Abstract
Introduction
Atopic dermatitis (AD), a relapsing and chronic inflammatory skin disorder, is featured by recurrent eczematous lesions and intense itch. It affects 3% of infants, 10–20% of children, and 3%–10% of adults and its disease burden ranks first among non-fatal skin diseases. 1 The pathogenesis of AD is complex, and it is reported that a strong genetic predisposition, epidermal dysfunction, microbial imbalance of skin microbiota and immune dysregulation are critical in AD development. 2 Current medications available for AD treatments are topical moisturizers, corticosteroids, calcineurin inhibitors for skin inflammation and systemic immunosuppressants. Nevertheless, these conventional traditional therapies might exhibit limited effectiveness and could be hindered by serious adverse effects. 3 The need is pressing for the creation of innovative treatment approaches for AD that offer superior effectiveness and lower toxicity.
Epigallocatechin gallate (EGCG) is the predominant catechin, constituting over half of the catechin content in both fresh tea leaves and green tea that has not undergone fermentation. It has garnered significant interest due to its various health benefits, including its anti-inflammatory, antioxidant, and angiogenic properties.4,5 Moreover, emerging evidence have shown that EGCG could facilitate the healing of skin wounds and avert skin photoaging triggered by UVB radiation through enhancing skin elasticity and collagen fibers, exhibiting its great potential in preventing AD.6,7 Herein, by virtue of the DNFB-induced AD mouse model, network pharmacology and molecular docking, we firstly assessed the therapeutic effects and potential mechanisms of EGCG against AD. The research methodology is graphically represented in Figure 1.
Schematic overview of the current research methodology.
Materials and Methods
Materials
Epigallocatechin gallate (EGCG, purity >95%) was supplied by Shanghai Aladdin Bio-Chem Technology Co. Ltd, China. DNFB (purity > 99%) was obtained from Shanghai Adamas Reagent Co., Ltd China. The mouse IgE ELISA kit was obtained from Quanzhou Ruixin Biological Technology Co., Ltd, China.
Experimental Procedure and the Establishment of AD Model
Female Kunming (KM) mice, weighing between 18–22 g, were acquired from the Laboratory Animal Center of Wuhan University and they were consistently housed and maintained under controlled environmental conditions (25 ± 2°C, 60 ± 10% humidity, a light/dark cycle of 12 h each). The experimental protocols followed in this research strictly adhered to the guidelines set forth in the “Guide for the Care and Use of Laboratory Animals,” Eighth Edition, 2011, published by the National Academies Press in Washington, USA.
After a week-long period of acclimatization, a total of 24 KM mice were randomly assigned to three groups with 8 mice per group: Normal group, AD group, and 100 mg/kg EGCG group. The induction of AD-like symptoms in the mice was achieved through repeated applications of DNFB, following methods from our previous studies.8,9 Briefly, the DNFB was dissolved in a solution of acetone and olive oil in a 3:1 ratio. Initially, the shaved dorsal area of the mice (3 cm×2 cm) was treated with 100 μL of a 1.0% DNFB solution on days 1 and 3 to sensitize them. Thereafter, twice-weekly applications of 100 μL of a 0.5% DNFB solution were administered for two weeks to elicit AD-like skin conditions. Mice in the various groups were administered either saline or 100 mg/kg EGCG via oral gavage at a dosage volume of 0.1 mL/10 g body weight. Throughout the entire experiment, the body weight, the mortality and food/water consumption of mice were meticulously monitored.
Evaluation of Skin Lesion and Measurement of Dorsal Skin Thickness
To assess the potential therapeutic impact of EGCG on clinical manifestations similar to AD, the severity scores for dermatitis in mice across various groups were evaluated. The dermatitis severity was macroscopically assessed on the 13th day, applying the four criteria previously outlined. In brief, the severity of symptoms was quantified on a scale ranging from 0 (none) to 3 (severe), considering the following clinical presentations: (1) erythema/hemorrhage, (2) edema, (3) scaling/dryness, and (4) erosion/excoriation.
Additionally, the extent of skin swelling was evaluated by determining the thickness of the mouse skin across different experimental groups. Upon completion of the study on day 14, the dorsal skin from each mouse was excised, from which circular samples (8 mm in diameter) were taken using a punch tool, and the thickness was subsequently gauged using a micrometer caliper. skin swelling was further examined by measuring the mouse skin thickness among different groups. Upon completion of the experimental procedures on day 14, the dorsal skin from each mouse was excised and punched (holes with the diameter of 8 mm), and then measured using a micrometer caliper.
Measurement of Visceral Organ Coefficient
On the 14th day, the mice were euthanized and the vital organs including heart, liver, spleen, lungs, kidneys, and thymus were swiftly extracted and accurately weighed using a precision balance. The visceral organ coefficient was given as organ/body ratio (100*g/g).
Histological Analysis
The dorsal skin of each mouse was excised and subsequently fixed in a 4% paraformaldehyde solution, prior to being embedded in paraffin. The paraffin-embedded skin samples were then cut into 4-micron slices and stained with either hematoxylin-eosin (H&E) to assess the thickness of the epidermis or toluidine blue (TB) to visualize the presence of mast cell infiltration. The microscopic examination was conducted using an Olympus microscope from Tokyo, Japan. Quantitative assessment of epidermal thickness and the count of mast cells were performed utilizing the ImageJ software developed by the National Institutes of Health in Maryland, USA.
Measurement of IgE Level in serum
Whole blood was collected from the ocular region, and then it was subjected to centrifugation at a rate of 3500 rpm for 15 min at 4°Cto isolate serum samples. The serum concentrations of immunoglobulin E (IgE), interferon-gamma (IFN-γ), and interleukin-4 (IL-4) were determined using enzyme-linked immunosorbent assay (ELISA) kits, strictly following the guidelines provided by the manufacturers.
Screening of EGCG-Related Targets
The Super-PRED and SwissTargetPrediction databases were utilized to identify potential targets for EGCG, specifying “Homo sapiens” as the species of interest. Subsequently, these candidate targets were consolidated and cross-referenced with the (https://www.uniprot.org/) database to confirm their validity and to standardize their nomenclature to the official abbreviations. Any targets that were duplicated, non-human, or did not adhere to the standard naming conventions were excluded.
Screening of AD-Related Targets and Overlapping Targets
“Atopic Dermatitis” OR “Atopic Eczema” were used as keywords in the DisGeNET (http://www.disgenet.org/), GeneCards (https://www.genecards.org) and OMIM (https://www.omim.org/) databases to search for AD-related targets, with the species set to “Homo sapiens”. The collected target data from these sources were then merged, with any redundant entries being discarded. Then, the AD-associated targets were compared with the EGCG candidate targets through Venn diagram analysis, and the intersecting targets were identified as potential therapeutic targets EGCG against AD.
Protein–Protein Interaction (PPI) Network Analysis and Screening of Hub Genes
The overlapping targets identified for EGCG in the treatment of AD were entered into the STRING 11.0 database to build a PPI network that maps out the interactions between them. A confidence score threshold of 0.7 was applied to the interactions. Following this, the PPI network analysis outcomes were imported into Cytoscape version 3.9.0 to graphically represent the connections between proteins. Cytohubba plug-in was then utilized to determine the degree centrality (DC) of the network, and the top 10 proteins with the highest “Degree” values were identified to form a Hub gene network.
GO Term and KEGG Pathway Enrichment Analysis
In our research, we harnessed the DAVID database and OmicShare cloud platform to conduct Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis for the intersecting targets. For GO functional analysis, the top 5 findings across biological processes (BP), molecular functions (MF), and cellular components (CC) were chosen based on the Q-value, which is the adjusted P-value. Similarly, the top 20 KEGG pathway results were selected using the same criterion and were depicted as bubble charts for visual representation.
Components-Targets-Pathways Network Construction
The “Component-Targets-Pathways” network, illustrating the interconnections among EGCG, AD, potential therapeutic targets, and associated signaling pathways, was constructed using Cytoscape 3.9.0. Within this network, nodes symbolize the components, targets, and pathways, and edges denote the interactions between them.
Molecular Docking
The 3D chemical structure of EGCG was obtained from downloaded from the PubChem (https://pubchem.ncbi.nlm.nih.gov/) database. The x-ray crystal structures of the core targets were obtained from the PDB(http://www.pdb.org/) database. AutoDockTools 1.5.6 software was used to add hydrogen atoms and set docking parameters for EGCG and the target proteins, respectively. The Autodock Vina software was employed to carry out semi-flexible molecular docking to obtain the optimal docking binding energy between the core targets and EGCG. The docking models that exhibited the strongest binding affinity were then depicted using PyMOL for a three-dimensional view and LigPlus for a two-dimensional representation.
Western Blotting (WB)
100 mg of dorsal skin lesions were lyzed in RIPA lysis buffer containing PMSF on ice. Following this, the protein extracts were harvested by centrifugation at 10,000 g at 4 C for 10 min. A 20 μg of total protein was then subjected to 10% SDS-PAGE gels and moved onto polyvinylidene difluoride (PVDF) membranes. After 1 h blocking with 5% BSA at room temperature, the membranes were then incubated with primary antibodies overnight at 4 C: RAGE (1:1000; ab216329), STAT1 (1:5000; 66545-1-Ig) and GADPH (1:50000; 60004-1-Ig). After blotting, the 1 h incubation of membranes was made at room temperature with the secondary antibody. The immune reactive bands were visualized using a Bio-Rad ChemiDoc MP Imaging System and the protein expression was quantified by Image J software.
Statistical Analysis
All data were statistically analyzed using GraphPad Prism 9.0/SPSS 24.0 software with one-way ANOVA followed by Dunnett's post-hoc test. Results are reported as means ± SD, and the differences were considered statistically significant at p < 0.05 or p < 0.01.
Results
Effect of EGCG on AD-Like Symptoms in DNFB-Induced AD Mice
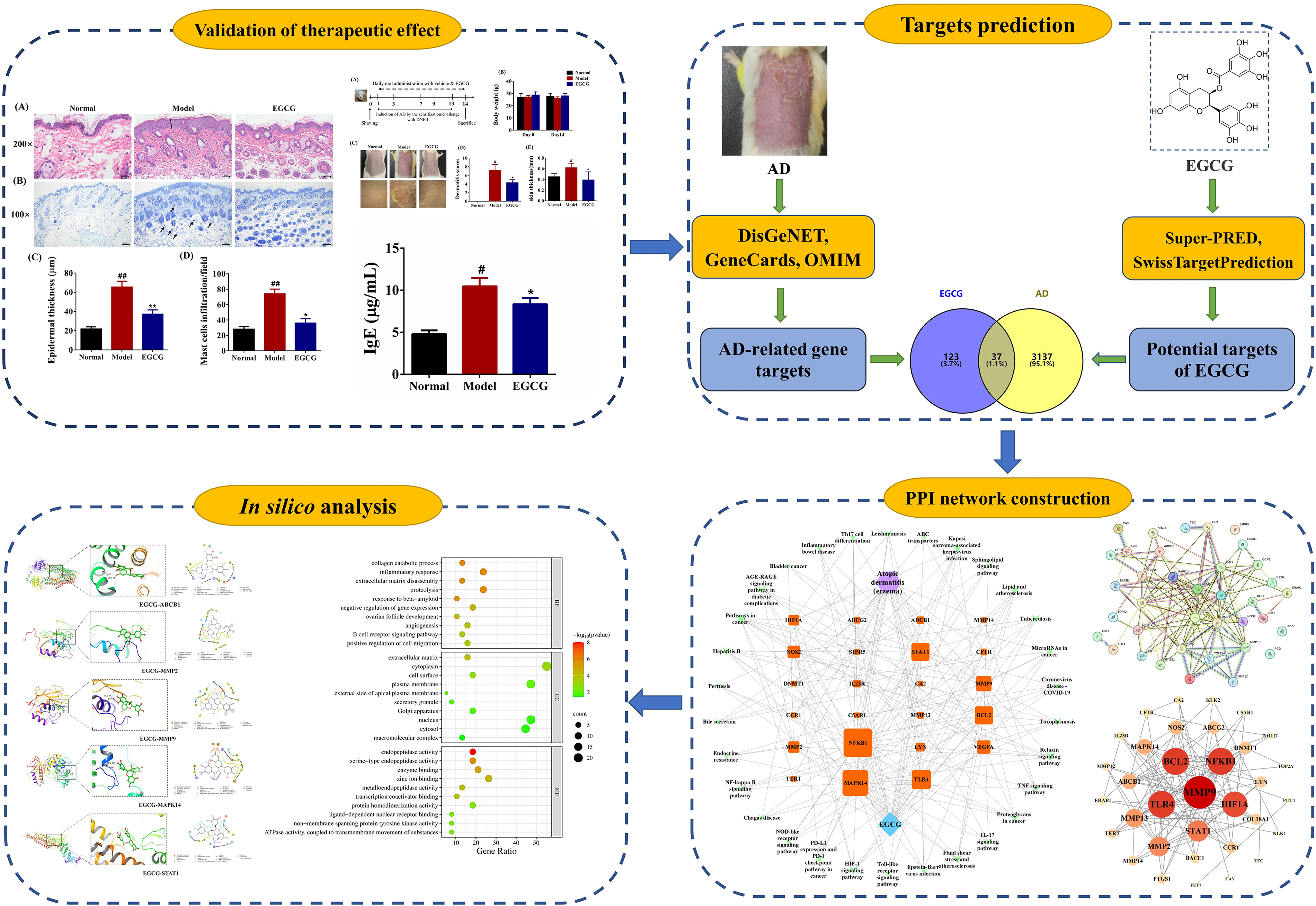
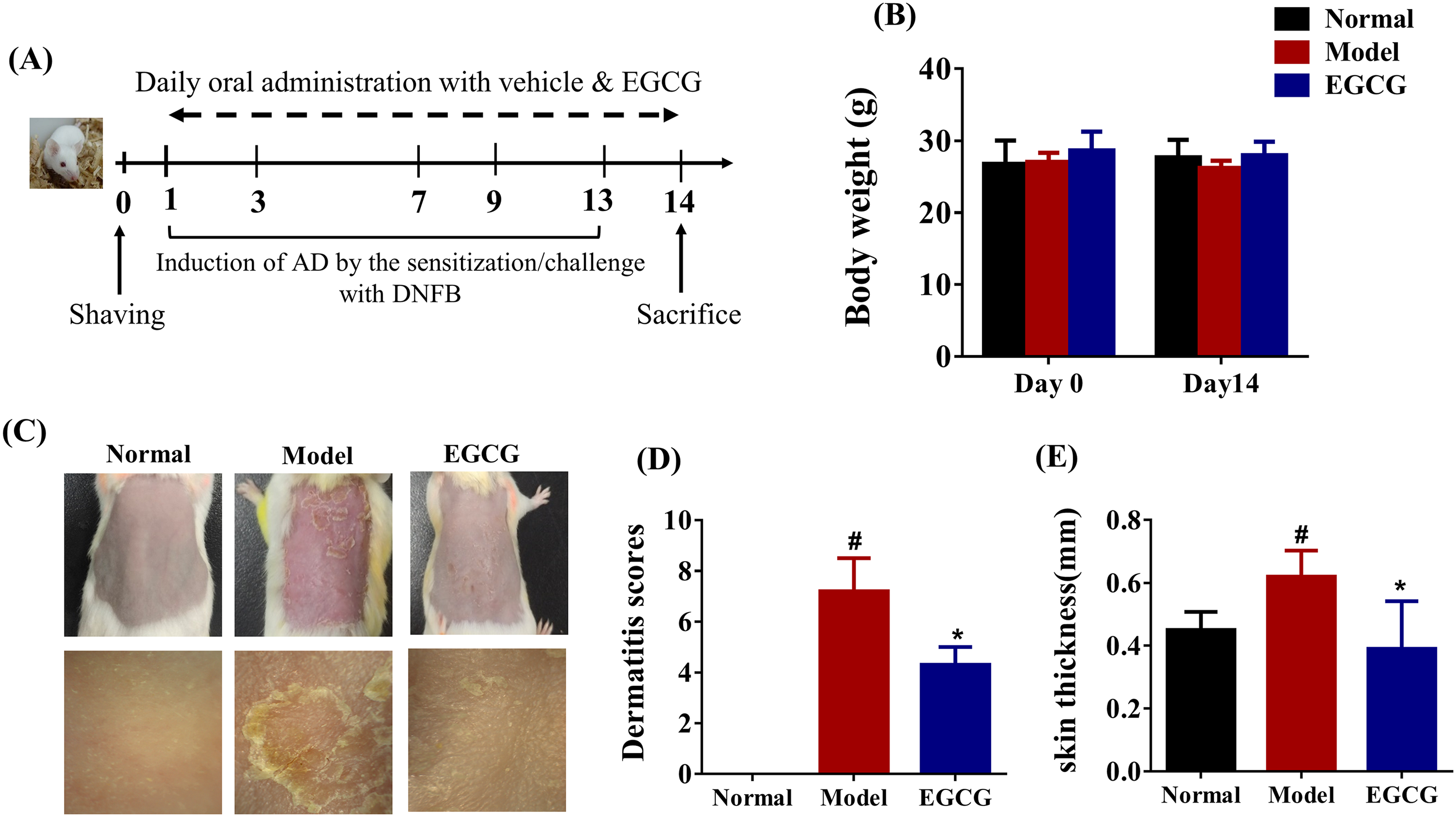
To investigate whether EGCG has the therapeutic potential to alleviate AD, the AD mouse model was induced by repeated exposure to DNFB on the dorsal surface of the skin once daily for 2 weeks (Figure 2A). During the experimental period, the body weight changes were not significantly different between vehicle- and EGCG-treated groups (Figure 2B, p>0.05). Furthermore, the findings from the organ weight assessments revealed no significant variations in the organ indices across the different groups, suggesting that the oral intake of EGCG did not exert any noticeable detrimental impact throughout the duration of the study (Figure 3).
EGCG alleviates AD-like skin lesions in mice. (A) A schematic diagram showing the induction and treatment of AD. (B) Body weights were measured for 2 weeks post oral application of EGCG. (C) Representative pictures of dorsal skin lesions among different groups on Day 14. (D) Dermatitis scores were examined among different groups on Day 14. (E) Skin swelling by thickness (mm) was measured on day 14. Data are presented as mean ± SD for each group (n =8). #p < 0.05, ##p < 0.01 in comparison to Normal group; * p < 0.05, ** p < 0.01 versus in comparison to Model group.
Effect of EGCG on visceral organ coefficient of mice (100*g per g body weight). Data are presented as mean ± SD for each group (n =8). #p < 0.05, ##p < 0.01 in comparison to Normal group; * p < 0.05, ** p < 0.01 versus in comparison to Model group.
A macroscopic analysis of the dorsal skin lesions was performed to evaluate the severity of the dermatitis and the representative photographs of mice in different group are shown in Figure 2C. In addition, EGCG markedly alleviated DNFB-triggered AD-like phenotypes quantified by skin thickness and dermatitis severity score (Figure 2D-E, p < 0.05). The application of 100 mg/kg EGCG led to a noticeable reduction in the dermatitis scores as compared to the AD model group (7.20 ± 1.30 vs 4.3 ± 0.71, p < 0.05). Consistently, the skin thickness markedly increased after topical treatment with DNFB (0.62 ± 0.08 mm vs 0.45 ± 0.06 mm, p < 0.05), and this increase was significantly attenuated by 100 mg/kg EGCG (p < 0.05).
Effect of EGCG on Skin Histopathology in DNFB-Induced AD Mice
Histological analysis of mouse skin tissues was performed using H&E staining. As depicted in Figure 4A&C, there was a significant increase in the thickness of the epidermis in the group treated with DNFB compared to the untreated control group (65.40 ± 6.10 μm vs 21.78 ± 2.26 μm, p < 0.01), which was attributed to the presence of edema, hyperkeratosis, and hyperplasia. Additionally, the AD mice induced by DNFB showed a pronounced escalation in the presence of inflammatory cells within the epidermal and dermal strata of the skin. Oral administration of EGCG was found to mitigate these histopathological changes, effectively restoring the skin to a normal state.

Effect of EGCG on skin histopathology in DNFB-triggered AD mice. (A) Typical photographs depicting H&E staining in various groups. (B) Typical photographs showing TB staining across different skin regions in various groups. (C) Measurement of epidermal thickness. (D) Quantification of mast cells. Data are presented as mean ± SD for each group (n = 3). #p < 0.05, ##p < 0.01 in comparison to Normal group; * p < 0.05, ** p < 0.01 versus in comparison to Model group.
Previous research indicates that the promotion of mast cells in skin lesions contribute to skin inflammation in the AD animal models and patients.10,11 In this study, TB staining was employed to evaluate the infiltration of mast cells in the dermis (Figure 4B&D). By TB staining, the number of mast cells were remarkedly elevated to (74.00 ± 6.13) cells/field in the inflamed skin lesions, compared with those of (28.00 ± 3.94) cells/field in the normal skin (p < 0.01). However, the elevation was significantly reduced in the EGCG-treated group (p < 0.05).
Effect of EGCG on Serum IgE Level
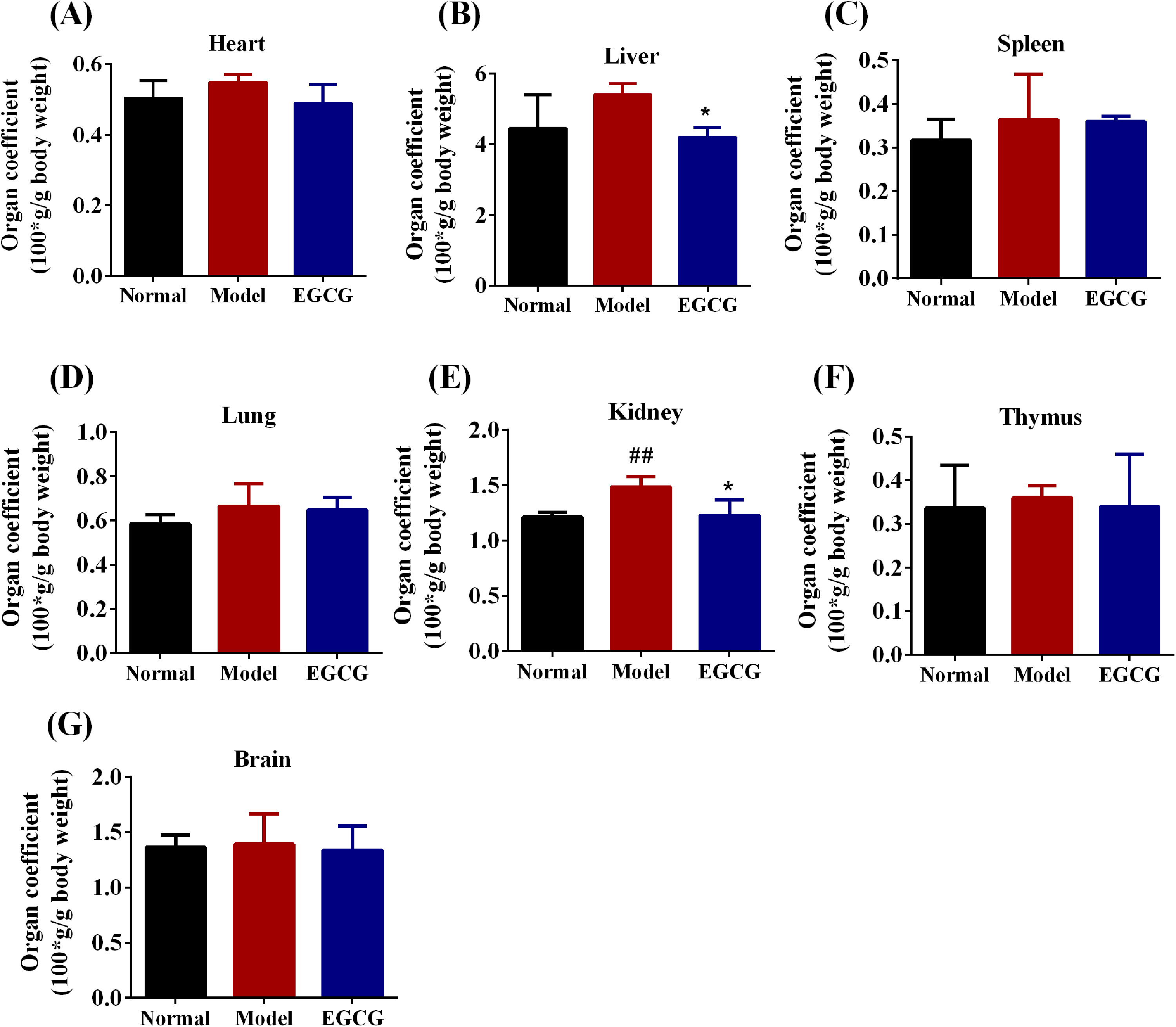
An elevated IgE level is the important characteristic of AD. 12 In our research, the serum IgE level in the DNFB-treated model group was prominently elevated compared to the normal group (10.45 ± 0.97 μg·mL−1 vs4.80 ± 0.42 μg·mL−1, p < 0.05). However, EGCG markedly inhibited the increased IgE level to (8.33 ± 0.65) μg·mL−1 (Figure 5, p < 0.05).
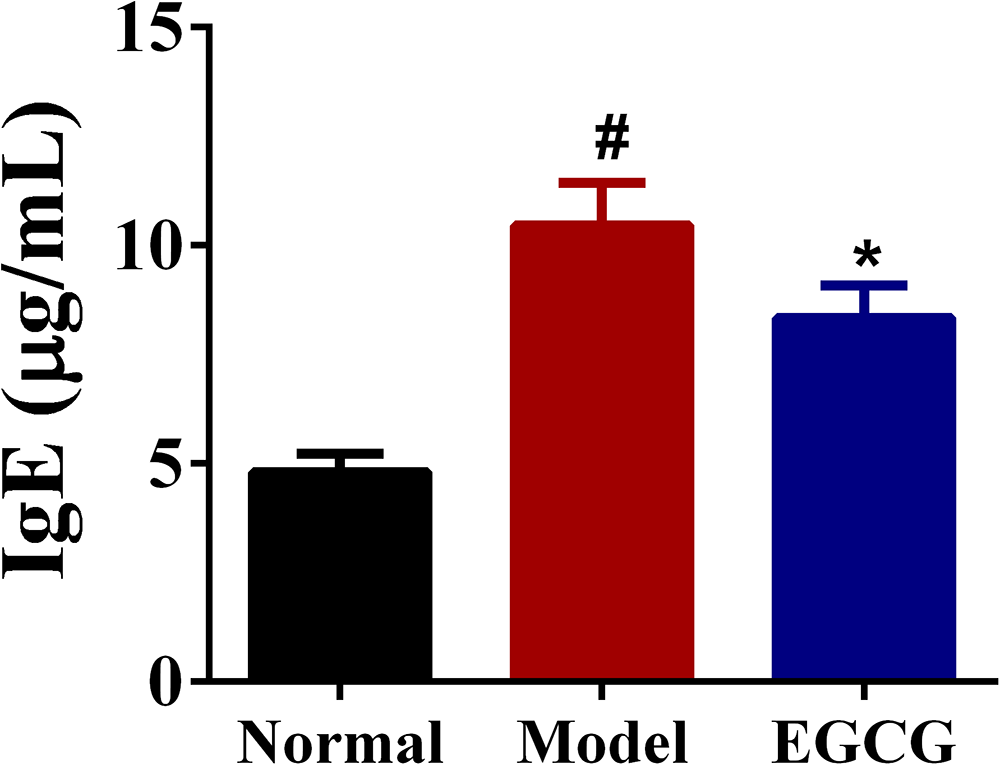
Effects of EGCG on serum IgE production in DNFB-induced AD mice. Data are presented as mean ± SD for each group (n = 6). #p < 0.05, ##p < 0.01 in comparison to Normal group; * p < 0.05, ** p < 0.01 versus in comparison to Model group.
Acquisition of the Targets Related to EGCG Against AD
160 candidate targets for EGCG were obtained after conducting a thorough search on SwissTargetPrediction and SuperPred databases and eliminating any duplicates (as illustrated in Figure 6). By accessing the DisGeNET, GeneCards, and OMIM databases, we compiled a list of 3174 targets associated with AD. Subsequently, 37 common targets between EGCG and AD were acquired by employing the Venny 2.1.0 tool.
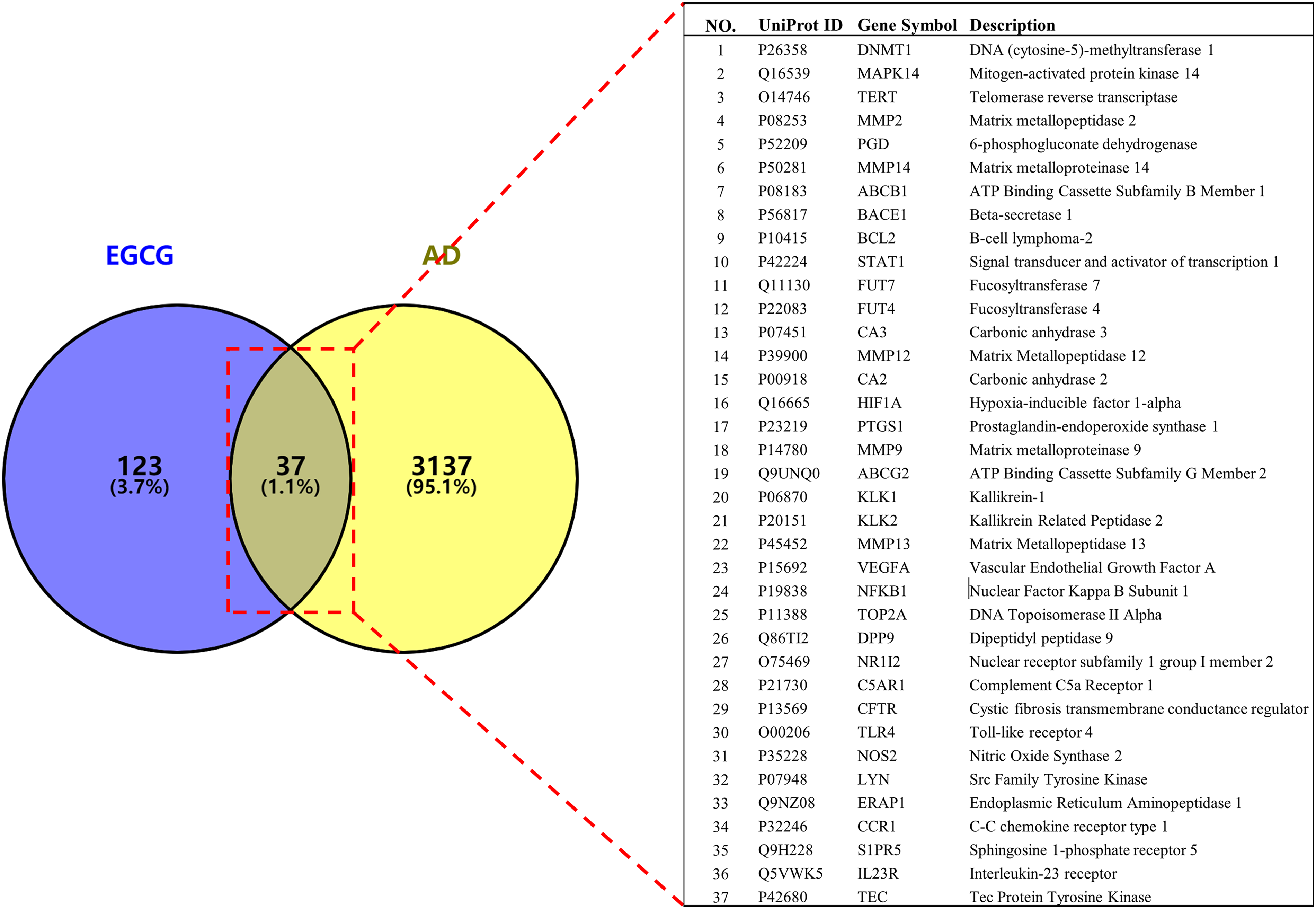
The overlapping targets of EGCG and AD identified by Venny 2.1.
PPI Network Construction and Topology Analysis
37 overlapping targets were analyzed within the STRING 11.0 platform to map out the protein-protein interaction (PPI) network, as depicted in Figure 7A. For the visualization of this network, Cytoscape version 3.9.0 was employed. The PPI network provided a comprehensive overview of the interconnectedness among the identified targets, comprising 34 nodes and 120 edges. In this network diagram, the circles correspond to the targets of EGCG against AD, while the edges signify the interactions among these targets. Top 10 nodes exhibiting the highest “Degree” values were further selected to construct a Hub gene network. This hub network is composed of 10 key proteins, including MMP9、NFKB1、BCL2、TLR4、HIF1A、STAT1、MMP2、MMP13、ABCB1、MAPK14 (Figure 7B&C), which are pivotal in the PPI network and may play significant roles in the mechanisms of EGCG's effects against AD.

Identification of key targets for EGCG against AD from a Protein–protein interaction (PPI) network. (A) PPI network of 37 common targets. (B) Top 10 hub genes identified from the application of CytoHubba within the Cytoscape 3.9.0 software.
Molecular Docking Between EGCG and Key Target Proteins
Molecular docking, a pivotal bioinformatics technique, is frequently utilized in the preliminary phases of drug development. 13 In this context, EGCG was subjected to molecular docking with 10 key targets to validate the interactions between the compound and these targets. The docking results are generally interpreted with binding energies, where values below −5.0 kcal/mol suggest a robust binding affinity between the ligand and the receptor.14,15 The results of the molecular docking between the Top 10 core targets and EGCG are detailed in Table 1. Notably, ABCB1, MMP2, MMP9, MAPK14, and STAT1 demonstrated enhanced binding affinities, with binding energies recorded at −8.85, −8.39, −8.11, −7.93, and −7.56 kcal/mol, respectively. The quintet of target proteins and active molecules, characterized by their lower binding energies, were chosen for the generation of a docking model diagram (Figure 8). Among which, there exists a hydrogen bonding interaction between EGCG and SER831 of ABCB1, and GLU402 of MMP9, and LEU289 of MAPK14, respectively.
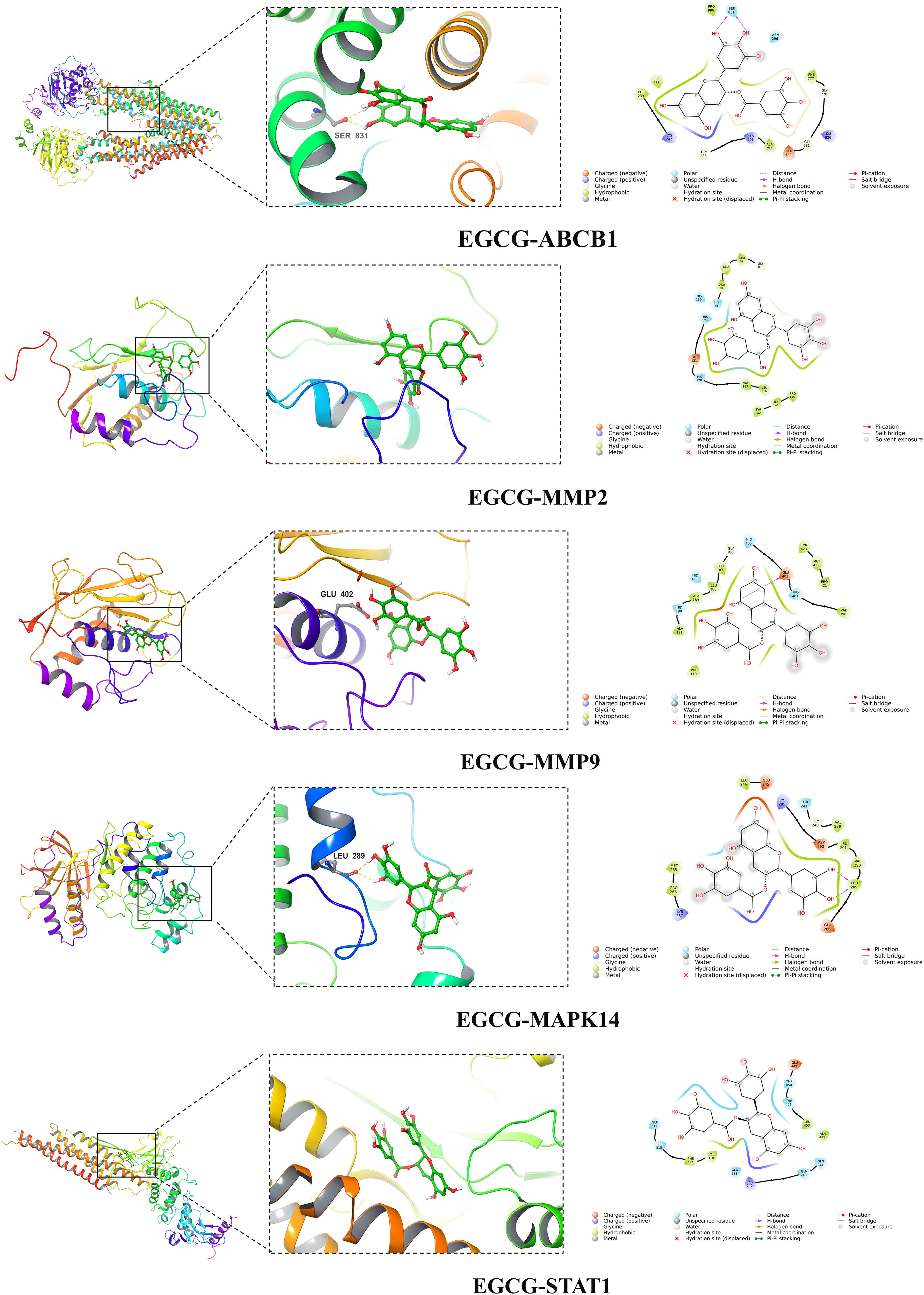
Three-dimensional (3D) and 2D maps of EGCG docking with the principal targets involved in the treatment of AD, including ABCB1, MMP2, MMP9, MAPK14, and STAT1.
The Results of the Molecular Docking Focusing on the Binding Affinity of EGCG to 10 Hub Target Proteins.

Gene Ontology (GO) Functional Analysis
37 overlapping targets were input into the DAVID database for GO functional analysis and the top 20 biological process (BP) enrichment items included collagen catabolic process, inflammatory response, extracellular matrix disassembly, proteolysis, angiogenesis, B cell receptor signaling pathway and positive regulation of cell migration as depicted in Figure 9. Cellular component (CC) enrichments involve extracellular matrix, cytoplasm, cell surface, plasma membrane and nucleus. Molecular function (MF) enrichments involve endopeptidase activity, enzyme binding, transcription coactivator binding and ligand-dependent nuclear receptor binding.

Graphical bubble chart depicting the GO enrichment analysis (top 10).
The Construction of EGCG-Target-Pathway Network
The KEGG pathway enrichment analysis uncovered that the 37 overlapping targets were predominantly involved in 30 distinct signaling pathways with statistical significance (P < 0.01). To delve deeper into the potential mechanisms by which EGCG could act as a preventative agent against AD, the top 30 pathways ranked by their P values, were graphically represented using the OmicShare tool. As depicted in Figure 10 and outlined in Table 2, the key pathways that may be influenced by EGCG in its anti-AD effects include the AGE-RAGE signaling pathway in diabetic complications, the Th17 cell differentiation, the IL-17 signaling pathway, the NF-κB signaling pathway, the Toll-like receptor signaling pathway, and the TNF signaling pathway, among others.
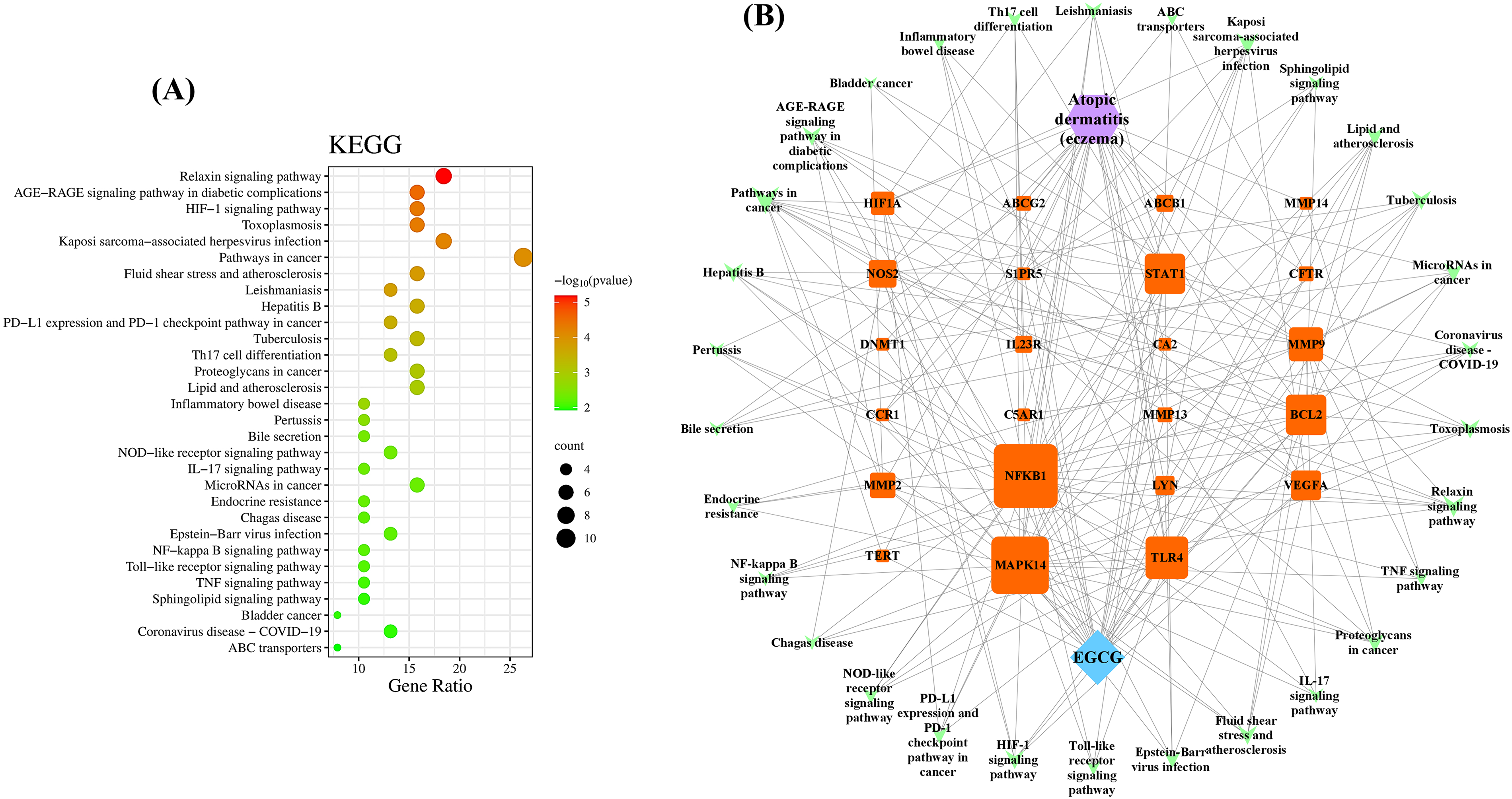
Bubble chart illustrating the KEGG pathway enrichment (Top 10). (A) In the KEGG enrichment analysis, the size of each bubble is proportional to the number of genes enriched within a pathway, and the color coding indicates the statistical significance of the enrichment. (B) The “component-target-pathway” network for EGCG against AD is depicted, where the outer green triangles represent the pathways of interest, and the inner orange squares denote the specific targets within these pathways.
Top 30 Significantly Enriched KEGG Pathways (P < 0.01).
EGCG Downregulated AGE-RAGE Signaling Pathway in DNFB-Induced AD-Like Mice
Based on the relative literature EGCG-target-pathway network, we determined the AGE-RAGE signaling pathway, which exhibited the lowest P-value and has been implicated in cell proliferation and inflammation, would be the crucial target for EGCG in the treatment of AD. In vivo, we used WB to detect the expression levels of RAGE (Receptor for AGEs) in the lesional skin. The expression of RAGE was significantly increased after DNFB induction, and decreased after the treatment of EGCG (Figure 11, p < 0.01 or < 0.05). STAT1 is one of the downstream targets in the AGE-RAGE signaling cascades and can be activated by the combination of AGE and RAGE, thus exacerbating the inflammatory response of AD. Similarly, according to the WB results, AD mice exhibited elevated expression of STAT1, which were significantly reversed by EGCG intervention (p < 0.01 or < 0.05).

(A) The protein expression levels of RAGE and STAT1 in the skin tissues were analyzed by WB, and the quantitative statistical analyses are shown in Figure (B). Data are presented as mean ± SD for each group (n = 3). #p < 0.05, ##p < 0.01 in comparison to Normal group; * p < 0.05, ** p < 0.01 versus in comparison to Model group.
Discussion
AD is an abnormal immune-mediated skin disease resulting from a combination of environmental, hereditary, and immunological factors. It is known for its chronic nature and the potential for persistent, long-term flare-ups. The hallmark clinical manifestations of AD are typically eczema-like rashes, dryness of the skin, and severe itching, all of which present significant obstacles in terms of therapeutic management. 16 In our research, we firstly established a DNFB-induced mouse model to assess the effectiveness of EGCG treating AD. Additionally, the comprehensive application of computational strategies encompassing network pharmacology and in silico molecular docking, was conducted to uncover the multi-target mechanisms of EGCG against AD.
In alignment with prior studies,17,18 DNFB-treated mice exhibited significantly elevated serum IgE levels, which confirmed the successful modeling of AD. The administration of EGCG notably lowered the serum IgE levels. Meanwhile, macroscopic assessments revealed that the AD-like lesional skin, the dermatitis score, and the degree of skin swelling were all mitigated by the oral intake of EGCG. These results suggests that EGCG could be a potential therapeutic agent for AD.
In addition, to delve deeper into the impact of EGCG on skin inflammation, we conducted H&E and TB assays to scrutinize the pathological alterations in the dorsal skin. Mice subjected to DNFB induction displayed significant thickening of the epidermis and a substantial infiltration of inflammatory cells, mirroring the histopathological characteristics observed in AD patients. Studies have indicated that there is an elevated presence of mast cells in the affected skin areas of individuals with AD. When IgE antibodies encounter allergens, they bind to the high-affinity IgE receptors on mast cells, prompting these cells to release granules containing histamine, lipid mediators, and IL-4. This release is instrumental in triggering the occurrence of intense itch and accelerates the progression of AD.19,20 Consistently, AD mice assembled more numbers of mast cells in their dorsal skin than normal mice. EGCG treatment markedly improved skin histopathology and caused a notable decrease in the accumulation of mast cells in the lesional skin.
Network pharmacology has played a pivotal role in providing a comprehensive understanding of the effects and underlying mechanisms of multi-component formulations by dissecting the interactions within chemical component-target networks. Molecular docking is a computer technology to predict interaction between small molecules with biomolecules, such as target proteins
In this research, we identified 37 shared targets of EGCG against AD. PPI analysis suggested that EGCG is capable of modulating multiple inflammation, immunity, and oxidative-associated targets simultaneously, thereby reducing AD-associated skin lesions, rather than focusing on a solitary target. Through topological analysis, the top 10 key targets were identified: MMP9, NF-κB1, BCL2, TLR4, HIF1A, STAT1, MMP2, MMP13, ABCB1, and MAPK14. Among these, ABCB1, also known as P-glycoprotein, is a 170-kDa protein with 1280 amino acids and two 6-transmembrane domains. It is well recognized for its function in transdermal drug delivery and has been involved in the genesis of various dermatological disorders. While direct evidence linking AD with ABCB1 in humans is limited, studies in mouse models hint at a potential protective role for ABCB1 against the itching and inflammation associated with AD.22,23 MMP9, MMP2, and MMP13 belong to matrix metalloproteinases (MMPs), which play critical role in tissue remodeling, cell migration, angiogenesis and epithelial apoptosis.24–26 MAPK14, a constituent of the MAPK family, serves as pivotal signaling pathways that govern a diverse array of cellular activities triggered by various stimuli, including proliferation, differentiation and stress response. 27 The transcription factor STAT1 can be phosphorylated and transferred into the nucleus, thus increasing the levels of pro-inflammatory cytokines. Suppression of STAT1 phosphorylation has been reported to ameliorate immune-mediated inflammatory dermatoses, such as AD and psoriasis. 28 Toll-like receptor 4 (TLR4) plays a crucial role in the innate immune system by initiating inflammatory reactions upon the detection of lipopolysaccharide (LPS) or other bacterial endotoxins. When TLR4 is excessively activated, it can lead to the release of cytokines, type I interferons, along with various other pro-inflammatory mediators, which acts as a significant catalyst for both acute and chronic inflammation. 29 Furthermore, the molecular docking data demonstrated that the 10 key targets had a strong binding affinity for EGCG, which validates the dependability of the network pharmacology approach.
To gain a deeper understanding of the biological roles that EGCG plays in treating AD, we conducted GO and KEGG enrichment analysis on the overlapping targets. GO analysis indicated that the potential targets of EGCG against AD refer to the modulation of various biological processes, such as collagen catabolic process, inflammatory response, proteolysis, angiogenesis, and positive regulation of cell migration. KEGG pathway enrichment analysis highlighted that these targets are predominantly involved in key pathways, including the AGE-RAGE signaling pathway in diabetic complications, the Th17/IL-17 signaling pathway, the NF-kappa B signaling pathway, the Toll-like receptor signaling pathway, and the TNF signaling pathway, among others. These pathways are considered pivotal mechanisms through which EGCG exerts its effects against AD. Advanced glycosylation end products (AGEs) are a series of complex molecules that are primarily formed under conditions of sustained hyperglycemia or during the aging process. When AGEs bind to their receptor RAGE, they could trigger the activation of downstream signaling pathways including NF-κB, MAPK, JAK/STAT, etc, leading to the generation of pro-inflammatory molecules (chemokines, cytokines, and adhesion molecules), the induction of oxidative stress, and the onset of inflammation.30–32 It has been documented that AGE/RAGE signaling pathway could be considered as an novel intervention strategy for skin diseases.31,33 Research has established that Th17 differentiation and the IL-17 signaling pathway are involved in the onset and progression of AD. Cytokines associated with Th17 cells, including IL-17A, IL-21, IL-22 as well as IL-6, are known to stimulate Th2-mediated immune response and impair the integrity of epidermal barrier.34,35
The “EGCG-target-pathway network”, which reflects correlations between the crucial pathways and the common targets of EGCG, suggest that EGCG might treat AD by acting on multiple molecular targets and biological pathways. Among them, the core target STAT1 and AGE-RAGE signaling pathway are closely related to inflammation and the regulation of immune response, which are considered to participate in various skin diseases.32,36 Further, we found that the targets of STAT1 and RAGE were both elevated in skin of AD model mouse. However, EGCG decreased the elevated expressions of STAT1 and RAGE. More importantly, the center target STAT1 selected by “EGCG-target-pathway network” was involved in the AGE-RAGE signaling pathway. 37
Undeniably, there are several limitations to the present study. First, the methodologies of network pharmacology and molecular docking are heavily dependent on the data and algorithms available, which means that their findings might not perfectly align with real-world outcomes due to the constraints of the databases and software used. Second, apart from the AGE-RAGE-STAT1 signaling, Th17 cell differentiation, IL-17 signaling pathway, NF-kappa B signaling pathway may also participate in EGCG against AD based on the network pharmacology analysis. Due to limitations in time and resources, we were not able to experimentally validate all the anticipated targets, or to integrate the cellular studies with clinical trials, which hindered us from fully elucidating the anti-AD mechanism of EGCG.
Conclusions
In summary, the present data have uncovered that EGCG could ameliorate AD-associated physical signs, clinical symptoms and pathological changes of AD mice induced by DNFB. Importantly, based on network pharmacology and molecular docking, we preliminary found that MMP9, NF-κB1, BCL2, TLR4, HIF1A, STAT1, MMP2, MMP13, ABCB1, and MAPK14 were the primary targets of EGCG acting on AD and the potential mechanisms of EGCG against AD were mainly related to the regulation of AGE-RAGE signaling pathway, Th17 cell differentiation, IL-17 signaling pathway, NF-kappa B signaling pathway. The animal experimental validation results further confirmed the significance of the core target STAT1 and the core pathway AGE-RAGE signaling pathway. These findings offered novel evidence and a new perspective for exploring the potential approach of EGCG against AD or even other immune-mediated skin diseases.
Footnotes
Acknowledgments
The authors would like to thank Yan Li from the Department of Medicine, Wuhan City College for her helpful advice on the network pharmacology analysis.
Author Contributions
Liu Tang and Xiao Jiang conceived and designed the whole experiment. Tianshuai Wang, Ruzhen Peng, and Jingyi Guo participated in the network pharmacology analysis and the completion of animal experiments. Liu Tang wrote the manuscript. Guangwen Luo and Shasha Fang reviewed and edited this manuscript. Liping Li and Benhong Zhou proposed modification suggestions. All the authors read and approved final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was granted approval by the Institutional Animal Care and Use Committee (IACUC) of the Animal Experiment Center of Wuhan University, Wuhan, China (Permit Number: 42010200006107).
Funding
This research was supported by “the Open Project of Hubei Key Laboratory of Wudang Local Chinese Medicine Research, Hubei University of Medicine (Grant No. WDCM2024011)”, “the Natural Science Foundation of Hubei Province (Grant No. 2023AFB239)”, “the Open Project of Hubei Key Laboratory (Grant No. 2023KFZZ015)”, “the Teaching and Research Project of Hubei association of Higher Education (Grant No. 2024XD216)”, and “China International Medical Foundation (Grant No. Z-2021-46-2101)”.
Statement of Human and Animal Rights
All the experimental protocols followed in this research strictly adhered to ethical guidelines of Wuhan University, China (Permit Number: 42010200006107) and the guidelines set forth in the “Guide for the Care and Use of Laboratory Animals,” Eighth Edition, 2011, published by the National Academies Press in Washington, USA.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
