Abstract
Objectives
The objective of this study was to illustrate the core mechanisms of pomegranate seed's anticancer effects beyond its antioxidant properties.
Methods
Using MCF-7 breast cancer cells, this study explores the impact of pomegranate seed on production of intracellular reactive oxygen species (ROS), cell survival, DNA damage, mitochondrial membrane potential, and apoptotic gene expression.
Results
Our findings revealed that pomegranate inhibited cell growth via promoting apoptosis. We observed noticeable enhancement in production of intracellular ROS in pomegranate seed-treated cells, along with increased of DNA damage. Additionally, pomegranate seed treatment led to a dose-dependent decrease in mitochondrial membrane potential, indicative of malfunction of mitochondria, resulted in initiation of apoptosis. Furthermore, we found differential regulation of apoptotic gene expression, with significant reductions in caspase 3 and Bcl-2 genes expression across all concentrations of pomegranate seed tested, and reductions in Bax and caspase 7 gene expression observed only with higher concentrations.
Conclusion
These results highlight the multifaceted mechanisms through which pomegranate seed exerts its anticancer effects, including ROS-mediated, DNA damage, dysfunction of mitochondria, and modulation of apoptotic pathways. Thus, pomegranate seed holds promise as a novel treatment option for therapeutic management of breast cancer.
Introduction
Breast cancer remains a significant global health concern, affecting women primarily between 40 to 55 years of age. Each year, approximately 2.3 million women receive a breast cancer diagnosis worldwide. 1 In the United States alone, nearly 281 550 new cases are anticipated in 2021, resulting in over 43 600 deaths. 2 Notably, the incidence of male breast cancer is also rising, with an estimated 2650 cases and around 530 deaths projected for 2021 in the United States. 3 These sobering statistics underscore the urgent need for continued research, early detection, and effective treatments to combat breast cancer. Despite advancements in therapeutic modalities, the search for novel and efficacious treatments continues, driven by the need to enhance patient outcomes and abate the disease burden.
Extensive research studies have highlighted the link between ingestion of fruits and vegetables rich diet to lower risk of development of cancer. These foods are abundant in phytochemicals, which possess potent anti-cancer properties.4,5 Moreover, natural products often exhibit multi-targeted effects while maintaining minimal side effects, positioning them as promising candidates for cancer treatment. 6 These substances, sourced from various organisms including microbes and plants, represent a vast repository of bioactive molecules with potential therapeutic applications. Polyphenols and flavonoid compounds, in fruits and vegetables, have been correlated to suppress genes expression, proteins, and signaling pathways involved in tumor growth and progression. 7 These natural compounds hold promise as potential therapeutic agents for cancer patients.
Among these natural compounds, pomegranate (Punica granatum) has garnered substantial attention for its potential anti-cancer properties. 8 The pomegranate fruit, rich in bioactive phytochemicals, has been revered for centuries in traditional medicine for its therapeutic benefits. Of particular interest are its seeds, which are a rich source of polyphenolic compounds such as ellagitannins, anthocyanins, and flavonoids, each with documented antioxidant, anti-inflammatory, and anti-carcinogenic properties. 9 In traditional medicine, pomegranate seeds have been historically documented to possess medicinal properties that regulate urine discharge and alleviate the burning sensation associated with urination as well as prevent the development of nephrotoxicity. 10 Additionally, pomegranate seeds have been used in the management of several ailments, including bronchitis, digestive issues, diarrhea, and parasite-infected wounds. 11 These traditional uses underscore the perceived therapeutic benefits of pomegranate seeds in managing various health conditions across different cultures and periods. For example, compounds originated from pomegranate peel, such as corilagin and pseudopelletierine, have been separated through isolation process and studied for their potential therapeutic effects. 12
Breast cancer is a heterogeneous disease, encompassing various subtypes with distinct molecular profiles and clinical behaviors. Among these, the MCF-7 cell line, derived from human breast adenocarcinoma, has been extensively studied and serves as a representative model for hormone-responsive breast cancer. 13 Its responsiveness to hormonal manipulation and well-characterized molecular features makes MCF-7 an invaluable tool for investigating potential therapeutic interventions.
Despite advances in understanding breast cancer biology and progression of targeted therapies, the quest for reliable and efficient treatments remains paramount. Consequently, exploring the therapeutic potential of natural compounds like pomegranate seed extract against breast cancer, particularly MCF-7 cells, holds significant promise. The multi-targeted actions of pomegranate compounds, including inhibition of proliferation, induction of apoptosis, and suppression of angiogenesis, 14 suggest their potential utility as adjuvant or standalone therapies in the management of breast cancer.
In this study, we comprehensively evaluated the therapeutic efficacy of pomegranate seed extract against the MCF-7 breast cancer cell line. Through number of in vitro laboratory assessment, we assessed its impact on cell viability, proliferation, molecular signaling pathways, and cell death implicated in breast tumors progression as a therapeutic target.
Materials and Methods
Pomegranate Seed Extract
Pomegranate seed extract capsules were purchased from (NeoCell, Irvine, California). It was dissolved in Dimethyl sulfoxide (DMSO) to obtain a final concentration of 50 mg/mL. DMSO is an effective solvent for a wide array of organic and inorganic materials. DMSO is miscible with water and most organic liquids.
Cell Culture
MCF-7 human breast cancer cell line was purchased from American Type Culture Collection (ATCC, Manassas, VA, USA) and was maintained in DMEM supplemented with 10% FBS and 1% penicillin/streptomycin solution. Cells Cultivation occurred under standard conditions of 37 °C and a 5% CO2 atmosphere until reaching confluence.
Evaluation of Cell Viability
To assess cell viability, a previously described methodology was employed. 15 Initially, cell seeding was performed at 5 × 105 cells/mL. Subsequently, the cells underwent exposure to different concentrations of pomegranate seeds (5, 20, 50, 125, 250 and 500 µg/mL) for another 48 h. Then, The MTT assay reagent poured and left to incubate for two hours at 37 °C. After incubation, the culture medium was removed and formed formazan crystals were dissolved in isopropanol followed by 15 min agitation before measuring absorbance at 540 nm using a spectrophotometer (Synergy-H1; BioTek).
Assessment of Natural Red Uptake (NRU)
In 96-well plates, initially MCF-7 cells were seeded at 5 × 105 cells/mL (density) and left for incubation (temp. 37 °C). Following this, MCF-7 cells were exposed to different amounts of pomegranate seeds up to 60 µg/mL, for 48 h. Subsequently, addition of 200 µL per well of Neutral Red Uptake (NRU) dye which was allowed to incubate (temp. 37 °C) for three hours. Afterwards, 100 µL of 0.5 g of CaCl2 was used to fixed cells, followed by a destaining step on a shaker for 15 min. Finally, microplate spectrophotometer was used to measure absorbance at 540 nm.
Analysis of Intracellular ROS Generation
Reactive oxygen species (ROS) assay was used to evaluate ROS generation from Sigma-Aldrich (St. Louis, MO, USA).
ROS production was checked by using 2’, 7-dichlorofluoresceindiacetate (DCFH-DA) also called (H2DCF-DA) dye as fluorescence agent measures the activity of ROS inside the cell. In 96-well black microplates, cells were cultured while 10% cultured medium was used for six well plates at concentration of 5 × 105 cells/mL, left for incubation (temp. 37 °C). After incubation, previous procedure was repeated as described earlier and allowed to incubate for another 48 h. The culture media was then aspirated and mixing of fresh cultural media with concentration of 100 µL agitated with 5 µg concentration of deep red solution in each well plate, while same solution with one milliliter was added per well of the 6-well plate. After addition, incubation of cells (temp. 37 °C) was done for about 1 h. Absorbance was subsequently measured using spectrophotometer at range of 485-528 nm. For imaging purposes, aspiration of nutrient broth from the 6-well plate and washing of cells for several times with 1X PBS was carried out. Finally, fluorescence microscope was used for taking images (DMLB, Leica, Germany).
Evaluation of Superoxide Dismutase (SOD) Activity
Cells were seeded at 1 × 106/mL and left for incubation (temp. 37 °C). Afterwards, the cells underwent a treatment of 5, 30 and 60 µg/mL of pomegranate seeds, followed by an additional 48-h incubation period (temp. 37 °C). After treatment, cells were gathered and rinsed with 1x PBS before being lysed using cold buffer and sonicated for about five minutes with one milliliter solution containing 210 mM mannitol, 1 mM EGTA, 20 mM HEPES, and 70 mM sucrose at pH 7.2. Then, centrifugation of lysed cells at 1500 rpm was carried out and supernatant was collected. Cayman Superoxidase dismutase (SOD) kit No.706002 was utilized to assessed SOD activity. In a 96-well plate, 200 µL of diluted radical detector was mixed with 10 µL of sample per well. The reaction was initiated by adding 20 µL of diluted Xanthine Oxidase in every well plate, accompanied by incubation of about 30 min with shaking on room temperature. Measurement of absorbance was performed at 440-460 nm. The SOD activity was estimated using the formular below:
SOD = [(sample LR − Y − (intercept/slope) × 0.23 mL/ 0.01 mL] × sample dilution.
Assessment of Glutathione (GSH) Levels
Cells were seeded at 1 × 106/mL and left for incubation (temp. 37 °C). Afterwards, these were exposed to the treatments mentioned above, followed by incubation for an additional 48 h (temp. 37 °C). After treatment, separation of cells through scraper, washing with 1X PBS, and sonication for about five minutes with 1 mL of PBS was carried out. Then, centrifugation of sonicated cells at 1500 rpm at 4 °C about five minutes, and transfer of supernatant to 5 mL tube was performed. The Glutathione (GSH) assay was conducted using the Cayman GSH kit No.703002 followed by manufacture's guidelines. Briefly, mixing of cocktail (150 µL) containing MES Buffer with sample (50 µL), reconstituted cofactor, reconstituted enzyme, deionized H2O, and 5,5’-dithio-bis-2-nitrobenzoic acid was executed. Then, incubation of plate in darkness for about 25 min, and finally measurement of absorbance was carried out at 410 nm.
Assessment of Mitochondrial Membrane Potential (MMP)
To investigate the significant effect of pomegranate seed on mitochondrial membrane permeability, JC-1 fluorescent dye was used. Coverslips were used to culture MCF-7 cells in 6-well plates. Also, black bottom plate was used at amount of 5 × 105 cells in medium (10%), followed by incubation period in 5% CO2 atmosphere at 37 °C. Afterwards, cells were treated with 5, 30 and 60 µg/mL of pomegranate seed for 48 h. After treatment, staining of with JC-1 for 20 min at 37 °C in 5% CO2. In the case of the microplate, monomer JC-1 fluorescence was measured on excitation and emission at 475 nm and 530 nm, respectively, while aggregate JC-1 fluorescence was measured on excitation and emission at 535 nm 590 nm, respectively. Following staining, cells were rinsed three times with PBS, and coverslips were transferred to microscopic slides for fluorescence imaging using CRCL's LSM780 NLO confocal microscope.
TUNEL Assay
MCF-7 cells were seeded on coverslips in 6-well plates and incubated as described above. Following this, cells were subjected to the pomegranate seeds for 48 h. Subsequently, 1% paraformaldehyde was used to fix the cells and washed with PBS. Abcam kit was used to execute tunnel assay, with cells incubated in 70% alcohol for 15 min. Following incubation, labeling solution of 51 µL of DNA was supplemented, accompanied by additional one hour incubation. Propidium Iodide/RNase staining buffer was included, and thirty min incubation of cells was ensured. After washing with rinse buffer solution and PBS, coverslips were inverted onto microscopic slides for visualization under a confocal microscope.
DAPI Staining for Apoptosis
In six well plates, cells were seeded on coverslips and incubated under standard conditions, accompanied by pomegranate seed at the above-mentioned concentrations for 48 h. 3% paraformaldehyde was used to fix cells and were then collected, centrifuged, and resuspended in binding buffer containing DNA dye (2.5 mg/mL), 4′,6-diamidino-2-phenylindole (DAPI). After a 15-min incubation in darkness, cells with nuclear damage were counted under fluorescence microscope.
Gene Expression Analysis
RNA samples were purified and used to synthesize cDNA using a cDNA kit (ThermoFisher USA, Cat No.00881457). Amplified PCR products were confirmed via agarose gel electrophoresis. RT-PCR was executed to find target sequences of apoptotic-related genes (Bcl2, Bax, p53, Cas-3 & 7, and TNF-α) following exposure to drugs for a day. Fluorescence detection was employed to visualize the accumulation of amplification products, along with, 2^- ΔΔCT method was used to calculate the fold expression.
Results
Assessment of Cell Viability
The significance of pomegranate seed extract on the growth and survival of MCF-7 breast cancer cells was investigated. MCF-7 cells were subjected to different amounts of pomegranate seed extract, ranging up to 500 µg/mL, for period of 48 h. Analysis depicted in Figure 1 demonstrated a gradual decrease in cell survival due to increasing concentrations of pomegranate seed extract, indicating a dose-dependent effect. An influential reduction in cell survival was evident at concentrations equivalent to or below 125 µg/mL. Origin Graphing Software was used to detect IC50 (80.9 µg/mL). Three selected concentrations blow the detected IC50 were selected for further investigations.

IC50 detection of investigated compounds. Cell viability of MCF-7 cells after treatment with different concentrations of pomegranate seeds for 48 h as evaluated by MTT assay. Each value represents the mean SE ± (n = 3), (*P < .05).
Assessment of Pomegranate Cytotoxicity at Non-Lethal Levels
To verify the cytotoxic effects of pomegranate seeds, the NRU assay was employed. This assay relies on a dye that identifies viable cells with intact membranes. By assessing the movement of lysosomal enzymes outside the cell, indicative of membrane damage, the dye quantification gauges cytotoxicity. MCF-7 cells were exposed to varying concentrations of pomegranate seeds (5, 30, and 60 µg/mL) for a duration of 48 h. As depicted in (Figure 2), pomegranate seeds elicited cytotoxic responses in MCF-7 cells across all tested concentrations. These findings suggest the potential of pomegranate seed extract to induce cytotoxic effects, underscoring its relevance for further exploration in cancer therapy.

Cytotoxicity (%) effect of pomegranate seeds on MCF-7 cells after 48 h as assessed by neutral red uptake (NRU) assay. Each value represents the mean SE ± (n = 3), (*P < 0.05, **P < .01, ***P < .001).
Assessment of Intracellular ROS Generation
The MCF-7 cell line was subjected to varied amounts of pomegranate seeds, as described previously, to assess their effect on intracellular ROS generation. Our findings revealed that treatment with 30 and 60 µg/mL of pomegranate seeds showed noticeable enhancement in ROS production than standard (*P < .05), as described in Figure 3. It was discovered that generation of ROS through cells at 5 µg/mL was lower compared to at 30 µg/mL. This may be due to hormetic conditions of seeds at lower concentration. Taken together, this observation suggests likelihood of pomegranate seeds in influencing oxidative stress levels within cells, highlighting their possible impact on oxidative stress associated disorders signaling pathways of cells.

Generated ROS in MCF-7 cells after subjected to pomegranate seeds for 48 h. Each value represents the mean SE ± (n = 3), (*P < .05).
Evaluation of Oxidative Stress Markers
Superoxide dismutase (SOD) is a crucial metalloenzyme that aids in the breakdown of superoxide anions into oxygen and hydrogen peroxide, serving as a vital cellular antioxidant defense mechanism against oxidative damage. When the MCF-7 cell-line was subjected to different concentration of pomegranate seeds to assess their impact on antioxidant activity, Figure 4 shows that SOD activity was notably reduced observed across all treatments than standard group (P < .01).

Superoxide dismutase activity in MCF-7 cells subjected to pomegranate seeds for 48 h. Each value represents the mean SE ± (n = 3), (*P < .05**P < .01).
Reduced GSH, a tripeptide with significant scavenging properties against various reactive species both intra- and extracellularly, was also assessed. As ROS levels were high and reduced with gradual increase in seed concentrations, the GSH levels in MCF-7 cells were measured post-exposure to the drugs. The outcomes highlighted a remarkable enhancement in cells subjected to all test groups than standard group (P < .01), as described in Figure 5. These findings underscore the potential of pomegranate seeds to modulate stress markers of oxidation, highlighting their role in defense mechanism of cellular antioxidant.
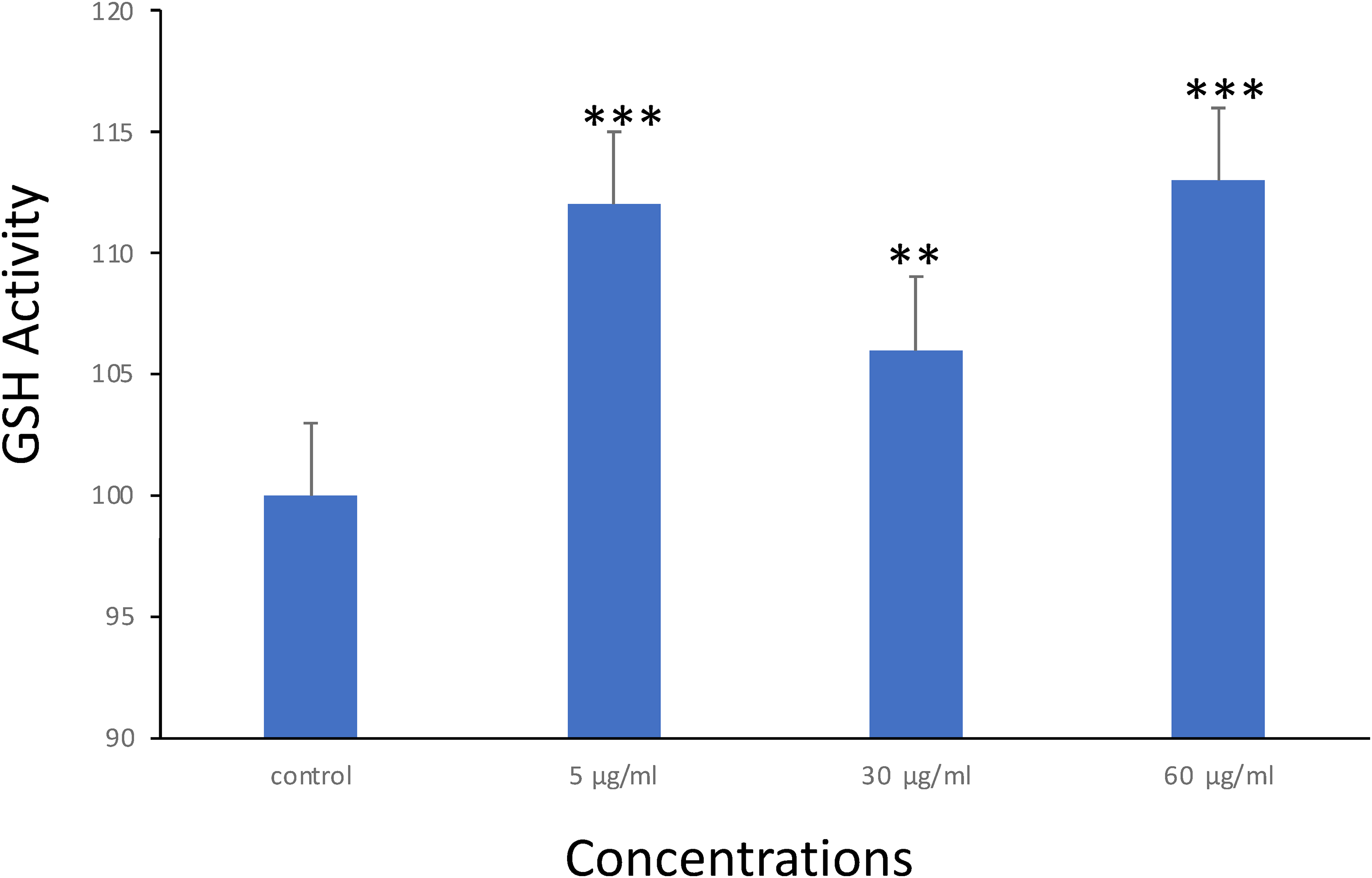
GSH activity in MCF-7 cells after treatment with pomegranate seeds for 48hr. Each value represents the mean SE ± (n = 3), (**P < .01, ***P < .001).
Assessment of Membrane Potential (MMP)
The evaluation of mitochondrial membrane potential serves as a valuable tool for analyzing changes in mitochondrial function and apoptosis. This assessment employs two fluorescent dyes: one stains healthy mitochondria (with high membrane potential) in red fluorescence, and the other one is damaged mitochondria (with low membrane potential) in green fluorescence. As depicted in (Figure 6), the ratio of JC-1 aggregates to monomer for treated cells to pomegranate seeds indicated a remarkable decline post incubation for 48 h in a dose-dependent manner. This observation suggests that pomegranate seed treatment results in disruption of mitochondrial membrane potential that could be indicative of mitochondrial dysfunction and significant initiation of cell death process.

Images representing MMP loss in MCF-7 cells after exposure to pomegranate seeds for 48 h.
Assessment of Nuclear Damage
DAPI staining was employed to detect nuclear damage post exposure to pomegranate seed. Based on the change in cell morphology, nuclear damage analysis revealed that exposure of MCF-7 cells to pomegranate seeds (5 and 60 µg/mL) led to remarkable rise in cell death process along with increment in number of cells exhibiting condensed chromatin within the nucleus, resulting in smaller, denser nuclei than standard unexposed cells, as illustrated (Figures 7A and B). This outcome indicated that pomegranate seed treatment induces apoptosis in MCF-7 cells, highlighting its novelty and significance as a therapeutic agent against breast cancer.

Evaluation of MCF-7 cells morphology under the fluorescence microscope after treated with pomegranate seeds. (A); DAPI staining (200X). Arrows indicate the cell death process. (B); mean fluorescence intensity. Each value represents the mean SE ± (n = 3), (*P < .05).
Assessment of Apoptosis
DNA damage following treatment was evaluated to elucidate the impact of pomegranate seeds on apoptotic events in MCF-7 cells. The TUNEL assay, which detects cells underwent death via labeling DNA breaks with dUTP, alongside propidium iodide stain (PI stain) labelling indicating cell membrane compromise, was conducted. As depicted in (Figure 8), the apoptotic-labelled MCF-7 cell's percentage was allowed to treat to pomegranate seeds was outstandingly increase than standard untreated group at both 5 and 30 µg/mL exposures (P < .001 and P < .05, respectively). Moreover, cell membrane integrity was assessed by PI staining revealed a significantly higher number of cells undergoing treatment with pomegranate seeds exhibiting compromised cell membranes. This is specified through an intense green fluorescence corresponding to PI entry into the cell via the compromised membrane. These outcomes concluded that pomegranate seed treatment cause DNA damage and compromises cell membrane integrity, contributing to apoptotic events in MCF-7 breast cancer cells.

TUNEL assay results showed the influence of pomegranate seeds on apoptotic cells (%). Experiments shown are in triplicates. (*P < .05, ***P < .001).
Gene Expression of Cancer Modulating Genes
Cancer development and progression modulates through intrinsic pathway of cell death and it involves various factors including caspase 3 and caspase 7, pro-apoptotic Bax, and anti-apoptotic Bcl-2. 16 To further understand the apoptotic mechanisms identified in the previous results, we evaluated the expression of these apoptotic genes. Our findings revealed significant reductions in the expression levels of caspase 3 and Bcl-2 genes across all concentrations tested. Conversely, Bax and caspase 7 genes expression levels revealed remarkable decline only at concentrations of 30 and 60 µg/mL (Figure 9). These outcomes highlight that pomegranate seed treatment influences key apoptotic genes expression, causing cell death via intrinsic pathway in MCF-7 cells. Furthermore, we assessed p53 and TNF-α gene expression as shown (Figure 9). Our findings also suggested that both p53 and TNF-α gene expression notably decrease in cells undergone treatment with pomegranate seeds at all concentrations tested. This further reiterates the suggestion that exposure to the seeds influences genes that modulate cancer likely via the pathway of cell viability.

mRNA levels of apoptotic genes in MCF-7 cells after treatments. Value in each MCF-7 cell indicates the mean of three experiments ± SE. n = 3, (*P < .05, **P < .01, ***P < .001).
Discussion
The present study explored the prospect of anti-cancer properties of pomegranate seeds in MCF-7 breast cancer cell line, focusing on various cellular mechanisms involved in apoptosis. Our findings shed light on the intricate molecular pathways through which pomegranate seed exerts its anti-cancer effects. The assessment of cell viability revealed a dose-dependent decrease in MCF-7 cell viability after exposure with pomegranate seeds. Moreover, the Neutral Red Uptake assay showed a substantial surge in cytotoxicity in cancer cells subjected to pomegranate seeds, further supporting their promise as anti-cancer agents. These observations align with preceding reports demonstrating the cytotoxic effects of pomegranate extract on cancer cell lines. Shirode et al (2014) showed that pomegranate extract from 20 µg/mL considerably antagonized MCF-7 cells proliferation. 17 Similar findings were communicated by Nasr et al (2023), who showed both seeds and peels of pomegranate significantly decreased cell viability of HepG2 cancer cells. 18 Similarly, Constantin et al (2014) illustrated that punicic acid and its derivatives caused a notable decline in cell viability in two breast cell lines, with corresponding incline in G0/G1 cell cycle phase in contrast to unexposed cells. 19
Our investigation into intracellular ROS production unveiled a notable enhancement in ROS generation in MCF-7 cells subjected to pomegranate seeds. Elevated ROS levels causes oxidative stress, leading to DNA injury and ultimately triggering apoptosis. 20 ROS, including superoxide anion (O2•−), hydrogen peroxide (H2O2), and hydroxyl radical (•OH), can directly attack DNA molecules, causing various types of damage such as single-stranded breaks, double-stranded breaks, and base modifications. 21 Additionally, ROS can indirectly induce damage to DNA by activating downstream signaling that eventually produce genotoxic compounds or by interfering with DNA repair mechanistic. 22 Consequently, accrual of ROS-induced DNA damage can overwhelm cellular repair mechanisms, triggering apoptotic pathways as a protective response to deter the propagation of injured DNA. 23 Consistent with this notion, our observations unveiled a heightened concentration of DNA injury in the MCF-7 cells following exposure to pomegranate seeds. This finding also corresponds with that of Shirode et al (2014), who also reported increased damage and to DNA ROS on treatment with pomegranate extracts. 17 This finding indicates that the cytotoxicity of pomegranate seeds may be facilitated, partly, by triggering of oxidative stress in cancer cells. Furthermore, our analysis regarding oxidative stress markers revealed intriguing results. While SOD activity decreased significantly in cells exposed to pomegranate seeds, indicating a drop in antioxidant defenses, the levels of reduced GSH increased significantly. These results infer a complex interplay between antioxidant defenses and oxidative stress as a result of pomegranate seed treatment. The evaluation of MMP provided additional understanding related to apoptotic mechanisms induced by pomegranate seeds. Treatment with pomegranate seeds prompted a dose-dependent decline in mitochondrial membrane potential, indicating mitochondrial dysfunction, a hallmark of apoptosis. 24 In line with this observation, flow cytometry analysis demonstrated a sizable rise in apoptotic cells following pomegranate seed treatment, as supported by DAPI staining. Altogether, it seems redox imbalance may have led to reduction in mitochondrial membrane potential thus resulting in mitochondrial outer membrane permeabilization (MOMP), similar to the findings reported by. 20 Heightened expression and stimulation of Bax destabilizes mitochondrial outer membrane, culminating in cytochrome C release and induction of downstream processes that precede the stimulation of caspase 3 and 7. 25 Bcl-2, an anti-apoptotic protein, binds to Bax to safeguard against mitochondrial dysfunction. However, an elevated expression of Bax relative to a low level of Bcl-2 expression results in an increased Bax/Bcl-2 ratio, thereby facilitating Bax-mediated MOMP. 20 This leads to cytochrome C release and subsequent caspases 3 and 7 activation along intrinsic pathway of apoptosis. Consequently, the Bax/Bcl-2 ratio has emerged as a prognostic indicator for survival in various cancers. It has been utilized as a predictive marker for cancer cells response to chemotherapy. Our investigation into the apoptotic genes expression revealed differential regulation of key apoptotic factors. While the Bcl-2 and caspase 3 expression levels were significantly reduced across all concentrations of pomegranate seeds tested, expression levels of Bax and caspase 7 was significantly reduced only at higher concentrations. It appears that the Bax to Bcl2 ratio was highest at 5 and 30 µg/mL, explaining the reason for the highest DNA damage observed respectively at these two concentrations. These highlight that pomegranate seed treatment modulates the apoptotic genes expression, promoting apoptosis in MCF-7 cells via the intrinsic apoptotic pathway. The observed decrease in p53 and TNF-α genes expression in MCF-7 cells subjected to pomegranate seeds at all tested concentrations is an encouraging finding that aligns with the overarching theme of present investigation. These further enlighten that exposure to pomegranate seeds affects key genes associated with cancer modulation, particularly through the cell survival pathway. Firstly, the p53 gene downregulation is noteworthy as p53 is a critical tumor suppressor gene involved in regulating cell cycle progression, repair of DNA, and apoptosis. 26 Reduced p53 expression can impair these regulatory functions, potentially promoting cell proliferation and survival, and ultimately contributing to tumorigenesis. Our findings of decreased p53 expression in pomegranate seed-treated cells support the notion that pomegranate seeds exert anti-cancer effects by modulating crucial signaling mechanisms concerned with cell survival and apoptosis. Similarly, the observed decrease in TNF-α gene expression further underscores the potential anti-cancer properties of pomegranate seeds. TNF-α is a pro-inflammatory cytokine with complex roles in cancer biology, including promoting cell survival or death based on cellular context. Although, TNF-α plays a dual function in cancer survival, downregulation of TNF-α expression may mitigate its pro-survival effects, potentially sensitizing cancerous cells to apoptotic signals and inhibiting tumor growth. 27 Our results intimate that pomegranate seeds may employ their anti-cancer effects, partly, by modulating TNF-α expression and its downstream cellular signaling.
Taken together, the decrease in expression of p53 and TNF-α genes in pomegranate seed-treated cells provides further evidence supporting the promising therapeutic utility of pomegranate seeds in patients with breast cancer. These results add to the growing insight of the molecular mechanistic inherent with anti-cancer effects of pomegranate seeds, highlighting their promise as a natural therapeutic agent for cancer treatment.
Limitation of the Study
The in vitro analysis demonstrated the anti-cancer effects of pomegranate seeds. However, several factors could have contributed to these effects including; in vitro conditions that may not fully reflect the complex microenvironment and interactions present in vivo. In addition, current study not has captured the full spectrum of all breast cancer cell lines, which could response differently to the pomegranate seeds. Moreover, applied different concentrations, exposure time, and other experimental models should be investigated in future work to explore potential effect of pomegranate seeds and resistance mechanisms. Taken together, we believe these limitations will provide valuable insights for further research and highlight the need for additional studies to address these challenges.
Conclusion
This study provides valuable understandings of multifaceted mechanisms underlying the anticancer effects of pomegranate seed in MCF-7 human breast cancer cell line through instigation of apoptosis. This apoptotic effect was preceded by a substantial rise in intracellular ROS production, suggesting oxidative stress in pomegranate seed-induced cytotoxicity. Furthermore, our findings revealed a dose-dependent decrease in mitochondrial membrane potential following pomegranate seed treatment, indicative of mitochondrial dysfunction and apoptosis initiation via intrinsic pathway. Moreover, our study highlighted the effect of pomegranate seed on apoptotic genes expression, with significant reductions witnessed for Bcl-2 and caspase 3 genes expression across all concentrations of pomegranate seeds tested. Additionally, reductions in Bax and caspase 7 gene expression were observed at high concentrations of pomegranate seed. These results imply that PSE modulates apoptotic pathways by disrupting the balance involving pro-apoptotic and anti-apoptotic factors. The identification of ROS-mediated DNA injury and mitochondrial impairment as key mechanisms underlying pomegranate seed-induced cytotoxicity provides valuable insights for future research aimed at optimizing the therapeutic efficacy of pomegranate-derived compounds. Overall, our study adds to the improving pool of evidence supporting utilization of natural compounds, like pomegranate seed, as alternative or adjunctive breast cancer treatments. Further investigations are warranted to elucidate the specific bioactive components of pomegranate seed carrying anticancer effects and to evaluate its efficacy in pre-clinical and clinical settings. Ultimately, establishing pomegranate-based therapies holds great promise for improving breast cancer outcomes.
Footnotes
Acknowledgements
This work was funded by Researchers Supporting Project number (RSP2024R26), King Saud University, Riyadh, Saudi Arabia. Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R62), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Author Contributions
R A. A and B. A. assessed the generation of iROS, and antioxidant assays. H. A., B. J, K. N. Y. evaluated apoptotic gene expression. M.Z. and A.A. A. evaluated performed Tunnel assay and cell morphology. A. A., N. S. J. and N. A. performed the cytotoxic assays. N. H. A. and M. Z. evaluated DNA damage and cell survival. S. A. and M.Z. wrote and edited the article. All authors have approved the final article.
Competing Interests
The authors declare no competing interests.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval Statement
This study did not require ethical approval as it did not involve human participants and animal studies.
Funding
This work was funded by Researchers Supporting Project number (RSP2024R26), King Saud University, Riyadh, Saudi Arabia. Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R62), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
