Abstract
Excessive accumulation of reactive oxygen species (ROS) has been regarded as a major contributor of pathogenesis in neurodegenerative diseases. N-trans-feruloyltyramine (NTF), an alkaloid isolated from several plants, has demonstrated an ability to be a potent antioxidant. In this study, the antioxidative and anti-apoptotic properties of NTF extracted from the stems of Polyalthia suberosa were investigated in the human neuroblastoma cell line SK-N-SH. NTF at concentrations ranging from 10 µM to 500 µM were not toxic to cells and reduced intracellular ROS levels significantly. Furthermore, pre-treatment of NTF significantly decreased H2O2-induced ROS generation and attenuated H2O2-mediated cytotoxicity. An increase in the expression of Bax and activated caspase-3 and reduction of Bcl-2 mediated by H2O2 was reversed by pre-treating the cells with 100 µM NTF. Likewise, NTF suppressed the increase of caspase-3 activity induced by H2O2. In conclusion, the findings reveal that NTF improves H2O2-induced intracellular ROS generation and decreases apoptosis. These protective effects of NTF could be useful for oxidative stress-related neurodegenerative conditions.
Introduction
Reactive oxygen species (ROS) are a group of reactive molecules containing oxygen, generated as a byproduct of several cellular metabolisms. Although beneficial when maintained at low concentration, high levels of ROS have several detrimental effects, including lipid peroxidation, and protein and DNA damage. 1 In order to prevent damage, cells utilize an antioxidant defensive system to scavenge ROS. However, in some conditions, production of ROS is much more pronounced, and can overwhelm cellular antioxidative capacity, leading to oxidative stress. Conditions known to be involved with oxidative stress include, but are not limited to, cardiovascular diseases, diabetes, cancer and neurodegenerative diseases.1, 2 Among these, diseases of the nervous system are of interest since neurons exhibit characteristics that can cause them to become severely damaged by ROS, such as high proportion of unsaturated fatty acids within membranes, high oxygen consumption and scarce amount of antioxidants.2, 3 Oxidative stress has been identified as a shared factor among neurodegenerative diseases such as Alzheimer's disease, amyotrophic lateral sclerosis and Parkinson's disease.4, 5 Thus, finding a way to reduce oxidative stress may pave the way to prevent or ameliorate disease progression.
N-trans-feruloyltyramine (NTF, Figure 1A) is an alkaloid extracted from several plants, including the stems of Polyalthia suberosa, 6 stems of Tinospora tuberculata, 7 branches of Enicosanthum membranifolium, 8 stems of Polygonum sachalinensis, 9 leaves of Solanum sordidum, 10 aerial parts of Triclisia sacleuxii, 11 seeds of Datura metel, 12 processed bulbs of Allium sativum, 13 and twigs of Celtis occidentalis. 14 Previous studies have reported that NTF is a potent antioxidant9, 10, 12, 13, 15, 16 and exhibits other pharmacological properties such as α-glucosidase inhibition 9 and anti-tumor activity. 13 In this study, we investigated the protective effects of NTF isolated from the stems of Polyalthia suberosa against H2O2-mediated cytotoxicity using an in vitro model of human neuroblastoma cell line SK-N-SH. The effect of NTF on ROS levels, cell viability, expression of pro-apoptotic as well as anti-apoptotic proteins and apoptotic activity were evaluated.

(A) Chemical structure of N-trans-feruloyltyramine (NTF), an alkaloid isolated from the stem of P. suberosa. (B-C) Effects of NTF on cell viability and ROS levels. (B) The SK-N-SH cells were treated with NTF at concentrations of 10, 25, 50, 100, 150, 250 and 500 μM for 24 h. Unlike H2O2, which significantly lowered cell viability, the viability was not altered by NTF treatment at tested concentrations. (C) Significant decrease in ROS levels in NTF-treated cells was observed at all concentrations tested. AA-treated cells was used as a positive control. **p < 0.01, ***p < 0.001, ****p < 0.0001 versus untreated control. Values are the mean
Results
NTF was Not Toxic to SK-N-SH Cells and Exhibited Antioxidative Properties
To assess whether NTF treatment would be detrimental to the SK-N-SH cells, varied concentrations of NTF were applied, ranging from 10 µM to 500 µM. A cell viability analysis revealed that addition of NTF at indicated concentrations did not affect viability of the SK-N-SH cells while addition of 150 µM H2O2 significantly decreased the viability (Table 1 and Figure 1B, one-way ANOVA: F = 28.39, p < 0.0001; Dunnett's multiple comparison test: p < 0.0001 for control vs. 150 µM H2O2). A significant reduction in ROS levels was found in samples treated with NTF at all tested concentrations and in a sample treated with well-known antioxidant ascorbic acid (AA) (Figure 1C, one-way ANOVA: F = 40.65, p < 0.0001; Dunnett's multiple comparison test: p < 0.0001 for control vs. 150 µg/ml AA, control vs. 10 µM NTF, control vs. 50 µM NTF, control vs. 100 µM NTF and control vs. 150 µM NTF; p = 0.0003 for control vs. 25 µM NTF and control vs. 250 µM NTF; p = 0.0038 for control vs. 500 µM). The findings demonstrate that NTF at concentrations between 1 µM to 500 µM is not toxic to the SK-N-SH cells and has antioxidative effects.
Percentage of Cell Viability in NTF Treated Cells Compared to Untreated Control.

Values are the mean ± SEM of three independent experiments.
NTF Ameliorated H2O2-Induced Cytotoxicity Through Inhibition of H2O2-Mediated ROS Induction
As a ROS, H2O2 causes reduction of cell viability in a concentration-dependent manner (Table 2, one-way ANOVA: F = 56.61, p < 0.0001; Dunnett's post-hoc test: p = 0.0253 for 37.5 μM H2O2 vs. control; p = 0.0031 for 75 μM H2O2 vs. control; p < 0.0001 for 150, 300, 600, 1200 and 2400 μM H2O2 vs. control). An H2O2 concentration that caused 50% reduction in viability of the cells (CC50) was 157 μM (Figure 2A). Thus, to test whether NTF would counteract H2O2-mediated reduction in cell viability, we pre-treated the cells with various concentrations (10, 25, 50, 100, 150, 250 and 500 µM) of NTF for three hours prior to the induction of cytotoxicity with H2O2. By using a phase contrast inverted microscope, we noticed morphological changes of the cells treated with H2O2 which unlike untreated cells (Figure 2B), shrunk and became detached, resembling dead cells (Figure 2C). Interestingly, pre-treatment with NTF prior to H2O2 addition lessened the number of detached cells (Figure 2D). A cell viability analysis consistently revealed a sharp decrease in viability of H2O2-treated cells (Figure 2E, one-way ANOVA: F = 49.46, p < 0.0001; Dunnett's test: p < 0.0001 for control vs. H2O2-treated group). Nevertheless, similar to pre-treatment with AA, the addition of NTF was able to help protect the cells against H2O2-induced toxicity as determined by a significant higher percentage of viability in cells pre-treated with NTF at all tested concentrations compared to the H2O2-treated group (Figure 2E, Dunnett's test: p < 0.0001 for H2O2-treated group vs. pre-treated AA and H2O2-treated group vs. pre-treated NTF at all concentrations). To further assess whether NTF-mediated protection against H2O2 is a consequence of a decrease in ROS levels induced by H2O2, the SK-N-SH cells were pre-treated with NTF at the indicated concentrations. As expected, H2O2 treatment caused a sharp rise in ROS levels (Figure 2F, one-way ANOVA: F = 17.69, p < 0.0001; Dunnett's test: p < 0.0001 for control vs. H2O2-treated group). Pre-treatment of NTF at all tested concentrations significantly improved an H2O2-mediated increase in ROS levels (Figure 2F, Dunnett's test: p < 0.0001 for H2O2-treated group vs. pre-treated AA and H2O2-treated group vs. pre-treated NTF at all concentrations).
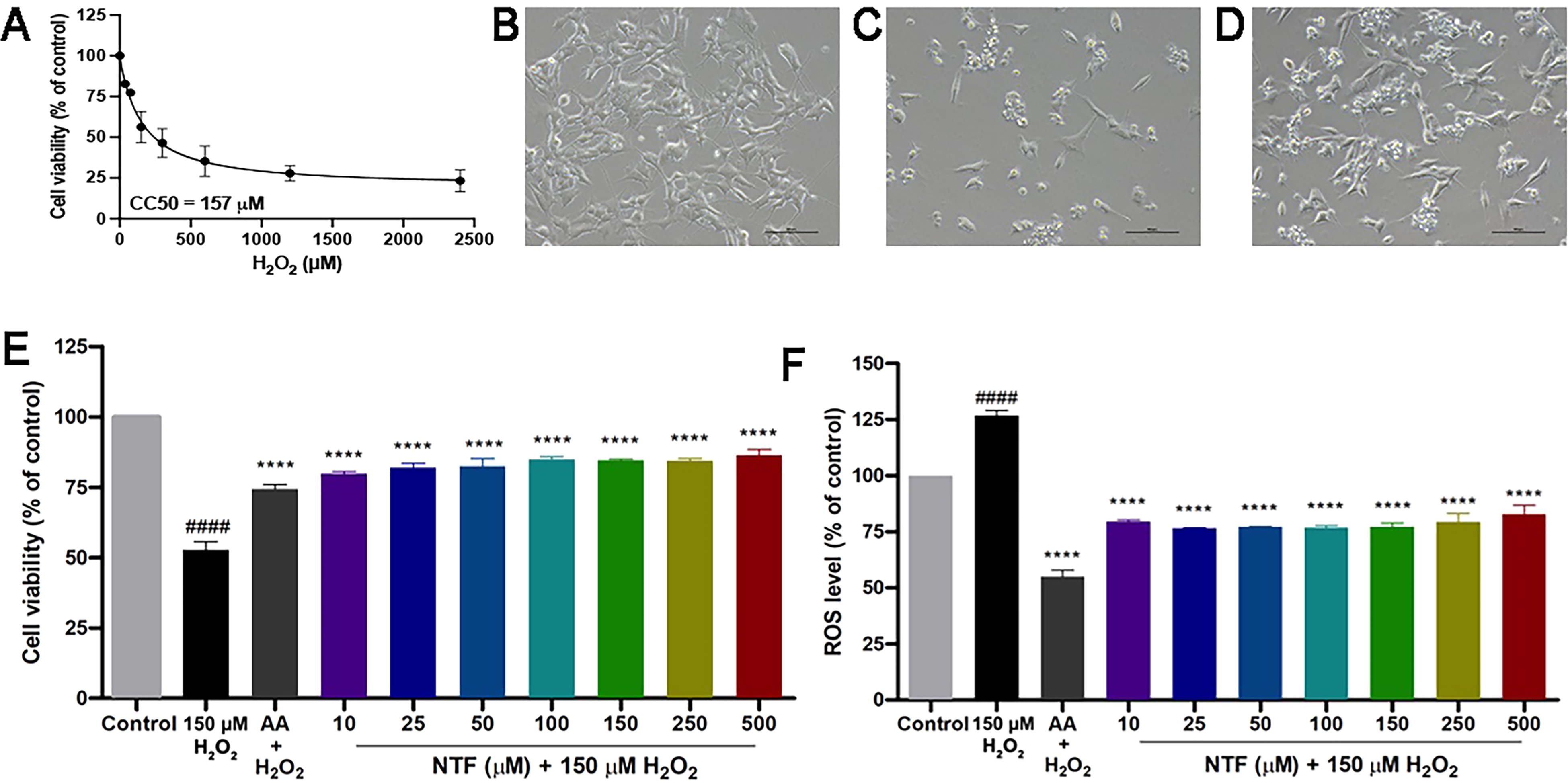
NTF reversed cytotoxicity induced by H2O2. (A) The 50% cytotoxic concentration (CC50) of H2O2 in SK-N-SH cells was 157 μM. (B-D) Morphology of untreated cells (B), H2O2-treated cells (C) and cells pretreated with NTF prior to H2O2 addition (D). Scale bars are 100 μm. (E) Treatment of H2O2 caused significant reduction in cell viability. The cytotoxic effect of H2O2 was abolished by pre-treatment of NTF at all tested concentrations. (F) An increase in ROS levels was observed in cells treated with 150 μM H2O2. Pretreatment of NTF decreased H2O2-induced ROS generation. ####p < 0.0001 versus untreated control. ****p < 0.0001 versus H2O2-treated control. Values are the mean + SEM of three independent experiments.
H2O2 Cytotoxicity in SK-N-SH Cells.

*p < 0.05, **p < 0.01, ****p < 0.0001 versus H2O2-treated control. Values are the mean ± SEM of three independent experiments.
NTF Inhibited H2O2-Mediated Apoptosis in SK-N-SH Cells
It is an established fact that apoptosis can be triggered by H2O2.17, 18 Hence, a decline in observed cell viability of H2O2-treated SK-N-SH cells is the result of apoptosis activation. A Western blot analysis demonstrated that levels of the pro-apoptotic protein Bax were elevated in H2O2-treated cells (Figure 3A, one-way ANOVA: F = 9.747, p = 0.0018; Dunnett's test: p = 0.0017 for control vs. H2O2-treated group). The H2O2-induced Bax expression was abolished when cells were pre-treated with 100 μM NTF (Figure 3A Dunnett's test: p = 0.0089 for H2O2-treated group vs. pre-treated NTF at 100 μM). Interestingly, levels of the anti-apoptotic protein Bcl-2, which was down-regulated in the cells treated with H2O2 (Figure 3B, one-way ANOVA: F = 8.405, p = 0.0031; Dunnett's test: p = 0.0236 for control vs. H2O2-treated group) was significantly elevated when the cells were pre-treated with NTF at 50 μM and 100 μM (Figure 3B, Dunnett's test: p = 0.0031, and p = 0.0012, respectively). To further determine anti-apoptotic effects of NTF, quantitation of activated caspase-3 levels as well as measurement of its activity was performed. In the presence of H2O2, up-regulation of activated caspase-3 was found (Figure 3C, one-way ANOVA: F = 9.361, p = 0.0021; Dunnett's test: p = 0.0137 for control vs. H2O2-treated group). This caused a significant increase in caspase-3 activity (Figure 3D, one-way ANOVA: F = 17.04, p = 0.0002; Dunnett's test: p < 0.0001 for control vs. H2O2-treated group). Consistent with Bax levels, pre-treatment of 100 μM NTF significantly decreased activated caspase-3 levels (Figure 3C, Dunnett's test: p = 0.0017 for H2O2-treated group vs. pre-treated NTF at 100 μM). Additionally, caspase-3 activity was significantly lower in cells pre-treated with NTF at all tested concentrations (Figure 3D, Dunnett's test: p = 0.0007 for H2O2-treated group vs. pre-treated NTF at 25 and 100 μM; p = 0.0016 for H2O2-treated group vs. pre-treated NTF at 50 μM). These findings indicate the effects of NTF against H2O2-mediated apoptosis.

Anti-apoptotic effects of NTF. (A and C) Pre-treatment of NTF at 100 μM significantly reduced Bax (A) and activated caspase-3 (C) levels in H2O2-treated SK-N-SH cells. (B) Cells pre-treated with 50 and 100 μM NTF prior to H2O2 addition showed significantly higher levels of Bcl-2. (D) Down-regulation of activated caspase-3 caused a decrease in caspase-3 activity in cells pre-treated with NTF prior to H2O2 treatment. #p < 0.05, ##p < 0.01, ####p < 0.0001 versus untreated control. **p < 0.01, ***p < 0.001 versus H2O2-treated control. Values are the mean + SEM of three independent experiments.
Discussion
NTF is an alkaloid found in various types of plants, such as tropical shrubs (Polyalthia suberosa), herbaceous plants (Tinospora tuberculata, Polygonum sachalinensis) and edible plants (Allium sativum). NTF is known for its broad ranges of biological effects such as being an antioxidant,10, 13, 15 anti-inflammation, 19 anti-melanogenesis, 8 α-glucosidase inhibition, 9 anti-cancer, 13 and anti-microbial.11, 20 Its availability in many plant species, together with its broad pharmacological effects, make NTF an attractive molecule for drug development. Antioxidative effects of NTF have been demonstrated in a number of studies. In a rat cortical neuronal culture, NTF at 25–250 μM ameliorated amyloid-β peptide (Aβ)-mediated ROS generation. 15 Likewise, reduction of ROS levels was observed in human hepatocyte cell line L02 treated with NTF. 13 In this study, we evaluated possible antiradical effects of NTF in the SK-N-SH cell lines. In conditions where the oxidative status was within normal limits, treatment of NTF ranging in concentration from 10 μM to 500 μM significantly decreased ROS levels (Figure 1C) without compromising cell viability (Figure 1B), demonstrating the antioxidative property of NTF. More importantly, in conditions where oxidative stress is induced by 150 μM H2O2, pre-treatment of NTF at all tested concentrations also significantly enhanced the oxidant scavenging capacity of the cells (Figure 2F). In addition to a ROS reduction effect in H2O2-treated cells, we also observed an increase in cell viability in cells pre-treated with NTF (Figure 2E).
It is common knowledge that H2O2 can induce apoptosis in various cell types via ROS-mediated activation of several mediators such as p53, p38 mitogen-activated protein kinase (MAPK), extracellular signal-regulated protein kinase 1/2 (Erk1/2) and c-Jun N-terminal kinase (JNK), which causes stimulation of pro-apoptotic proteins including Bax and inhibition of anti-apoptotic proteins such as Bcl-XL and Bcl-2.18, 21–23 Consequently, a release of cytochrome c triggers the intrinsic pathway of apoptosis via activation of caspase-3.24, 25 Thus, it is highly likely that a decline in cell viability in H2O2-treated SK-N-SH cells (Figure 2E) would be a result of apoptosis. Although H2O2 can also induce other types of cell death such as necroptosis, this usually occurs when high dosage of H2O2 is used.25, 26 The use of antioxidant N-acetyl-cysteine (NAC) has been shown to reverse H2O2-mediated neuronal apoptosis by impeding JNK, Erk1/2 and p38 activation in the differentiated neuroblastoma cell line SH-SY5Y. 27 Likewise, pre-treatment of an iridoid glycoside loganin alleviated H2O2-induced phosphorylation of JNK, Erk1/2 and p38, augment the Bcl-2/Bax ratio, leading to decreased activated caspase-3 levels. 28 Consistent with previous studies, our data also showed evidence of increased apoptosis as determined by increased levels of Bax (Figure 3A), a decrease in Bcl-2 levels (Figure 3B), and an increase in levels and activity of activated caspase-3 (Figure 3C and D) in cells treated with H2O2. Collectively, a rise in cell viability in samples pre-treated with NTF prior to induction of cytotoxicity by H2O2 (Figure 2E) would result in reduced ROS levels, which in turn prevent apoptosis as evidenced by a restoration of Bax, Bcl-2 and activated caspase-3 levels (Figure 3A-C) as well as caspase-3 activity (Figure 3D). The anti-apoptotic effects of NTF reported in this study correlates well with other studies. NTF has demonstrated an ability to mitigate Aβ1−42-induced apoptosis in primary rodent neuronal cell culture. 15 A protective effect of NTF against H2O2-induce cytotoxicity in fetal hepatocyte cell line L02 has also been reported. 13
Although our study demonstrated protective effects of NTF against ROS-induced apoptosis in a cell line with neuronal phenotype, these effects would need to be tested in vivo. A bioavailability study conducted in a rat model revealed that, after oral administration of NTF at a dose of 20 mg/kg, its metabolites could be detected in plasma and urine. 12 Interestingly, the hydroxy groups at 4-position of benzene rings, which is important for ROS scavenging, 29 were well preserved in these metabolites. 12 This supports a possibility of using NTF as an antioxidative agent via oral administration. Nevertheless, recent evidence has shown that when using a parallel artificial membrane permeability assay (PAMPA), NTF is unable to passively cross the blood-brain barrier. 14 However, with the aid of novel strategies such as nanotechnology-based delivery, molecules could be engineered to cross the blood-brain barrier.30, 31 This would offer an option to determine the usefulness of NTF in protecting ROS-mediated neurodegeneration in vivo.
Conclusion
To summarize, our findings demonstrate the antioxidative effects of NTF in an in vitro SK-N-SH model. NTF treatment for up to 500 µM was not toxic to the SK-N-SH cells. Moreover, pre-treatment of NTF successfully prevented H2O2-induced ROS generation and resulted in inhibition of H2O2-mediated apoptosis. Given its antioxidative property, NTF may be useful in preventing and/or ameliorating severity of conditions where oxidative stress is involved in the pathophysiology.
Experimental
Chemicals
N-trans-feruloyltyramine (NTF) was isolated from a stem of Polyalthia suberosa, grown in Kalasin, Thailand, by chromatography using hot acetone extract. 6 Hydrogen peroxide was purchased from Merck. L-ascorbic acid, 2′,7′-dichlorofluoescin diacetate (DCFH-DA), dimethyl sulfoxide (DMSO) and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were obtained from Sigma.
Cell Culture and NTF Treatment
The SK-N-SH cell line, originating from human neuroblastoma, was obtained from ATCC. The cells were maintained in MEM (Gibco) supplemented with 10% (v/v) fetal bovine serum (FBS), 100 units/ml penicillin and 100 µg/ml streptomycin (all from Gibco) at 37 °C with 5% CO2. For each experiment, cells were dissociated with 0.25% trypsin-EDTA, counted, plated at 2 × 105 cells/ml density and maintained at 37 °C with 5% CO2 for 24 h. The cells were treated with NTF at indicated concentrations (10, 25, 50, 100, 150, 250 and 500 µM) for three hours before 150 µM H2O2 was added. Cells treated with 150 µg/ml ascorbic acid (AA) was used as a positive control. Analyses were performed 24 h after the addition of H2O2.
Cell Viability Analysis
Spectrophotometric analysis of MTT was used as described previously to determine the viability of SK-N-SH cells. 15 After being treated with H2O2 for 24 h, 20 µl of 5 mg/ml MTT was added to the cells in each well of a 96-well plate. The cells were kept at 37 °C with 5% CO2 for four hours. The medium was then removed and DMSO was added (150 µl for each well) to solubilize the formazan crystals. The absorbance was measured at 570 nm with a microplate reader (BioTek). Cell viability was presented as a percentage of untreated cells.
Measurement of ROS Production
Intracellular ROS levels were measured using DCFH-DA. After adding H2O2 for 24 h, 10 µM DCFH-DA was applied to the cells within each well of a 96-well plate before incubating at 37 °C with 5% CO2 for 30 min. Quantification of fluorescence from DCF was done using an excitation wavelength at 485 nm and emission wavelength at 530 nm with a Multi-Detection microplate reader (BioTek). ROS levels were presented as a percentage of untreated cells.
Western Blotting Analysis
Western blotting was performed as described previously with some modifications. 32 The SK-N-SH cells were cultured at a density of 1 × 106 cells/cm2. The cells were treated with NTF and H2O2 as described in the previous section. Twenty-four hours after being treated with H2O2, the cells were lysed using lysis buffer. The lysate was incubated on ice for 30 min before being centrifuged. The supernatant was obtained and the concentration of protein was determined using a nanophotometer (Implen). Samples were electrophoresed on 10% SDS–PAGE before being transferred to a nitrocellulose membrane (Millipore). The membranes were blocked with buffer comprising 5% bovine serum albumin (BSA) for 1 h at room temperature. The membranes were applied with primary antibodies (mouse anti-human ß-actin antibody, Cell signaling; mouse anti-human Bax antibody, Santa Cruz; rabbit anti-human Bcl-2 antibody, Cell signaling; rabbit anti-human activated caspase-3 antibody, Cell signaling) at 1:1000 dilution overnight at 4 °C. Then, the membrane was treated with appropriate HRP-conjugated secondary antibodies (goat anti-mouse IgG and goat anti-rabbit IgG, Santa Cruz) at 1:1000 dilution for two hours at room temperature. The membranes had enhanced chemiluminescence substrate solution (Biorad) applied to them and were visualized on x-ray film. The band density was analyzed using densitometric quantification by ImageJ software (National Institutes of Health).
Determination of Caspase-3 Activity
After being treated with H2O2 for 24 h, the cells were harvested and lysed. Approximately 50 µg of protein from the lysate of SK-N-SH cells were used to measure caspase-3 activity with the CaspACETM assay system (Promega). Briefly, 2 µl of the DEVD-pNA substrate was added into the lysate before incubating it at 37 °C for four hours. Absorbance of pNA, a product cleaved by activated caspase-3, was measured at 405 nm with a microplate reader (BioTek). Caspase-3 activity was presented as a percentage of controls.
Statistical Analyses
All experiments were conducted in three independent replicates. Results are presented as the mean ± SEM. Statistical differences were determined by one-way ANOVA with the Dunnett's multiple comparison test done to analyze differences between the H2O2-treated group and NTF-pre-treated groups using Prism version 9 (GraphPad Software). Statistical significance was accepted when a p-value was less than 0.05.
Footnotes
Acknowledgements
EPM is supported by Siriraj Graduate Scholarship, Faculty of Medicine Siriraj Hospital, Mahidol University. CV is supported by the Chalermphrakiat Grant, Faculty of Medicine Siriraj Hospital, Mahidol University.
Author Contributions
RS provided conceptual input, designed and performed experiments, analyzed data and drafted the manuscript. EPM and NT designed and performed experiments. WT provided conceptual input, analyzed data and edited the manuscript. CM designed experiments and analyzed data. PT & SN provided conceptual input. CV provided conceptual input, designed experiments, analyzed data and wrote the manuscript. All authors approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Faculty of Medicine Siriraj Hospital, Mahidol University, (grant number R016032001, R016333045).
Trial Registration
Trial registration is not applicable for this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
