Abstract
Introduction
Memecylon scutellatum (Lour.) Hook. & Arn. (Melastomataceae) is a lesser-known plant species indigenous to tropical and subtropical regions. This species has attracted scientific interest due to its potential medicinal properties, primarily driven by its traditional use in folk medicine across Southeast Asia. Ethnobotanical studies have documented its application in treating various ailments, including wounds, infections, and inflammation. 1 Despite its extensive use in traditional medicine, there is a paucity of scientific literature that thoroughly examines the chemical composition and biological activities of M. scutellatum.
In recent years, the growing interest in natural remedies and the urgent need for new drugs to combat emerging diseases have intensified the search for plants with significant bioactive properties. Memecylon species reported to possess potential pharmacological effects. 2 Previous studies on M. malabaricum, 3 M. umbellatum, 4 and M. edule 5 have identified compounds with antidiabetic, antimicrobial, and anti-inflammatory activities. However, chemical components and biological investigations of M. scutellatum have not been studied yet. Herein, we report the chemical constituents and anti-inflammatory activity of M. scutellatum.
Materials and Methods
General Experimental Procedures
All NMR spectra were recorded on a Bruker 600 MHz (Bruker, Massachusetts, United States). High-resolution mass data were obtained from Korea Basic Science Institute (KBSI, Chuncheon Center). Column chromatography (CC) was performed on silica-gel (Kieselgel 60, 230-400 mesh, Merck, Darmstadt, Germany), RP-18 resins (30-50 μm, Fuji Silysia Chemical Ltd, Kasugai, Japan) or HP-20 diaion (Mitsubishi Chemical Corporation, Tokyo, Japan). For thin-layer chromatography (TLC), pre-coated silica-gel 60 F254 (0.25 mm, Merck, Darmstadt, Germany) and RP-18 F254S (0.25 mm, Merck, Darmstadt, Germany) plates were used. HPLC was carried out using an AGILENT 1100 HPLC system using J'sphere H-80 column (250 × 20 mm), a flow rate of 3.0 mL, and a DAD detector.
Plant Material
The leaves of M. scutellatum were collected at Phuc Yen, Vinh Phuc, Vietnam in September 2022 and identified by Dr Nguyen The Cuong, Institute of Ecology and Biological Resources, VAST. A voucher specimen (USTH-MS01) was deposited at the Institute of Ecology and Biological Resources, VAST.
Extraction and Isolation
The dried powder of M. scutellatum leaves (9.5 kg) was sonicated with methanol (15 L MeOH at 45 °C, 2 times, each 2 h) and then filtered through filter paper, removed solvent under reduced pressure to yield 950 g of MeOH extract. The MeOH extract was suspended in water and successively partitioned with n-hexane and ethyl acetate, giving n-hexane (MS1, 45.1 g), ethyl acetate extracts (MS2, 66.6 g), and water layer (MS3).
The water layer (MS3) was chromatographed on a Diaion HP-20 column chromatography (CC) eluting with water to remove polar components, then increase the concentration of MeOH in water (25, 50, 75% and 100% MeOH, each 1 L) to obtain four fractions, MS3A (59.2 g), MS3B (25.3 g), MS3C (23.5 g), and MS3D (2.0 g). MS3A was chromatographed on a silica gel CC eluting with gradient solvents of dichloromethane/MeOH (20/1, 10/1, 5/1, 2.5/1, v/v) to give four fractions, MS3A1–MS3A4. MS3A1 was chromatographed on a silica gel CC eluting with CH2Cl2/MeOH (7/1, v/v) to give four fractions, MS3A1A-MS3A1D. MS3A1A was chromatographed on an RP-18 CC eluting with MeOH/water (1/3, v/v) to obtain two fractions, MS3A1A1 (80.0 mg) and MS3A1A2 (80.0 mg). MS3A1A1 was purified by an HPLC using 18% MeCN in water to yield
2-Hydroxy-6-Methylhept-5-en-3-yl) 3-O-[β-D-Apiofuranosyl-(1′′→6′)]-β-D-Glucopyranoside (1)
White amorphous powder;
Acid Hydrolysis
Compound
Cell Culture
Murine macrohage RAW 264.7 cells were provided by Professor Lee Jeong Hyung, Kangwon National University, Gangwo-do, Korea. Cells were cultured at 37 °C in a humidified 5% CO2 incubator in DMEM (Gibco, Invitrogen, USA), 10% FBS, 1% penicillin/streptomycin solution.
Cell Viability
Cytotoxicity of compounds was determined by MTT assay.6,7 Cells were grown in 96-well plates (50,000 cells/ 200 μL/ well). Cells were incubated in 24 h (37 °C, 5% CO2) before treated with the MTT solution (0.5 mg/mL) in 4 h. The formazan crystals formed were dissolved with isopropanol. Absorbance was measured at 570 nm using a Biotek microplate reader (Biotek Instruments, USA).
Estimation of Nitric Oxide Level
Murine macrophage RAW 264.7 cells were grown in 96-well plates at a concentration of 50,000 cells/200 μl/well. After an incubation in a humidified incubator at 37 °C, 5% CO2 for 24 h, cells were pre-activated for 30 min with compounds, then added 1 µg/mL lipopolysaccharide (Escherichia coli 0111: B4; Sigma Aldrich, USA). Cells were incubated for 24 h, and nitric oxide level was evaluated by Griess assay with a ratio of 1:1 (v/v) and calculated by standard curve of NaNO2. Optical density was read at 540 nm wavelength by Biotek microplate reader (Biotek Instruments, USA).
Statistical Analysis
The results are presented as mean ± SD of three independent experiments. Statistical significance between multiple groups was evaluated using ANOVA, followed by the Dunnett method for comparisons. P value of < 0.05 was considered statistically significant.
Results and Discussion
Dried leaves of M. scutellatum were extracted with MeOH and fractionated with dichloromethane, ethyl acetate (EtOAc), and water. From the water layer and subsequently separation, one new and fourteen known compounds were isolated.
Compound
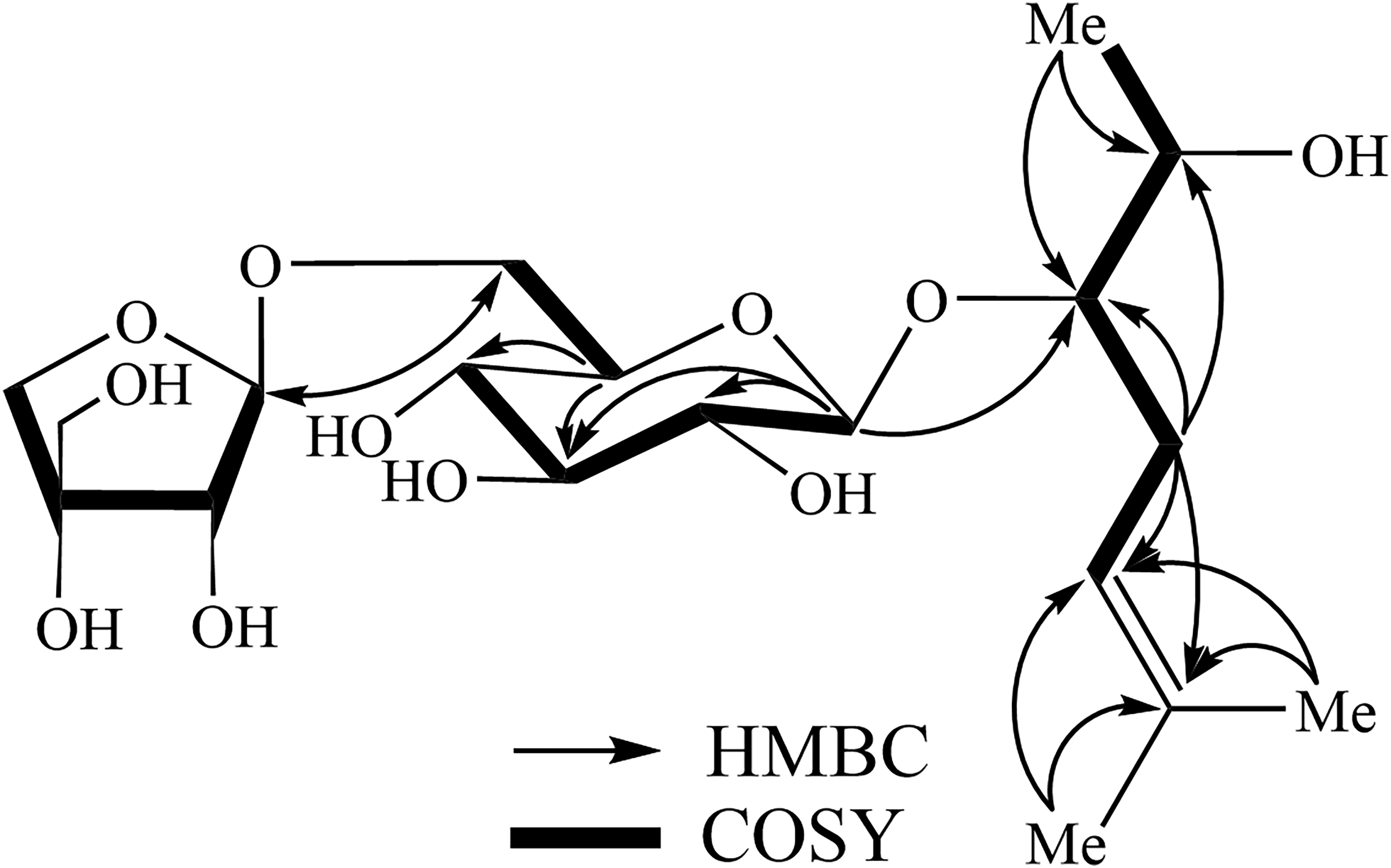
The key HMBC and COSY correlations of compound
The known compounds were identified to be 1-O-(β-D-glucopyranosyl)-2-[2-methoxy-4-(ω-hydroxypropyl)-phenoxy]-propan-3-ol (

The chemical structure of compound
Nitric oxide (NO) is a signaling molecule that plays a crucial role in the development of inflammation and is recognized as a pro-inflammatory mediator. NO inhibitors represent a significant therapeutic advancement in the treatment of inflammatory diseases.
20
To assess the cytotoxic effects of all isolated compounds, we conducted the MTT assay. The results showed that compounds

Effect of compounds
Inhibitory Effect of NO Production of Compounds

Cardamonin was used as positive control, showed NO inhibition of 52.0 ± 0.5% at a concentration of 3 μM. Each bar show mean ± SD of three independent experiments performed in triplicate (***P < 0.001 compared LPS). Values are shown as means ± SD (n = 3).
Conclusions
In our study, we identified one new compound, named memeloside (
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Vietnam Academy of Science and Technology (VAST) under grant number THTEXS.05/22-25.
