Abstract
Objectives
Tang Du Qing granules, a traditional Chinese medicine for diabetes, may alleviate insulin resistance induced by persistent organic pollutant exposure. This study aims to explore the mechanism by which Tang Du Qing granules improve insulin resistance in rats exposed to PCB126.
Methods
Male ZDF (fa/fa) rats were divided into a PCB126-exposed group and a model control group. Male ZDF (fa/+) rats served as the normal control group. Normal controls received a regular diet with olive oil via gavage, while the model control group received special feed with olive oil. PCB126 group received special feed with PCB126 in olive oil for 8 weeks. Post-exposure, the positive drug treatment group received CH223191, and Tang Du Qing granules were administered at high, medium, and low doses for 4 weeks. Relevant biochemical indicators and molecular signaling changes were assessed.
Results
PCB126 exposure increased levels of FBG, FINS, HOMA-IR and elevated serum TG, TC, and FFA in ZDF (fa/fa) rats compared to the model control group (
Conclusion
Tang Du Qing granules ameliorate glucose and lipid toxicity and insulin resistance induced by PCB126 exposure in ZDF (fa/fa) rats by modulating the imbalance between AhR and PPARs and related molecular signals.
Introduction
Since the 1970s, although interventions have been implemented globally to regulate the production and use of persistent organic pollutants (POPs), their harmful effects persist due to their high lipophilicity and bio accumulative nature. In China, pollution remains a significant concern, with regions such as the Yangtze River Delta and the Pearl River Delta exhibiting significantly higher levels of POPs than foreign countries, including the United States, as indicated by risk thresholds.1-4 In the past six years, the human body burden of POPs in Hebei Province has increased by 25%. Meanwhile, over the past two decades, there has been a rapid increase in the prevalence of insulin resistance (IR) and diabetes patients in China. 5
POPs, a class of environmentally stable, difficult-to-degrade, highly lipophilic, and bio accumulative synthetic substances, not only contribute to environmental pollution but are also ingested through diet. 6 Their role as significant factors in the development of insulin resistance and diabetes has been well established. The previous research has indicated an association between POPs, including polychlorinated biphenyls (PCBs), and the occurrence of type 2 diabetes and insulin resistance. Numerous epidemiological studies have also demonstrated a link between persistent organic pollutants and type 2 diabetes resulted in metabolic syndrome.7,8 Additionally, some experimental studies have provided further evidence supporting the notion that exposure to POPs is associated with type 2 diabetes or insulin resistance. 9
Insulin resistance and abnormal lipid levels are key components of metabolic syndrome. Metabolic syndrome encompasses a group of risk factors including hypertension, abnormal blood sugar levels, increased waist circumference, and abnormal lipid levels, all of which collectively heighten the risk of cardiovascular diseases and type 2 diabetes.10-12 Exposure to POPs increases the toxic burden in the body, leading to insulin resistance and consequently raising the risk of type 2 diabetes.9,13 Undertaking in-depth research on the role of POPs in causing insulin resistance is crucial, and exploring the significance of pharmaceutical interventions for insulin resistance holds great importance.
POPs primarily accumulate in human adipose tissue and liver, where they exert their effects by specifically activating the Aryl hydrocarbon receptor (AhR). AhR, a ligand-activated transcription factor, is highly expressed in the liver and to a lesser extent in adipose tissue. 14 The mechanism of POPs in relation to the development of insulin resistance is not entirely clear. Studies suggest that AhR independently affects glucose and lipid metabolism and can trigger inflammatory responses, leading to insulin resistance. 15 POPs disrupt glucose metabolic balance and interfere with glucose utilization and insulin signaling by activating AhR. They also impact the maturation and differentiation of fat cells and the metabolism of fatty acids.16,17
Additionally, the peroxisome proliferator-activated receptors (PPARs) also play a crucial role in maintaining energy balance and suppressing inflammation, regulating glucose and lipid metabolism, and maintaining insulin sensitivity. 18 The combined effect of AhR and PPARs under the exposure to POPs can lead to insulin resistance. Exposure to PCB126 in mice would activate AhR in the liver while decrease the PPARα expression, resulting in fatty liver and disturbed glucose metabolism. 19 AhR may also interfere with PPARs’ functions, affecting downstream targets in glucose and lipid metabolism and inflammatory responses. 15 PPARs can activate the glucose transporter type 4 (GLUT4), regulate the cluster of differentiation 36 (CD36) expression, affecting fatty acid uptake, while the fibroblast growth factor 21 (FGF21) and the tumour necrosis factor α (TNFα), among other inflammatory factors, are regulated by PPARs. 20 Thus, AhR and PPARs play significant roles in insulin resistance induced by POPs.
During our preliminary research, we discovered that Tangduqing granules (TDQ), the in-hospital clinical formulation, could regulate glucose and lipid metabolism and improve insulin resistance by modulating the expression of PPARγ. 21 In this study, we aimed to utilize pharmacological and molecular biology research methods to establish an insulin resistant animal model exposed to organic pollutants through the PCB induction. We would conduct preliminary pharmacological experiments to investigate the efficacy of TDQ in mitigating insulin resistance caused by POPs. Simultaneously, we explored its potential molecular signaling mechanisms, further elucidating the underlying mechanisms of the anti-insulin resistance effects of TDQ.
Materials and Methods
Experimental Drug
Tangduqing granules (Jin Pharm. Z20190058) was purchased from the First Affiliated Hospital of Tianjin University of Traditional Chinese Medicine. This has been reported from our previous research. 21
Experimental Environment
Temperature: 20–25 °C; Relative Humidity: (45 ± 5)%; Noise: not exceeding 85 decibels; Maintain ventilation and air exchange. The animal room should have a light/dark cycle every 12 h (setting the daily light period from 7:00 to 19:00). Throughout the experimental period, rats should have free access to food and water, ensuring that food and water are sufficient. Bedding in the cages should be changed daily, and drinking utensils should be cleaned and disinfected regularly to keep the cages dry and tidy.
Animals and Treatment
The SPF-grade male ZDF (fa/fa) rats were selected as the model group (MG), aged 8 weeks, with a weight range of (180-200) g. The SPF-grade male ZDF (fa/+) rats were chosen as the normal control group (NG), also aged 8 weeks, with a weight range of (140-160) g. Both strains were purchased from Beijing Vital River Laboratory Animal Technology Company Limited, with license number SCXK 2016–0006. The rats were fed with PMI Lab diet 5008, consisting of crude protein (24%), crude fat (7.1%), crude fiber (3.2%), nitrogen-free extract (48.9%), minerals (organic) (6.7%), and vitamins (3.2%), purchased from Hongli (Shanghai) Biotech Company Limited.
According to the results of our previous experiments, seventy male ZDF (fa/fa) rats and ten male ZDF (fa/+) rats (used as the normal control group) were fed a standard diet for approximately one week. Subsequently, the seventy male ZDF (fa/fa) rats were randomly divided into a model group (10 rats) and a poisoned group (60 rats) using a random drawing method. The normal group received a regular diet with an equivalent volume of olive oil administered by gavage. The model group received a specialized diet (PMI Lab diet 5008) with an equivalent volume of olive oil administered by gavage. The poisoned group received the same specialized diet with PCB126 (250 ng/kg) dissolved in an equivalent volume of olive oil administered by gavage. Gavage was performed once daily for a total of 8 weeks. The high, medium, and low doses of TDQ were determined based on our previous cellular and animal experiments, with doses set at 6.0, 3.0, and 1.5 g/kg & day, respectively. 21 The experimental grouping and dosing information could be referred to Figure 1 and Table 1.

Schematic diagram showing the drug treatment in ZDF mice.
the Dosage, Frequency, and Total Duration Schedule for Experimental Drug Administration.
Sample Collection and Biochemical Index
Blood samples for biochemical marker testing in mice were drawn from the inner canthus of the eye, with each sample amounting to approximately 1.5 mL. Each biochemical marker test required 100–200 μL of blood, with all tests conducted according to the reagent kit instructions.
Fasting Blood sugar was tested by the blood glucose meter systems (Roche, Swiss). The concentrations of fasting serum insulin (FINS) in the serum were determined with radioimmunoassay by using kits from Beijing Sino-UK institute of Biological Technology (Beijing, China). The homeostasis model of assessment for insulin resistance was computed as follows: HOMA-IR = [fasting glucose (mmol/L) × fasting insulin (μU/mL)]/22.5. The homeostasis model of assessment for insulin sensitivity was computed as follows: HOMA-I = Ln [1/(FBG × FINS)]. 22
Free fatty acids (FFA) in the serum were measured by using an ELISA kit according to the manufacturer's instructions (Jiancheng Corporation, Nanjing, China). Leptin and adiponectin levels in the serum were measured by using an ELISA kit according to the manufacturer's instructions (Invitrogen Corporation, Carlsbad, CA, USA). Concentrations of triglyceride (TG) and total cholesterol (TC) in the serum and TG in the liver were determined using colorimetric kits (BioSino Bio-Technology & Science Inc. and Beijing Sino-UK Institute of Biological Technology) with a spectrophotometer, according to the manufacturer's instructions.
Hepatic Histology Analysis
After fixation with 4% paraformaldehyde, liver tissues underwent routine dehydration, transparency, and paraffin embedding. Tissues were sectioned into 5 μm thick slices, followed by standard dewaxing and hematoxylin and eosin (H&E) staining. The morphology of liver tissues from each group was observed under a Leica microscope.
Real-Time PCR
Liver tissues were harvested from rats, followed by grinding and total RNA extraction. The reverse transcription enzyme was employed to synthesize cDNA, serving as the template for PCR amplification using Taq polymerase. A standard curve was established, and RT-PCR was utilized to obtain Ct values for the detection of gene expression, including AhR, PPARγ, PPARα, GLUT4, FGF21, CD36, and TNFα. Primer sequences were designed using Primer Premier 5.0 based on the mRNA sequences of rat genes retrieved from the NCBI GenBank. The relevant primer sequences were provided in Table 2.
Primer Sequence for Real-Time PCR.

Western Blotting
The antibodys, such as AhR (ER62618, Huaan Biotechnology, China). PPARα (ab24509, Abcam, USA), PPARγ (ab272718, Abcam, USA), CD36 (ab252923, Abcam, USA), GLUT4 (ab33780, Abcam, USA) FGF21 (ab171941, Abcam, USA), TNFα (ab66579, Abcam, USA), were used for Western blot are performed according to the manufacturer's instructions. The main procedure is as follows:
Total proteins were extracted from each group of rat liver tissues. Protein concentrations were determined using the BCA method. Proteins were separated by SDS-PAGE gel electrophoresis, transferred onto the PVDF membrane using a transfer apparatus. After blocking with 5% skimmed milk powder for 2 h. Then, they were respectively added diluted rabbit polyclonal antibodies against β-actin (1:1000), AhR (1:1000), CD36 (1:1000), PPARα (1:1000), PPARγ (1:1000), FGF-21 (1:1000), GLUT4 (1:1000), and TNFα (1:1000) in TBST. The membranes were incubated overnight at 4 °C. Subsequently, the membranes were incubated with corresponding secondary antibodies (1:10 000) for 2 h and then exposed to the developing solution. The target bands were exposed using an ultra-sensitive multifunctional imaging system, and the grayscale values of each target protein/β-actin were analyzed using Image J software.
Statistical Analysis
All statistical analyses were performed using Prism 9 (GraphPad). Data are expressed as mean ± standard deviation, and comparisons of measures were made using One-Way ANOVA. A value of
Results
The Changes in various Biochemical Indicators in Rats Before and After Administration
Prior to administration, this suggested that, compared to the normal group, the model group showed significant increases in the FBG, FINS, HOMA-IR, FFA, TG and TC (

TDQ mitigated the insulin resistance of type 2 diabetic rats resulted from the PCB126-exposed. A. The effects of TDQ on Fasting Blood Glucose in each group (n = 10). B. The effects of TDQ on fasting serum insulin in each group (n = 10). C. The effects of TDQ on homeostasis model assessment-insulin resistance in each group (n = 10). D. The effects of TDQ on free fatty acids in each group (n = 10). E. The effects of TDQ on triglyceride in each group (n = 10). F. The effects of TDQ on total cholesterol in each group (n = 10).
Meanwhile, after treated with drugs, when compared to the poisoned group, the group treated with TDQ and the positive poisoned group showed a significant decrease in the above-mentioned indicators (
The Histopathological Morphological Changes in Liver Tissues of Rats After Administration in Each Group
In the normal group, hepatocytes exhibited uniform size with no cellular degeneration. The cytoplasm appeared red, and the cell nucleus was centrally located, staining blue. The hepatic lobule structure was clear, with a radial arrangement of hepatic cords extending from the central vein towards the periphery. The liver cords were neatly arranged, and there was no proliferation of fibrous tissue (Figure 3A). In comparison to the normal group, both the model and poisoned groups displayed diffuse hepatocyte swelling, unclear cell contours, and reduced cytoplasm (Figure 3B and C). Additionally, hepatocyte nuclei exhibited lightening or disappearance, or were compressed to one side. The hepatic lobule lost its radial arrangement, and there was loose connection between hepatocytes. The hepatic sinusoids were enlarged, and numerous fat vacuoles were visible within the cytoplasm of hepatocytes. Severe hepatocellular steatosis was evident, accompanied by scattered infiltration of inflammatory cells (black arrow in Figure 3C). The poisoned group exhibits more severe pathological changes.
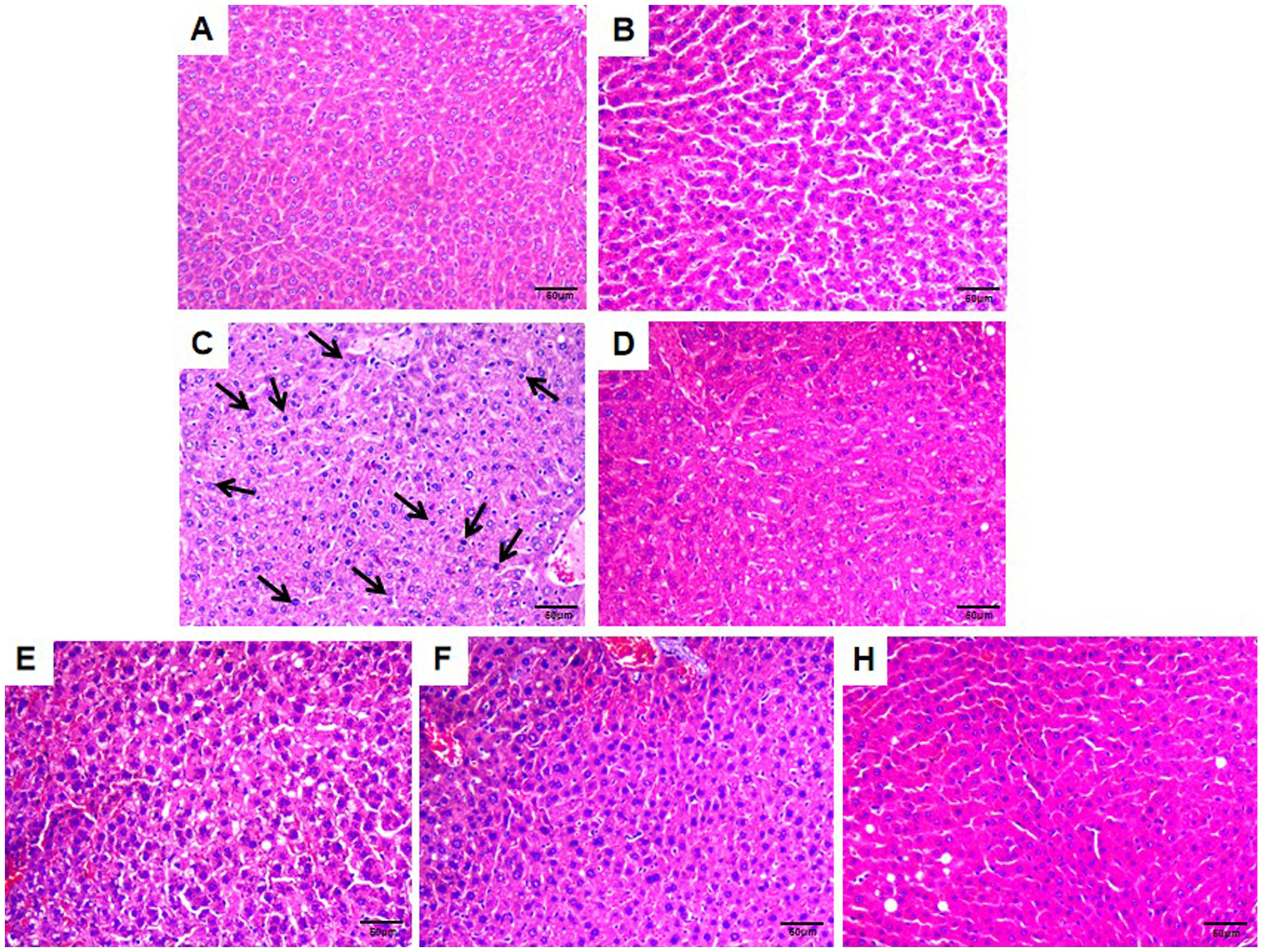
TDQ improved the pathological changes in rat liver cells induced by the PCB126 exposure. The conventional H&E staining was performed (magnification: 400x). A. Normal group. B. Model group. C. Poisoned group. D. Positive poisoned group. E. The low dose of Tang Du Qing granules treated group. F. The medium dose of Tang Du Qing granules treated group. G. The high dose of Tang Du Qing granules treated group.
After administration, compared to the poisoned group, the pathological features in the liver tissues of the drug-treated group rats, including cellular swelling, fat vacuoles, and inflammation, showed significant improvement. Moreover, the effects were more pronounced in the medium and high-dose TDQ groups as well as the positive drug group (Figure 3D–G).
The mRNA Expression of key Genes in Each Group of Rats After Administration
According to the results of the RT-PCR (Figure 4), compared to the normal group, the expression levels of AhR mRNA, CD36 mRNA, and TNFα mRNA in the liver tissues of the model group rats were significantly higher, while the expression levels of the PPARα mRNA, PPARγ mRNA, GLUT4 mRNA, and FGF21 mRNA were significantly lower, with all differences being statistically significant (

The signaling pathways mediated through AhR and PPARs play crucial roles in high-fat diet and the PCB126-exposed insulin resistance and abnormal glycolipid metabolism diabetic. The quantitative real-time PCR was performed. A. The qRT-PCR analysis of mRNA levels of AhR (n = 5). B. The qRT-PCR analysis of mRNA levels of PPARα (n = 5). C. The qRT-PCR analysis of mRNA levels of PPARγ (n = 5). D. The qRT-PCR analysis of mRNA levels of CD36 (n = 5). E. The qRT-PCR analysis of mRNA levels of GLUT4 (n = 5). F. The qRT-PCR analysis of mRNA levels of FGF21 (n = 5). G The qRT-PCR analysis of mRNA levels of TNFα (n = 5).
Remarkably, following treatment with TDQ, compared to the poisoned group, the expression levels of these seven genes exhibited a trend of reverse changes. Furthermore, with an increase in the dosage of TDQ, a certain dose-response relationship was observed. In the high-dose group, the expression levels of various genes were comparable to the levels influenced by the positive drug group.
The Protein Expression of Key Genes in Each Group of Rats After Administration
The results of the western blot experiment showed that, compared to the normal group, the protein expression levels of AhR, CD36, and TNFα in the liver tissues of the model group rats were significantly higher, while the protein expression levels of PPARα, PPARγ, GLUT4, and FGF21 were significantly lower, all exhibiting statistical significance (

The signaling pathways mediated through AhR and PPARs play crucial roles in high-fat diet and the PCB126-exposed insulin resistance and abnormal glycolipid metabolism diabetic. The Western Blot was performed. A. Western blotting showing differential gene protein expression in liver. B. The quantitative analysis of AhR protein expression relative to β-actin (n = 3). C. The quantitative analysis of PPARα protein expression relative to β-actin (n = 3). D. The quantitative analysis of PPARγ protein expression relative to β-actin (n = 3). E. The quantitative analysis of CD36 protein expression relative to β-actin (n = 3). F. The quantitative analysis of GLUT4 protein expression relative to β-actin (n = 3). G The quantitative analysis of TNFαprotein expression relative to β-actin (n = 3). H. The quantitative analysis of FGF21protein expression relative to β-actin (n = 3).
Subsequently, following treatment with TDQ, compared to the poisoned group, there was a trend of reverse changes in the protein levels of various genes in the medium and high-dose groups as well as the positive drug group. However, the changes in protein levels of various genes in the low-dose group of TDQ was not as evident. It is noteworthy that, with an increase in the dosage of TDQ, the certain dose-response relationship was also observed in the protein levels of various genes. Furthermore, in the high-dose TDQ group, the protein levels of various genes were essentially consistent with the levels influenced by the positive drug group.
Discussion
The PCBs are a class of persistent organic pollutants that are also classified as endocrine-disrupting chemicals (EDCs) and metabolic-disrupting chemicals (MDCs). EDCs interfere with normal hormone functions, while MDCs can cause metabolic abnormalities, leading to obesity, diabetes, fatty liver, or metabolic syndrome. 23 Based on structural characteristics and toxicological mechanisms, PCBs can be divided into two categories: non-dioxin-like (NDL) PCBs and dioxin-like (DL) PCBs. DL PCBs have a coplanar structure that allows them to bind to and activate AhR, with PCB126 being the most toxic and environmentally relevant DL PCBs. 24 Studies have shown that PCB126 may account for 26% of AhR activation effect from environmental pollutants. 25 In contrast, NDL PCBs, due to their non-coplanar structure, cannot activate AhR. The toxicological mechanisms of NDL PCBs are mainly attributed to their interactions with various cellular receptors, including nuclear receptors such as the pregnane X receptor and the constitutive androstane receptor. Although DL PCBs may also interact with these receptors, the biological significance of these interactions remains unclear, as the concentration of DL PCBs in the human body is much lower than that of NDL PCBs.
AhR, also known as the dioxin receptor, is a ligand-activated transcription factor primarily expressed in the liver and adipose tissue. PCB126 may specifically activate AhR, influencing glucose and lipid metabolism, triggering inflammation, and consequently leading to insulin resistance.26-28 PPARs, which play a central role in maintaining energy balance and inhibiting inflammation, are crucial in regulating factors that maintain metabolic balance, insulin sensitivity, and the suppression of inflammation. The subtypes of particular interest in current research are PPARα (mainly expressed in the liver) and PPARγ (mainly expressed in adipose tissue, also in the liver).29,30 Both AhR and PPARs are predominantly expressed in the liver and adipose tissues. 31 Therefore, under PCB126 exposure, it is likely that AhR and PPARs jointly regulate glucose and lipid metabolism, as well as inflammatory responses, leading to insulin resistance.
While AhR plays a significant role in glycogenolysis, glycogenesis, and fatty acid oxidation (ie, energy production and balance regulation), studies have shown that different concentrations of PCB126 induce distinct physiological responses. High doses of PCB126 can lead to acute toxicity in animals. For instance, Shi HX et al 32 reported that feeding male C57BL/6J mice with a diet containing PCB126 (20 μg/kg) for two weeks significantly impacted hepatic lipid metabolism, resulting in increased hepatic lipid input, accompanied by hypolipidemia, and ultimately causing hepatic steatosis. Additionally, Eti NA et al 33 found that feeding wild-type male and female rats a diet containing PCB126 (5 µmol/kg body weight; 1.63 mg/kg) in corn oil for 28 days caused significant weight loss in the rats, a reduction in relative thymus weight, and an increase in relative liver weight. However, the results from these high-dose studies differ from our experimental findings. We used a low-dose model (PCB126 in corn oil, 250 ng/kg) that more closely mimics exposure to persistent environmental pollutants, representing a subtoxic dose. Under these low-dose conditions, we observed that ZDF (fa/fa) rats showed short-term weight gain, elevated the FBG, FINS, and HOMA-IR index, impaired glucose tolerance, and increased levels of the TG, TC, and FFA. These metabolic disturbances are likely attributed to the activation of various signaling pathways, including its receptor gene AhR, triggered by different doses of PCB126. This dose-dependent variability highlights the complexity and diversity of PCB126's toxicological effects.
Therefore, it is important to note that the effects of different doses of PCB126 on normal rats and diabetic rats are not consistent. In this section, our discussion will focus on the effects of high doses of PCB126 on diabetic rats and how TDQ exerts its regulatory mechanisms. In our study, when the rat exposed to PCB126, the activation of AhR and decreased expression of the PPARα are observed in the liver, leading to hepatic steatosis and disrupted glucose metabolism. 33 CD36, a highly glycosylated single-chain transmembrane protein, belonging to the class B scavenger receptor family, has a unique neck-loop structure. CD36 has been implicated in lipid metabolism disruption and the occurrence of inflammation.34,35 The activation of AhR by POPs increases the peripheral FFA, inhibits the hepatic fatty acid oxidation, induces CD36 expression, leading to excessive fatty acid intake and triglyceride storage in the liver. 36 TNFα is a central mediator in insulin resistance occurring in obesity-related type 2 diabetes. POPs binding to the aromatic hydrocarbon receptor AhR upregulates TNFα expression, contributing to the onset of insulin resistance and type 2 diabetes. 37 The GLUT4 is a key regulatory protein for glucose uptake from the periphery, playing a crucial role in maintaining glucose homeostasis. 38 It has been shown that increasing the expression and translocation of GLUT4 in tissues can alleviate symptoms of insulin resistance in type 2 diabetes, ultimately achieving preventive and therapeutic effects. Therefore, the development of new drugs targeting GLUT4 has become a hotspot in drug research.39-41 AhR disruption disturbs the balance of glucose and lipid metabolism, inhibits the expression and activity of GLUT4 associated with glucose utilization, and induces insulin resistance.42,43 FGF21 is a cytokine family with various biological functions, participating in substance metabolism to maintain the balance of fat and glucose metabolism in the body. 44 Studies indicate that FGF21, functioning as an endocrine factor, is involved in substance metabolism, and under the PPARα regulation, it acts as a stable lipid regulator in the liver, preventing the development of fatty liver.45,46 FGF21, regulated by both the PPARα and AhR, plays a potent role in regulating glucose and lipid metabolism.47,48
POPs selectively activate AhR, affecting several downstream targets involved in glucose and lipid metabolism as well as inflammatory responses, which are also regulated by PPARs. 49 on one hand, PPARs are regulated by AhR, and on the other hand, they have a modulatory effect on the downstream genes regulated by AhR. 50 They can co-activate GLUT4 to improve glucose metabolism and regulate the activation of genes such as CD36 and FGF21, impacting lipid metabolism. 51 In this process, as the peroxisome proliferator responsive element, the inflammatory factor TNFα, acting as a response element for peroxisome proliferation, is also involved in glucose and lipid metabolism under the joint regulation of the PPAR agonists. 52 Consequently, POPs lead to an increase in the levels of the TC, TG, and FFA. The FFA and the TG, in turn, are central substances causing lipotoxicity, which promotes inflammation and insulin resistance. Then the insulin resistance exacerbates lipotoxicity, forming a vicious cycle. Insulin resistance is a key factor causing metabolic disorders, resulting in elevated blood glucose and disrupted lipid metabolism.53-55 It is a crucial factor in triggering lipotoxicity and various complications. Therefore, PCB126 may contribute to insulin resistance through lipotoxicity (Figure 6).

The signaling pathways mediated through AhR and PPARs resulted from the PCB126-exposed may contribute to IR through gluco-lipotoxicity. In the figure, Gluco-lipotoxicity can represent the metabolic toxicity induced by a high-glucose, high-fat diet, as well as the metabolic toxicity exacerbated by organic pollutants such as the PCB126. TDQ can alleviate the effects caused by PCB126 through the AhR-related signaling pathway.
Diabetes is a metabolic disorder characterized by elevated blood glucose levels, with its complex etiology being influenced by multiple genetic and environmental factors.56,57 The intricate pathogenesis of diabetes determines the complexity of its treatment. Currently, diabetes medications primarily developed based on single-target approaches face significant limitations.58-60 Traditional Chinese medicine (TCM) offers a unique therapeutic mechanism for treating diabetes, exhibiting the ability to improve organ and tissue functions through multiple targets, pathways, and stages.61,62 TCM interventions in diabetes provide significant improvements in clinical symptoms and associated complications. 63 The use of TCM for an extended period is associated with minimal side effects, conferring irreplaceable roles and distinct advantages compared to Western medicin. Pharmacological research indicates that the main active components in TCM for treating diabetes include flavonoids, alkaloids, polysaccharides, and saponins.64-66
In our previous studies, we found that TDQ could counteract glucolipotoxicity and improve insulin resistance by regulating the expression of PPARγ. 21 In this study, we intervened with rats exposed to POPs based on this granule. We explored the regulatory effects of TDQ on POPs-exposed rats and their potential mechanisms, further elucidating its target actions. The results indicated that in rats exposed to POPs, the expression levels of AhR in liver tissues were downregulated after treatment with TDQ and the positive drug. Post-treatment, the levels of glucolipid metabolism indicators related to insulin resistance, the inflammatory genes TNFα and CD36 mRNA, as well as protein expression levels, were reduced compared to the poisoned group. The mRNA and protein levels of the PPARα, PPARγ, GLUT4, and FGF21, which played a powerful regulatory role in glucolipid metabolism and insulin resistance, are increased. The insulin resistance index decreases, with significant effects observed in the medium and high-dose TDQ groups as well as the positive poisoned group. This suggested that TDQ had a vital role in improving insulin resistance in POPs-exposed rats. The mechanism may be related to its regulation of AhR/PPARs pathway in liver tissues, affecting the expression of CD36, TNFα, GLUT4, and FGF21. Through bidirectional signal regulation, this ultimately leaded to a balance in blood glucose and lipids.
The positive drug CH223191 was a discovered effective and specific AhR antagonist in vertebrates, capable of efficiently inhibiting PCB126-mediated nuclear translocation and AhR binding to DNA.67,68 Therefore, CH223191 could effectively block the activation of AhR genes induced by PCB126, thereby improving insulin resistance. TDQ demonstrated an equivalent effect and exhibited a certain dose-response relationship, indicating that the granules possessed a role similar to an ‘AhR receptor antagonist’. Thus, TDQ also exhibited a potent detoxifying effect against the ‘toxins of environmental pollution’ entering the body.
Finally, it was important to note that our study utilized only one toxic chemical, PCB126, at a single dose to induce a model in rats. In reality, different types of organic pollutants could lead to varying metabolic mechanisms and cause diverse molecular signaling changes. Even different doses of PCB126 might result in distinct physiological alterations in diabetic rats. From this perspective, future experiments would benefit from extending the administration period, using various organic pollutants, and applying different doses of PCB126 to observe the regulatory effects of TDQ on AhR/PPARs pathway in toxin-exposed rats. This approach could provide significant insights into how TDQ improves insulin resistance in rats exposed to POPs.
Conclusion
In this study, we observed that exposure to PCB126 could induce glucose and lipid toxicity as well as insulin resistance in the type 2 diabetic rats. This potential molecular mechanism might be associated with the dysregulation of AhR and PPARs, influencing the expression of downstream targets related to glucose and lipid metabolism and inflammatory responses.
Supplemental Material
sj-xlsx-1-npx-10.1177_1934578X241310007 - Supplemental material for Effects of Tangduqing Granules on Insulin Resistance and Their Association with AhR and PPARs Regulation in Type 2 Diabetic Rats Exposed to Persistent Organic Pollutants
Supplemental material, sj-xlsx-1-npx-10.1177_1934578X241310007 for Effects of Tangduqing Granules on Insulin Resistance and Their Association with AhR and PPARs Regulation in Type 2 Diabetic Rats Exposed to Persistent Organic Pollutants by Lu Zhang, Shengnan Guo, Yue Gao, Murong Jiang, Qiumei Liu, Wenli Yang, Yao Liu, Rui Qian, Shentao Wu and Hongyi Liu in Natural Product Communications
Supplemental Material
sj-xlsx-2-npx-10.1177_1934578X241310007 - Supplemental material for Effects of Tangduqing Granules on Insulin Resistance and Their Association with AhR and PPARs Regulation in Type 2 Diabetic Rats Exposed to Persistent Organic Pollutants
Supplemental material, sj-xlsx-2-npx-10.1177_1934578X241310007 for Effects of Tangduqing Granules on Insulin Resistance and Their Association with AhR and PPARs Regulation in Type 2 Diabetic Rats Exposed to Persistent Organic Pollutants by Lu Zhang, Shengnan Guo, Yue Gao, Murong Jiang, Qiumei Liu, Wenli Yang, Yao Liu, Rui Qian, Shentao Wu and Hongyi Liu in Natural Product Communications
Supplemental Material
sj-xlsx-3-npx-10.1177_1934578X241310007 - Supplemental material for Effects of Tangduqing Granules on Insulin Resistance and Their Association with AhR and PPARs Regulation in Type 2 Diabetic Rats Exposed to Persistent Organic Pollutants
Supplemental material, sj-xlsx-3-npx-10.1177_1934578X241310007 for Effects of Tangduqing Granules on Insulin Resistance and Their Association with AhR and PPARs Regulation in Type 2 Diabetic Rats Exposed to Persistent Organic Pollutants by Lu Zhang, Shengnan Guo, Yue Gao, Murong Jiang, Qiumei Liu, Wenli Yang, Yao Liu, Rui Qian, Shentao Wu and Hongyi Liu in Natural Product Communications
Footnotes
Abbreviations
Acknowledgments
The authors express their gratitude to the National Natural Science Foundation of China and the Yunnan Science and Technology Department Project for their primary support and assistance in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
We clarified that all animal experimentation procedures were conducted in accordance with the guidelines for the care and use of laboratory animals, and approval was obtained from the Institutional Animal Care and Use Committee (IACUC). The experimental protocols conducted in this article with animals were approved by the Ethical Committee of Yunnan University of Traditional Chinese Medicine (No. DW-2022-019). All the participants provided written informed consent. All methods were carried out in accordance with the relevant guidelines and regulations. All methods are reported in accordance with ARRIVE guidelines.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81760856, 82360919), Yunnan Science and Technology Department Project (2017FD174, 2018FF001(-053), 202001AZ070001-054, 202001AZ070001-060), the Second Level Candidates of 131 Innovative Talents Training Project of Tianjin (2018), The Fifth Batch of National Traditional Chinese Medicine Excellent Clinical Talents Training Project. (Announcement from the Personnel and Education Department of the National Administration of Traditional Chinese Medicine. No. 2022-1).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
