHEA has been a topic of considerable debate in the literature.
19
DOR is one of the most difficult challenges in ART.
15
Accordingly, the search for effective therapeutic strategies remains the main focus of reproductive medicine today. Traditional Chinese Medicine (TCM) is widely used in China to treat DOR. Based on TCM theories, oocytes originate from the kidney essence, and kidney-yang deficiency may be one of the root causes of DOR.
20
Yougui pills (YGP) is a Chinese medicinal compound consisting of Cortex Cinnamomi Cassiae
In the present study, the cortisol-induced DOR rat model was used to evaluate the therapeutical effects of YGP. OS and apoptosis in the ovary were studied to investigate mechanisms of YGP on impaired ovarian function. These results provide more research foundations for the efficacy of YGP.
Materials and Methods
Chemicals and Reagents
YGP (Z11021040) was purchased from Beijing Tongrentang Co., Ltd. Cortisol (S31360) was purchased from Shanghai Yuanye Biotechnology Co., Ltd. Estradiol Valerate Tablets (Progynova, J20130009) was obtained from Delpharm Lille Sas. The papanicolaou stain and HE stain were purchased from biosharp. The RIPA lysis buffer, PMSF, and BCA protein assay kit were purchased from Byeotime. Enzyme-linked immunosorbent assay kits for FSH (MM-70867R1), LH (MM-0624R1), AMH (MM-0219R1), and Cortisol (MM-0574R1) were obtained from Wuhan Bioqiandu Technology Co., Ltd. Primary antibodies of Keap1 (1:1000, WL02135), Nrf2 (1:1000, WL02117), SOD2 (1:1000, WL02506), NQO1 (1:1000, WL04860), Bcl-2 (1:1000, WL01556), Bax (1:1000, WL01637), and Caspase3 (1:1000, WL02117), β-tubulin (1:1000, WL01931), and antirabbit secondary antibody (1:2000, WLA023) were purchased from Wanlei Biochemical Technology Co., Ltd. NOX4 (1:1000, 14347-1-AP) was purchased from proteintech.
Animal
Female SD rats weighing 180 to 200 g (age 6 weeks) were purchased by Nanjing Kaisijia Biochemical Technology Co., Ltd and were adaptively fed for 1 week (room temperatures of 25 °C ± 2 °C, under controlled relative humidity levels of 50% ± 5% and photoperiods of 12 h daylight cycle). They were given free food and water. All animal procedures were approved by the Animal Ethics Committee of Jiangsu Province Academy of Traditional Chinese Medicine (AEWC-20220615-214).
YGP Administration
The animals were randomized into 5 groups (

YGP restored weight loss and ovarian index. (A) Experiment design. (B) Ovarian index. (C and D) Changes in body weight. ###
Estrous Cycle Monitoring
The estrous cycle was measured before the experiment to ensure that all groups were normal. From day 6 of the experiment, the estrous cycle was checked daily. The stage of the estrous cycle was assessed by cytological analysis of vaginal smears. The vaginal smear was taken at 9 a.m. The vaginal cavity was flushed with 1 mL of normal saline. The collected solution was distributed uniformly across glass slides, and fixed for 30 min in 95% ethanol. Then, the samples were stained with papanicolaou staining, washed under running water and dried. Finally, light microscopy (GMBH37081; Carl Zeiss) was used to examine the morphology and number of exfoliated cells.
Ovarian Index
The body weights of the rats were recorded daily throughout the course of the experiment. After 28 days of administration, blood samples of rats were collected from the rats under isoflurane anesthesia, after which the rats were sacrificed. The ovaries were subsequently collected and weighed. A portion of the ovarian tissues were fixed in 4% paraformaldehyde for histopathological examination, and the remaining ovaries were stored at −80 °C. Ovarian index = wet weight of ovary (mg)/body weight (g).
Enzyme-Linked Immunosorbent Assays
According to the instructions of the manufacturer, serum levels of FSH, AMH, LH, and cortisol were determined. Blood samples were obtained from the abdominal aorta, and coagulated for 3 h at room temperature. The serum was then collected by centrifugation at 3000×g for 20 min and stored at −80 °C.
Tissue Histopathological Analyses
Portions of the ovarian tissues isolated from rats were fixed in 4% paraformaldehyde, embedded in paraffin, and cut into 4 µm thick slices for subsequent analysis. Subsequently, the sections were then deparaffinized, rehydrated, and finally stained with H&E. The ovarian changes were examined using a microscope (GMBH37081; Carl Zeiss).
Western Blot
Ovarian tissues were homogenized using RIPA lysis buffer containing with 1% PMSF, and then the concentration of ovarian protein was measured by the BCA method. The protein was separated by SDS-PAGE electrophoresis and subsequently transferred to a polyvinylidene fluoride membrane (IPFL00010, Merck KGaA, Germany). After blocking with 5% nonfat milk in TBST for 2 h at room temperature, the membranes were incubated with the appropriate primary antibodies overnight at 4 °C. The membranes were washed on 3 times with TBST, and then an antirabbit secondary antibody was added for 2 h. ECL reagent was used to detect the blots. Image J software was applied for semiquantify analysis of the Azure Biosystems C600 (Azure Biosystems Inc., USA) visualized proteins.
Statistical Analysis
All results were presented as mean ± SD and were performed with GraphPad Prism 8.0.2. One-way ANOVA or Student
Results
YGP Restored Weight Loss and Ovarian index
To explore the pharmacological efficacy of YGP, a cortisol-induced DOR rat model was used in this study. Glucocorticoids such as cortisol and corticosterone have been found to promote apoptosis in ovarian cells, and impair the developmental potential of oocytes.25,26 The experiment design is shown in Figure 1A. The status of the rats was monitored daily. Weight loss in the DOR group was significant in comparison to control (Figure 1C and D), whereas it was markedly attenuated after 28 days of YGP and progynova intervention. Figure 1B shows a significant reduction in ovarian index in the model group. In response to YGP and progynova, there was a marked increase in the ovarian index in rats.
YGP Alleviated Ovarian Morphological Changes
The follicular stage was classified according to the method described previously. 27 To assess microscopic changes in ovarian morphology, H&E stain was performed on ovarian sections (Figure 2A). The number of primary follicles was significantly decreased in the model group, while it was higher after YGP treatment (Figure 2B). Cortisol-induced reduction in the number of secondary follicles and antral follicles is reversed by HYGP and progynova administration (Figure 2C and D). H&E analysis of ovarian sections displayed a marked increase in the number of atretic follicles responding to cortisol, while the abnormalities were ameliorated after administration of YGP and progynova (Figure 2E). The number of various follicles was dose-dependently elevated in the high and low experimental groups compared to the model group. Overall, YGP could alleviate cortisol-induced ovarian dysfunction.

YGP alleviated ovarian morphological changes and estrous cycle. (A) Representative micrographs of H&E. PrF: primary follicles; SF: secondary follicles; GF: antral follicles; AF: atretic follicles; CL: corpus luteum (magnification ×100). (B) The number of primary follicles. (C) The number of secondary follicles. (D) The number of antral follicles. (E) The number of atretic follicles. (F) Example images of estrous cycle phases. Black arrow showed the nucleated epithelial cells; blue arrow showed the nonnucleated cornified epithelial cells; red arrow showed the leukocytes (magnification ×100). (G) Changes in the estrous cycles between 21 and 28 days of the experiment. ##
YGP Restored the Estrous Cycle
To evaluate the estrous cycle, this study used cytological evaluation of vaginal smears. The estrous cycle stages were identified as previously described methods. 28 In brief, proestrus: mainly nucleated epithelial cells (black arrow). Estrus: mainly nonnucleated cornified epithelial cells (blue arrow), irregular shape, cell density is large. Metestrus: cornified epithelial cells accompanied by nucleated epithelial cells and a large number of leukocytes (red arrow). Diestrus: primarily leukocytes, cell density is average (Figure 2F). All rats had normal estrous cycles before modeling. After cortisol treatment, the number of rats showed irregular estrous cycles. However, half of the rats in the HYGP and progynova groups returned to their normal estrous cycles (Figure 2G).
YGP Regulated Serum Hormone Levels
Cortisol, a glucocorticoid, is a biological marker for responding to stress. 29 As shown in Figure 3A, the concentration of cortisol in the serum was significantly lower after treatment with YGP. The reliable markers of ovarian reserve function include FSH, LH, and AMH. 30 Significant changes in FSH, LH, and AMH levels were observed in the model group. Specifically, in response to cortisol, there was an increase in FSH and LH, while the AMH levels declined. Compared to the model group, FSH and LH levels were significantly reduced and AMH levels significantly elevated after YGP intervention. In addition, the changes in hormone levels showed a significant dose–effect relationship between the YGP groups (Figure 3B-D). The results suggest that YGP may facilitate the regulation of serum hormone levels, thereby sustaining ovarian function.
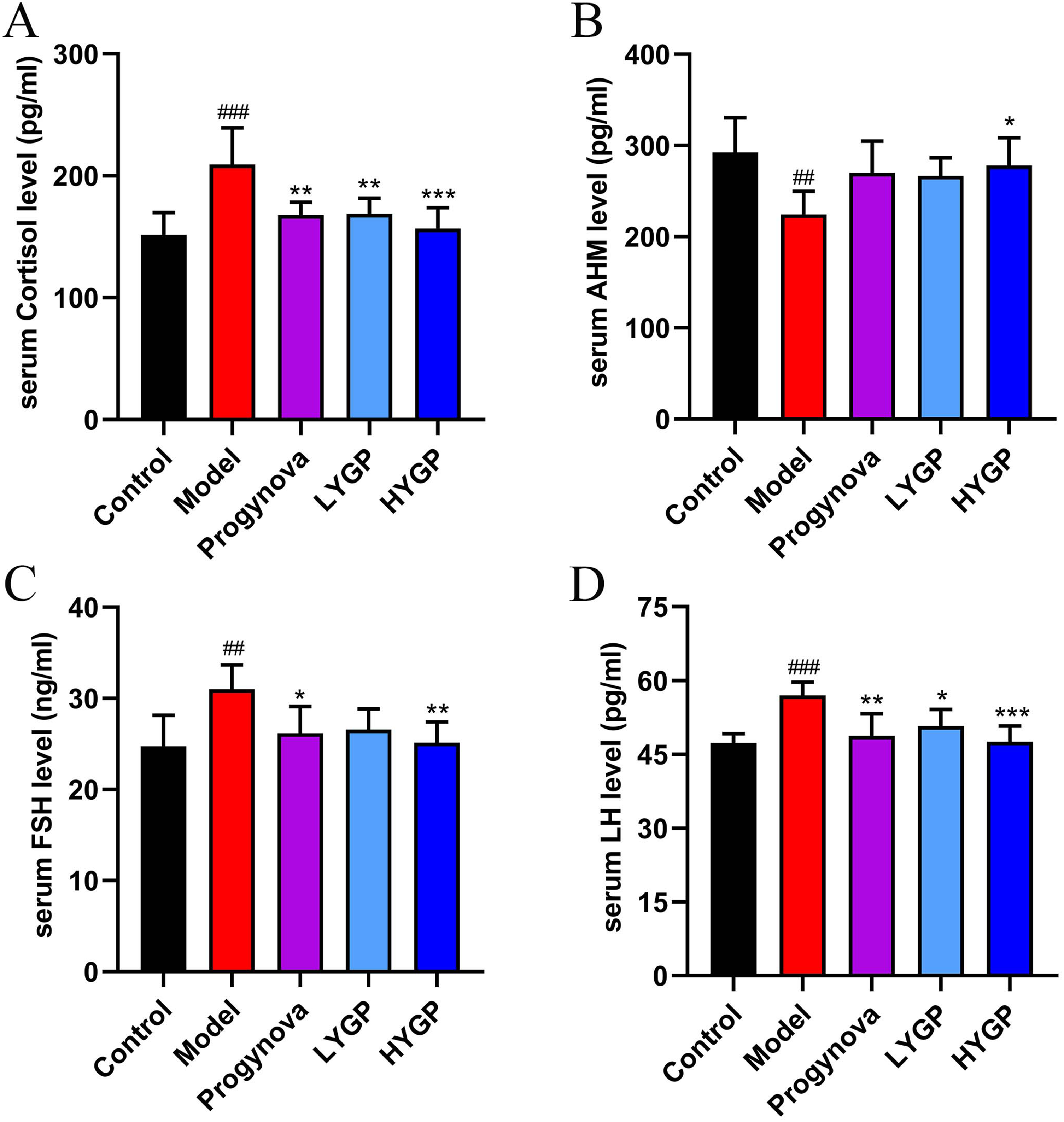
YGP regulated serum hormone levels. Serum hormone levels of (A) cortisol, (B) AMH, (C) FSH, (D) LH. ##
YGP Reduced OS by Regulating Keap1/Nrf2
OS has a meaningful impact on the microenvironment of the follicle, the maturation and fertilization of the oocyte and the development of the embryo. 31 To determine whether the Keap1/Nrf2 pathway, a vital pathway in the defense against OS, 32 is affected in the DOR model, this study used WB to measure the levels of Nrf2, Keap1, SOD2, and NQO1 which are proteins associated with the pathway in ovarian tissue. The expression of Keap1 and Nrf2 was markedly reduced in the model group. YGP significantly upregulated the cortisol-induced decline of Nrf2 and Keap1 expression (Figure 4A-C). After cortisol treatment, SOD2 and NQO1 protein expression were markedly lower, whereas NOX4 expression was markedly higher. However, treatment with YGP and progynova largely abolished the abnormal expression of SOD2, NQO1, and NOX4 (Figure 4D-F). The expression of proteins associated with the pathway were dose-dependently elevated in the low and high dose experimental groups compared to the model group. In conclusion, YGP could alleviate OS in ovarian tissue by regulating the Keap1/Nrf2 signaling pathway.

YGP reduced oxidative stress by regulating Keap1/Nrf2. The protein levels of (A and B) Keap1, (A and C) Nrf2, (A and D) SOD2, (A and E) NQO1 and (A and F) NOX4 were determined using immunoblots. ##
YGP Decreased Apoptosis Levels in Ovarian Tissues
To demonstrate the role of apoptosis in the cortisol-induced DOR model, this study detected the ovarian levels of Bcl-2, Bax, and Caspase3. As shown in Figure 5C-E, the expression of Bax and Caspase3 were markedly upregulated, whereas the Bcl-2 expression was downregulated after cortisol treatment. After the YGP treatment, the abnormal expression of Bcl-2, Bax, and Caspase3 induced by cortisol were attenuated in a dose-dependent manner. Additionally, the Bcl-2/Bax ratio was significantly downregulated in DOR rats, whereas it was significantly increased in the YGP group (Figure 5B). In conclusion, YGP could decrease apoptosis levels in ovarian tissues.

YGP decreased apoptosis levels in ovarian tissues. The protein levels of (A and C) Bax, (A and D) Bcl-2 and (A and E) Caspase3 were determined using immunoblots. (B) The Bcl-2/Bax ratio. ##
Discussion
Previous studies have shown that YGP could alleviate DOR in a clinical setting. 22 However, it remains unclear whether YGP could exert a therapeutic effect in a rat DOR model and, if so, by what mechanism. Our studies showed that YGP significantly restored body weight, the ovarian index, ovarian morphology, and the estrous cycle. YGP was observed to regulate serum levels of cortisol, FSH, AMH, and LH. Moreover, YGP alleviated cortisol-induced DOR by reducing OS and apoptosis. YGP inhibited OS by regulating Keap1/Nrf2 signaling pathway. Meanwhile, YGP reduced apoptosis and upregulated the Bcl-2/Bax ratio. Overall, YGP could alleviate DOR by regulating OS and apoptosis in rats.
Women tend to be more susceptible to stressful situations due to changes in society and lifestyle. 33 Psychological stress has been identified as a significant contributing factor in the pathogenesis of diseases affecting the female reproductive system. 34 Several researches have indicated that psychological stress could lead to the development of DOR in female rodent models.35,36 Hypothalamic–pituitary–adrenal (HPA) dysregulation is induced by repeated and prolonged stress, leading to the secretion of various stress hormones, including cortisol. 37 An increased cortisol level is generally regarded as a specific indicator of stress. In addition, the HPA axis is suppressed, resulting in the development of a stress response after glucocorticoid injection. 38 Meanwhile, cortisol impairs oocyte function through induction of ovarian cell apoptosis. 25 It has been shown that cortisol is used to construct DOR model, 39 the cortisol-induced model also exhibited symptoms of DOR in this study. Therefore, it is important to explore the mechanism of YGP in preventing cortisol-induced DOR and thus reducing stress-related adverse effects. The modeling of cortisol, although it causes ovarian damage, is a less mature modeling approach, we will therefore further investigate modeling methods for cortisol-induced DOR.
AMH is a hormone produced by granulosa cells in ovarian follicles. 40 AMH levels are directly proportional to ovarian reserve, therefore, AMH has been used to evaluate ovarian reserve function. 41 This study confirmed that YGP significantly improved body weight, ovarian index, and AMH levels. A dose-dependent boost in the numbers of primary, secondary, and antral follicles in response to YGP was observed, suggesting that YGP could protect granulosa cells and improve ovarian reserve. The hypothalamic–pituitary–gonadal (HPG) axis is a complete and coordinated neuroendocrine system. The hypothalamus regulates pituitary gland to secrete LH and FSH through GnRH, which connects the brain to the pituitary gland, and thereby controls gonadal development and sex hormone secretion. 42 LH and FSH have a synergistic effect on the maturation of the follicle and corpus luteum, estrogen secretion as well as ovulation. 43 Meanwhile, AMH strongly increases the activation of GnRH neurons and effectively stimulates GnRH secretion. 42 This research confirmed that YGP could restore serum levels of AMH, LH, and FSH. Taken together, YGP may improve ovarian reserve function by modulating the HPG axis, thereby facilitating the repair of disrupted gonadal hormones.
We further investigated the possible mechanisms based on the protective effect of YGP in DOR rats. Apoptosis is a form of cell death that is highly regulated and necessary for many homeostatic and pathological. 44 Follicle fate is determined by ovarian apoptosis with the balance between antiapoptotic and proapoptotic molecules. Studies have shown that apoptosis is associated with oocyte loss and atresia. 45 The Bcl-2 family proteins are key regulators of apoptosis, which include both proapoptotic (Bax) and antiapoptotic (Bcl-2) members. Cell fate is largely controlled by the balance between these 2 Bcl-2 proteins. 46 Caspase-3 is the predominant mediator of apoptosis. 47 The present study demonstrated that YGP may upregulate Bcl-2 expression while downregulate the expression of Bax, caspase-3. To sum up, YGP could restore DOR by inhibiting apoptosis.
Psychological stress has been demonstrated to induce the release of cortisol, which reduces the quality and number of oocytes. 7 Psychological stress also triggers the production of ROS, which accumulate in the ovary leading to OS. 7 An imbalance between prooxidants and antioxidants in the body can cause ovarian damage, which may subsequently lead to the development of a range of reproductive diseases. 48 It has been shown that high levels of cortisol and OS could induce the death of granulosa cells. 49 OS triggers apoptosis in most germ cells and even in ovulating oocytes. OS can activate the mitochondria-mediated apoptosis pathway, resulting in reduced ovulation rate and oocyte quality.50,51 In this study, cortisol was shown to induce apoptosis and OS in the ovary, which is consistent with the above studies. Here, we found that treatment with YGP ameliorated OS in ovarian tissue. Overall, YGP could improve DOR by reducing OS-mediated apoptosis.
Nrf2 and its key negative regulator Keap1, are critical for maintaining oxidation-reduction equilibrium, metabolic homeostasis, and modulating inflammation. 52 The Keap1-Nrf2 system is the main mode for organisms to resist oxidation, which includes both exogenous and endogenous origin. 14 Keap1 works as a redox sensor, and Nrf2 is a transcription factor that modulates various cytoprotective signals. Keap1 exerts an inhibitory effect on Nrf2 activity at homeostatic conditions. Conversely, upon exposure to stress, Nrf2 is released. Loss of Nrf2 function led to premature follicle activation and an increased the sensitivity of the ovaries to environmental pollutants.5,53 The abnormal expression of Nrf2 induced by cortisol was prevented by YGP. Meanwhile, the abnormal expression of NQO1 and SOD2, which are downstream proteins of the Keap1/Nrf2 pathway, were also restored after YGP administration. These findings confirmed that YGP could regulate the Keap1/Nrf2 pathway to attenuate ovarian oxidative damage.
Conclusion
In conclusion, this study confirmed that YGP could ameliorate ovarian reserve against psychological stress-induced DOR. YGP could reduce ovarian OS and apoptosis in cortisol-induced rats via regulating the Keap1/Nrf2 signaling pathway, which leads to the enhancement of ovarian reserve function. This provides an experimental basis for the clinical application of YGP.
Bei-Bei Jiao, Tian Li, Bei-Bei Zhou, Si Chen, Yue Chen, Yan Lu, Jian-Min Liu, Wei-Bo Zhao, Jian Zhang, and Pei-Juan Wang
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241296409 - Supplemental material for Yougui Pills Alleviates Diminished Ovarian Reserve Through Regulating Oxidative Stress and Apoptosis in Rats
Supplemental material, sj-docx-1-npx-10.1177_1934578X241296409 for Yougui Pills Alleviates Diminished Ovarian Reserve Through Regulating Oxidative Stress and Apoptosis in Rats by Bei-Bei Jiao, Tian Li, Bei-Bei Zhou, Si Chen, Yue Chen, Yan Lu, Jian-Min Liu, Wei-Bo Zhao, Jian Zhang and Pei-Juan Wang in Natural Product Communications
Footnotes
Authors’ Contributions
Bei-Bei Jiao: conceptualization, data curation, formal analysis, methodology, validation, and writing—original draft; Tian Li: methodology and data curation. Bei-Bei Zhou: methodology and visualization. Si Chen: formal analysis, investigation, and methodology. Yue Chen: data curation, formal analysis, and software. Yan Lu: methodology and data curation. Jian-Min Liu: methodology and data curation. Wei-Bo Zhao: conceptualization, investigation, and supervision. Jian Zhang: conceptualization, investigation, and supervision. Pei-Juan Wang: conceptualization, resources, and writing—review and editing.
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animals were granted approval for the experimental protocol by the Animal Ethics Committee of Jiangsu Province Academy of Traditional Chinese Medicine at Nanjing city on June 15, 2022, with registration number AEWC-20220615-214.
Funding
Financial support for this work was provided by the Science and Technology Program for Traditional Chinese Medicine of Zhejiang Province (Grant No. 2023ZL639); Natural Science Foundation of Nanjing University of Chinese Medicine (Grant No. XZR2021003); National Natural Science Foundation of China (Grant No. 82374511); and Wang Peijuan Famous Traditional Chinese Medicine Expert Inheritance Studio Project of Jiangsu Province (Grant No. 20SG2S001).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
