Abstract
Three dihydrostilbene derivatives (
The genus Pouzolzia (Urticaceae family) comprises around 40 flowering species. Most of them are shrubs and distribute mainly in tropical region. 1 Several of the species are herbs such as P. hirta, P. indica, P. sanguinea, and P. zeylanica. They have been used in traditional medicines for the treatment of inflammation, diarrhea, and ulcers. 2 Phytochemical studies of Pouzolzia species have not been intensively investigated and mostly focused on P. zeylanica. 3 -5 Phenolic compounds in terms of flavonoids and lignans were isolated from P. zeylanica. Some of these were reported to be potent anti-inflammatory agents by inhibiting either nitric oxide production or activation of peroxisome proliferator-activated receptor beta and peroxisome proliferator-activated receptor gamma enzymes. 4,5 With the aim to find anti-inflammatory constituents from Pouzolzia species, we describe herein the elucidation of a new dihydrostilbene derivative and a new neolignan, together with 6 known compounds, from the aerial part of P. sanguinea. Anti-inflammatory activity of the isolated compounds was evaluated by their inhibitory effect on NO production in lipopolysaccharide (LPS)-activated BV2 cells.
Results and Discussion
Compound
Nuclear Magnetic Resonance Spectral Data for Compounds 1, 2, and 4.

Measured at aCD3OD, b125 MHz, c500 MHz.
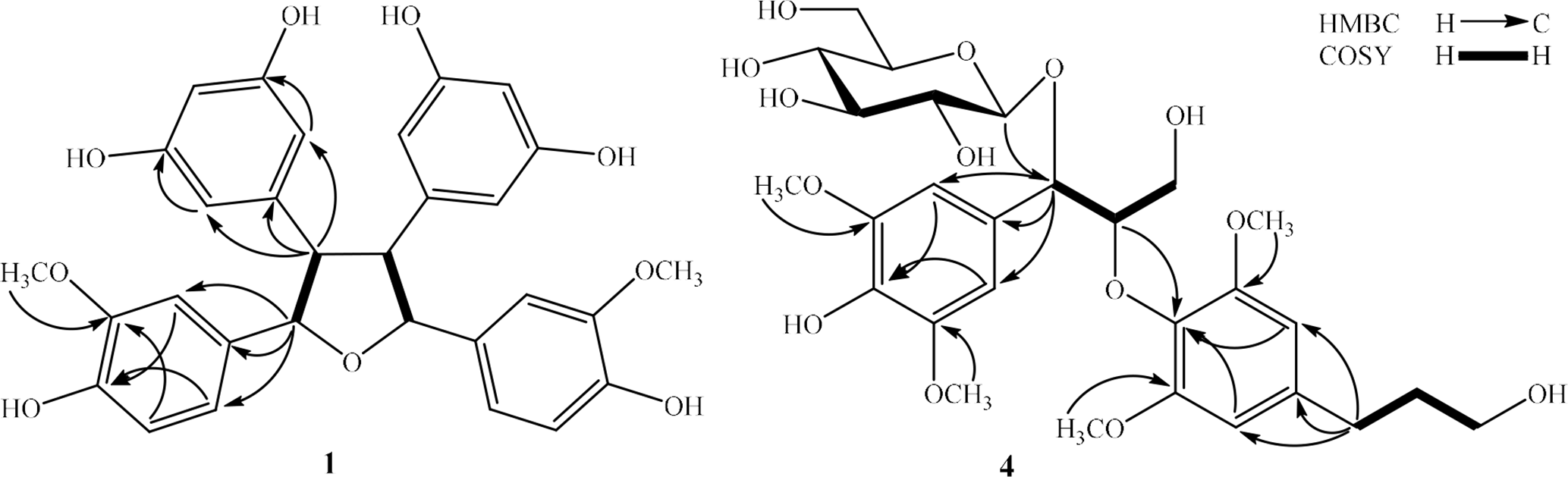
Key HMBC and COSY correlations of compounds 1 and 4. COSY, correlation spectroscopy; HMBC, heteronuclear multiple bond correlation.

Chemical structure of compounds 1-
Compound
The other isolated compounds were determined to be pouzolignan K (
Neolignans and stilbene derivatives are naturally occurring polyphenols found in the plant kingdom. Neolignans are formed by dimerization of 2 phenylpropanoid (C6-C3) units. However, stilbene derivatives also occur in the form of monomeric stilbenes (1,2-diphenylethylene core) and oligomeric stilbenes. Their chemical structures are characterized by the presence of terminal phenyl groups, which are functionalized with various numbers of hydroxyl and/or methoxy groups. Neolignans and stilbenes have a wide range of biological properties such as antioxidant, anti-inflammatory, anti-neurodegenerative, antitumor, antiviral, and antimicrobial activities.
13,14
In this work, the anti-inflammatory activity of compounds
Inhibitory Effect of Compounds 1-

Abbreviations: Abbreviations: NT, not tested; LPS, lipopolysaccharide; L-NMMA, NG-monomethyl-L-arginine.
aCell viability was evaluated at compound concentration of 80 µM.
bL-NMMA was used as a positive control.
Material and Methods
General Experimental Procedures
Optical rotation was recorded using a Jasco P-2000 digital polarimeter. HR-ESI-MS was measured on an Agilent 6530 Accurate Mass Q-TOF instrument, and 1-dimensional (1D)- and 2D-NMR spectra on a Bruker 500 MHz spectrometer; data acquisition was analyzed by MestReNova software. The circular dichroism spectrum was measured on a Chirascan spectrometer. Preparative HPLC was performed on an Agilent 1100 system using a YMC J’sphere ODS-H80 column (4 µm, 20 × 250 mm). Thin-layer chromatography was carried out on precoated silica gel 60 F254 and/or RP-18 F254S plates. Compounds were detected by spraying with H2SO4 solution (5%) followed by heating on a hot plate.
Plant Material
The plant samples were collected at Lam Dong province, Vietnam in March 2018. P. sanguinea (Blume) Merr. was identified by Dr Nguyen The Cuong, Institute of Ecology and Biological Resources, Vietnam Academy of Science and Technology (VAST). A voucher specimen (number: NCCT0318) was deposited at the Institute of Ecology and Biological Resources (VAST).
Extraction and Isolation
The dried and powdered aerial parts of P. sanguinea (15 kg) were macerated 3 times with methanol in an ultrasonic bath (each 20 L, in 60 minutes at room temperature). The solution was evaporated under reduced pressure to give the methanol extract (730 g). This was suspended in water and extracted in turn with n-hexane, methylene chloride, and ethyl acetate to yield the corresponding n-hexane, methylene chloride, and ethyl acetate extracts, and the water layer. The ethyl acetate extract (34 g) was chromatographed on a silica gel column, eluting with methylene chloride and methanol (40/1, 20/1, 10/1, 5/1, 3/1, 1/1, v/v) to give 6 fractions E1-E6. Fraction E2 was separated on a reverse-phase C-18 column chromatography, eluting with methanol/water (1/1, v/v) to give 3 fractions E2A-E2C. Fraction E2A was purified by pre-HPLC using acetonitrile in water (22%) as mobile phase to give compounds
Pouzobistilbene A (1)
Yellow amorphous powder,
Pouzosanoside A (4)
Yellow amorphous powder,
Nitric Oxide Assay
NO production was determined by nitrite concentration in the culture medium using Griess reaction. BV2 cells were maintained in Dulbecco’s modified Eagle medium supplemented with 5% fetal bovine serum and 1% penicillin-streptomycin medium. The cells were dispensed into a 96-well culture plate (4 × 104 cells/well) and incubated at 37 °C in a humidified atmosphere (5% CO2 and 95% air). After 24-hour incubation, the cells were treated with/without compounds and then stimulated with LPS (100 ng/mL) for the next 30 minutes. After an additional 24-hour incubation, the cell culture medium (50 µL) was mixed with an equal volume of Griess reagent for 10 minutes. Absorbance was read at 520 nm on an Emax reader. The amount of nitrite accumulated in the medium was obtained from a standard curve, which was built by NaNO2 serial dilution. NG-monomethyl-L-arginine was used as a positive control. After medium collection for the Griess test, cell viability was estimated by adding MTT (0.2 mg/mL) and incubating for 2 hours. The supernatant was carefully aspirated and the formazan crystals were dissolved in 200 µL of dimethyl sulfoxide. Absorbance was read at 540 nm and compared with the control group. Data are expressed as mean ± SD of triplicate experiments. Statistical analysis was performed by GraphPad Prism software.
Conclusion
Two new compounds pouzobistilbene A (
Supplemental Material
Figure S1 - Supplemental material for New Neolignan and Dihydrostilbene Derivatives from Pouzolzia sanguinea Inhibit NO Production in LPS-Activated BV2 Cells
Supplemental material, Figure S1, for New Neolignan and Dihydrostilbene Derivatives from Pouzolzia sanguinea Inhibit NO Production in LPS-Activated BV2 Cells by Le Thi Hong Nhung, Phan Thi Quyen, Trinh Thi Thuy, Nguyen Thanh Tam, Nguyen Thi Hoang Anh, Do Thi Thanh Xuan, Nguyen Xuan Nhiem, Bui Huu Tai and Phan Van Kiem in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the Graduate University of Science and Technology, Vietnam Academy of Science and Technology under grant number GUST.STS.ĐT2019/HH03.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
